Abstract
Heart failure (HF) is one of the leading causes of morbidity and mortality worldwide. Sacubitril/valsartan, an angiotensin receptor-neprilysin inhibitor, has been approved for the treatment of HF. At present, there have been few systematic and detailed reviews discussing the efficacy and safety of sacubitril/valsartan in HF. In this review, we first introduced the pharmacological mechanisms of sacubitril/valsartan, including the reduction in the degradation of natriuretic peptides in the natriuretic peptide system and inhibition of the renin-angiotensin system. Then, we summarized the efficacy of sacubitril/valsartan in HF patients with reduced ejection fraction (HFrEF) or preserved ejection fraction (HFpEF) including the reduction in risks of mortality and hospitalization, reversal of cardiac remodeling, regulation of biomarkers of HF, improvement of the quality of life, antiarrhythmia, improving renal dysfunction and regulation of metabolism. Finally, we discussed the safety and tolerability of sacubitril/valsartan in the treatment of HFrEF or HFpEF. Compared with ACEIs/ARBs or placebo, sacubitril/valsartan showed good safety and tolerability, although the risk of hypotension might be high. In conclusion, the overwhelming majority of studies show that sacubitril/valsartan is effective and safe in the treatment of HFrEF patients but that it has little benefit in HFpEF patients. Sacubitril/valsartan will probably be a promising anti-HF drug in the near future.
Introduction
Heart failure (HF), as a complex clinical syndrome that is rooted in any structural or functional impairment of ventricular filling or ejection of blood, is one of the leading causes of morbidity and mortality worldwide. 1 In the United States, more than 5.1 million people have clinically manifested HF and 650,000 new HF patients are diagnosed with HF annually. 1 Compared with patients with other heart diseases, HF patients have more severe risks of mortality and hospitalization. 1 A study enrolled 39,982 HF patients who were admitted with worsening HF or developed HF symptoms during a hospitalization for which HF was the primary discharge diagnosis; the findings showed that the 5-year mortality rate was 75.4% and that the 5-year readmission rate was 80.4% for the total cohort from January 1, 2005, through December 30, 2009, with 5 years of follow-up through the end of December 2014. 2 In addition, HF imposes a tremendous economic burden on the world, estimated at 108 billion dollars per annum in 2012. 3 According to the measurement of left ventricular ejection fraction (LVEF), the types of HF mainly include HF with preserved EF (HFpEF, EF ≥ 50%) and HF with reduced EF (HFrEF, EF < 40%). 1 Additionally, HF with mid-range ejection fraction (HFmrEF, 50% > EF ≥ 40%) was introduced by the European Society of Cardiology (ESC) in 2016 and the 2021 ESC HF guidelines showed a new change of the term “heart failure with mid-range ejection fraction” to “heart failure with mildly reduced ejection fraction” (HFmrEF). Along with implantable cardioverter-defibrillator (ICD) and cardiac resynchronization therapy, the multidrug combinations, including beta-blockers (BBs), angiotensin-converting enzyme inhibitors (ACEIs)/angiotensin receptor blockers (ARBs), and aldosterone receptor antagonists (MRAs) remain a cornerstone of the treatment of HF. Conventional drugs (BBs, ACEIs/ARBs and MRAs) have decreased the mortality and hospitalization rates of HF patients, mainly by regulating the renin-angiotensin system (RAS) and sympathetic nervous system. However, the outcomes of HF patients still remain unsatisfactory and include high mortality, high rehospitalization rates, poor quality of life, malignant arrhythmia, and renal dysfunction. 4 The most recent ESC-HF pilot study showed that the 12-month all-cause mortality rate for hospitalized HF patients was 17% and that the 12-month hospitalization rate for HF patients was 44%. 5 To overcome the limitations of current drug therapy for HF and improve the prognosis of these patients, many new compounds have been developed. The DAPA-HF study showed that dapagliflozin, a sodium-glucose cotransporter 2 (SGLT2) inhibitor, decreased the rates of HF deterioration and death from cardiovascular causes in HFrEF patients irrespective of the presence of diabetes mellitus. 6 The VICTORIA study showed that vericiguat, a novel oral soluble guanylate cyclase stimulator, decreased the composite of death from any cause or hospitalization among high-risk HF patients. 7 Importantly, sacubitril/valsartan, a first-in-class angiotensin receptor-neprilysin inhibitor (ARNI, formerly known as LCZ696), has gained much attention in the treatment of HF due to its dual effects on the RAS and natriuretic peptide system. 8
Studies have shown that sacubitril/valsartan can improve cardiac function and exercise capacity, reverse cardiac remodeling, and reduce the risks of cardiovascular mortality and hospitalizations in HF patients (Figure 1). The ESC and the American College of Cardiology (ACC) guideline committees have awarded a class 1B recommendation for sacubitril/valsartan in reducing the risks of cardiovascular death and hospitalization in HFrEF patients who remain symptomatic after treatment with ACEIs/ARBs, BBs and MRAs. 1,5 In addition, a meta-analysis provided evidence that sacubitril/valsartan had fewer drug-risks compared with ACEIs/ARBs, such as angioedema, hyperkalemia, cough, dizziness, renal dysfunction and arterial hypotension. 9 However, some issues regarding sacubitril/valsartan treatment for HFrEF, HFmrEF or HFpEF patients still need to be investigated and reviewed.
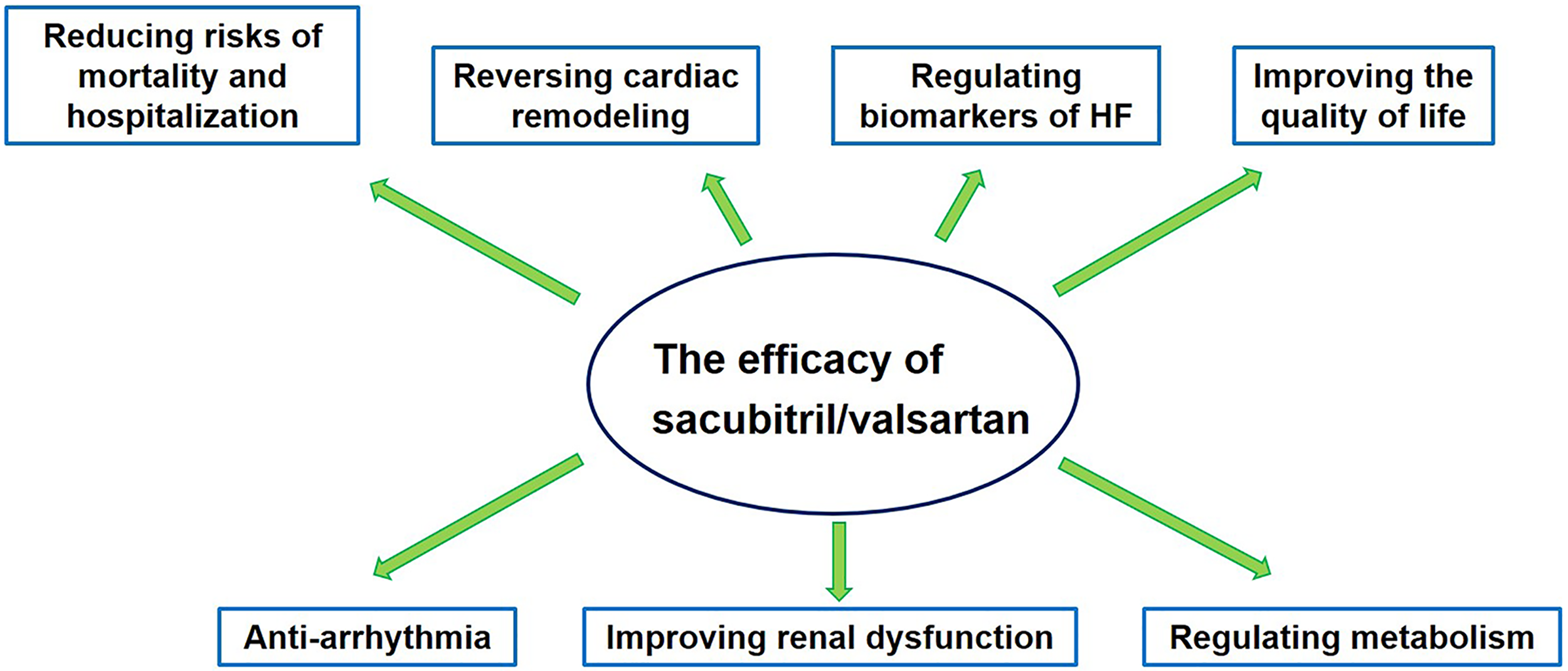
The efficacy of sacubitril/valsartan in the treatment for HF patients.
The Mechanism of Sacubitril/Valsartan in the Treatment of HF
Sacubitril/valsartan contains valsartan and sacubitril in a 1:1 mixture by molecule count and has 2 therapeutic targets in the treatment of HF: the natriuretic peptide system for sacubitril and the RAS for valsartan (Figure 2). The natriuretic peptide system mainly comprises of atrial-derived natriuretic peptides (ANP), B-type (brain) natriuretic peptides (BNP), and C-type natriuretic peptides (CNP). BNP is secreted mainly from the left ventricle and is markedly increased in HF patients, compared with ANP and CNP. 10 BNP is a biomarker for predicting the severity of HF and guiding therapy for HF; moreover, it has the marked benefit of increasing sodium excretion in the treatment of HF, compared with ANP and CNP. 11 Pro-BNP degrades into BNP and N-terminal pro-BNP (NT-pro-BNP) (Figure 2). BNP binds to natriuretic peptide receptor B on the cell membrane, which induces cleavage of guanosine triphosphate to cyclic guanosine monophosphate (cGMP) by cytoplasmic G proteins and initiates an intracellular cGMP signaling cascade involving protein kinase G, ultimately regulating downstream expression of genes involved in smooth muscle cell relaxation, diuresis, and natriuresis (Figure 2). 12 Sacubitril, as a neprilysin inhibitor, is converted to sacubitrilat by esterase and regulates the natriuretic peptide system by inhibiting the degradation of BNP (Figure 2). 12 Therefore, sacubitril increases the level of BNP resulting in vasodilation, sodium excretion and improvement of cardiac remodeling. In addition, sacubitril inhibits the degradation of angiotensin, endothelin 1, adrenomedullin, opioids and amyloid-β peptide, which might also participate in the beneficial effect in HF patients. 13 However, considering that sacubitril increases the plasma angiotensin II (Ang II) concentration, sacubitril alone does not have an obvious superiority and should be combined with ARBs such as valsartan in the treatment of HF. 14 The reason why sacubitril combines with ARBs rather than ACEIs is because both sacubitril and ACEIs inhibit the degradation of bradykinin which induces angioneurotic edema. 15 The RAS is activated in HF, enhancing sympathetic nerve activity and inducing cardiac remodeling, which aggravates the progression of HF. 16 In the RAS, Ang II is synthesized from angiotensinogen by renin and angiotensin-converting enzyme (Figure 2). 16 Ang II binds to the Ang II type 1 receptor on the cell membrane, which regulates arterial vasoconstriction, renal tubular reabsorption of sodium and water, vascular smooth muscle contraction and aldosterone release. 17 Valsartan inhibits the effects of Ang II by selectively blocking the type-1 receptor, which improves cardiac remodeling and dysfunction in HF patients. 18

The mechanism of sacubitril/valsartan in the treatment for HF patients through regulating natriuretic peptide system and renin-angiotensin system. BNP indicates B-type (brain) natriuretic peptides; NT-pro-BNP, N-terminal pro-BNP; cGMP, cyclic guanosine monophosphate.
Methods and Materials
To perform a review of the available literature, a search of the published English literature was conducted in the Medline, Embase, the Cochrane Library, ClinicalTrials.gov database and Google Scholar databases through February of 2021, using the following keywords: (“sacubitril-valsartan” OR “sacubitril/valsartan” OR “LCZ696” OR “LCZ” OR “ARNI”) AND (“heart failure” OR “cardiac dysfunction”).
Studies that were included for review met the following criteria: (I) the type of trial had to be an observational study, randomized controlled trial, meta-analysis or other types of clinical studies; (II) enrolled patients had to be diagnosed with HF; (III) patients had to be assigned to sacubitril/valsartan treatment orally; and (IV) all studies had to include data on mortality and hospitalization, cardiac remodeling, the regulation of biomarkers of HF, quality of life in HF patients, antiarrhythmia, renal function, the regulation of metabolism, and adverse events, etc. Studies were excluded if they did not have a measurement of these indices.
Risks of bias, namely, random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessors, incomplete outcome data, selective reporting bias, for-profit bias and other bias, were evaluated with the Cochrane Risk of Bias tool. 19
Efficacy of Sacubitril/Valsartan in the Treatment of HF
The efficacy of sacubitril/valsartan in the treatment of HF is discussed from the following 7 key perspectives: reduction in the risks of mortality and hospitalization, reversal of cardiac remodeling, regulation of biomarkers of HF, improvements of the quality of life, decrease in arrhythmia, improvement of renal function and regulation of metabolism in HF patients. We assessed the risk of bias according to the Cochrane Risk of Bias tool and listed this information in the Supplementary Table 1.
Reduction in the Risks of Mortality and Hospitalization
We listed the evidence for sacubitril/valsartan decreasing the risk of mortality and hospitalization among HFrEF patients, HFpEF patients and HFmrEF patients, respectively. These studies are listed in Supplementary Table 2.
Two randomized controlled trials (RCTs), 3 observational trials and 3 meta-analyses evaluated the mortality and hospitalization in HFrEF patients treated with sacubitril/valsartan. One RCT, the PARADIGM-HF trial, was designed to compare the risks of mortality and hospitalization between HFrEF patients treated with sacubitril/valsartan and those treated with ACEIs. 20 The study indicated that compared with enalapril, sacubitril/valsartan reduced the risk of the primary composite endpoint (cardiovascular death or HF hospitalization) by 20% (hazard ratio, HR, 0.80; 95% CI, 0.73 to 0.87; P < 0.001), the risk of cardiovascular death by 20% (HR, 0.80; 95% CI, 0.71 to 0.89; P < 0.001), the risk of first HF hospitalization by 21% (HR, 0.79; 95% CI, 0.71 to 0.89; P < 0.001), and the risk of all-cause mortality by 16% (HR, 0.84; 95% CI, 0.76 to 0.93; P < 0.001) after a median follow-up of 27 months. In this study, sacubitril/valsartan was more effective in reducing the risk of cardiovascular mortality or HF hospitalization than enalapril in HFrEF patients. The PIONEER-HF trial, a multicenter, randomized, double-blind, active-controlled trial, evaluated the effect of sacubitril/valsartan in HFrEF patients who were hospitalized for acute decompensated HF. 21 Sacubitril/valsartan was more effective than enalapril in reducing the risk of HF rehospitalization or cardiovascular death (9.2% vs. 15.2%; HR, 0.58; 95% CI, 0.39 to 0.87; P = 0.007) in enrolled patients after an 8-week follow-up. 22 A retrospective cohort study compared the effectiveness of sacubitril/valsartan with that of ACEIs/ARBs in HFrEF patients treated in routine clinical practice. 23 A total of 7,893 HFrEF patients were included and the mean follow-up time was 6.3 months. Compared with the ACEIs/ARBs group, the sacubitril/valsartan group had a lower all-cause mortality rate (HR, 0.80; 95% CI, 0.66 to 0.97; P = 0.027), lower all-cause hospitalization rate (HR, 0.86; 95% CI, 0.80 to 0.91; P < 0.001) and similar HF hospitalization rate (HR, 1.07; 95% CI, 0.96 to 1.19; P = 0.260). This study provided real-world effectiveness data outside of a clinical trial that compared sacubitril/valsartan with ACEIs/ARBs. A recent crossover trial evaluated the effects of comprehensive disease-modifying pharmacological therapies (sacubitril/valsartan, BBs, MRAs, and SGLT2 inhibitors) versus conventional therapy (ACEIs/ARBs and BBs) in HFrEF patients by making indirect comparisons of 3 pivotal studies, including the EMPHASIS-HF (n = 2,737), PARADIGM-HF (n = 8,399), and DAPA-HF (n = 4,744) trials. 24 The primary endpoint of cardiovascular death or HF hospitalization was decreased in the comprehensive disease-modifying therapy group versus the conventional therapy group (HR, 0.38; 95% CI, 0.30 to 0.47). The combination of sacubitril/valsartan, BBs, MRAs and SGLT2 inhibitors might be recommended as a new therapeutic standard for the treatment of HFrEF. A meta-analysis showed that sacubitril/valsartan decreased all-cause mortality (13.8% vs. 16.1%; risk ratio, RR, 0.86; 95% CI, 0.79 to 0.94; P = 0.0008) and hospitalization (12.6% vs. 16.1%; RR, 0.79; 95% CI, 0.72 to 0.87; P ≤ 0.00001) in HFrEF patients, compared with ACEIs/ARBs. 25 Another meta-analysis evaluated the risks of HF hospitalization and all-cause mortality between HFrEF patients treated with sacubitril/valsartan or another neprilysin inhibitor, lisinopril, and those treated with ACEIs. This meta-analysis included 3 RCTs, including the IMPRESS, OVERTURE, and PARADIGM-HF trials. IMPRESS and OVERTURE evaluated the effect of omapatrilat or ACEIs in HFrEF patients treated by ACEIs/ARBs. This meta-analysis showed that compared with ACEIs, combined neprilysin/RAS inhibition significantly decreased the risks of the composite outcome of death or HF hospitalization (HR, 0.86; 95% CI, 0.76 to 0.97; P = 0.013) and the risk of all-cause mortality (HR, 0.88; 95% CI, 0.80 to 0.98; P = 0.021) in HFrEF patients. 26
One RCT and 2 meta-analyses evaluated the mortality and hospitalization in HFpEF patients treated with sacubitril/valsartan. In the PARAGON-HF trial, compared with valsartan, sacubitril/valsartan did not decrease the risk of overall HF hospitalization (RR, 0.85; 95% CI, 0.72 to 1.00) or death from cardiovascular causes (HR, 0.95; 95% CI, 0.79 to 1.16) after a 35-month follow-up. 27 Death from any cause occurred in 342 patients (14.2%) from the sacubitril/valsartan group and in 349 patients (14.6%) from the valsartan group (HR, 0.97; 95% CI, 0.84 to 1.13). 27 A meta-analysis found no benefit for HFpEF patients treated with sacubitril/valsartan, compared with those treated with ACEIs/ARBs by assessing all-cause mortality (13.3% vs. 14.0%, RR, 0.95; 95% CI, 0.83 to 1.09; P = 0.47). 25 Another meta-analysis showed that sacubitril/valsartan did not significantly decrease the risk of all-cause mortality (HR, 0.97; 95% CI, 0.84 to 1.13; P = 0.58) in HFpEF patients. 28
One meta-analysis and one RCT assessed the effects of sacubitril/valsartan on the mortality and hospitalization in HEmrEF patients. The meta-analysis showed that in patients with HF, including HFmrEF and HFpEF, the rate of HF hospitalization was significantly lower in the sacubitril/valsartan group than in the ACEIs/ARBs group (RR, 0.84; 95% CI, 0.77 to 0.91; P < 0.001). 29 Notably, sacubitril/valsartan did not significantly improve the cardiovascular mortality in these patients (RR, 0.94; 95% CI, 0.79 to 1.12; P = 0.481). Furthermore, all-cause mortality was not significantly different between the sacubitril/valsartan group and the ACEIs/ARBs group (RR, 0.95; 95% CI, 0.83 to 1.08; P = 0.417). However, the meta-analysis did not divide patients into subgroups according to the type of HF. A prespecified pooled analysis, combining data from PARADIGM-HF (LVEF eligibility ≤ 40%; n = 8,399) and PARAGON-HF (LVEF eligibility ≥ 45%; n = 4,796), showed that sacubitril/valsartan might benefit individuals with an EF below the normal range (<50%, HFrEF and HFmrEF). 30 In a subgroup analysis of PARAGON-HF, sacubitril/valsartan improved the composite of total hospitalizations for HF and death from cardiovascular causes in the below-median LVEF (<57%, but not those with LVEF above the median) subgroup (RR, 0.78; 95% CI, 0.64 to 0.95). 27 These studies implied that sacubitril/valsartan was likely to have a benefit of reducing the risks of mortality and hospitalization in HFmrEF patients.
In summary, evidence from RCTs, meta-analyses and observational studies indicated that sacubitril/valsartan decreased the risks of all-cause mortality and hospitalization in HFrEF patients but not in HFpEF patients. Sacubitril/valsartan might also benefit in HFmrEF patients. Few studies have evaluated the effects of sacubitril/valsartan in HFpEF or HFmrEF patients, and these effects should be explored further in the future.
Reversal of Cardiac Remodeling
Cardiac remodeling is one of the most important mechanisms in the progression of HF. Recently, researchers have paid considerable attention to improving the prognosis of HF patients by inhibiting or reversing cardiac remodeling. 31 We present the evidence of the ability of sacubitril/valsartan to reverse left ventricular remodeling in HFrEF or HFpEF patients in Supplementary Table 3. We also discussed the effects of sacubitril/valsartan on the reversal of right ventricular and atrial remodeling in HF patients.
One RCT, 10 observational studies and 2 meta-analyses showed the reversal of left ventricular remodeling in HFrEF patients treated with sacubitril/valsartan. One RCT showed that the decrease in effective regurgitant orifice area in HFrEF patients with functional mitral regurgitation was significantly greater in the sacubitril/valsartan group than in the valsartan group (−0.058 ± 0.095 vs. −0.01 ± 0.105 cm2; P = 0.032). 32 In the PROVE-HF trial, sacubitril/valsartan increased LVEF by 9.4% (P < 0.001) and decreased left ventricular end-diastolic volume index by 12.25 mL/m2 (P < 0.001), left ventricular end-systolic volume index by 15.29 mL/m2 (P < 0.001), left atrial volume index by 7.57 mL/m2 (P < 0.001) and the E/e′ ratio by 1.3 (P < 0.001) in 794 HFrEF patients after 12 months of treatment. 33 These changes showed obvious improvement in the structure and function of left ventricular remodeling in HFrEF patients treated with sacubitril/valsartan. A meta-analysis that included 16 observational trials and 4 RCTs evaluated the effect of sacubitril/valsartan on reversing cardiac remodeling in HF patients. 34 This meta-analysis indicated that compared with ACEIs/ARBs in HFrEF patients, sacubitril/valsartan significantly improved left ventricular hypertrophy and function, which manifested as LVEF (mean difference, MD, 4.64%; 95% CI, 3.93 to 5.35), end-systolic volume (MD, −18.23 ml; 95% CI, −27.25 to −9.20), end-diastolic volume (MD, −21.6 ml; 95% CI, −24.32 to −18.88), end-systolic dimension (MD, −3.50 mm; 95% CI, −5.56 to −1.44), end-systolic dimension (MD, −2.42 mm; 95% CI, −3.06 to −1.78), left atrial volume (MD, −7.59 mL; 95% CI, −14.03 to −1.14), and left ventricular mass index (MD, −14.44 g/m2; 95% CI, −22.61 to −6.27). Another meta-analysis showed a beneficial effect of sacubitril/valsartan compared with ACEIs/ARBs on increasing LVEF (RR, 0.80; 95% CI, 0.72 to 0.88; P < 0.0001) in HFrEF patients. 25
One RCT, the PARAMOUNT-HF trial, evaluated the effects of sacubitril/valsartan on reversing cardiac remodeling in HFpEF patients. After 36 weeks, although reductions in left atrial volume (P = 0.003), left atrial width (P = 0.03) and left atrial volume index (P = 0.007) were observed, LVEF (P = 0.69), E/A (P = 0.43) and E/e′ (P = 0.42) were not significantly improved by sacubitril/valsartan, compared with valsartan. 35 There may be no benefit of sacubitril/valsartan in improving cardiac remodeling in HFpEF patients.
Three observational studies evaluated the effects of sacubitril/valsartan on reversing right ventricular or atrial remodeling in HF patients, respectively. Right ventricular recovery is associated with decreased mortality in HF patients. 36 In the Daunia HF registry study, HFrEF patients treated by sacubitril/valsartan for 12 months clearly showed improvements in right ventricular function, including the pulmonary artery systolic pressure (31.0 ± 12.8 vs. 34.7 ± 12.5 mmHg, P < 0.05), the tricuspid annular plane systolic excursion (17.8 ± 3.9 vs. 16.5 ± 4.0 mm, P < 0.001), reduced mean pulmonary artery systolic pressures (3.7 ± 11.4 mmHg, P < 0.05) and increased mean tricuspid annular plane systolic excursion (1.3 ± 2.5 mm, P < 0.001). 37 For the first time, a beneficial effect of sacubitril/valsartan on right ventricular function was reported. In addition, sacubitril/valsartan improved atrial remodeling, which could be reflected by the P-wave on electrocardiogram. 38 In a retrospective cohort study, treatment with sacubitril/valsartan for 1 month favorably altered P-wave dispersion (28.6 ± 5.3 vs. 33.6 ± 7.9, P = 0.006) and maximum P-wave duration (116.1 ± 14.1 vs. 135.6 ± 32.1, P = 0.041) on standard 12-lead electrocardiograms in HFrEF patients. 39 Another retrospective cohort study found that sacubitril/valsartan treatment for 12 months obviously increased the reservoir function of the atrial chambers (P < 0.001) compared with ACEIs/ARBs treatment in HF patients, as detected by the peak atrial longitudinal strain. 40 In this study, the reservoir function of the atrial chambers was associated with a reduced risk of atrial fibrillation (AF) recurrence. Sacubitril/valsartan might benefit HF patients with AF by reversing atrial remodeling. However, few subjects were enrolled in these 3 observational studies, and the results should be verified in RCTs.
In brief, current evidence from RCTs, meta-analyses and observational studies indicated that sacubitril/valsartan played an important role in reversing cardiac remodeling and improving cardiac function in HFrEF patients, but not in HFpEF patients. RCTs and meta-analyses should be implemented to explore the effects of sacubitril/valsartan on reversing right ventricular or atrial remodeling in HF patients.
Regulation of Biomarkers of HF
Biomarkers of HF provide support for clinical decision making regarding the diagnosis of HF and aid in the evaluation of the prognosis or disease severity. The primary biomarkers of HF include BNP, NT-pro-BNP, cardiac troponin, soluble suppression of tumorigenicity 2 (sST2), catecholamines and galectin-3.1 We list the main studies on exploring how sacubitril/valsartan regulates biomarkers of HF in HFrEF or HFpEF patients in Supplementary Table 4.
Two RCTs, 10 observational studies and 2 meta-analyses explored the effects of sacubitril/valsartan on regulating biomarkers of HF in HFrEF patients. One RCT, the PIONEER-HF trial, showed that compared with enalapril, sacubitril/valsartan significantly reduced the level of NT-pro-BNP (ratio of change, 0.76; 95% CI, 0.69 to 0.85) after 1 week of treatment, and reduced the levels of high-sensitivity cardiac troponin (P < 0.001) and sST2 (P < 0.001) after 4 weeks of treatment. 21 Moreover, treatment with sacubitril/valsartan led to a significant increase in the level of urinary cyclic guanosine (P < 0.001, 1 week). 21 One retrospective cohort study showed that after 6 months follow-up, epinephrine levels were significantly increased in HFrEF patients treated with sacubitril/valsartan in comparison with baseline levels (66 (37 to 93) pg/mL vs. 38 (18 to 74) pg/mL, P < 0.001). 41 Until now, this has been the only study on the regulation of the level of catecholamines by sacubitril/valsartan in HF patients. A prospective cohort study evaluated the effects of sacubitril/valsartan on the levels of the natriuretic peptide system in HFrEF patients. 42 In this study, the average follow-up time was 22, 46, and 84 days. The level of ANP rapidly and substantially increased with the initiation and titration of sacubitril/valsartan; the level of MR-pro-ANP, NT-pro-BNP, and pro-BNP1-108 variably declined after treatment with sacubitril/valsartan, whereas CNP concentrations showed no consistent change. The rapid and significant increase in ANP by sacubitril/valsartan implied that ANP could be regarded as a biomarker for identifying the response to sacubitril/valsartan therapy. 42 A meta-analysis showed a beneficial effect of sacubitril/valsartan of decreasing the level of NT-pro-BNP (MD, 3.3; 95% CI, 2.23 to 4.38; P < 0.0001) in HFrEF patients relative to that of ACEIs/ARBs. 25 Another meta-analysis showed that compared with ACEIs/ARBs, sacubitril/valsartan reduced the level of NT-pro-BNP (HFrEF, MD, −243.00 pg/mL; 95% CI, −264.26 to −221.74; HFpEF, MD, −111.00 pg/mL; 95% CI, −157.92 to −64.08) in both HFrEF patients and HFpEF patients. 34 Moreover, sacubitril/valsartan reduced sST2 levels in HFrEF patients (MD, −1.60 ng/mL; 95% CI, −2.61 to −0.59). 34
Two RCTs and 2 meta-analyses evaluated the effects of sacubitril/valsartan on regulating biomarkers of HF in HFpEF patients. In the PARAMOUNT-HF trial, there was a more significant reduction in the level of NT-pro-BNP (ratio of change, 0.77; 95% CI, 0.64 to 0.92, P = 0.005) after 12 weeks and the level of hs-TnT (P = 0.03) after 36 weeks for HFpEF patients in the sacubitril/valsartan group compared with those in the valsartan group. 35 In the PARAGON-HF trial, the primary endpoint, the change in NT-pro-BNP from baseline to 12 weeks, was significantly decreased in the sacubitril/valsartan group compared with the valsartan group (ratio of change, 0.77; 95% CI, 0.64 to 0.92; P = 0.005). A meta-analysis showed that sacubitril/valsartan did not decrease the level of sST2 (MD, 3.80 ng/mL, 95% CI, −8.67 to 1.07) in HFpEF patients. 34 Another meta-analysis showed that sacubitril/valsartan led to a minor incremental reduction in NT-pro-BNP concentration (mean ratio, 0.83; 95% CI, 0.80 to 0.86) in HFpEF patients. 28
The evidence from RCTs, meta-analyses and observational studies indicated that sacubitril/valsartan significantly decreased the level of NT-pro-BNP in HFrEF patients and HFpEF patients, compared with ACEIs/ARBs. Sacubitril/valsartan also obviously decreased the level of sST2 in HFrEF patients but not in HFpEF patients.
Improvements in Quality of Life
HF obviously reduces health-related quality of life (HRQL), especially physical function and vitality. 1 One of the most important aims of the treatment of HF is to reduce symptoms and enhance the HRQL. We list studies on how sacubitril/valsartan influences the quality of life in HFrEF or HFpEF patients assessed by the Kansas City Cardiomyopathy Questionnaire (KCCQ), Minnesota Living with HF questionnaire scores, the six-minute walk test distance and New York Heart Association (NYHA) class in Supplementary Table 5.
The KCCQ is a systematic and readily available HRQL evaluation instrument for HF patients. 43 One RCT and 3 observational studies evaluated the effects of sacubitril/valsartan of improving KCCQ scores in HFrEF patients. In the PARADIGM-HF trial, the reduction in KCCQ scores was significantly lower in HFrEF patients treated with sacubitril/valsartan after the 8-month follow-up than those treated with enalapril (−2.99 vs. −4.63; P = 0.001). In the CHAMP-HF trial, a significant improvement was found in the 12-item KCCQ score in HFrEF patients initiating with sacubitril/valsartan treatment compared with patients who had not started with sacubitril/valsartan therapy (5.3 ± 19.0 vs. 2.5 ± 17.4, P < 0.001). 44 Another observational study showed that there were also statistically significant differences in the quality of life domain scores (P < 0.001), symptom frequency domain scores (P = 0.02) and social limitation domain scores (P = 0.007) of the KCCQ scores. 45 An observational study used the Minnesota Living with HF questionnaire to evaluate the quality of life in HFrEF patients switched from valsartan to sacubitril/valsartan treatment. 39 Minnesota Living with HF questionnaire scores were improved in HFrEF patients at the 1-month follow-up (23.2 ± 7.0 vs. 31.2 ± 6.2, P < 0.001). 39 A meta-analysis assessed the Minnesota Living with HF Questionnaire score of HFrEF patients and showed evidence that compared with ACEIs/ARBs, sacubitril/valsartan was beneficial to these patients (MD, −5.19; 95% CI, −8.37 to −2.01; P = 0.001). 25 These studies implied that sacubitril/valsartan improved quality of life in HFrEF patients, as evaluated by KCCQ scores or Minnesota Living with HF questionnaire scores.
The six-minute walk test is a submaximal and simple exercise test for evaluating physical functional capacity in chronic HF patients. 46 Five observational studies and one meta-analysis evaluated the effects of sacubitril/valsartan on the six-minute walk test in HFrEF patients. A prospective cohort study showed that sacubitril/valsartan treatment significantly increased the six-minute walk test distance (+Δ = 41.8 m (33.4 to 50.2); P < 0.001) in HFrEF patients after a 30-day follow-up (P < 0.001). 47 Another prospective cohort study assessed the effect in HFrEF patients listed for heart transplantation. It was shown that sacubitril/valsartan increased the six-minute walk test distance (P < 0.001) and improved the NYHA class (P < 0.001) in advanced HFrEF patients who were waiting for heart transplantation and followed up until heart transplantation, device implantation, or the last follow-up visit after a 2-year follow-up. 48 A meta-analysis that included 20 trials, showed significant improvement in six-minute walk test distance (MD, 27.62 m; 95% CI, 15.76 to 39.48; P < 0.00001) in HFrEF patients treated with sacubitril/valsartan. 34 The evidence from meta-analysis and observational studies implied that the six-minute walk test distance was increased by sacubitril/valsartan in HFrEF patients.
The NYHA classification system is another important evaluation index of quality of life in HF patients. 49 Whether sacubitril/valsartan improved the NYHA class of HFrEF patients was studied in 3 cohort studies. A retrospective cohort study reported significant improvements in NYHA class (P < 0.05) and the quality of life scores on the EQ5D-VAS (standardized instrument to measure health outcomes) (P < 0.05) in HFrEF patients treated with sacubitril/valsartan after 6 to 12 months of follow-up. 50 In another retrospective cohort study, sacubitril/valsartan therapy obviously improved NYHA class from 2.3 ± 0.6 to 1.8 ± 0.5 (P < 0.001), increased LVEF from 31.2 ± 7.0% to 37.3 ± 10.5% (P < 0.001) and decreased the level of NT-pro-BNP from 3884 ± 4871 pg/ml to 1975.3 ± 3006.6 pg/ml (P = 0.0001) in HFrEF patients after 316.1 ± 155.9-day follow-up. 51 However, given the weak level of evidence from observational studies, RCTs and meta-analyses should be implemented.
Few studies have evaluated whether sacubitril/valsartan affects the quality of life of HFpEF patients. In the PARAMOUNT-HF trial, HFpEF patients in the sacubitril/valsartan group showed no improvement in NYHA class compared with those in the valsartan group after the 12-week follow-up and 36-week follow-up (P = 0.11 and P = 0.05). In addition, no difference in the KCCQ scores of these 2 groups was found. 35 A meta-analysis showed that there was no significant effect on the change in NYHA functional class (OR, 1.23; 95% CI, 0.86 to 1.74) in HFpEF patients treated with sacubitril/valsartan. 28 Therefore, sacubitril/valsartan may have no effect on the quality of life in HFpEF patients.
Thus, sacubitril/valsartan was found to improve the quality of life of HFrEF patients, as assessed by the quality of life scores, the six-minute walk test distance and NYHA class. However, these beneficial effects of sacubitril/valsartan were not observed in HFpEF patients.
Antiarrhythmia
HF patients suffer from many kinds of arrhythmia, including AF, ventricular arrhythmias, symptomatic bradycardia, pauses and atrioventricular block. Ventricular arrhythmias are closely associated with high mortality in HF patients. Whether sacubitril/valsartan has a role in antiarrhythmia has been reported. We showed these studies in Supplementary Table 6.
Sacubitril/valsartan might decrease the risk of ventricular arrhythmia in HFrEF patients. Four observational studies evaluated this effect in HFrEF patients treated with sacubitril/valsartan. An observational study showed that sustained ventricular tachycardia (VT, defined as >30 s)/VT episodes (15 vs. 4, n; P = 0.03), nonsustained VT (22 vs. 8, n; P = 0.01), appropriate shocks (13 vs. 3, n; P = 0.02) and paroxysmal atrial tachycardia/AF episodes (34 vs. 19, n; P = 0.03) were significantly decreased in HFrEF patients after 12 months of follow-up. 52 Another observational study evaluated the effects of sacubitril/valsartan on ventricular arrhythmias in HFrEF patients with ICD and remote monitoring over a 9-month follow-up period. 53 This study indicated that sacubitril/valsartan reduced nonsustained VT episodes (5.4 ± 0.5 vs. 15 ± 1.7, P < 0.002), sustained VT (0.8 vs. 6.7%, P < 0.02) and adequate ICD shocks (0.8 vs. 6.7%, P < 0.02) in HFrEF patients, compared with ACEIs/ARBs. A retrospective analysis enrolled HFrEF patients who received sacubitril/valsartan for a class-I indication and were implanted with an ICD or underwent cardiac resynchronization therapy with remote monitoring. 54 The study found that initiation of sacubitril/valsartan was associated with a lower risk of VT/ventricular fibrillation (P < 0.001), which resulted in a lower incidence of ICD-interventions. Further findings from another observational study showed that sacubitril/valsartan reduced the recurrence risk of AF. In HFrEF patients with a history of AF, sacubitril/valsartan decreased the risk of AF recurrence (P = 0.001) after the 12-month follow-up, compared with ACEIs/ARBs. 40
QTc interval prolongation is associated with a high risk of malignant arrhythmias in HF patients. 55 Whether sacubitril/valsartan affects the QTc interval has remained controversial. Two studies explored the effects of sacubitril/valsartan on the QTc interval. A prospective cohort study showed that the QTc interval (451.9 vs. 426.0 ms, P < 0.001), QRS duration (125.1 vs. 120.8 ms, P = 0.033) and mechanical dispersion index (88.4 vs. 78.1 ms, P = 0.036) were significantly reduced in HFrEF patients treated with sacubitril/valsartan after 6 months, which might be the mechanism by which sacubitril/valsartan reduced the risk of sudden death. 56 A single-center, randomized, positive- and placebo-controlled, partially blinded study indicated that sacubitril/valsartan at single therapeutic (194/206 mg) and supratherapeutic (582/618 mg) doses had no effect on cardiac repolarization in healthy subjects by assessing corrected QTcF (ΔΔQTcF; Fridericia correction) on electrocardiograms. 57 In brief, these studies indicated that sacubitril/valsartan did not delay the QT interval which reflected the depolarization and repolarization process of the ventricles.
These observational studies implied that sacubitril/valsartan might have an antiarrhythmic effect in HFrEF patients, including a reduction in ventricular arrhythmias. However, no RCTs or meta-analyses have investigated this. Given the weak evidence level of observational studies, the effects of sacubitril/valsartan on antiarrhythmia in HF patients should be further explored in RCTs.
Improvement of Renal Dysfunction
A large number of patients have renal dysfunction at the terminal stage of HF and renal dysfunction deteriorates the prognosis of HF patients. Whether sacubitril/valsartan could improve renal dysfunction or inhibit the deterioration of renal dysfunction in HFrEF or HFpEF patients has been studied. These studies are shown in Supplementary Table 7.
One RCT and one retrospective study evaluated the effects of sacubitril/valsartan on renal function in HFrEF patients. The PARADIGM-HF trial reported that the progression of renal dysfunction in HFrEF patients was slower in the sacubitril/valsartan group than in the enalapril group (change in estimated glomerular filtration rate (eGFR), −1.61 ml/min/1.73 m2/year vs. −2.04 ml/min/1.73 m2/year, P < 0.001). 58 A retrospective study showed that the renal function was improved after 12 months of treatment with sacubitril/valsartan compared to baseline (P < 0.001) and a greater improvement was found in HFrEF patients aged < 65 years (P = 0.002) and those with chronic kidney disease (P = 0.009). 59 This was the first real-life clinical study that verified the benefit of sacubitril/valsartan in HFrEF patients with renal impairment after a 12-month follow-up. Overall, these studies implied that sacubitril/valsartan improved the kidney function in HFrEF patients with chronic kidney disease.
Two RCTs evaluated the kidney protective effects of sacubitril/valsartan in HFpEF patients. In the PARAMONT-HF trial, compared with those in the valsartan group, HFpEF patients in the sacubitril/valsartan group had a smaller decrease in eGFR (sacubitril/valsartan, −1.6 mL/min per 1.73 m2 vs. valsartan, −5.2 mL/min per 1.73 m2; P = 0.007) and a greater increase in the urinary albumin-creatinine ratio (sacubitril/valsartan, 1.9 mg/mmol at baseline, 2.9 mg/mmol at week 36; valsartan, 2.0 mg/mmol at baseline, 2.0 mg/mmol at week 36; P = 0.02) at the 36-week follow-up. 35 In the PARAGON-HF study, the decline in eGFR was lower in HFpEF patients treated with sacubitril/valsartan than in those treated with valsartan (−1.5 vs. −5.2 mL/min per 1.73 m2; P = 0.002). 60 The 2 RCTs implied that sacubitril/valsartan had a protective effect on kidney function in HFpEF patients.
Three RCTs and one observational study provided evidence that sacubitril/valsartan had a beneficial effect of renal protection in HFrEF or HFpEF patients compared with ACEIs/ARBs. A meta-analysis that included 6 randomized trials showed that sacubitril/valsartan significantly reduced the risk of renal function deterioration (OR, 0.77; 95% CI, 0.61 to 0.97, P = 0.02) in HFrEF and HFpEF patients, compared with ACEIs/ARBs. 61 Considering the effects of ACEIs/ARBs on renal function deterioration, sacubitril/valsartan would be a superior choice in the treatment of HF with renal dysfunction.
Regulation of Metabolism
Studies have reported whether sacubitril/valsartan regulates the metabolism of blood glucose and uric acid. In the PARADIGM-HF trial, 3,778 HFrEF patients, diagnosed with diabetes or HbA1c ≥ 6.5% at screening, were randomly assigned to a sacubitril/valsartan group or an enalapril group. The study found that HbA1c concentrations obviously decreased by 0.26% in the sacubitril/valsartan group, compared with 0.16% in the enalapril group (P = 0.0023) during the first year. The HbA1c concentrations continuously decreased in the sacubitril/valsartan group over the 3-year follow-up. 62 In addition, the ability of sacubitril/valsartan to regulate serum uric acid was explored in the PARADIGM-HF trial. Sacubitril/valsartan reduced serum uric acid by 0.24 (0.17 to 0.32) mg/dL over 12 months (P < 0.0001), compared with enalapril in HFrEF patients. 63 In an observational study, sacubitril/valsartan reduced uric acid (7.5 to 7.1 mg/dL, P = 0.009) in HFrEF patients after 12 months of treatment. 64
At present, few studies have evaluated the effects of sacubitril/valsartan on metabolism. The available evidence from the PARADIGM-HF trial indicated that sacubitril/valsartan might improve the metabolism of blood glucose and uric acid in HFrEF patients but the mechanism was unclear. Whether and how sacubitril/valsartan regulates metabolism should be studied further.
Safety and Tolerability
There are differences in safety and tolerability between sacubitril/valsartan and ACEIs/ARBs in the treatment of HF. This is usually assessed by the adverse events of sacubitril/valsartan, which include hypotension, hyperkalemia, dizziness, cough, liver related adverse events, headaches, angioedema and renal impairment (Figure 3). In this review, we mainly discussed the safety and tolerability of sacubitril/valsartan in HFrEF or HFpEF patients. We list these studies in Supplementary Table 8.

The safety of sacubitril/valsartan in the treatment for HF patients.
Three RCTs, 2 observational studies and 2 meta-analyses evaluated the safety and tolerability of sacubitril/valsartan in HFrEF patients. In the PARADIGM-HF trial, 12.3% of subjects in the enalapril group and 10.7% of subjects in the sacubitril/valsartan group withdrew due to an adverse event during the run-in phase (P = 0.03). 20 The incidences rates of hypotension, hyperkalemia (>6.0 mmol/L), renal impairment (serum creatinine ≥ 2.5 mg/dL), and cough were 14%, 4.3%, 3.3% and 11.3% respectively in the sacubitril/valsartan group, while they were 9.2%, 5.6%, 4.5% and 14.3% respectively in the enalapril group (P < 0.05, respectively). Another RCT, the PIONEER-HF trial, showed that the rates of worsening renal function (13.6% vs. 14.7%, RR, 0.93; 95% CI, 0.67 to 1.28), hyperkalemia (11.6% vs. 9.3%, RR, 1.25; 95% CI, 0.84 to 1.84), and symptomatic hypotension (13.3% vs. 14.0%, RR, 0.95; 95% CI, 0.83 to 1.09) did not differ significantly between the sacubitril/valsartan group and the enalapril group. 21 One observational study evaluated the difference in adverse effects between HFrEF inpatients and HFrEF outpatients treated with sacubitril/valsartan. The incidence rates of discontinuation (17.0% vs. 11.5%, P = 0.13), hypotension (16.0% vs. 16.7%, P = 0.88), renal dysfunction (7.0% vs. 6.8%, P = 0.94), and hyperkalemia (1.0% vs. 4.9%, P = 0.09) were similar between the 2 groups. 65 Additionally, the rate of discontinuation due to hypotension-related adverse events was similar (0.9% vs. 0.7%; P = 0.38). A meta-analysis, including 3 studies, compared the safety in HFrEF patients treated with sacubitril/valsartan or ACEIs. 26 Sacubitril/valsartan was associated with an increased risk of hypotension, but reduced risks of renal dysfunction and hyperkalemia in all 3 trials, compared with ACEIs. Another meta-analysis showed that sacubitril/valsartan had a beneficial effect on serious adverse events compared with ACEIs/ARBs in HFrEF patients (37.4% vs. 41.8%, RR, 0.89; 95% CI, 0.86 to 0.94; P ≤ 0.00001), including the risk of hyperkalemia (RR, 0.44; 95% CI, 0.26 to 0.76; P = 0.003), fatigue (RR, 0.10; 95% CI, 0.10 to 0.79; P = 0.03) and syncope (RR, 0.62; 95% CI, 0.43 to 0.91; P ≤ 0.01). 25 The evidence from RCTs, meta-analyses and observational studies indicated that sacubitril/valsartan had a good tolerability in HFrEF patients compared with ACEIs/ARBs.
Two RCTs and one meta-analysis investigated the safety and tolerability of sacubitril/valsartan in HFpEF patients. In the PARAGON-HF trial, the incidences rates of hypotension, angioedema and hyperkalemia (>6.0 mmol/L) were 15.8%, 0.6%, and 3.1% respectively in HFpEF patients treated with sacubitril/valsartan, while they were 10.8%, 0.2%, and 4.3% respectively in the valsartan group (P < 0.001, P = 0.02, and P = 0.04, respectively). 27 However, liver-related adverse events (6.3% vs. 7.5%, P = 0.11) were similar between the 2 groups. In the PARAMOUNT-HF trial, the incidences rates of any adverse events (64% vs. 73%, P = 0.14), hypotension (19% vs. 18%, P = 0.88), renal dysfunction (2% vs. 5%, P = 0.34), and hyperkalemia (8% vs. 6%, P = 0.50) were similar between HFpEF patients treated with sacubitril/valsartan or valsartan. 35 A meta-analysis showed no evidence of a difference in serious adverse events between the sacubitril/valsartan group and the ACEIs/ARBs group (55.5% vs. 56.1%; RR, 0.99; 95% CI, 0.94 to 1.04; P = 0.63) in HFpEF patients. 25 Although few studies have explored the safety and tolerability of sacubitril/valsartan in HFpEF patients, these clinical studies supported the fact that the safety and tolerability might be similar between sacubitril/valsartan and valsartan in HFpEF patients.
Although some studies did not distinguish the type of HF, they still provided valuable evidence for comparing the safety between sacubitril/valsartan and ACEIs/ARBs or placebo in the treatment of HF patients. A meta-analysis showed that sacubitril/valsartan significantly decreased the risk of discontinuation of treatment for any adverse events (RR, 0.97; 95% CI, 0.80 to 1.17, P = 0.75), compared with ACEIs/ARBs or placebo. Sacubitril/valsartan increased the risk of angioedema (RR, 1.93; 95% CI, 1.02 to 3.68, P = 0.04) and dizziness (RR, 1.28; 95% CI, 1.08 to 1.52, P = 0.004) and decreased the risk of renal dysfunction (RR, 0.73; 95% CI, 0.59 to 0.91, P = 0.004) and bronchitis (RR, 0.82; 95% CI, 0.68 to 0.98, P = 0.03) in HF or hypertension patients. 9 No significant difference was found in the incidence rates of hypotension, hyperkalemia, cough, upper respiratory tract inflammation, diarrhea, back pain, nasopharyngitis, headache or influenza between the sacubitril/valsartan group and the ACEIs/ARBs group. The meta-analysis concluded that sacubitril/valsartan was associated with lower drug-risks than ACEIs/ARBs or a placebo.
These studies implied that compared with ACEIs/ARBs, sacubitril/valsartan was safe and well-tolerated in the treatment of HFrEF or HFpEF. However, the incidence of hypotension or dizziness might be high.
The Indication and Titration of Sacubitril/Valsartan
According to the 2021 ACC expert consensus, the indications for the use of sacubitril/valsartan include HFrEF (EF ≤ 40%), NYHA class II-IV HF, and administration in conjunction with a background of guideline-directed medical therapy for HF in place. 66 The starting dose and target dose of sacubitril/valsartan for the treatment of HF are 24/26 mg to 49/51 mg twice daily and 97/103 mg twice daily, respectively. 66 The tolerability of sacubitril/valsartan is assessed in 2 to 4 weeks after the starting dose. If tolerated, it is up-titrated to achieve maximally tolerated dose. 67
Conclusion
In conclusion, RCTs, meta-analyses and observational studies reported that sacubitril/valsartan had benefits in reducing the risks of mortality and hospitalization, reversing cardiac remodeling, decreasing the levels of HF biomarkers, improving the quality of life and antiarrhythmia, improving renal dysfunction and regulating metabolism in HFrEF patients. However, the benefits of sacubitril/valsartan in HFpEF patients are few. Although studies on HFmrEF are limited, they imply that sacubitril/valsartan is likely to have benefit to HFmrEF patients. Moreover, evidence from RCTs, meta-analyses and observational studies indicated that sacubitril/valsartan was safe and well-tolerated in HFrEF or HFpEF patients, compared with ACEIs/ARBs.
Supplemental Material
Supplemental Material, sj-rar-1-cpt-10.1177_10742484211058681 - The Efficacy and Safety of Sacubitril/Valsartan in Heart Failure Patients: A Review
Supplemental Material, sj-rar-1-cpt-10.1177_10742484211058681 for The Efficacy and Safety of Sacubitril/Valsartan in Heart Failure Patients: A Review by Rui Zhang, Xiaotong Sun, Ya Li, Wenzheng He, Hongguang Zhu, Baoshan Liu and Aiyuan Zhang in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Author Contributions
Aiyuan Zhang, Baoshan Liu, and Rui Zhang participated in the conception, design, writing, and critical revision of the manuscript for important intellectual content and final approval of the manuscript submitted. Xiaotong Sun, Ya Li, Wenzheng He, and Hongguang Zhu participated in the writing and critical revision of the manuscript for important intellectual content and final approval of the manuscript submitted.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Shandong Province Natural Science Foundation (ZR2020QH005, ZR2021QH262).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
