Abstract
Aims:
In recent large trials, sacubitril/valsartan demonstrated favorable effects in patients with HF. However, many patients do not achieve the target dose of treatment. This study investigated the factors linked to up-titration of sacubitril/valsartan in patients with heart failure and preserved ejection fraction (HFpEF).
Methods:
Using a multicenter retrospective database, 204 consecutive patients with HFpEF (left ventricular ejection fraction ≥ 40%) who were treated with sacubitril/valsartan between October 2020 and March 2022 were analyzed. Up-titration was defined as an increase in dosage above 24/26 mg BID beyond 12 weeks after the initiation of sacubitril/valsartan.
Results:
Among the patients, 55% underwent up-titration, and 8% discontinued the drug. The baseline systolic blood pressure (SBP) was higher in patients with up-titration than in those with no up-titration; SBP values similar to that at baseline were observed between the 2 groups at 2 to 4 weeks and at 12 weeks after the commencement of sacubitril/valsartan treatment. The majority of those who discontinued sacubitril/valsartan did so because of hypotension. The multivariable logistic regression model showed that a history of hypertension, history of atrial fibrillation, baseline SBP, and baseline estimated glomerular filtration rate <60 mL/min/1.73 m2 were associated with sacubitril/valsartan up-titration.
Conclusion:
Approximately half of all patients did not undergo up-titration, and 8% of those with HFpEF discontinued the sacubitril/valsartan therapy. For aggressive up-titration and continuation of sacubitril/valsartan, patients with lower baseline SBP, renal dysfunction, absence of a history of hypertension, and presence of atrial fibrillation may require more careful monitoring.
Keywords
Introduction
Despite remarkable progress in drug therapies, patients with HF still account for high mortality and HF rehospitalizations worldwide. 1 Sacubitril/valsartan has emerged as a new therapeutic agent to improve the long-term prognosis of HF patients. In the PARADIGM-HF trial, sacubitril/valsartan showed lower cardiovascular death and HF rehospitalization rates than enalapril in HF with reduced ejection fraction (HFrEF). 2 PARAGON-HF trial investigated the efficacy of sacubitril/valsartan in patients with heart failure and preserved ejection fraction (HFpEF); no significantly prognostic benefit of sacubitril/valsartan was found when compared to ARBs. However, a subanalysis found that sacubitril/valsartan had a prognostic benefit in women and patients with left ventricular ejection fraction (LVEF) <57%. 3 In addition, a recent meta-analysis found that sacubitril/valsartan was associated with a significant reduction in the risk of HF hospitalization in patients with HFpEF when compared to placebo. 4 Therefore, the AHA/ACC/HFSA Guideline will be updated in 2022 to recommend sacubitril/valsartan treatment for patients with HFpEF as a class IIb indication. 5
Sacubitril/valsartan is started at a low dose and up-titrated to the target dose with consideration of safety and tolerability. However, randomized controlled trials have demonstrated that many patients have difficulty with up-titrating to the target dose owing to the associated side effects and intolerance. 6,7 Similarly, according to real-world data from a retrospective analysis, up-titration was not attempted in approximately 60% of the patients 6 months after the first sacubitril/valsartan prescription in patients with HFrEF. 8 Therefore, achieving the up-titration of sacubitril/valsartan and identifying predictors of up-titration is essential. The PARAGON-HF implemented dose escalation to the target dose before randomization; hence, there is no evidence for factors related to up-titration in patients with HFpEF. This study investigated the rate of up-titration, change in systolic blood pressure (SBP), and underlying factors associated with up-titration of sacubitril/valsartan in patients with HFpEF using a multicenter retrospective database.
Methods and Materials
Study Population
This multicenter cohort study was conducted at the Kindai University Hospital, Tokai University Hospital, Arida City Hospital, Hirakata Kohsai Hospital, and Yao City Hospital. A total of 231 consecutive patients with HFpEF who were initiated on sacubitril/valsartan during HF hospitalization or outpatient visits between October 2020 and March 2022 were retrospectively enrolled. The study protocol was approved by the ethics committee of Kindai University (R03-150) and was registered at the University Hospital Medical Information Network (UMIN) Clinical Trial Registry (unique identifier: UMIN000044351). Informed consent was obtained from all the recruited patients. The study conformed to the principles outlined in the Declaration of Helsinki.
Study Protocol and Treatment
This multicenter, retrospective study included patients hospitalized with acute decompensated HF and outpatients with chronic HF who were being treated according to the current guidelines. 5 They were directed or switched from ACEIs or ARBs to sacubitril/valsartan, which was selected at the physician’s discretion. Patients who previously received ACEI were initiated on sacubitril/valsartan more than 36 hours after withdrawal. Patients on ARBs were directly switched to sacubitril/valsartan. All patients were started on a dose of 24/26 mg BID sacubitril/valsartan, with up-titration considered by cardiologists every 2 to -4 weeks based on patient tolerability. Patients were observed 12 weeks after the initiation of sacubitril/valsartan; up-titration was defined as a dose of sacubitril/valsartan above 24/26 mg BID. Patients who were administered sacubitril/valsartan during the intravenous diuretic period (n = 15) or missed follow-up within the 12 weeks observational period (n = 9), or hospitalized due to worsening HF during the 12 weeks study period (n = 3) were excluded from this study.
Data Collection
The prescribed doses of sacubitril/valsartan after a 12-week observation period were extracted. When patients discontinued treatment within 12 weeks of sacubitril/valsartan initiation, reasons for the discontinuation were extracted from medical records. Additionally, patient characteristics and medical history were obtained from the medical records. The use of calcium channel blockers (CCBs) was defined as the administration of dihydropyridine CCBs. SBP levels were obtained from medical records at the time of sacubitril/valsartan initiation and 2, 4, and 12 weeks after initiation. Data on body mass index, laboratory parameters, and medications were collected at the time of sacubitril/valsartan initiation. The eGFR was calculated using the following formula: eGFR (mL/min/1.73 m2) = 194 × serum creatinine1.094 × age−0.287 (if female; × 0.739). Transthoracic echocardiography was performed by expert sonographers before sacubitril/valsartan initiation, and LVEF was calculated using the modified Simpson method. 9 LVEF ≥ 40% was defined as HFpEF.
Statistical Analysis
Continuous variables are presented as medians with interquartile ranges (IQR). Categorical variables are presented as numbers and percentages (%). The equality of median between the 2 groups was determined using the Mann-Whitney U test for continuous variables and the chi-squared test for categorical variables. Univariate and multivariable logistic regression analyses were conducted. Covariates with P < .10 in univariable analysis were entered into the subsequent multivariable analysis. The JMP 14.2.0 software (SAS Institute Inc., Cary, NC, USA) was used for all statistical analyses. Statistical significance was set at P < .05.
Results
Baseline Patients’ Characteristics
The dose distribution of sacubitril/valsartan after a 12 weeks observation period is as follows: discontinuation, 8.3% (17 patients); 24/26 mg BID, 36.8% (75 patients); 49/51 mg BID, 38.2% (78 patients); and 97/103 mg BID, 16.7% (34 patients) (Figure 1). Of all patients, 54.9% (112/204) were uptitrated with sacubitril/valsartan. Patients without up-titration had a lower body mass index, a lower rate of hypertension, and a higher rate of atrial fibrillation than those with up-titration (Table 1). HF severity, history of cardiovascular disease, and initiation of sacubitril/valsartan therapy during hospitalization were comparable between the 2 groups (patients without up-titration vs patients with up-titration).

Dose distribution after 12 weeks of sacubitril/valsartan initiation.
Patient Characteristics.a

Abbreviations: HF, heart failure; NYHA, New York Heart Association.
a Data are presented as median (25th-75th percentiles) or number (%).
The baseline laboratory parameters were similar between the 2 groups (Table 2). Patients with up-titration had a higher rate of prescribed ARBs and CCBs, and a lower rate of loop diuretic administration than those without up-titration. Dose reduction of CCBs from baseline to 12 weeks was observed in 12% (11 of 92) of those without up-titration and 16% (18 of 112) of those with up-titration (P = .40).
Parameters and Medications at the Timing of Sacubitril/Valsartan Initiation.a

Abbreviations: ACEi, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; BNP, B-type natriuretic peptide; CCB, calcium channel blocker; eGFR, estimated glomerular filtration rate; LVEF, left ventricular ejection fraction; MRA, mineralocorticoid receptor antagonist; SGLT2i, sodium-glucose cotransporter 2 inhibitor.
a Data are presented as median (25th-75th percentiles) or number (%).
Systolic Blood Pressure
Patients without up-titration had a significantly lower baseline SBP than those with up-titration [124 (110-139) mmHg vs 137 (122-150) mmHg; P < .0001; Figure 2]. SBP at 2 to 4 weeks [118 (101-128) mmHg vs 128 (115-140) mmHg, P < .0001] and 12 weeks [118 (100-130) mmHg vs 127 (118-144) mmHg, P < .0001] after initiation were significantly lower in patients without up-titration than in those with up-titration. In contrast, the change in SBP between baseline and after 2 to 4 weeks of initiation did not differ between the 2 groups [patients without up-titration: −9.0 (−23.5 to 0) vs patients with up-titration −6.0 (−17.5 to 1); P = .28]. In patients who were prescribed CCBs before the initiation of sacubitril/valsartan, the number of patients experiencing CCB dose reduction during the 12 weeks period tended to be higher in patients without up-titration (56% [9/16]) than in those with up-titration (33% [13/40]), but this difference was not statistically significant (P = .10).
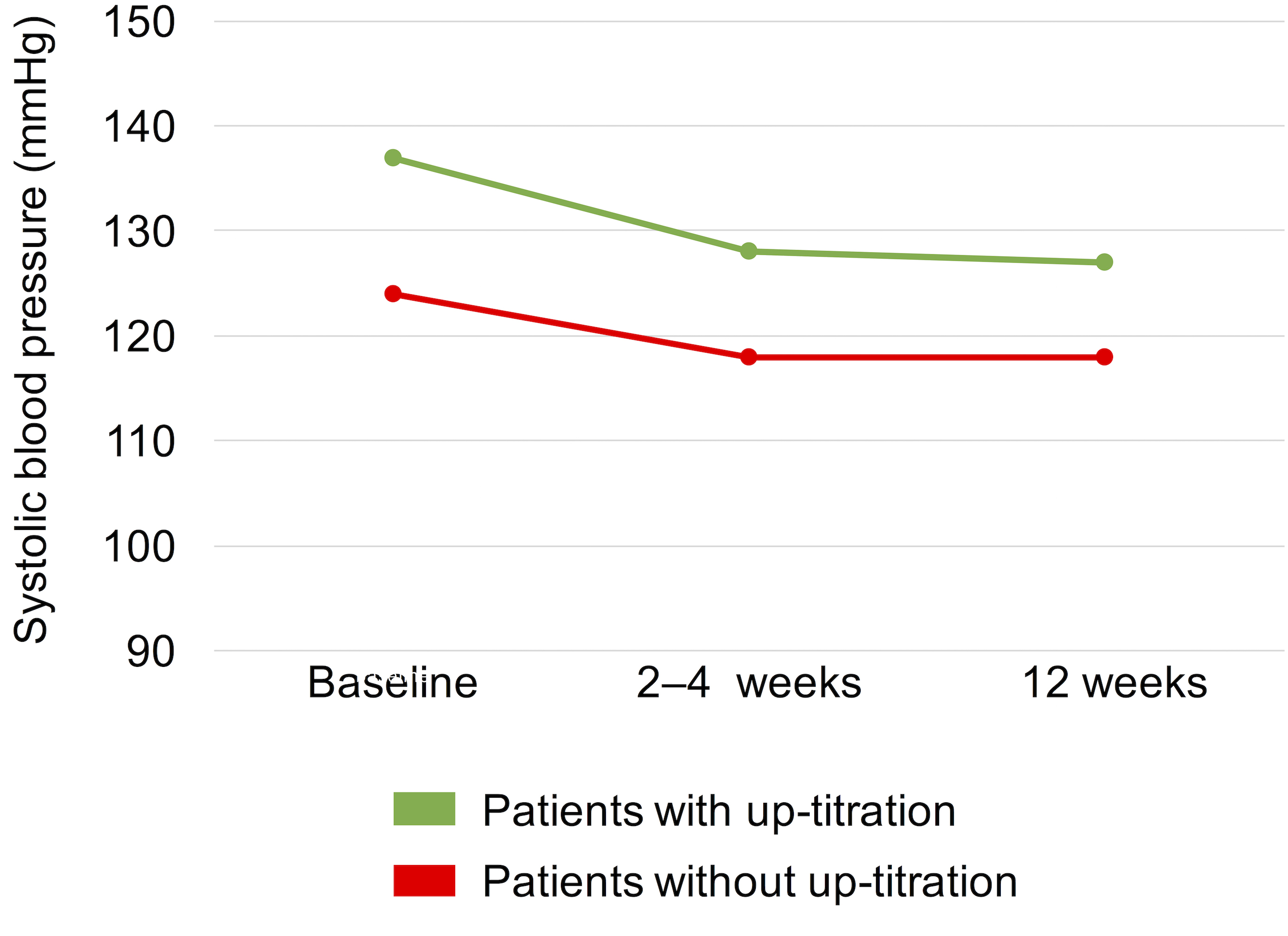
Changes in systolic blood pressure at baseline, 2 to 4 weeks, and 12 weeks after the initiation of sacubitril/valsartan treatment between patients with and without up-titration.
Factors Related to Up-Titration
Univariate logistic regression analyses indicated that age ≥65 years, male sex, body mass index, history of hypertension, history of atrial fibrillation, baseline SBP, baseline eGFR <60 mL/min/1.73 m2, conversion from ARB, use of mineralocorticoid receptor antagonist, use of CCBs, and use of loop diuretics were P < .01 (Table 3). These covariates were entered into multivariable logistic regression analyses, which revealed that a history of hypertension, history of atrial fibrillation, baseline SBP, and baseline eGFR <60 mL/min/1.73 m2 were associated with the up-titration of sacubitril/valsartan. Conversely, age, baseline serum potassium level, and conversion from ARB were not associated with up-titration.
Logistic Regression Model for Up-Titration of Sacubitril/Valsartan.

Abbreviations: ARB, angiotensin receptor blocker; CCB, calcium channel blocker; CI, confidence interval; eGFR, estimated glomerular filtration rate; HF, heart failure; MRA, mineralocorticoid receptor antagonist; NYHA, New York Heart Association; OR, odds ratio; SGLT2i, sodium-glucose cotransporter 2 inhibitor.
Discontinuation
During the 12 weeks after initiation of treatment, 17 patients discontinued sacubitril/valsartan administration (Figure 1). Of the 17 patients, 13 discontinued treatment because of symptomatic hypotension, 3 had an interruption, and 1 had hyperkalemia. None of the patients developed angioedema. The median time from initiation to discontinuation was 34 (12-72) days. The baseline SBP, SBP at 2-4 weeks after initiation, and SBP at 12 weeks after initiation in patients who discontinued treatment were 120 (105-135) mmHg, 118 (99-130) mmHg, and 120 (104-132) mmHg, respectively. Female patients and patients with previous cardiac surgery had a higher rate of discontinuation of treatment than men and patients without previous cardiac surgery (Supplementary Table 1). Age, history of hypertension, serum potassium level, eGFR, and prescribed ARBs or calcium channel blockers were comparable between the patients who discontinued treatment and those who continued. The univariate logistic regression analysis revealed that previous cardiac surgery and baseline SBP were related to sacubitril/valsartan discontinuation (Supplementary Table 2).
Discussion
In real-world data from a multicenter, retrospective study, 55% of patients with HFpEF were up-titrated during the 12-week observation period, and 8% of those with HFpEF discontinued. Patients who did not up-titrate had a significantly lower SBP at baseline, and SBP intergroup differences similar to that at baseline were observed at 2 to 4 weeks as well as at 12 weeks after the commencement of sacubitril/valsartan treatment. Of those who discontinued sacubitril/valsartan, most were due to hypotension. The multivariable analysis showed that a lower baseline SBP, renal dysfunction, absence of a history of hypertension, and presence of atrial fibrillation were negative determinants of up-titration. Previous large trials included preselected patients to tolerate the maximum dose being tested; hence, titration and tolerability of sacubitril/valsartan to the maximum dose are not well understood. Thus, our findings emphasize the significance of appropriate sacubitril/valsartan dose titrations in patients with HFpEF.
Failure to achieve a higher target dose has been reported in real-world data, with a retrospective analysis of 12 082 patients with HFrEF from the German database showing that out of 7760 patients started on sacubitril/valsartan at 24/26 mg BID, 41% underwent up-titration for 18 weeks from first sacubitril/valsartan prescription. 8 In our study, 55% of patients with HFpEF received up-titration indicating a relatively higher up-titration rate when compared to real-world data in the HFrEF setting. One factor to consider is that patients with HFpEF have higher baseline SBP than those with HFrEF. When comparing PARADIGM-HF and PARAGON-HF, patients with HFpEF had a baseline SBP 8-9 mmHg higher than those with HFrEF. 2,3 Kim et al investigated the dose–response relationship of renin-angiotensin system blockers, including ACEIs and ARBs, in patients with acute decompensated HF. They concluded that patients who received high doses of renin-angiotensin system blockers had a higher EF and approximately 10 mmHg higher SBP than those who received low doses. 10 Predictors of successful up-titration in our study, such as blood pressure, renal function, and atrial fibrillation, have also been noted as predictors of successful up-titration in patients with HFrFE. 6 In contrast, age, severity of HF, initiation during hospitalization, and serum potassium level were not associated with up-titration. The predictors of up-titration in patients with HFpEF require further investigation.
During the run-in period in PARAGON-HF trial, 16% of patients discontinued sacubitril/valsartan. 11 In their study, the reasons for discontinuation were hypotension, renal impairment, and hyperkalemia. The mean baseline SBP was 135 mmHg in patients who discontinued sacubitril/valsartan, which was relatively high; nevertheless, most patients discontinued because of hypotension. Although the reason for the large difference in discontinuation rates between PARAGON-HF during the run-in period and our results is unclear, a starting dose of 49/51 mg BID during the run-in period in PARAGON-HF may have caused a higher discontinuation rate. Sacubitril/valsartan prescriptions in the real world can be continued more easily than the discontinuation rates expected from randomized controlled clinical trials.
Strength and Limitations
In our study, we investigated the rates of sacubitril/valsartan up-titration and discontinuation, the causes of discontinuation, changes in SBP after initiation, and predictors of up-titration in patients with HFpEF. The results of this study may be useful in clinical practice when considering the initiation of sacubitril/valsartan therapy and during up-titration. Our study had 3 limitations. First, although this study used real-world data from a multicenter setting, the sample size was relatively small. However, to the best of our knowledge, this is the first report that compares real-world data and up-titration of sacubitril/valsartan in patients with HFpEF, suggesting the need for further large-scale studies. Second, sacubitril/valsartan up-titration was performed at the discretion of the attending physicians, and selection bias may exist. A large retrospective study suggested that patients prescribed sacubitril/valsartan by cardiologists have reported higher rates of up-titration within the initial 6 months of treatment than those prescribed by general physicians or physicians of other specialties. 8 The descriptions and up-titration of sacubitril/valsartan were managed by cardiologists in this study; therefore, we believe that the influence of selection was relatively small due to only cardiologist being involved in the treatment. Moreover, all patients were followed-up every 2 to 4 weeks, suggesting equal opportunity for medication changes among them. Third, this was a retrospective observational study, and complete follow-up data for BNP or NT-proBNP were lacking. A confirmation of the association between sacubitril/valsartan up-titration and these biological biomarkers in patients with HFpEF would have increased the clinical significance of the study and should be considered for future investigation.
Conclusion
In this multicenter retrospective database of patients with HFpEF, approximately half did not undergo up-titration, and 8% of the patients discontinued sacubitril/valsartan. More careful monitoring may be needed in patients with lower baseline SBP, renal dysfunction, absence of a history of hypertension, and presence of atrial fibrillation for aggressive up-titration and continuation of sacubitril/valsartan. Further studies on factors related to the up-titration of sacubitril/valsartan in patients with HFpEF are needed.
Supplemental Material
Supplemental Material, sj-docx-1-cpt-10.1177_10742484221146375 - Up-Titration of Sacubitril/Valsartan Among Patients With Heart Failure and Preserved Ejection Fraction
Supplemental Material, sj-docx-1-cpt-10.1177_10742484221146375 for Up-Titration of Sacubitril/Valsartan Among Patients With Heart Failure and Preserved Ejection Fraction by Koichiro Matsumura, Takeshi Ijichi, Junko Morimoto, Kensuke Takabayashi, Mitsunori Miho, Keisuke Ueno, Eijiro Yagi, Toru Takase, Masafumi Ueno and Gaku Nakazawa in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Author Contributions
Conception and design: KM, TI, JM, KT, MM, KU, and GN. Data acquisition: TI, JM, KT, KU, and EY. Data analysis/interpretation: KM, TT, MU, and GN. All authors participated in the writing, reviewing, and editing of the manuscript and approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
