Abstract
Aim:
Changes in QRS duration in patients with heart failure with reduced ejection fraction (HFrEF) after sacubitril/valsartan therapy is not fully understood. This study aimed to assess the association of duration of HFrEF diagnosis with electrocardiographic and echocardiographic outcomes between before and after sacubitril/valsartan.
Methods:
We included HFrEF patients who received naïve sacubitril/valsartan therapy for ≥3 months, between January 2016 and March 2018. All patients were divided into 2 groups based on their duration of HFrEF. Generalized linear models were analyzed the cardiac outcomes after sacubitril/valsartan therapy by HFrEF duration.
Results:
Among these, 42 patients were HFrEF duration of <1 year and 47 patients were ≥1 year. The mean difference of QRS duration was lesser in the <1-year group than in the ≥1-year group (−2.3 msec vs 6.3 msec;
Conclusion:
Prolonged QRS durations and unfavorable LVEF recoveries after sacubitril/valsartan therapy were observed in patients with HFrEF duration of ≥1 year. Earlier diagnosis of HFrEF and appropriate medication treatment may be beneficial in the improvement of QRS duration and LVEF recovery.
Keywords
Introduction
Guideline-directed medical therapy (GDMT) for heart failure with reduced ejection fraction (HFrEF) is recommended to reduce morbidity and mortality, regardless of the patient’s background, etiology, and pathogenesis. 1,2 A prolonged QRS duration appears common in patients with HFrEF and is an independent predictor of high mortality. 3 Reverse remodeling of left ventricular ejection fraction (LVEF) could be deteriorating if the QRS duration progresses. 4 Patients with a wide QRS complex, especially a left bundle branch block (LBBB), had a lower LVEF recovery after GDMT and needed to be treated sooner with cardiac resynchronization therapy (CRT). 4
Sacubitril/valsartan, which is an angiotensin receptor neprilysin inhibitor (ARNI), could be used to improve cardiac reverse remodeling by blocking natriuretic peptide (NP) degradation, decreasing cardiac fibroblast proliferation, and modulating cardiac electrophysiology. 5 The timing of sacubitril/valsartan treatment plays an essential role in the recovery of LVEF and echocardiographic change patterns. 6 However, whether the sacubitril/valsartan therapy for HFrEF patients had an influence on biomedical, cardiac function outcomes remains inconclusive.
However, few studies have focused on cardiac electrocardiography changes during sacubitril/valsartan treatment. 7 The evaluation of electrocardiogram and echocardiographic changes could be a useful way to identify the electrophysiological effects associated with sacubitril/valsartan therapy. Therefore, this study aimed to examine the relationship between duration of HFrEF diagnosis and electrocardiographic and echocardiographic outcomes before and after sacubitril/valsartan treatment.
Methods
Data Source and Study Patients
In this multicenter retrospective cohort study, we enrolled patients with a diagnosis of New York Heart Association functional class II-III and left ventricular ejection fraction ≤35%. We included patients who had sacubitril/valsartan therapy for ≥3 months between January 2016 and March 2018. The data for each patient were retrospectively reviewed from the electronic medical records of the patients who were followed up until March 31, 2018 or until their death. The treatment decisions regarding the GDMT for HFrEF was made by the attending physicians. Only those patients who have received sacubitril/valsartan therapy for at least 3 months were included in this study. The Institutional Review Board of Mackay Memorial Hospital approved the study protocol (no. 18MMHIS115e) and written informed consent was waived.
A total of 156 patients with HFrEF were enrolled during the study period. We excluded 28 patients who had incomplete information and received sacubitril/valsartan therapy for less than 3 months. Ten patients with permanent pacemaker, including single/dual chamber pacemaker, cardioverter defibrillator (ICD), cardiac resynchronization pacemaker (CRT-P) and cardiac resynchronization defibrillator pacemaker (CRT-D) were excluded. In addition, 29 patients with atrial fibrillation, beta blockers or interventricular conduction delay were also excluded. Our final sample included 89 eligible patients had received sacubitril/valsartan therapy for at least 3 months. There were early (HFrEF duration <1 year, n = 42) and late groups (≥1 year, n = 47) (Figure 1).

Flow diagram of patient selection.
Outcome Measures
The primary outcomes were the changes in electrocardiographic and echocardiographic parameters between before and after at least 3 months of sacubitril/valsartan therapy, including electrocardiogram (ECG) PR interval, QRS duration, corrected QT (QTc) intervals, left ventricular ejection fraction (LVEF), and left atrial diameter, and left ventricular end diastolic diameter (LVEDD). All patients underwent a standard 12-lead ECG before and after sacubitril/valsartan therapy were measured, which was conducted using the GE Marquette MAC 3500 machine (GE Marquette Inc., Milwaukee, WI, USA). In addition, all ECG measurements in this study were performed by 2 independent cardiologists blinded to the patient information.
Definition of the Study Groups and Baseline Measurement
The main independent variable of interest was the duration of HFrEF diagnosis before sacubitril/valsartan therapy. The date of initial HFrEF diagnosis was collected by reviewing the electronic medical records of each patient, and the HFrEF duration before sacubitril/valsartan initiation was calculated in reference to initial HF diagnosis. Given that a greater LVEF is achieved with earlier GDMT initiation, 8 patients were classified into the early (HFrEF duration of <1 year) and late (HFrEF duration of ≥1 year) groups.
The following covariates included age, sex, body mass index (BMI), heart rate, alcohol drinking, ischemic cardiomyopathy, diabetes mellitus (DM), hypertension, myocardial infarction, atrial fibrillation, moderate to severe aortic regurgitation (AR) and mitral regurgitation (MR), heart rate, blood urea nitrogen (BUN), creatinine, N-terminal brain natriuretic peptide (NT-proBNP), and duration of sacubitril/valsartan exposure. Treatment duration was calculated from the start date of the sacubitril/valsartan therapy to its end date or until March 31, 2018.
Statistical Analysis
All data analyses were performed using IBM SPSS 22.0 for windows (IBM Corp., Chicago, IL, USA). Distributions of baseline patient demographics, medical conditions, and electrocardiographic parameters between the early and late groups before initiating sacubitril/valsartan were examined using the Fisher’s exact test for categorical variables, and the Student’s
Results
Baseline demographics and characteristics of the patients are shown in Table 1. The majority of the 58 patients were male (65.2%), with a mean age of 66.8 years. Baseline demographics, medical conditions, and echocardiographic parameters were similar between the 2 groups, except for a lower LVEF level (30.5% vs 33.8%,
Patient Demographics and Clinical Characteristics.

Abbreviations: AR, aortic regurgitation; ACEi, angiotensin-converting enzyme inhibitors; ARB, angiotensin receptor blocker; BMI, body mass index; DM, diabetes mellitus; eGFR, estimated glomerular filtration rate; HFrEF, heart failure with reduced ejection fraction; HTN, hypertension; LVEDD, left ventricular end diastolic diameter; LVEF, left ventricular ejection fraction; MI, myocardial infarction; MR, mitral regurgitation; MRA, mineralocorticoid receptor antagonist; NOAC, novel oral anticoagulant; NT-proBNP, N-terminal brain natriuretic peptide; SAC/VAL, sacubitril/valsartan; SD, standard deviation.
The outcomes related to the electrocardiographic and echocardiographic parameters after the sacubitril/valsartan therapy in the 2 groups are presented in Table 2. In early group, the baseline and follow-up QRS durations were 106.2 msec and 103.9 msec respectively, with a mean change of −2.3 msec. In late group, the baseline and follow-up QRS duration was 121.9 msec and 128.2 msec respectively, with a mean change of 6.3 msec. Late group patients had a significantly increased QRS duration (differential change of 8.6 msec, 95% CI: 0.9-16.3,
Electrocardiographic and Echocardiographic Outcomes Before and After of Sacubitril/Valsartan Therapy Between Patients With Early and Late Duration of HFrEF Diagnosis.

Abbreviations: CI, confidence index; ECG, electrocardiogram; HFrEF, heart failure with reduced ejection fraction; LVEDD, left ventricular end diastolic diameter; LVEF, left ventricular ejection fraction.
a 31 patients in early group and 34 patients in late group.
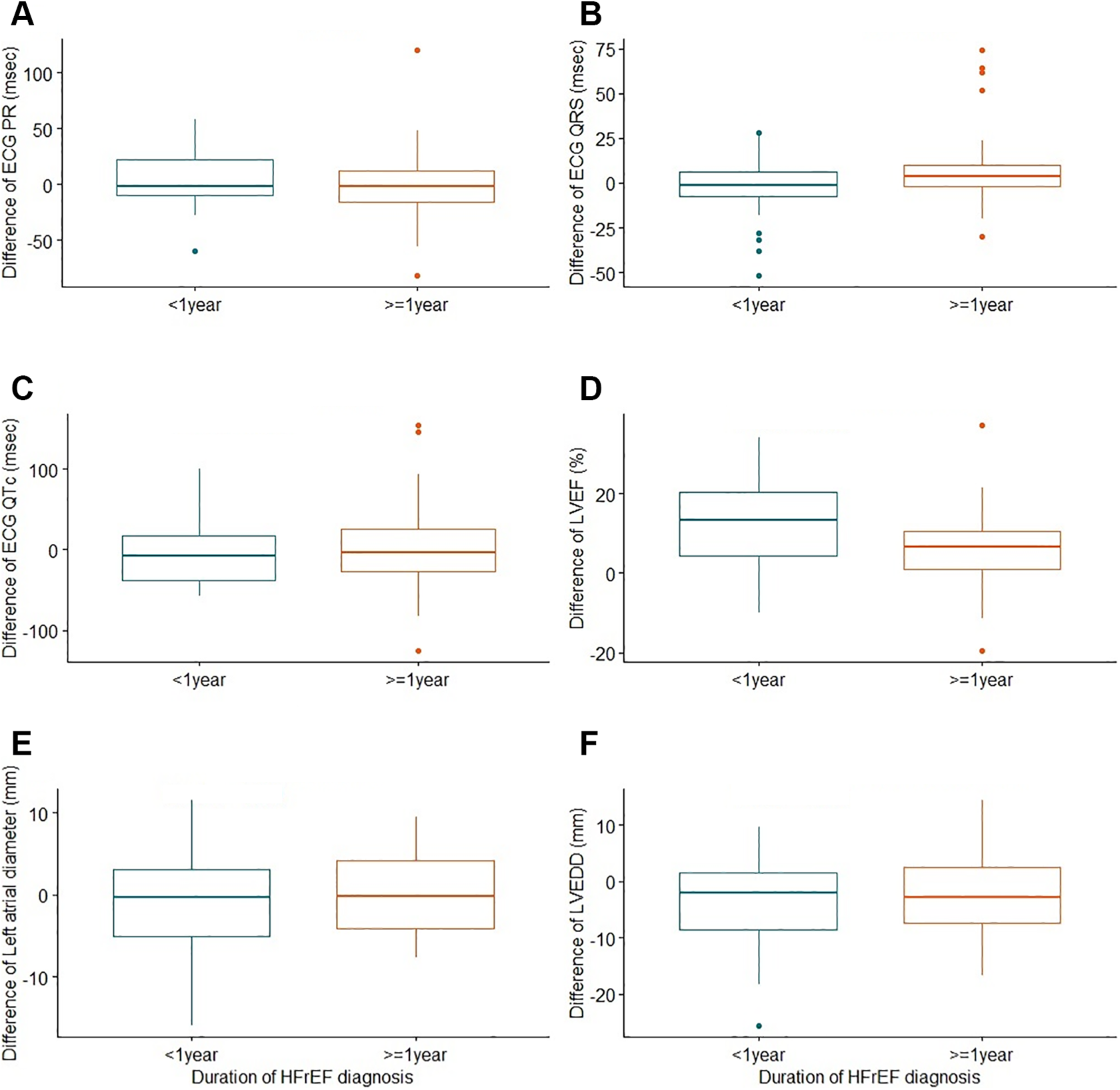
Differences of electrocardiographic and echocardiographic outcomes between before and after at least 3 months of sacubitril/valsartan therapy by duration of HFrEF diagnosis: (A) ECG PR interval (
In univariable analyses, late group patients had significantly more prolonged QRS durations after the sacubitril/valsartan therapy (coefficient = 8.6; 95% CI, 1.2-16,
Univariable and Multivariable Analyses of Factors Associated With Cardiac Function Outcomes After At Least 3 Months of Sacubitril/Valsartan Therapy for Late Group Patients (HFrEF Duration ≥1 Year).

Abbreviations: BUN, blood urea nitrogen; CI, confidence interval; ECG, electrocardiogram; HFrEF, heart failure with reduced ejection fraction; LVEDD, left ventricular end diastolic diameter; LVEF, left ventricular ejection fraction; NT-proBNP, N-terminal brain natriuretic peptide.
a The model was adjusted for patient’s age, gender, BMI, alcohol drinking, ischemic cardiomyopathy, DM, hypertension, myocardial infarction, atrial fibrillation, moderate to severe AR, moderate to severe MR, NT-proBNP, BUN, eGFR, total daily dosage and duration of SAC/VAL exposure, and current medications as compared with early group patients (HFrEF duration <1 year).
Discussion
The main findings of the present study indicated that QRS durations were significantly prolonged after sacubitril/valsartan therapy in late group patients (HFrEF durations ≥1 year) compared with early group patients (HFrEF durations <1 year), even after adjusting for clinical variables. However, the better reverse remodeling in LVEF after sacubitril/valsartan therapy was significantly noted in patients with HFrEF durations of <1 year after the adjustments.
Sacubitril/valsartan, a composite ARNI, represents a novel pharmacological strategy for managing HFrEF patients. Adjusted cardiovascular outcomes were similar across different etiologic categories including idiopathic, ischemic, or hypertensive causes when using sacubitril/valsartan over enalapril. 9 In this study, there were no significant differences in underlying diseases and clinical conditions before the sacubitril/valsartan treatment between the 2 groups. Serial echocardiographic follow-ups showed significant LVEF enhancement and LVEDD shortening after sacubitril/valsartan, especially in the early group patients. Cardiac reverse remodeling, manifested by LVEF increases ≥10% or LVEDD decreases ≤55 mm, is associated with an improved prognosis of HF. 10,11 Cardiac reverse remodeling responses after sacubitril/valsartan therapy could be due to smaller baseline LVEDD, higher initial doses, or longer sacubitril/valsartan treatment durations. 10 In addition, we found the converse relationship between HFrEF duration and cardiac reverse remodeling responses. Early group patients had more significant LVEF increases (mean +13.7%) and LVEDD decreases (from 59.8 to 56.0 mm, SD: −3.8 mm) compared with late group patients.
LV remodeling is a major mechanism of underlying disease progression in HFrEF patients. 12 Angiotensin converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) have anti-fibrotic effects via the RAAS blockade. Neprilysin also inhibits cardiac fibroblast proliferation. This is significant as cardiac fibroblasts can impact molecular and cellular events that collectively determine cardiac structure and electric activity. Dual RAAS and neprilysin inhibition that attenuates cardiac reverse remodeling may then be contributed to by the superior effects of cardiac fibrosis and hypertrophy. 11,13 The PIONEER-HF trial demonstrated that sacubitril/valsartan could reduce myocardial injury and hemodynamic stress, as reflected by decreases seen in NT-proBNP after stabilization of acute decompensated HF. 14 A meta-analysis combing 20 trials investigating cardiac reverse remodeling reported sacubitril/valsartan could improve LV remodeling as early as at least 3 months compared with ACEI/ARB in HFrEF patients. 15 Therefore, sacubitril/valsartan could be used in HFrEF patients to limit LV remodeling while precluding non-responses or intolerance.
A progressive increase in QRS duration altered LVEF recovery and deteriorated patient prognoses. 3 In patients with HFrEF, those with wide QRS durations had lower LVEF recovery after GDMT compared to those with a narrow QRS duration. 4 CRT could correct QRS prolongations and should be initiated earlier in patients with LVEF ≤35% and prolonged QRS durations following optimal GDMT. 2 In the post hoc analysis of the PARADIGM-HF trial, sacubitril/valsartan reduced the incidence of cardiovascular death or HF hospitalization despite different background diseases, but information relating to cardiac electrophysiology during the sacubitril/valsartan treatment was not included. 16,17 The effect of sacubitril/valsartan on ECG QRS durations and on QTc intervals was uncertain, 7 and little research has focused on the association between cardiac electrocardiographic changes and clinical outcomes during sacubitril/valsartan treatment. Okutucu et al 1 demonstrated that switching from ramipril to sacubitril/valsartan could alter the myocardial electrophysiological properties in HFrEF. In another prospective study, anti-arrhythmia effects seen as shortened QRS complexes and QTc intervals were notable after sacubitril/valsartan therapy. 18 Shorter QRS durations, which reflect favorable cardiac reverse remodeling processes and LV synchrony, may be associated with responses to ARNIs in patients with HFrEF, compared with non-QRS shortening. 19 Besides, greater LVEF is achieved with earlier initiation of GDMT in patients with HF duration <1 year, compared with those with ≥1 year. 8 In our study, we also found different alterations in the electrophysiological parameters after sacubitril/valsartan therapy. Reductions in QRS and QTc durations were estimated only in patients with HFrEF durations of <1 year before sacubitril/valsartan treatment. Delayed administration of sacubitril/valsartan therapy in patients with longer HFrEF diagnosis durations may lead to prolonged QRS durations and worse LVEF reverse remodeling. Our results imply that (i) ECG QRS changes were favorable after switching to sacubitril/valsartan therapy in patients with HFrEF durations <1 year; (ii) more cardiac reverse remodeling was noted in patients with shorter HFrEF durations.
In the present study, the timing of initiating the sacubitril/valsartan therapy could have influenced the electrocardiographic and echocardiographic parameters; however, the possible mechanism remains uncertain. Sacubitril/valsartan could improve cardiac remodeling by blocking natriuretic peptide (NP) degradation, thereby decreasing cardiac fibroblast proliferation and modulating cardiac electrophysiology. 5 After inhibiting neprilysin, high circulating NP levels exert multiple physiologic effects, thereby enhancing diuresis and natriuresis, and by modifying ventricular arrhythmia substrates by reducing myocardial fibrosis. 1,12,20 In advanced cardiovascular disease, hyperactivity of cardiac fibroblasts resulted in myocardial fibrosis, with adverse effects on cardiac structure and electrical activities, and then finally contributed to the development of HF. 5,21 Myocardial fibrosis is a complex phenomenon underlying the development of HF, resulting from the aberrant activation of various cell types and signaling pathways. 21 Sacubitril/valsartan might reduce profibrotic signaling, which may contribute to a greater frequency of reverse remodeling compared to enalapril. 22 Furthermore, sacubitril/valsartan should be evaluated as a direct antifibrotic therapeutic for HF with preserved ejection fraction. 23 This was the first study to evaluate the relationship between the timing of prescribing sacubitril/valsartan in patients with different HFrEF durations and changes in electrophysiological properties. Further research may investigate the remodeling response following sacubitril/valsartan treatment over time.
Several limitations of this study should be noted. Although we have controlled for numerous covariates, other unmeasured factors may influence the results. In addition, this study was not prospectively powered and the sample sizes used may not have been large enough to show a difference between the 2 groups. Moreover, we cannot treat the intervention as an independent variable, since there was no control group and no levels of intervention. We have added this content to the limitations and future research directions section to describe more sophisticated analyses that could be used with alternative research designs. According to Taiwan’s National Health Insurance reimbursement, sacubitril/valsartan could be used in patients with HFrEF who received ACEIs/ARBs at least 1 month. Therefore, clinical cardiologist favored sacubitril/valsartan rather than ACEIs/ARBs in the long-term treatment of HFrEF. However, since the setting of CRT could influence the duration of QRS, we excluded patients who were indicated for CRT implantation. Future studies could focus on the consequences of the differences in patients with HFrEF who require CRT.
Conclusion
Our study provides evidence that late group patients (HFrEF durations ≥1 year) had prolonged QRS durations and reduced degrees of LVEF improvement after sacubitril/valsartan therapy compared with early group patients (<1 year). However, the impaired response of the late HFrEF group to sacubitril/valsartan treatment in terms of improvement in electrographic and echocardiographic outcomes may reflect progression of the underlying disease. Our findings suggest that sacubitril/valsartan therapy may improve LV systolic remodeling and prevent QRS prolongation, especially in patients with shorter durations of HFrEF. Future researches should include a control group design for the evaluation of treatment response in patients with HFrEF.
Footnotes
Acknowledgments
The authors are very grateful to all of those associated with the study for their important contribution in gathering, managing, and processing the data.
Author Contributions
PLL: Conceptualization, data curation, resources, writing—original draft, supervision; YHL: Writing—review and editing; LYML: Writing—review and editing; CTT: Writing—review and editing; TFY: Writing—review and editing; WRC: Writing—review and editing; MYH: Writing—review and editing; HYC: Writing—review and editing; CCH: Conceptualization, formal analysis, writing—review and editing, supervision.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
