Abstract
Background:
Angiotensin receptor neprilysin inhibitor (ARNI) was recommended by major guidelines as the frontline therapy for heart failure with reduced ejection fraction (HFrEF) since its clinical benefit was proved in the Prospective comparison of ARNI with ACEI to Determine Impact on Global Mortality and morbidity in Heart Failure (PARADIGM-HF) trial. However, little is known about its safety and effectiveness in real-world practice, often with sicker and more fragile patients. In addition, East Asia population is underrepresented in PARADIGM-HF trial.
Methods:
We performed a retrospective analysis of patients who received ARNI in 3 medical institutes located in Northern Taiwan. Patients who received a prescription of at least 30 days of ARNI were enrolled. The date of first prescription was defined as the index date, and a period of 12 months preceding the index date was defined as the baseline period.
Results:
A total of 452 patients were identified (age: 61.9 ± 15.0, male: 79.4%). Compared to PARADIGM-HF populations, our patients had higher values of baseline serum creatinine (mean: 1.5 vs 1.1 mg/dL) and B-type natriuretic peptide (BNP; median: 554.5 vs 255 pg/mL). After 12 months, 41.6% of the patients received less than half of the standard dose. Overall, all-cause death, cardiovascular death, and heart failure readmission rate were 3.0%, 1.1%, and 6.9% in 12 months, respectively. In those who had both baseline and 12-month data, renal function did not change (1.7-1.8 mg/dL,
Conclusion:
In our study, patients with HFrEF had higher BNP and serum creatinine level at baseline and had received lower dose of ARNI than the PARADIGM-HF populations. Angiotensin receptor neprilysin inhibitor appeared to be safe as regard renal function and effective in real-world practice. Left ventricular reverse remodeling was observed 1 year after heart failure medication treatment, including ARNI.
Keywords
Introduction
Sacubitril/valsartan, an angiotensin receptor neprilysin inhibitor (ARNI), was recommended by major guidelines 1,2 as the frontline therapy for heart failure with reduced ejection fraction (HFrEF) since its clinical benefit was proved by the Prospective comparison of ARNI with ACEI to Determine Impact on Global Mortality and morbidity in Heart Failure (PARADIGM-HF) trial. 3 Compared to enalapril, ARNI further reduced cardiovascular death or heart failure (HF) hospitalization and all-cause death in this pivotal trial.
However, patients with HFrEF in real world often have worse symptoms, worse renal function, and suboptimal medication prescription. 4,5 In addition, Asian population was underrepresentative in PARADIGM-HF trial, which accounted for only about 18% in the ARNI group. The purpose of the present study is to investigate the safety and effectiveness of ARNI in a tertiary medical center in Taiwan.
Methods
Data Source and Study Design
This is a single-arm observational study which uses retrospective collected data. The data included for analysis were obtained from the Chang Gung Research database (CGRD). Chang Gung Research database is a deidentified database derived from original medical records of Chang Gung Memorial Hospital, which includes 3 medical institutes in Northern Taiwan (Linkou, Taipei, and Taoyuan branches). 6 Figure 1 shows the patient identification in this study. Data of patients who received a prescriptions of ARNI for at least 30 days during January 2016 to May 2018 were extracted from CGRD. The date of first prescription was defined as the index date, and a period of 12 months preceding the index date was defined as the period for detecting comorbidities. Each patient was followed until the day of outcome occurrence or death within 1 year after the index date or May 31, 2018, whichever came first.

Patient inclusion criteria.
End Points
End points observed included all-cause death, cardiovascular death, HF readmission, composite of all-cause death or HF readmission, and composite of cardiovascular death or HF readmission. Other end points including all-cause readmission, nonfatal myocardial infarction, nonfatal stroke, and new renal replacement therapy were reported in the Supplementary Material. Subgroup analysis to assess the association between the composite end point (all-cause death or HF readmission) and baseline renal function was performed.
Statistics
Interval changes of echocardiographic parameters and laboratory data were compared from patients with available data between baseline and 12 months after the index date. Changes of continuous variables and categorical variables between baseline and 12th month were compared using paired sample
Results
A total of 452 patients with prescriptions for at least 30 days were included for analysis. Of them, 17 patients had only baseline data, so end point analysis was available for 435 patients. Finally, there were 192 patients alive and had echo examination data at 12-month follow-up (Figure 1).
Table 1 demonstrates the baseline characteristics of the study population. Mean age was 61.9 years (range: 21.2-99.6 years), and male was predominant (79.4%). Hypertension, hyperlipidemia, diabetes mellitus, chronic kidney disease (CKD; eGFR < 60 mL/min/1.73 m2), old myocardial infarction, and atrial fibrillation were the most prevalent comorbidities (all above 20%). The most common etiology for heart failure was ischemic cardiomyopathy. At baseline, mean creatinine level was 1.5 mg/dL, mean eGFR was 68.1 mL/min/1.73 m2, and median BNP level was 554.5 pg/mL. Mean left ventricular ejection fraction (LVEF) was 31.8%, with mean left ventricular end-diastolic diameter 61.6 mm, end-systolic diameter 51.8 mm, and left atrium diameter 46.1 mm at baseline. There were 23.5% of the patients with moderate or severe mitral regurgitation (MR). As to medication, 90.5% of the patients received β-blockers and 58.4% had mineralocorticoid receptor antagonists (MRAs) at baseline. Dosage of baseline HF medication is listed in the Supplementary Materials. Available baseline characteristics of the ARNI population in PARADIGM-HF trial are also listed in the table for comparison.
Baseline Characteristics.a
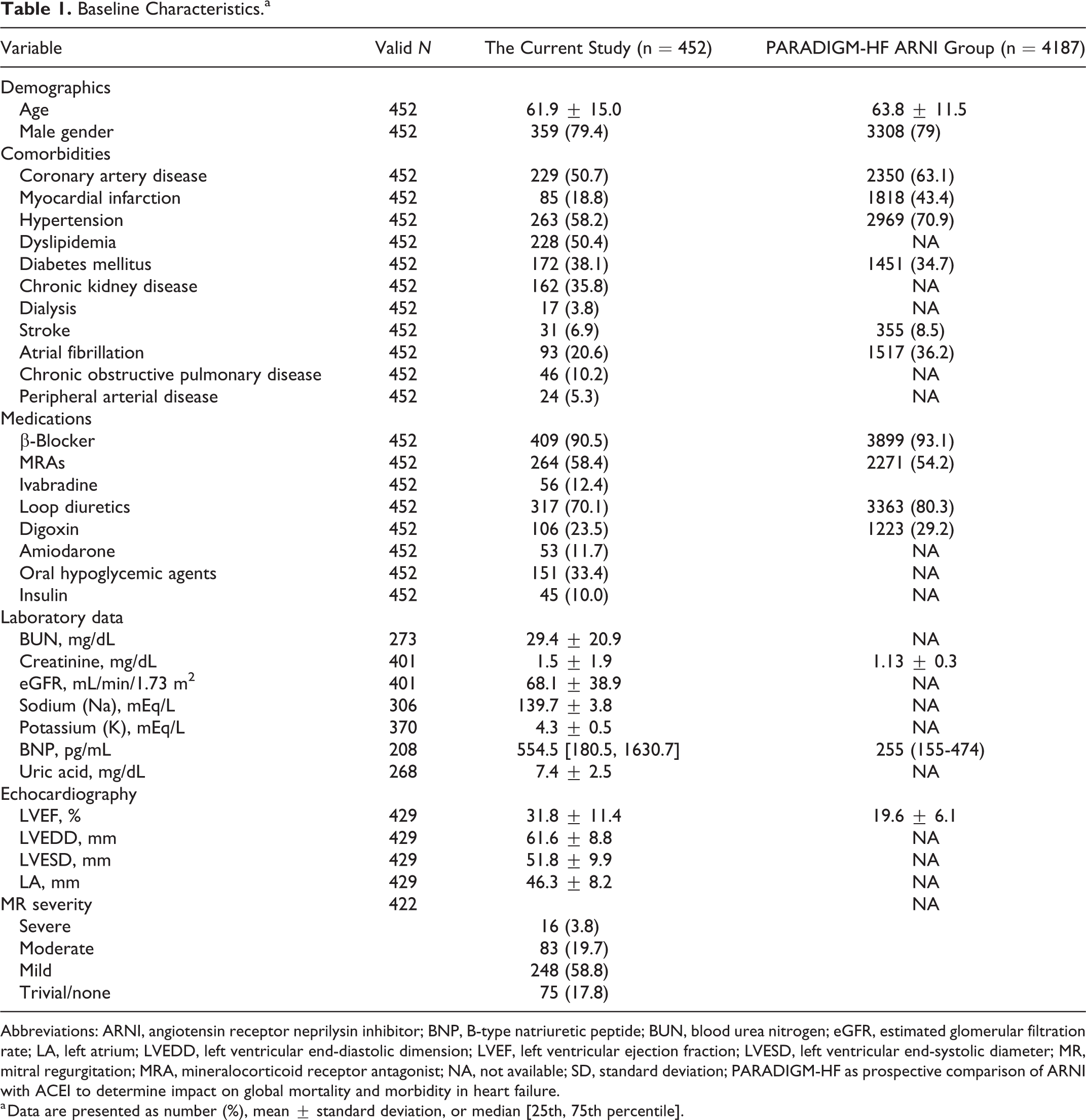
Abbreviations: ARNI, angiotensin receptor neprilysin inhibitor; BNP, B-type natriuretic peptide; BUN, blood urea nitrogen; eGFR, estimated glomerular filtration rate; LA, left atrium; LVEDD, left ventricular end-diastolic dimension; LVEF, left ventricular ejection fraction; LVESD, left ventricular end-systolic diameter; MR, mitral regurgitation; MRA, mineralocorticoid receptor antagonist; NA, not available; SD, standard deviation; PARADIGM-HF as prospective comparison of ARNI with ACEI to determine impact on global mortality and morbidity in heart failure.
a Data are presented as number (%), mean ± standard deviation, or median [25th, 75th percentile].
Mean dose of ARNI was 131.8 mg/d at baseline and 187.1 mg/d at 12th month. Figure 2 reported the percentage of different ARNI doses at start, 6th month, and 12th month. Sixty-five percent of the physicians used 100 mg/d as the starting dose. After 12 months, 15.8% of the patients received standard dose of 400 mg/d, 42.6% of them received 200 mg/d, and 41.6% of the population received less than half of the standard dose; 7.6% and 9.9% of the patients did not receive a prescription of ARNI at 6th month and 12th month, respectively.

Dosage for ARNI at baseline and follow-up. The valid sample size was 435, 402, and 392 at the baseline, 6 months, and 12 months, respectively. ARNI indicates angiotensin receptor-neprilysin inhibitor.
Table 2 shows the clinical outcomes at 1 year. The cumulative event rate for HF readmission, cardiovascular death, all-cause death, the composite of cardiovascular death or HF readmission, and the composite of all-cause death or HF readmission was 6.9%, 1.1%, 3.0%, 7.1%, and 8.5% in 12 months, respectively.
Clinical End Points During a 12-Month Follow-Up of the Current Study and a Median of 27-Month Follow-Up of PARADIGM-HF ARNI Group.a
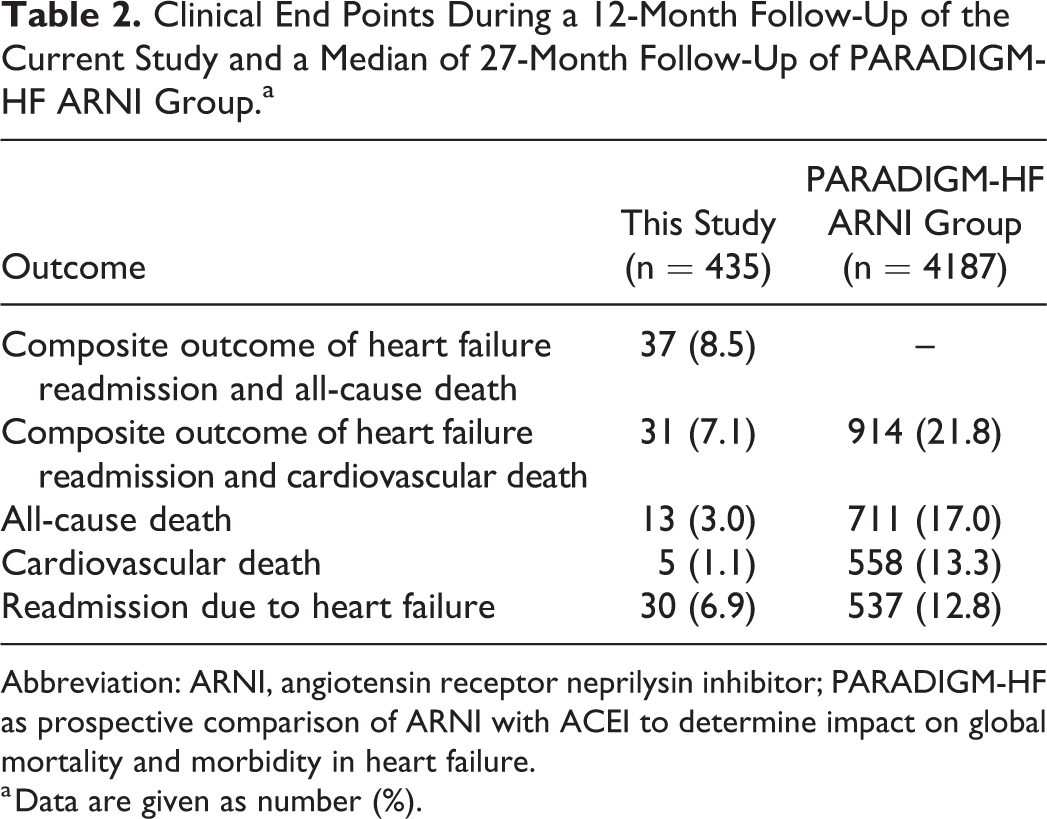
Abbreviation: ARNI, angiotensin receptor neprilysin inhibitor; PARADIGM-HF as prospective comparison of ARNI with ACEI to determine impact on global mortality and morbidity in heart failure.
a Data are given as number (%).
Table 3 shows the interval changes between baseline and 12 months after ARNI treatment. Significant improvement in LVEF and reduction in left ventricular size were observed after 1 year. The prevalence of moderate to severe MR did not change at 1 year. B-type natriuretic peptide significantly decreased and potassium level significantly increased. In addition, the level of uric acid significantly reduced. Blood urea nitrogen and creatinine did not change. Table 4 shows the BNP level at baseline and 12th month, according to an increase (>10%), unchange (−10% to 10%), or decrease (<−10%) at 1 year. Approximately 30% of the patients had increased, 44% had unchanged, and 26.3% had decreased levels.
Changes of Echocardiographic and Laboratory Data Between the Baseline and 12-Month Follow-Up.a

Abbreviations: BNP, B-type natriuretic peptide; BUN, blood urea nitrogen; LA, left atrium; LVEDD, left ventricular end-diastolic dimension; LVESD, left ventricular end systolic diameter; LVEF, left ventricular ejection fraction; MR, mitral regurgitation.
a Data are expressed as number (%), or mean ± standard deviation, or median [25th, 75th percentile].
BNP Change Between Baseline and 12 Months.a

Abbreviation: BNP, B-type natriuretic peptide.
a Data are expressed as median (interquartile range).
Supplemental Figure 1 demonstrates the lower eGFR group had higher risks of cumulative composite events. Supplemental Table 1 compares the baseline characteristics of patients with eligible follow-up echo data (n = 192) and those without (n = 243). The results revealed that patients with eligible follow-up echo data were more likely to be prescribed with ivabradine and have greater left ventricular end-systolic diameter value.
Discussion
In this retrospective, single-arm, observational study from tertiary medical centers in Northern Taiwan, we found that gender distribution, age, prevalence of major comorbidities, and LVEF were similar to PARADIGM-HF populations. The prescription rate of β-blockers and MRAs in the present study was also close to PARADIGM-HF trial, which was higher than other real-world HF registries. 5,7 Three differences in baseline characteristics were found. Both baseline BNP and serum creatinine level were higher in the present study than in the PARADIGM-HF (554.5 vs 255 pg/mL; 1.5 vs 1.1 mg/dL, respectively). Four weeks of stable condition with maximally tolerated dose of β-blocker and angiotensin converting enzyme inhibitor (ACEi) or angiotensin receptor blocker (ARB) was required before the prescription of ARNI in Taiwan. It was possible that the majority of baseline BNPs were checked in the setting of acute decompensation in the CGRD population (may be few weeks before the initiation of ARNI) because recheck biomarkers for stable patients in clinics are not a routine practice. In PARADIGM-HF, patients received a mean dose of 394 mg/d of ARNI at the last assessment, which means almost every participants could tolerate full dose. In the present study, the mean dose at 12th month was 187.1 mg/d, which was much lower than in the PARADIGM-HF. In addition, the dosage for ACEi, ARB, β-blocker, and MRA was all about half of the guideline-recommended dose in the baseline period. A post hoc analysis from PARADIGM-HF by Vardeny et al 8 reported that medication dose reduction identified patients who had more comorbidities and increased frailty, thus associated with higher risk of major cardiovascular event. In addition, the efficacy of ARNI relative to enalapril became insignificant if the dose was reduced to less than 50% of the maximal dose (hazard ratio: 0.79, 95% confidence interval: 0.58-1.07). With smaller body size, more prevalent CKD and less prevalent hypertension in Asian HF population may be the standard or effective dose of ARNI is lower than in Western population. Due to the lack of medication prescription protocol, physicians being reluctant to titrate up the dose without specific reasons was also a possible cause. However, because of single-arm retrospective study with multiple unadjusted confounding factors, this observation is just hypothesis generating and needs more evidence to prove.
The baseline mean creatinine level was 1.5 mg/dL with a mean eGFR of 68 mL/min/1.73 m2, and 35.8% of the patients had CKD. Renal function did not change significantly after 1 year of ARNI treatment. A post hoc analysis demonstrated that compared to enalapril, ARNI led to a slower rate of decrease in the eGFR, even in patients with CKD. 9 In addition, Packer et al reported that the addition of neprilysin inhibition attenuates the effect of diabetes to accelerate the deterioration of renal function that occurs in patients with chronic HF. 10 In summary, ARNI protects renal function for patients having HFrEF with CKD. Thus, CKD should not be an obstacle for the prescription of ARNI.
The level of BNP decreased modestly but significantly (777-655 pg/mL,
Serum uric acid level significantly decreased after ARNI therapy in our study. An analysis by Mogensen et al 12 showed that serum uric acid concentration was an independent predictor of worse outcomes after multivariable adjustment in patients with HFrEF. Compared with enalapril, sacubitril/valsartan reduced serum uric acid and improved outcomes irrespective of serum uric acid. LV reverse remodeling with improved LVEF and reduced LV size was observed after 1 year of HF treatment in our study. Several reports 13,14 also showed similar findings about the early benefit of ARNI on left ventricular function and geometry. In the previous study, 15 a double-blind randomized trial, ARNI reduced functional MR to a greater extent than did valsartan. In CGRD population, the etiology and severity of MR was not well characterized and quantified, so it was difficult to make any conclusion about the effect of ARNI on MR.
A post hoc analysis from PARADIGM-HF trial by Damman et al 9 reported that patients with CKD had higher risk of primary outcome (cardiovascular death or HF readmission) than patients without CKD (30% vs 21%). However, the relative risk reduction of ARNI, compared with enalapril, was similar in patients with CKD and without CKD. In addition, the absolute risk reduction was even greater in patients with CKD (3.7 vs 2.1 fewer patients per 100 patient-years). A cohort study by Smith et al 16 showed that CKD is common and an important independent predictor of death and hospitalization in adults with HF across the spectrum of left ventricular systolic function. Our study also suggested that lower eGFR may be associated with higher risks of death and HF readmission. In summary, ARNI is feasible and beneficial in patients with CKD. However, improving the outcomes for patients with both HFrEF and CKD is still an unmet need.
As this retrospective study was based on a claims database, there were inherent limitations. For example, coding errors and typos are not uncommon in real-world practice. Some important clinical information was not available in the database, including blood pressure, heart rate, and functional class. In addition, variable missing rate was noticed among certain important echocardiographic and laboratory data during follow-up. Because of retrospective design, it was impossible to investigate the reason for lower dosage than clinical trial.
Conclusion
Patients with HFrEF in CGRD had higher baseline values of BNP and serum creatinine and had received lower dose of ARNI than the PARADIGM-HF populations. Angiotensin receptor neprilysin inhibitor appeared to be safe as regards renal function and effective in real-world practice. LV reverse remodeling was observed after 1 year of HF treatment including ARNI.
Supplemental Material
Supplemetal_Figure_1 - Angiotensin Receptor Neprilysin Inhibitor for Patients With Heart Failure and Reduced Ejection Fraction: Real-World Experience From Taiwan
Supplemetal_Figure_1 for Angiotensin Receptor Neprilysin Inhibitor for Patients With Heart Failure and Reduced Ejection Fraction: Real-World Experience From Taiwan by Fu-Chih Hsiao, Chun-Li Wang, Po-Cheng Chang, Yu-Ying Lu, Chien-Ying Huang and Pao-Hsien Chu in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Supplemetal_Tables - Angiotensin Receptor Neprilysin Inhibitor for Patients With Heart Failure and Reduced Ejection Fraction: Real-World Experience From Taiwan
Supplemental Material, Supplemetal_Tables for Angiotensin Receptor Neprilysin Inhibitor for Patients With Heart Failure and Reduced Ejection Fraction: Real-World Experience From Taiwan by Fu-Chih Hsiao, Chun-Li Wang, Po-Cheng Chang, Yu-Ying Lu, Chien-Ying Huang and Pao-Hsien Chu in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Acknowledgments
The authors thank the support of the Chang Gung Medical Foundation and Yuka Chu’s critical reading. All authors have read the journal’s authorship agreement and reviewed and approved the manuscript.
Author Contributions
Fu-Chih Hsiao and Pao-Hsien Chu contributed to conception and design, acquisition, analysis, or interpretation, drafted the manuscript, gave the final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy; Chun-Li Wang, Po-Cheng Chang, Yu-Ying Lu, and Chien-Ting Huang contributed to acquisition, analysis, and interpretation, and agrees to be accountable for all aspects of work ensuring integrity and accuracy; Fu-Chih Hsiao and Chien-Ting Huang contributed to acquisition, analysis, and interpretation, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Fu-Chih Hsiao was supported by Chang Gung Memorial Hospital by the grant (CORPG5G0011); and Pao-Hsien Chu by the grant (CORPG3H0091).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
