Abstract
Over the past decade, soluble guanylate cyclase (sGC) activators and stimulators have been developed and studied to improve outcomes in patients with heart failure with reduced ejection fraction (HFrEF). The sGC enzyme plays an important role in the nitric oxide (NO)-sGC-cyclic guanosine monophosphate (cGMP) pathway, that has been largely untargeted by current guideline directed medical therapy (GDMT) for HFrEF. Disruption of the NO-sCG-cGMP pathway can be widely observed in patients with HFrEF leading to endothelial dysfunction. The disruption is caused by an oxidized state resulting in low bioavailability of NO and cGMP. The increase in reactive oxygen species can also result in an oxidized, and subsequently heme free, sGC enzyme that NO is unable to activate, furthering the endothelial dysfunction. The novel sGC stimulators enhance the sensitivity of sGC to NO, and independently stimulate sGC, while the sGC activators target the oxidized and heme free sGC to stimulate cGMP production. This review will discuss the pathophysiologic basis for sGC stimulator and activator use in HFrEF, review the pre-clinical and clinical data, and propose a place in the HFrEF armamentarium for this novel pharmacotherapeutic class.
Keywords
Introduction
Despite advances in guideline directed medical therapy (GDMT), heart failure (HF) remains a significant burden on the US healthcare system with 6.2 million aged 20 or older reporting the diagnosis in 2016. 1 That same year, HF comprised over 800,000 US hospital discharges. 1 More recently in the EMPEROR-Reduced trial, patients on quadruple backbone GDMT still had 13.2 HF hospitalizations (HFH) per 100 patient years. 2 One pathway not targeted effectively with current therapies is the nitric oxide (NO)-soluble guanylate cyclase (sGC)-cyclic guanosine monophosphate (cGMP) pathway. 3 Two novel classes of medications, sGC stimulators and sGC activators, present further opportunities to optimize HF with a reduced ejection fraction (HFrEF) medical therapy. In this paper, the pathophysiologic basis for use of sGC stimulators and activators will be reviewed, along with a summary of clinical data and a proposed place in therapy for these agents in patients with HFrEF. Although dysfunction in the NO-sGC-cGMP pathway can adversely affect both patients with HFrEF and HF with preserved ejection fraction (HFpEF), current data do not support their use in the latter population. 4
Pathophysiology of the NO-sGC-cGMP Pathway in HF
The NO-sGC-cGMP pathway plays an important role in both endothelial and myocardial function. In functional endothelium, endothelial nitric oxide synthase (eNOS) is stimulated by shear stress from laminar flow to convert L-arginine to NO. 5,6 NO then distributes into various tissues and stimulates sGC to produce cGMP, an important secondary messenger in normal cardiac and vascular function. In smooth muscle tissue, the increase in cGMP leads to an increase in cGMP-dependent protein kinase G (PKG), resulting in a decrease of intracellular calcium. 6,7 This reduction in calcium decreases cross-bridging of actin and myosin due to the decreased phosphorylation of myosin, resulting in smooth muscle relaxation. 6,8 In cardiac tissue, cGMP plays a concentration-dependent role in myocardial contractility. 9,10 Ventricular relaxation and remodeling have also been reported to be modified by sGC expression. 11,12 NO donors positively affect ventricular relaxation and improve diastolic distensibility, allowing for improved coronary perfusion with prolonged diastolic intervals. 13 Other downstream effects of cGMP include inhibition of smooth muscle proliferation, leukocyte recruitment, and platelet aggregation. 14,15
Oxidative stress has been shown to induce cardiovascular disease pathology, including HF. 16 Increased oxidative stress can affect the NO-sGC-cGMP pathway in numerous ways (see Figure 1). NO rapidly reacts with superoxide and other reactive oxygen species (ROS) to produce peroxynitrite, 17 decreasing the NO available to activate sGC. Increased levels of peroxynitrite can also mediate oxidation of tetrahydrobiopterin (BH4). In the absence of adequate levels of BH4, eNOS will undergo uncoupling and convert molecular oxygen to superoxide rather than L-arginine to NO. 17 Finally, the binding of NO to sGC depends on the state of the sGC receptor. NO binds with the Fe2+ of the heme group located on sGC, causing a conformational change to increase the conversion of guanosine triphosphate to cGMP. 18 If the Fe2+ is oxidized to Fe3+ and subsequently loses the heme moiety, NO is unable to induce activation of the enzyme. 19 Peroxynitrite and other ROS can play a role in reducing the iron to its ferrous state and render the sGC inactive. 17,20 These mechanisms lead to decreases in the production and bioavailability of NO as well as the production of cGMP from inactive sGC, resulting in endothelial dysfunction.

The effect of oxidative stress on the NO-sGC-cGMP pathway. Oxidative stress leads to increased levels of reactive oxygen species including superoxide (O2-). Superoxide reacts with NO to produce peroxynitrite (ONOO-). Increased levels of peroxynitrite can lead to eNOS uncoupling further increasing O2- concentration. Peroxynitrite can also oxidize the NO-sensitive sGC with a Fe2+ moiety to the NO-insensitive with a Fe3+ moiety. The sGC stimulators can independently activate sGC along with also having a synergistic effect with endogenous NO in activating sGC. The sGC activators can activate both the oxidized sGC and heme free sGC to produce cGMP. L-Arg indicates L-arginine; eNOS, endothelial nitric oxide synthase; NO, nitric oxide; O2, molecular oxygen; O2-, superoxide; ONOO-, peroxynitrite; sGC, soluble guanylate cyclase; GTP, guanosine triphosphate; cGMP, cyclic guanosine monophosphate.
HF pathology itself can also induce endothelial dysfunction. Increased levels of tumor necrosis factor-α (TNF-α), a common finding in HF, has been identified as a potential factor in downregulation of eNOS. 21 Decreased NO production may also contribute to reduced exercise capacity as NO contributes significantly to limb blood flow during exercise. 22 Decreased exercise capacity can lead to a sedentary lifestyle, which can further increase levels of ROS and decrease activity of eNOS due to decreased shear stress from lower heart rates. 23 -25 The resultant endothelial dysfunction can add to increased left and right ventricular afterload and dysregulation of both renal and coronary circulation. 23
Endothelial dysfunction is a significant independent contributor to mortality, cardiovascular events, and morbidity in patients with HF. 26,27 Common comorbid disease states such as diabetes, hyperlipidemia, and hypertension can also add to endothelial dysfunction. 28 The associated poor outcomes make it an attractive target for pharmacological therapy in this population. Studies have been conducted on various parts of the NO-sGC-cGMP pathway in HF. The NO donor isosorbide dinitrate reduced mortality in black patients when added to standard therapy in HFrEF, 29 however it requires multiple times a day dosing and concomitant use of hydralazine to prevent nitrate tolerance. 30 No large randomized trials have assessed the efficacy of phosphodiesterase-5 (PDE5) inhibitors in patients with HFrEF, but the low bioavailability of cGMP in endothelial dysfunction may limit the efficacy of PDE5 inhibitors. However, a meta-analysis did find improvement in clinical outcomes for patients with HFrEF that utilized PDE5 inhibitors. 31
Two novel classes of medications, sGC stimulators and sGC activators, have become an attractive target for HF pharmacotherapy. The sGC activators, such as cinaciguat, can activate both the oxidized (Fe3+) and heme-free sGC to produce cGMP by mimicking the heme group. 18,32 This allows for the production of cGMP even in an environment under oxidative stress. Additionally, sGC activators may also prevent the degradation of sGC and increase sGC protein levels. 32 On the other hand, sGC stimulators, such as riociguat and vericiguat, require a reduced heme (Fe2+) moiety to stimulate cGMP production independent of NO. 18 sGC stimulators have also been shown to have a synergistic effect with endogenous NO by stabilizing sGC in its active configuration. 18,33
Clinical Data in HfrEF
A summary of the clinical trials evaluating sGC stimulators and activators can be found in Table 1.
Evidence From Clinical Trials of sGC Activators and Stimulators.
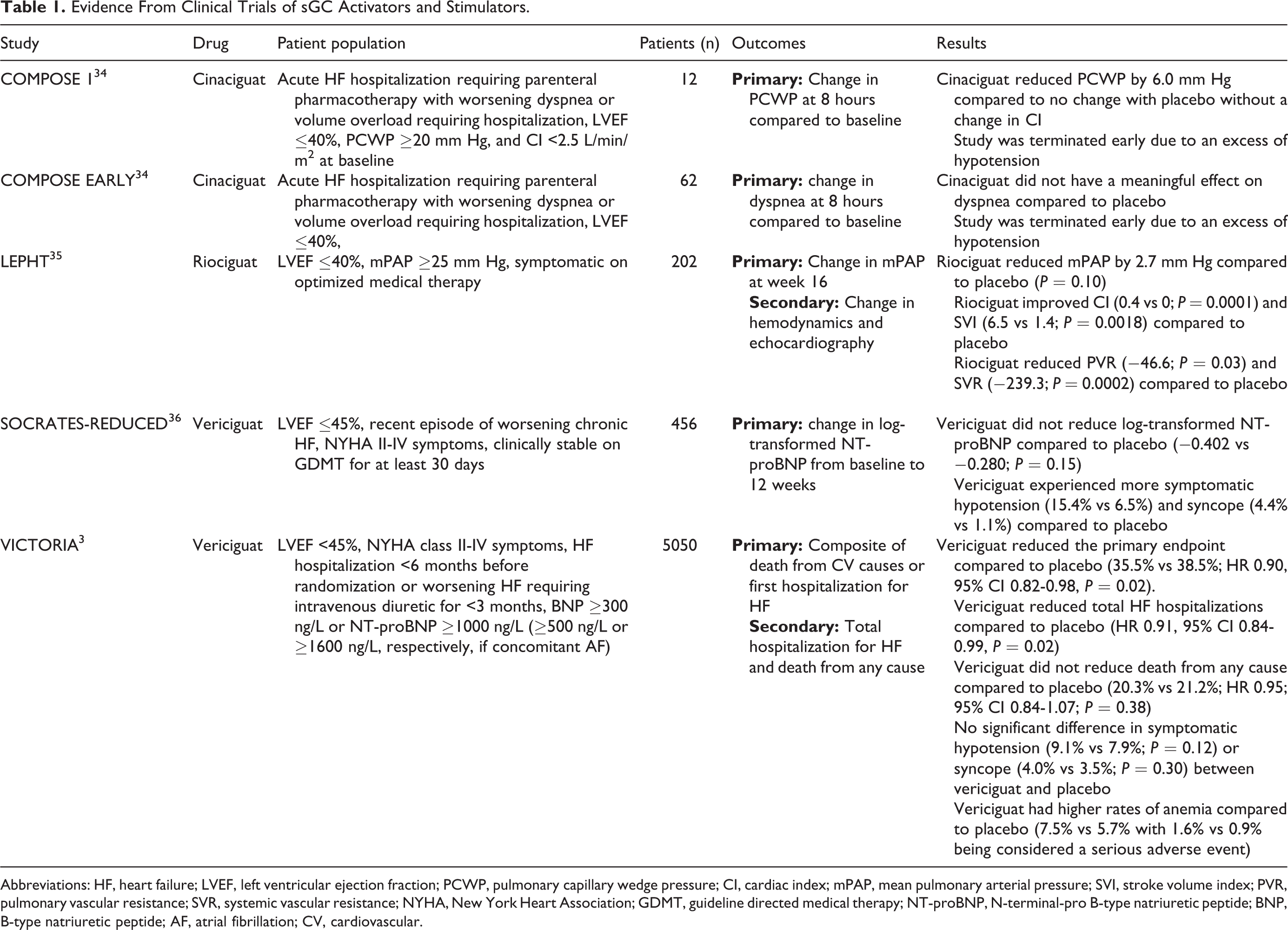
Abbreviations: HF, heart failure; LVEF, left ventricular ejection fraction; PCWP, pulmonary capillary wedge pressure; CI, cardiac index; mPAP, mean pulmonary arterial pressure; SVI, stroke volume index; PVR, pulmonary vascular resistance; SVR, systemic vascular resistance; NYHA, New York Heart Association; GDMT, guideline directed medical therapy; NT-proBNP, N-terminal-pro B-type natriuretic peptide; BNP, B-type natriuretic peptide; AF, atrial fibrillation; CV, cardiovascular.
The sGC activator cinaciguat, administered as an intravenous (IV) infusion, showed initial promise in improving hemodynamics in acute decompensated heart failure (ADHF). 37 This nonrandomized, uncontrolled, unblinded, phase II study included an initial dose-finding phase followed by proof-of-concept (POC) to evaluate the safety and hemodynamic efficacy of cinaciguat given as a 6-hour infusion in 60 patients (27 dose-finding, 33 POC) admitted with ADHF in whom invasive hemodynamic monitoring was indicated. Doses were titrated and ranged from 50 to 400 mcg/h, with an initial dose of 100 mcg/h selected for POC based upon pre-defined improvements in pulmonary capillary wedge pressure (PCWP). The primary outcome of change in PCWP from baseline to the end of the 6-hour infusion was significantly improved from baseline to the end of infusion (mean −7.9 mm Hg ± 3.0, P < 0.0001), and a variety of other hemodynamic parameters were also improved including reductions in systemic and pulmonary vascular resistance, and increases in cardiac output, with no significant change in heart rate (HR), renal function, N-terminal pro-brain natriuretic peptide (NT-proBNP), or plasma renin activity. Hemodynamics trended back toward baseline at the 2-hour post-infusion assessment. Hypotension was the most frequent adverse event (AE), reported in 10% of the overall population.
The COMPOSE program was subsequently developed based upon these data together with unpublished placebo-controlled data in 139 patients showing improvements in PCWP with higher incidence of hypotension at doses ≥ 200 mcg/h. 34 These 3 phase IIb studies intended to evaluate the effects of fixed-dose cinaciguat at doses < 200 mcg/h for 24-48 hours in patients with HFrEF (EF ≤ 40%) requiring hospitalization for acute decompensation. Due to concerns of confounding of dyspnea assessment when invasive hemodynamics are known, separate evaluations of hemodynamic (COMPOSE 1 and 2) and dyspnea (COMPOSE EARLY) outcomes were developed. The primary hemodynamic endpoint was change in PCWP at 8 hours, evaluated at different fixed doses in COMPOSE 1 (placebo, cinaciguat 50, 100, or 150 mcg/h) and COMPOSE 2 (placebo, cinaciguat 10, or 25 mcg/h). In COMPOSE EARLY the primary outcome was change in dyspnea at 8 hours from baseline based on visual analog scale with the same dosing groups as used in COMPOSE 1.
All 3 trials were terminated early: COMPOSE 1 [intention to treat (ITT) population n = 12] and COMPOSE EARLY (ITT population n = 62) due to an excess of hypotension with cinaciguat; and COMPOSE 2 (ITT population n = 4) due to futility due to slow enrollment and unclear risk: benefit of the lower dosing strata. A summary of descriptive data showed that cinaciguat was associated with numerically lower PCWP (mean −6.0 mm Hg vs no change) but no difference in cardiac index in COMPOSE 1. Cinaciguat was also associated with non-dose-related reductions in systolic blood pressure (SBP) (−15.3, −14, and −12.3 mm Hg in the 50, 100, and 150 mcg/h groups, respectively, vs −3.3 mm Hg with placebo). While there were no differences in treatment emergent AE in this trial, there was a higher incidence of hypotension and one death in the 150 mcg/h dose. COMPOSE EARLY showed a lack of severe dyspnea at baseline with improvements prior to cinaciguat initiation with standard therapy alone and no improvements from baseline after treatment. Hypotension was also significant (27.9% vs 5.3% placebo), resulting in study withdrawal of 11 patients in the cinaciguat group. Based upon these descriptive data showing a lack of clear symptomatic or hemodynamic benefit, coupled with the significant hypotensive effects, the authors concluded that it was unlikely for cinaciguat to provide benefit in further studies.
The first experience with oral sGC stimulators in HFrEF came from the Left Ventricular Systolic Dysfunction Associated with Pulmonary Hypertension Riociguat Trial (LEPHT). 35 This phase IIb dose-finding study investigated the effects of various doses of riociguat compared to placebo over 16 weeks on mean pulmonary artery pressure (mPAP) in 201 patients with pulmonary hypertension secondary to left ventricular dysfunction. In the 160 patients with evaluable repeat hemodynamics at 16 weeks, riociguat was not associated with improvement in placebo-corrected change in mPAP regardless of target dose. Several secondary outcomes were improved with riociguat at the highest target dose (2 mg three times daily) including cardiac index, systemic vascular resistance, and Minnesota Living With Heart Failure Questionnaire. AE of concern were dizziness and hypotension, with serious AE including syncope, hypotension, and cardiac failure in the highest dose group, albeit in low numbers. Based upon these findings and the relative safety, further investigation targeting this pathway was explored in the HFrEF population. 38
Vericiguat’s role in the treatment of worsening heart failure (WHF) was first investigated in the parallel SOluble guanylate Cyclase stimulatoR in heArT failurE Studies (SOCRATES) trials. 36 The trials were designed to evaluate the effects of vericiguat on biomarkers after 12 weeks of treatment in patients with WHF requiring recent intervention. SOCRATES-REDUCED was a randomized, double-blind, placebo controlled, dose finding study that enrolled patients with a left ventricular ejection fraction (LVEF) less than 45%, New York Heart Association (NYHA) class II-IV symptoms, and deemed clinically stable after recent WHF despite receipt of GDMT for at least 30 days. WHF was defined as requirement for hospitalization or need for IV diuretic within 4 weeks in the outpatient setting. Natriuretic peptide cut-offs were determined based upon presence or absence of atrial fibrillation (AF) [normal sinus rhythm (NSR) BNP ≥ 300 pg/mL or NT-proBNP ≥ 1000 pg/mL; AF BNP ≥ 500 pg/mL or NT-proBNP ≥ 1600 pg/mL]. Clinical stability was determined based on lack of need for IV vasodilator therapy for more than 24 hours and IV diuretic therapy for more than 12 hours. Key exclusion included hypo- or hypertension (SBP < 110 or > 160 mm Hg), brady- or tachycardia [HR < 50 or ≥ 100 bpm], acute coronary syndrome or cardiac resynchronization therapy defibrillator placement within 60 days, concomitant nitrate or PDE5 inhibitor use, and hepatic (Child-Pugh B or C) or renal impairment [estimated glomerular filtration rate (eGFR) < 30 mL/min/1.73m2 ]. Patients were randomized equally to placebo or one of four vericiguat groups based on target dose (1.25, 2.5, 5, or 10 mg) within 4 weeks of discharge. Sample size was calculated based on an assumed treatment effect of −0.187 log (pg/mL) for NT-proBNP in the pooled vericiguat group including the 3 highest doses, which was prespecified to be compared to placebo for all endpoints.
A total of 456 patients were randomized a mean of 14.4 days after clinical stabilization and final analysis for efficacy included 351 patients who received study drug per protocol with evaluable 12-week NT-proBNP levels. The groups were relatively well matched with mean age of 68 years, 80% male, and notably less than 4% black. Median baseline NT-proBNP was 3076 pg/mL in the entire study population and was higher in the placebo and lowest dose vericiguat groups. The majority of patients had been hospitalized for WHF (78%) and were predominantly NYHA functional class I or II (53%). Use of GDMT was reasonable with 90% on a beta blocker, 61% angiotensin-converting enzyme inhibitor (ACEi), 23% angiotensin receptor blocker (ARB), and 62% mineralocorticoid receptor antagonist (MRA). Given the timing of the trial there was no use of neprilysin inhibition (ARNI) and no data on sodium-glucose co-transporter-2 (SGLT2) inhibitor use. Blood pressure was robust [mean SBP 126 mm Hg, diastolic BP (DBP) 75 mm Hg], with a high prevalence of concomitant AF (34%), diabetes mellitus (48%), hypertension (78%), and chronic kidney disease (41%).
The primary outcome was the change in log-transformed NT-proBNP level from baseline to 12 weeks in the pre-specified per-protocol pooled vericiguat group (2.5, 5, and 10 mg doses) compared to placebo. The 1.25 mg dose was excluded due to presumed lack of effect. There was no difference in the pooled vericiguat group versus placebo (mean difference −0.402 vs −0.280; 90% CI −0.32 to 0.07, P = 0.15). A number of exploratory secondary outcomes were also evaluated and suggested a dose-response relationship for the primary outcome with increasing dose of vericiguat and, notably, with the 10 mg dose versus placebo in pairwise comparison (mean difference −0.250; 90% CI −0.50 to 0.00, P = 0.048). This was not seen with other dose groups.
Safety outcomes were evaluated in the modified ITT population of 455 patients. Vericiguat was fairly well tolerated without significant differences in AE, including those leading to discontinuation, or the development of acute kidney injury. The pooled vericiguat group did, however, experience more symptomatic hypotension (15.4% vs 6.5%) and syncope (4.4% vs 1.1%). These AE tended to emerge during the first 2 weeks of therapy in the vericiguat group when patients would have been receiving 2.5 mg based upon the titration schedule.
The authors concluded that, despite a significant improvement in NT-proBNP, given the potential dose-response and overall tolerability vericiguat warranted additional study in a phase 3 trial to determine its potential role in patients with HFrEF and recent WHF.
Based upon these early phase data, the VICTORIA trial was designed to evaluate the effects of vericiguat on clinical outcomes in a high-risk HFrEF population. 39,40 This multinational, double-bind, event driven trial enrolled adult patients with a LVEF less than 45%, NYHA class II-IV symptoms, and evidence of WHF with elevated natriuretic peptides despite receipt of GDMT. WHF was defined as requirement for intervention within the preceding 6 months and divided into 3 cohorts: hospitalization within the past 3 months, hospitalization within the past 3-6 months, or need for IV diuretic within 3 months in the outpatient setting. Enrollment of patients with more remote hospitalization was capped at 20% of the overall population. Natriuretic peptide cut-offs were determined based upon presence or absence of AF (NSR: BNP ≥ 300 pg/mL or proBNP ≥1000 pg/mL; AF: BNP ≥500 pg/mL or NT-proBNP ≥1600 pg/mL). Key exclusion included hypotension (SBP < 100 mm Hg), concomitant nitrate or PDE5 inhibitor use, use of IV inotropes within 24 hours, and severe renal impairment based on eGFR (< 15 mL/min/1.73m2). Patients were randomized to receive vericiguat 2.5 mg titrated to 10 mg once daily or placebo after a 30-day screening period and were followed until the prespecified number of cardiovascular deaths occurred. Sample size was calculated based on an expected 1-year cardiovascular mortality rate of 11% and assumed 20% reduction in the intervention group.
A total of 5050 patients were randomized and the final analysis for efficacy included 2526 patients who received vericiguat (median achieved dose 9.2 mg) and 2524 placebo. The groups were well matched with mean age of 67 years, 76% male, and notably less than 5% black. The majority of patients had been hospitalized within 3 months of enrollment (67%) with nearly equal distribution in the remaining cohorts and were predominantly NYHA functional class II (59%) or III (40%). Use of GDMT from the 3 major classes (beta blockade, ACEi/ARB/ARNI, and MRA) was present in 60% patients, with only 15% receiving ARNI and no data on SGLT-2 inhibitor use. Blood pressure was robust (mean SBP 121 mm Hg, DBP 73 mm Hg), with a high prevalence of concomitant diabetes (47%), coronary artery disease (58%), and hypertension (79%).
Vericiguat use over the median follow-up of 10.8 months was associated with a 3% absolute reduction in the primary composite outcome of time to cardiovascular death or first HFH (35.5% vs 38.5% with placebo; HR 0.90, 95% CI 0.82-0.98, P = 0.02). This effect was driven by a reduction in HFH (27.4% vs. 29.6%) and appeared to become apparent within 3 to 4 months of randomization. A number of secondary outcomes were also evaluated, demonstrating vericiguat’s impact on total HFH (HR 0.91, 95% CI 0.84-0.99, P = 0.02) with no significant improvement in cardiovascular mortality (HR 0.93, 95% CI 0.81-1.06). These results were consistent across a number of prespecified subgroups outside of patients 75 years of age or older, with baseline eGFR of 30 mL/min/1.73 m2, in the highest quartile of baseline NT-proBNP, or LVEF 40%-45%.
Vericiguat was fairly well tolerated without significant differences in AE defined as serious (34.8% vs 32.8%) or overall AE (80.5% vs 81%). Based upon concerns raised in the SOCRATES-REDUCED trial symptomatic hypotension and syncope were predetermined to be AE of interest and were found to occur numerically but not statistically more with vericiguat (9.1% vs 7.9%, P = 0.12 and 4% vs 3.5%, P = 0.30, respectively). Other notable differences included higher rates of anemia and GI side effects.
The authors concluded that vericiguat was associated with a reduction in time to cardiovascular death or HFH in comparison with placebo in a high-risk HFrEF population.
There are several important limitations to consider. First, background GDMT with triple therapy including beta blockade, ACEi/ARB/ARNI and MRA was relatively low at just under 60% with low ARNI utilization and lack of data on SGLT2 inhibitor use to determine incremental benefit of vericiguat. Additionally, those patients with the highest NT-proBNP, older age, and severe renal impairment did not appear to benefit. Benefit did appear to be driven by HFH, which differs from data with other more novel HFrEF therapies 2,41,42 and may align more with ivabradine and digoxin, although cross trial comparisons must be undertaken with careful consideration for varying baseline characteristics. The authors proposed that the lack of mortality benefit may be related to the higher than anticipated event rate resulting in a shorter follow-up.
A subsequent analysis of outcomes in relation to baseline NT-proBNP as a continuous variable provided additional insights into the benefits of vericiguat. 43 After adjustment for the Meta-Analysis Global Group in Chronic Heart Failure (MAGGIC) risk score, patients in the lowest (≤4000 pg/mL) group derived the most benefit from vericiguat for both the primary composite (HR 0.77, 95% CI 0.68-0.88) as well as individual components of HFH (HR 0.78, 95% CI 0.67-0.90) and cardiovascular death (HR 0.75, 95% CI 0.60-0.94). Benefit was maintained, albeit diminished, when including patients with higher baseline NT-proBNP (≤8000 pg/mL), but those with the highest levels (>8000 pg/mL) did not derive benefit. The increased severity of illness in this latter population could have resulted in lack of benefit with vericiguat and should be considered during treatment selection.
There are a number of supplementary studies planned for the VICTORIA population that may help to further define the optimal patient population who may benefit from treatment. 40 These include a pharmacokinetic analysis, evaluation of a variety of biomarkers, a genome-wide association study, and a number of imaging modalities. Additionally, the North American cohort will undergo both an economic and quality-of-life evaluation.
Place in Therapy
The 2021 American College of Cardiology/American Heart Association/Heart Failure Society of America Expert Decision Pathway for HFrEF Treatment Optimization now recommends quadruple backbone therapy for patients with HFrEF, which includes an ACEi/ARB/ARNI, MRA, beta-blocker, and SGLT2 inhibitor. 44 Vericiguat is only briefly mentioned, but with no formal recommendation for use. Given the recency of the VICTORIA-HF data and the latest European heart failure guideline release in 2016, there is also no formal recommendation from the European Society of Cardiology. Vericiguat was officially approved for use in the United States by the Food and Drug Administration in January of 2021. The official labeling recommends vericiguat may be considered for use in patients with HFrEF that are persistently symptomatic despite optimization of other heart failure therapies. The recommended starting dose is 2.5 mg orally once daily with food, with the goal of titrating in 2-week intervals to a maximum dose of 10 mg once daily. The package labeling also gives recommendations for decreasing (if dose is currently at 5 or 10 mg) or interrupting therapy (if dose is at 2.5 mg) if the patient’s SBP is < 90 mm Hg. Vericiguat should be avoided in patients currently maintained on long-acting nitrates or PDE5 inhibitors, however, patients utilizing short-acting nitrates were included in VICTORIA-HF. 39,45
The role of vericiguat in the management of patients with HFrEF moving forward will likely be limited. Clinicians will need to discern its place in therapy given the more recent, positive data with SGLT-2 inhibitors and the fact that nearly none of the VICTORIA-HF population was simultaneously receiving this now GDMT. 2,41 Moreover, only approximately 15% of the VICTORIA-HF population was on ARNi therapy, begging the question of how impactful vericiguat would have been if patients were ultimately maintained on quadruple backbone therapy. With the current struggles initiating and titrating GDMT based on registry data, adding an additional medication with questionable benefits to the clinical armamentarium seemingly will complicate polypharmacy, and increase pill burden and cost for patients. 46
Given that sGC stimulators offer a unique mechanism compared to existing HFrEF therapies and the VICTORIA-HF population was considered to be more acutely ill based on required enrollment BNP levels, recent hospitalization, and/or the need for recent IV diuretic administration, there may be a potential role for vericiguat in these patients that are either already maximized on GDMT or with pre-existing preclusions to their use. 39 Though differing mechanisms of action, digoxin or ivabradine may be viewed as more palatable options for reducing HFH given their overall neutral effect on blood pressure, the most common side effect of vericiguat, which can ultimately inhibit the up-titration of concomitant GDMT. While not FDA approved yet, omecamtiv mecarbil, a cardiac myosin activator studied in the GALACTIC-HF trial, will also add an additional morbidity reducing option to currently available HFrEF therapies. 47 Vericiguat may potentially be a preferred agent in patients with PH secondary to HFrEF, though data is currently lacking, and no studies are in progress. While the overall use case for vericiguat seems minimal, the advent of machine learning, artificial intelligence, and neural networks may aid clinicians in determining which patients would potentially benefit the most from therapy. Parallel to this thought, precision medicine tailored not only to the patient’s genome, but heart failure etiology, offers the potential to craft designer heart failure regimens for each unique patient.
Conclusion
The sGC stimulators and activators offer a unique mechanism of action compared to currently available HFrEF therapies. While a mortality benefit has not yet been established with these agents, the VICTORIA-HF trial demonstarted a morbidity benefit with oral vericiguat that led to it’s FDA approval in January 2021. Future studies are needed to determine potentially niche roles for vericiguat and other sGC-NO pathway agents that will ultimately aid clinicians in determining the most appropriate patient populations to deploy these agents.
Footnotes
Acknowledgments
Figure 1 was created with ![]() .
.
Author Contributions
Theodore J. Berei and Kristen T. Pogue conceived the idea for this work. All authors contributed to the literature search, data analysis, manuscript preparation, and critical revision of the work.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Theodore J. Berei is an editorial board member of the Journal of Clinical Pharmacy and Therapeutics.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
