Abstract
Policosanol (POL) is a hypocholesterolemic drug of natural origin and has been shown to reduce circulating levels of proprotein convertase subtilisin/kexin type 9 (PCSK9) in healthy participants. Recently, we have reported that POL can attenuate aortic calcification in diabetic dyslipidemic rats; however, the underlying mechanism is not fully elucidated. We aimed to investigate the effect of POL on aortic calcification and whether PCSK9 has a contributory role and also to examine whether the combination of POL with pentoxifylline (PTX) as anti–tumor necrosis factor α would offer additional benefits. Thirty adult male New Zealand rabbits weighing 1.5 to 2 kg were randomly assigned to 5 groups. One group received standard chow diet and served as normal control group (NC). The other 4 groups received 0.5% wt/wt cholesterol-rich diet for 12 weeks and concurrently treated with placebo, POL, PTX, or a combination of POL and PTX. Sera samples and aortic tissue were collected for biochemical measurements and histological assessment. Rabbits fed a cholesterol-rich diet demonstrated dyslipidemia, increased inflammatory state, and elevated serum levels of PCSK9, compared to the NC group. Aortic calcification was evident in dyslipidemic rabbits, represented by increased calcium deposition and osteopontin expression in aortic tissue, along with elevated serum levels of alkaline phosphatase and osteocalcin. Dyslipidemic rabbits showed a significant upregulation of wingless-type MMTV integration site family 3A and bone morphogenetic protein 2 genes in their aortic tissue. Policosanol significantly reduced circulating PCSK9 levels, suppressed calcification markers, and attenuated aortic calcification. Combination of POL with PTX alleviated aortic calcification to a greater extent than either monotherapy, which may be attributed to further suppression of PCSK9 and calcification markers. These findings suggested that POL exerted anticalcifying effect partly via inhibition of PCSK9. Combination of POL and PTX offered additional benefits and might represent a promising therapeutic option for aortic calcification.
Introduction
Vascular calcification is a major health problem which contributes to arterial stiffness, hypertension, and aortic valve stenosis. The latter is a common cause for aortic valve replacement worldwide. Two types of vascular calcification have been identified according to the site of calcium deposition in blood vessels. Intimal calcification is usually associated with atherosclerotic plaques, whereas medial calcification occurs mostly with diabetes, chronic kidney disease, and aging. Both types represent a leading cause of cardiovascular mortality and morbidity. 1 Vascular calcification has been believed to be a degenerative process characterized by a passive calcium deposition in the vasculature, until many lines of evidence confirm that it is a highly regulated process that mimics bone mineralization. It has been suggested that the interplay between lipid accumulation, inflammatory cytokines, and vascular cells undergoing osteogenesis can drive the intimal calcification. 2
Low-density lipoprotein cholesterol (LDL-C) has been thought to be a key determinant in the pathogenesis of intimal calcification because it was extensively detected in sites of calcification in aorta and aortic valves 3 ; however, this postulation has been challenged recently. 4,5 Low-density lipoprotein cholesterol–lowering medications including statins and ezetimibe fail to prevent the development or progression of vascular calcification in patients with hypercholesterolemia, despite effective improvement in lipid profile and cardiovascular outcomes. 6 Moreover, extracorporeal LDL removal technique, known as LDL apheresis, significantly attenuates the progression of atherosclerosis but appears to be of limited clinical benefit regarding aortic calcification in patients with familial hypercholesterolemia (FH). 7 Based on these observations, it seems that the calcification process proceeds independently of plasma total cholesterol (TC) or LDL-C levels, once an intimal injury has taken place. Alternatively, emerging evidence suggests a mutual link between LDL receptors (LDLRs) and intimal calcification. 4 Low-density lipoprotein receptors regulate plasma cholesterol levels through endocytosis of LDL particles, and their significance has been evidenced from Ldlr knockout (Ldlr−/− ) mice which display extensive aortic calcification. 8 Furthermore, mutation of Ldlr gene in patients with homozygous FH has been associated with premature aortic calcification and accelerated aortic valve stenosis, and to a lesser extent in patients with heterozygous FH. In those patients, no significant correlations were found between calcification scoring and plasma cholesterol levels. 9 Taken together, these data markedly support a potential role for LDLRs, but not LDL-C levels, in modulation of vascular calcification. Proprotein convertase subtilisin/kexin type 9 (PCSK9) is a key LDLR-degrading protein which acts through binding to hepatic and extra-hepatic LDLRs leading to endocytosis and lysosomal degradation, thus increasing circulating LDL-C levels. 10 A cross talk has been suggested between PCSK9 and vascular calcification. Transgenic mice overexpressing Pcsk9 gene that encodes the PCSK9 protein exhibit aortic calcification along with hypercholesterolemia when fed a high-fat diet, compared with their age- and diet-matched wild-type controls. 8 Therefore, inhibition of PCSK9 and the subsequent increase in LDLRs may hold the promise for vascular calcification therapy, beyond cholesterol-lowering effect. Noteworthy, PCSK9 inhibitors have been recently developed in the form of PCSK9 monoclonal antibodies, for example, evolocumab and alirocumab. These drugs are currently undergoing phase III clinical studies and appear to be promising in reducing elevated LDL-C levels in patients with hypercholesterolemia. However, there are still considerable concerns regarding their safety, tolerability, and efficacy especially during long-term use. 11
Policosanol (POL) is a safe, well-tolerated natural hypocholesterolemic drug isolated from the purified wax of sugar cane. It is composed of a mixture of long-chain primary aliphatic alcohols, particularly hexacosanol, octacosanol, and triacosanol. Several reports have demonstrated that POL can improve lipid profile through reducing hepatic cholesterol biosynthesis, raising high-density lipoprotein cholesterol (HDL-C) levels, and enhancing LDL-C clearance from the circulation. Beneficial effects of POL on platelet aggregation and LDL peroxidation have been also reported. 12 Of particular importance, POL has been shown to mildly attenuate serum PCSK9 levels in healthy participants without affecting lipid profile. 13 Recently, we have demonstrated that POL can alleviate arterial calcification in diabetic dyslipidemic rats 14 ; however, the exact molecular mechanism underlying its anticalcifying effect is still unknown. On the other hand, tumor necrosis factor α (TNF-α) is a pro-inflammatory cytokine that exerts a potential role in vascular calcification. 15 A previous study has shown that incubation of calcifying vascular cells with different concentrations of TNF-α induces an osteoblast-like phenotype coupled with a dose-dependent increase in alkaline phosphatase (ALP) activity and calcium deposition, suggesting the pro-osteogenic properties of TNF-α. 16 Therefore, inhibition of TNF-α may be a therapeutic target for aortic calcification. One of the drugs having well-established inhibitory effect on TNF-α is pentoxifylline (PTX), which is commonly used in the treatment of intermittent claudication. Experimental and clinical studies have demonstrated a beneficial effect of PTX on atherosclerosis and vascular health as a whole. 17 The present study was undertaken to investigate the effect of POL on aortic calcification in cholesterol-rich diet–fed rabbits and to examine whether the inhibition of TNF-α by PTX along with POL might offer additional benefits.
Methods and Materials
Experimental Design
Thirty adult male New Zealand white rabbits, weighing 1.5 to 2 kg, obtained from Faculty of Agriculture (Zagazig University, Egypt), were acclimatized for 1 week in the animal facility of Faculty of Pharmacy, Zagazig University at standard environmental conditions with free access to standard chow and tap water. Thereafter, rabbits were randomly assigned to 5 groups (n = 6/group) as follows: Normal control group (NC): Rabbits received standard chow diet.
Placebo group: Rabbits received 0.5% wt/wt cholesterol-rich diet (El-Gomhouria Company, Egypt) daily for 12 weeks and simultaneously treated with placebo. 18
POL group: Rabbits received 0.5% wt/wt cholesterol-rich diet and simultaneously treated with POL (Organika Health Products Inc., Richmond, British Columbia, Canada) in a dose level 5 mg/kg body weight/day for 12 weeks. 19
PTX group: Rabbits received 0.5% wt/wt cholesterol-rich diet and simultaneously treated with PTX (Trental tablets 400 mg; Sanofi Aventis, Laval, Quebec , Canada) in a dose level 40 mg/kg body weight/day for 12 weeks. 20
POL + PTX group: Rabbits received 0.5% wt/wt cholesterol-rich diet and simultaneously treated with a combination of PC and PTX for 12 weeks.
All experimental protocols were performed in accordance with the National Institutes of Health (NIH) Guidelines for handling of laboratory animals and approved by the animal care committee of Faculty of Pharmacy, Zagazig University.
Blood and Tissue Sampling
At the end of experimental treatments, blood samples were collected from a marginal ear vein of rabbits after overnight fasting. Serum was prepared from blood by direct centrifugation and processed for determination of lipid profile, PCSK9, ALP, and osteocalcin (OCN) levels. Rabbits were killed and aortas were harvested, washed in ice-cooled normal saline, and divided into 2 parts. One part was snap-frozen in liquid nitrogen and stored at −80°C for biochemical analyses. The second part was placed in 10% neutral-buffered formalin at 4°C for 72 hours and processed for histological analyses of calcium deposition and osteopontin (OPN) immunohistochemistry.
Measurement of Serum Lipids
Total cholesterol (TC), triglycerides (TG), and HDL-C were estimated in serum using the commercial kits (Spinreact Co., Sant Esteve de Bas, Girona, Spain), according to the manufacturers’ instructions. Friedewald equation was applied to calculate LDL-C levels, where LDL-C = TC − [HDL-C + TG/5]. 21
Determination of Serum PCSK9 and Aortic TNF-α Levels
Serum PCSK9 and aortic TNF-α levels were determined by rabbit enzyme-linked immunosorbent assay (ELISA) kit (MyBiosource Inc., San Diego, California, USA) following the manufacturer’s protocols.
Assessment of Bone Morphogenetic Protein 2 and Wingless-Type MMTV Integration Site Family 3A Gene Expressions by reverse transcription polymerase chain reaction (RT-PCR)
Total RNA was isolated from aortic tissue by tissue extraction kit (Qiagen, Hilden, Germany). Total RNA was then converted into complementary DNA (cDNA) by high-capacity reverse transcription kit (Fermentas, Waltham, Massachusetts, USA). Complementary DNA was amplified by reverse transcription polymerase chain reaction (RT-PCR) kit (Stratagene, La Jolla, San Diego, California, USA), then quantified by using an Applied Biosystem with software version 3.1 (StepOne, Foster city, USA). Relative messenger RNA (mRNA) expression was calculated by cycle threshold method (2−ΔΔCt). 22 Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) mRNA was used as an internal control for all experiments. The sequences of primers for target genes were as follows: bone morphogenetic protein 2 (BMP-2): forward, 5′-ATTAGCAGGTCTTTGCACCAA-3′, reverse, 5′-TTATTTTGCTGTGCTAACGAC-3′; wingless-type MMTV integration site family 3A (Wnt3a): forward, 5′-ATTGAATTTGGAGGAATGT-3′, reverse, 5′-CTTGAAGTACGTGTAACG-3′; GAPDH: forward, 5′-GTGAAGGTCGGAGTCAAG-3′, reverse, 5′-GAGATGATGACCCTTTTGC-3′.
Biochemical Analysis of Calcification Markers
Serum OCN levels were measured by rabbit ELISA kits (Lifespan Biosciences, Inc., Seattle, WA). Serum activity of ALP was measured colorimetrically using the commercially available kit (Vitro Medical Co., Hannover, Germany).
Immunohistochemical Analysis of OPN Expression in Aortic Tissue
Osteopontin expression was evaluated by immunohistochemistry, as described before. 23 Aortic sections were dewaxed with xylene and rehydrated with phosphate buffered saline (PBS), then boiled in 10 mM citrate buffer (pH = 6) for 15 minutes for retrieval of antigen. Thereafter, sections were incubated for 30 minutes at room temperature with 3% hydrogen peroxide diluted in methanol for inhibition of endogenous peroxidase. To reduce nonspecific background staining, slides were incubated in a solution containing PBS, 2% bovine serum albumin (BSA), and 0.01% Triton for 1 hour at room temperature. Slides were incubated with anti-OPN rabbit polyclonal antibody (RB-9097-P0; Thermo Fisher Scientific, Massachusetts) diluted in 1:50 10 mM PBS, 0.2% BSA and 15 mM sodium azide, overnight at 4°C. After several washes in PBS, a biotinylated antirabbit secondary antibody was added to the slides for 30 minutes at room temperature. Slides were incubated with the avidin–biotin–horseradish peroxidase (HRP) complex for 1 hour, and the reaction was developed with diaminobenzidine reagent (Santa Cruz Biotechnology, Dallas Texas, USA), according to the supplier’s instructions. Negative controls consisted of tissue sections processed in the same manner, but incubated with normal rabbit serum. Images were taken at 40× magnification using a digital camera (AmScope 5.0 MP Microscope, Irvine, California, USA). Expression of OPN in aortic tissue was determined as the mean area percentage of OPN-immunopositive cells (dark brownish in color). Three aortic sections from 3 different rabbits were used in each group. Five high-power microscopic fields were randomly selected, and an average value was taken for each animal, using customized automated image processing software (ImageJ, version 1.47d; NIH, Bethesda, Maryland, USA).
Histological Assessment of Calcium Deposition in Aortic Tissue
Following dehydration with ascending grades of ethanol (75%-100%), aortic tissue specimens were embedded in paraffin and sliced into 2-µm cross sections using a microtome (Leica RM 2155, England, United Kingdom). Specimens were stained thereafter with Alizarin red solution to examine calcium deposition, which appeared as orange-red deposits under light microscopy. 24 Calcium deposition was quantified using a scoring system, giving 0 = no calcium deposits; 1 = 10% to 20% of aortic wall contained calcium deposits; 2 = 30% to 40% of aortic wall contained calcium deposits, while 3 = more than 50% of aortic wall contained calcium deposits. The investigation was performed by one researcher who was blinded to different groups’ assignment. Three aortic sections were used from 3 different rabbits from each group. At least 5 microscopic fields were used and averaged for each animal.
Statistical Analysis
Results were statistically analyzed by Prism 5 GraphPad (San Diego, California, USA) and expressed as mean ± (standard deviation). One-way analysis of variance followed by Tukey-Kramer test was performed to examine the significance of differences between groups. Correlations between PCSK9 and studied parameters were performed by nonparametric Pearson correlation analysis. P < .05 was considered statistically significant.
Results
Serum Lipid Profile
Parameters of lipid profile are summarized in Table 1. A marked dyslipidemia was evident in cholesterol-rich diet–fed rabbits (placebo group) after 12 weeks, represented by significant increases in serum levels of TG, TC, LDL-C, and TC/HDL-C ratio, whereas there was a significant decrease in serum HDL-C levels compared to the NC group. On the other hand, concurrent treatment of cholesterol-rich diet–fed rabbits by either POL or PTX resulted in a marked improvement in lipid profile, while the effect of POL on TC, HDL-C, and LDL-C levels was more superior to PTX. Combination of both drugs modulated TG and HDL-C levels greater than either monotherapy.
Serum Lipids.a

Abbreviations: NC, normal control rabbits fed standard chow diet; Placebo: 0.5% wt/wt cholesterol-rich diet fed–rabbits treated concurrently with placebo; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; POL, cholesterol-rich diet–fed rabbits treated concurrently with policosanol (5 mg/kg body weight/day) for 12 weeks; PTX, cholesterol-rich diet–fed rabbits treated concurrently with pentoxifylline (40 mg/kg body weight/day) for 12 weeks; POL + PTX, cholesterol-rich diet–fed rabbits treated concurrently with policosanol and pentoxifylline for 12 weeks; TG, triglycerides; TC, total cholesterol.
a Data are represented as mean ± (standard deviation), n = 6/group.
Ψ P < .001 versus NC.
β P < .001 versus placebo.
∞ P < .01, P < .001 versus POL.
¥ P < .001 versus PTX.
Serum PCSK9 Level
As shown in Figure 1A, serum PCSK9 levels dramatically increased in dyslipidemic rabbits by 80-fold when compared to the NC group. Concurrent treatment with POL and PTX either individually or in a combined form significantly lowered serum PCSK9 levels. On the other hand, the combined therapy (POL + PTX) showed more significant decrease than either individual treatment. Serum PCSK9 (A) and aortic TNF-α (B) levels in experimental rabbits. NC indicates normal control rabbits fed standard chow diet; Placebo: 0.5% wt/wt cholesterol-rich diet–fed rabbits concurrently treated with placebo; POL: cholesterol-rich diet–fed rabbits treated concurrently with policosanol (5 mg/kg body weight/day) for 12 weeks; PTX: cholesterol-rich diet–fed rabbits treated concurrently with pentoxifylline (40 mg/kg body weight/day) for 12 weeks; POL + PTX: cholesterol-rich diet–fed rabbits treated concurrently with policosanol and pentoxifylline for 12 weeks; PCSK9, proprotein convertase subtilisin/kexin 9; TNF-α: tumor necrosis factor-α. Vertical columns represented mean while error bars represented standard deviation, n = 6/group; Ψ
P < .001 vs NC; β
P < .001 vs placebo; Δ
P < .05, *P < .001 vs POL; $
P < .01, ¥
P < .001 vs PTX.
Aortic TNF-α Level
Tumor necrosis factor-α was assessed as an inflammatory marker (Figure 1B). Tumor necrosis factor-α level markedly increased in aortic tissue of dyslipidemic rabbits by 5-fold compared to the NC group and significantly attenuated by POL and PTX. However, the inhibitory effect of PTX on aortic TNF-α was much greater than POL. Combination of POL and PTX induced additional inhibition of TNF-α than either monotherapy.
Wingless-Type MMTV Integration Site Family 3A and BMP-2 Gene Expressions
Both Wnt3a and BMP-2 were evaluated as osteoblast differentiation markers (Figure 2). There was a significant upregulation of Wnt3a and BMP-2 genes in aortic tissue of dyslipidemic rabbits by 10.4- and 12-folds, respectively, compared to the NC group. Cotreatment with either POL or PTX significantly decreased aortic Wnt3a and BMP-2 mRNA levels and further decreased by the combined therapy. Of note, POL therapy achieved greater effect than PTX regarding both genes. Gene expressions of Wnt3a (A) and BMP-2 (B) in aortic tissue of experimental rabbits. NC indicates normal control rabbits fed standard chow diet; Placebo: 0.5% wt/wt cholesterol-rich diet–fed rabbits concurrently treated with placebo; POL, cholesterol-rich diet–fed rabbits treated concurrently with policosanol (5 mg/kg body weight/day) for 12 weeks; PTX: cholesterol-rich diet–fed rabbits treated concurrently with pentoxifylline (40 mg/kg body weight/day) for 12 weeks; POL + PTX, cholesterol-rich diet–fed rabbits treated concurrently with policosanol and pentoxifylline for 12 weeks; Wnt3a, wingless-type MMTV integration site-3a; BMP-2: bone morphogenetic protein-2. Vertical columns represented mean while error bars represented standard deviation, n = 6/group; Ψ
P < .001 versus NC; β
P < .001 vs Placebo; Δ
P < .05, ∞
P < .01 vs POL; ¥
P < .001 vs PTX.
Serum ALP and OCN Levels
Both ALP and OCN represent relevant indicators for vascular calcification (Table 2). Dyslipidemic rabbits (placebo group) displayed significant increases in serum levels of ALP and OCN, compared to the NC group. Both markers were significantly lowered by POL or PTX. The effect of POL on serum OCN was significantly greater than PTX. Coadministration of PTX with POL significantly reduced circulating ALP and OCN levels to greater extent than either monotherapy.
Serum ALP and OCN Levels.a

Abbreviations: ALP, alkaline phosphatase; NC, normal control rabbits fed standard chow diet; Placebo, 0.5% wt/wt cholesterol-rich diet–fed rabbits treated concurrently with placebo; OCN, osteocalcin; POL, cholesterol rich diet–fed rabbits treated concurrently with policosanol (5 mg/kg body weight/day) for 12 weeks; PTX, cholesterol-rich diet–fed rabbits treated concurrently with pentoxifylline (40 mg/kg body weight/day) for 12 weeks; POL + PTX, cholesterol-rich diet–fed rabbits treated concurrently with policosanol and pentoxifylline for 12 weeks; .
a Data are represented as mean ± (standard deviation), n = 6/group.
Ψ P < .001 versus NC.
β P < .001 versus placebo.
Δ P < .05, *P < .001 versus POL.
$ P < .01, ¥ P < .001 versus PTX.
Osteopontin Immunohistochemistry
Osteopontin expression in aortic tissue was examined by immunohistochemical staining (Figure 3). Examination of representative aortic sections of dyslipidemic rabbits displayed extensive OPN expression in tunica intimal–medial cells, which was markedly attenuated by either POL or PTX. Policosanol produced better effect relative to PTX. The coadministration of POL with PTX induced greater effect on OPN expression than either monotherapy. Representative photomicrographs showing OPN IHC-stained aortic sections and quantification analysis for area percentage of OPN-immunopositive cells in aortic sections of experimental rabbits. Osteopontin IHC-stained aortic sections showed extensive OPN expression in tunica intimal–medial cells in Placebo (B) that was significantly attenuated in POL- (C) and PTX-treated groups (D). Osteopontin expression was nearly undetected in POL- and PTX-treated group (E). Scale bar = 50 μm. For quantification analysis, expression of OPN was determined as the mean area percentage of OPN-immunopositive cells (F). Five microscopic fields were investigated, and an average value was calculated for each animal. Three aortic sections from 3 different rabbits were used for each group. (F) Vertical columns represented mean while error bars represented standard deviation. OPN, osteopontin; IHC, immunohistochemical; NC, normal control rabbits fed standard chow diet; Placebo: 0.5% wt/wt cholesterol-rich diet–fed rabbits concurrently treated with placebo; POL, cholesterol-rich diet–fed rabbits treated concurrently with policosanol (5 mg/kg body weight/day) for 12 weeks; PTX, cholesterol-rich diet–fed rabbits treated concurrently with pentoxifylline (40 mg/kg body weight/day) for 12 weeks; POL + PTX: cholesterol-rich diet–fed rabbits treated concurrently with policosanol and pentoxifylline for 12 weeks. Ψ
P < .001 versus NC, β
P < .001 vs Placebo; *P < .001 vs POL; ¥
P < .001 vs PTX.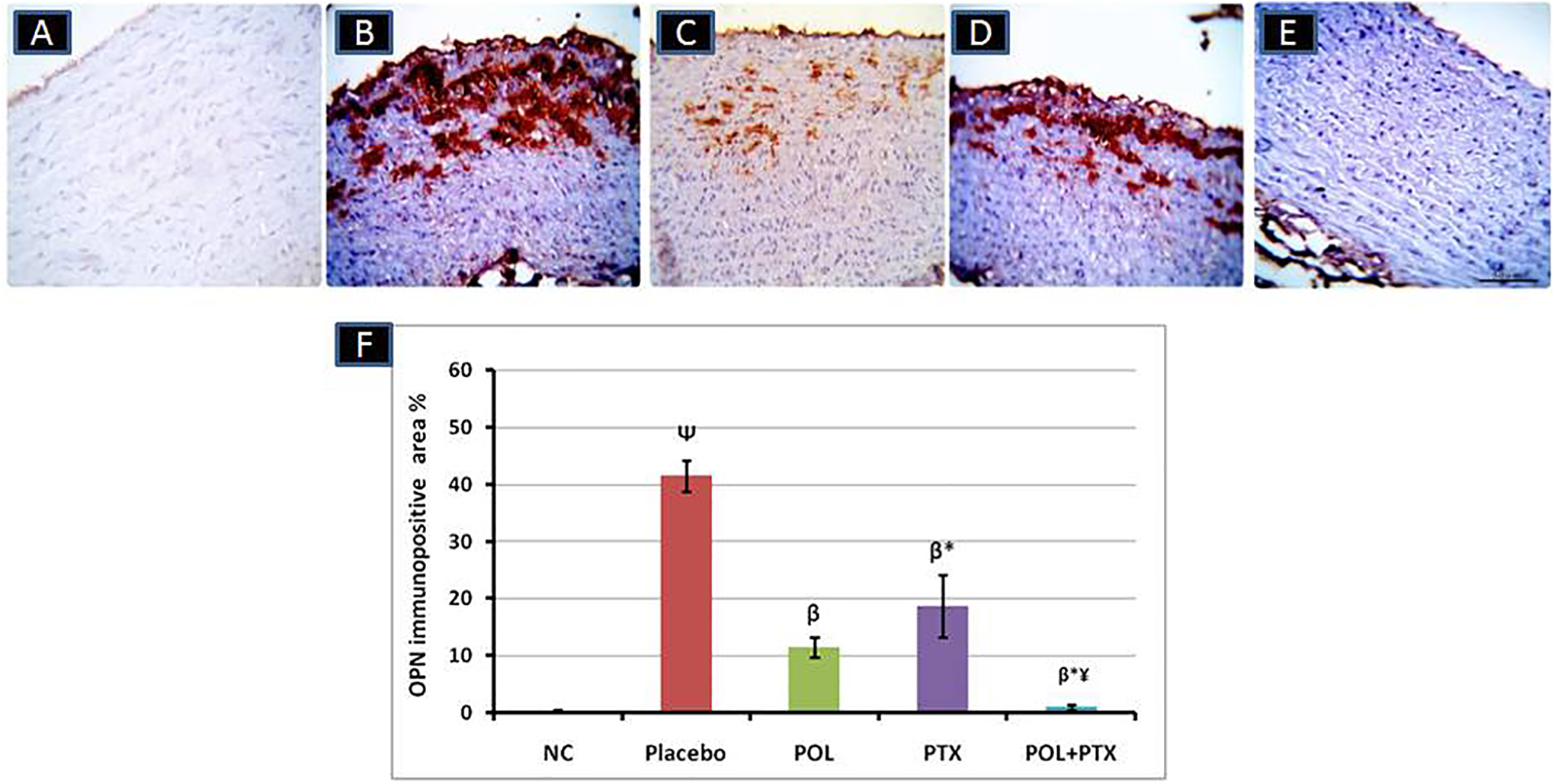
Calcium Deposition
Alizarin red staining of aortic tissue was used to detect aortic calcification (Figure 4). Calcium deposits appeared as irregular red-orange granules distributed in the tunica media. Ectopic calcification was significantly evident in aortic sections of cholesterol-rich diet–fed rabbits, which was significantly attenuated by POL and to a lesser extent by PTX. The cotherapy of POL and PTX effectively abrogated aortic calcification, compared to each individual drug. Representative photomicrographs showing Alizarin red-stained aortic sections and quantification analysis for area of deposited calcium in aortic sections of experimental rabbits. Alizarin red-stained aortic sections revealed subintimal and interstitial calcium deposition as irregular red granular materials between dissociated elastic fibers in Placebo (B), significantly attenuated in POL- (C), and PTX-treated groups (D). Calcium deposition was very low or even absent in POL and PTX- treated group (E). Scale bar = 50 μm. For quantification analysis, calcium deposition was expressed as the percentage of calcium staining (F) using a semiquantitative scale, giving 0 = no calcium deposits; 1 = 10% to 20% of aortic wall contained calcium deposits; 2= 30% to 40% of aortic wall contained calcium deposits, 3 = more than 50% of aortic wall contained calcium deposits. Five microscopic fields were investigated, and an average value was calculated for each animal. Three aortic sections from 3 different rabbits were used for each group. (F) Vertical columns represented mean while error bars represented standard deviation. NC, normal control rabbits fed standard chow diet; Placebo, 0.5% wt/wt cholesterol-rich diet–fed rabbits concurrently treated with placebo; POL, cholesterol-rich diet–fed rabbits treated concurrently with policosanol (5 mg/kg body weight/day) for 12 weeks; PTX, cholesterol-rich diet–fed rabbits treated concurrently with pentoxifylline (40 mg/kg body weight/day) for 12 weeks; POL + PTX: cholesterol-rich diet–fed rabbits treated concurrently with policosanol and pentoxifylline for 12 weeks; Ψ
P < .001 vs NC; β
P < .001 vs Placebo; *P < .001 vs POL; ¥
P < .001 vs PTX.
Biochemical Correlations
Nonparametric Pearson correlation analysis was conducted to investigate the association between PCSK9 and calcification markers, as well as TNF-α using the combined data from all the experimental groups. As illustrated in Figure 5, serum levels of PCSK9 were positively correlated with aortic levels of TNF-α (r = 0.98, P < .0001), BMP-2 mRNA (r = 0.89, P < .0001), and Wnt3a mRNA (r = 0.93, P < .0001), as well as serum levels of OCN (r = 0.66, P < .0001) and ALP (r = 0.96, P < .0001). Nonparametric Pearson correlation analysis. ALP indicates alkaline phosphatase; BMP-2, bone morphogenetic protein-2; OCN, osteocalcin; PCSK9, proprotein convertase subtilisin/kexin 9; TNF-α, tumor necrosis factor α; Wnt3a, wingless-type MMTV integration site-3a.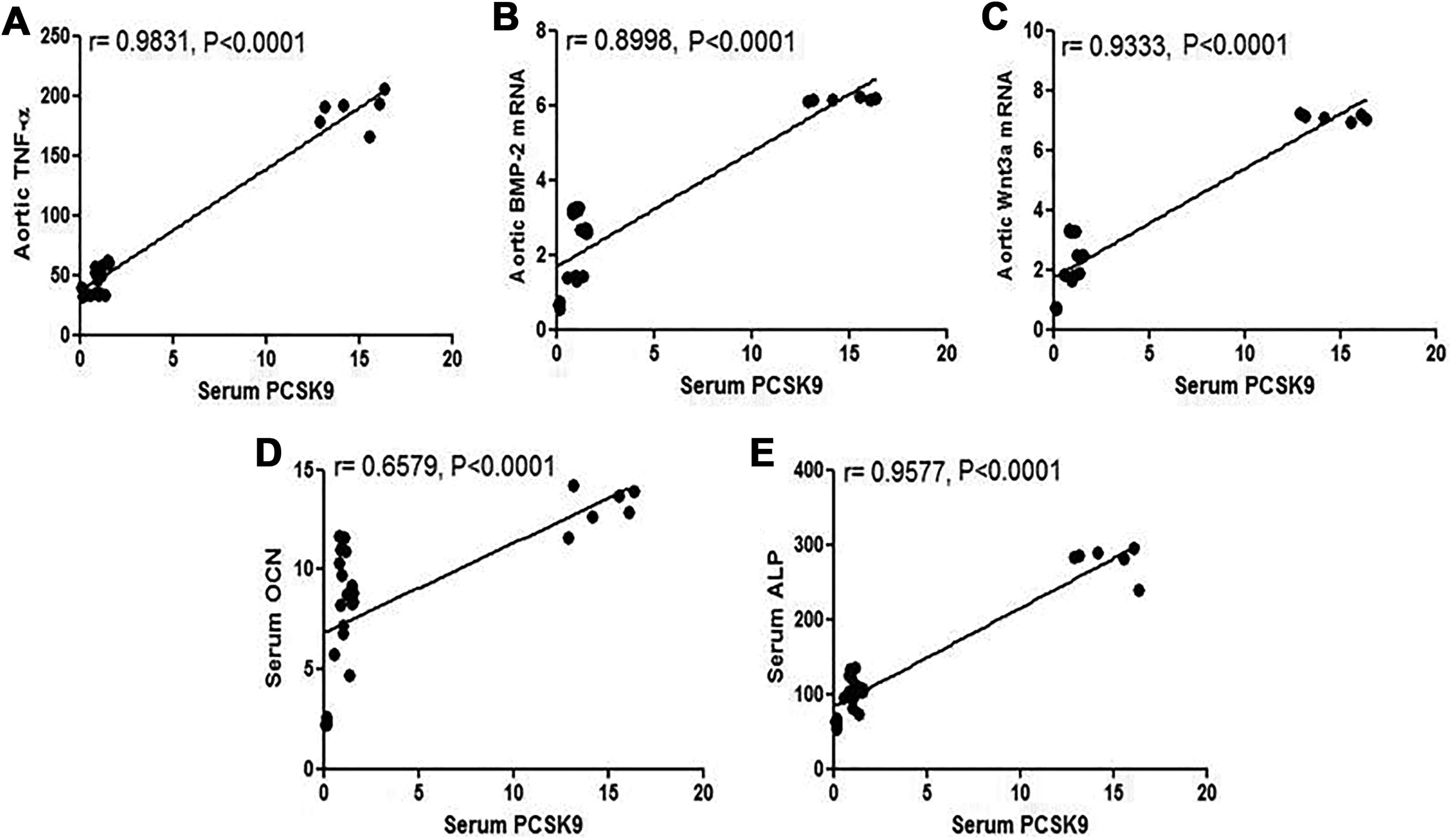
Discussion
Aortic calcification is widely common with hypercholesterolemia and significantly compromises the cardiovascular outcomes. The present study demonstrated that POL could attenuate aortic calcification likely via inhibition of PCSK9. Moreover, combination of POL with PTX provided additional benefits on aortic calcification.
In this study, high cholesterol diet–fed rabbits were used as well-established model for hypercholesterolemia and atherosclerosis due to its reproducibility, close similarity to human lipoprotein metabolism, and reasonable cost compared to the genetically manipulated animal models. 25 Feeding rabbits a cholesterol-rich diet for 12 weeks induced marked dyslipidemia and inflammation along with ectopic calcification in the subintimal layer of aorta of dyslipidemic rabbits, in agreement with others. 18,26 Furthermore, concurrent treatment with POL significantly attenuated aortic calcium deposition, in consistent with our previous report. 14 These findings underscored the anticalcifying effect of POL; however, the molecular mechanisms remain to be determined.
A key finding of the current study was the significantly elevated serum PCSK9 levels in dyslipidemic rabbits. The role of PCSK9 in cholesterol homeostasis as proatherogenic factor has been well established. 10 Our data showed that POL effectively attenuated serum PCSK9 and in turn LDL-C levels in dyslipidemic rabbits, suggesting that POL could serve as a PCSK9 inhibitor. This might be another possible mechanism for the hypolipidemic effect of POL. On the contrary, statins, as HMG-CoA reductase inhibitors, reduce intracellular cholesterol biosynthesis leading to increased nuclear translocation of sterol-regulatory element binding protein-2, which upregulates PCSK9 gene expression and increases circulating PCSK9 levels. 27 –29 The increased PCSK9 enhances lysosomal degradation of LDLRs and hence limiting the efficacy of statins-induced LDL-C clearance. This may explain the drawbacks of statins therapy, particularly “statins escape phenomenon” and “residual cardiovascular risk,” in which plasma levels of LDL-C may increase after prolonged use of statins. 30 Interestingly, combination of POL with atorvastatin has been shown to offset statin-induced elevation of serum PCSK9 levels in patients with hypercholesterolemia, thus improving their efficacy. 13
More importantly, evidence of a potential link between PCSK9 and vascular calcification has emerged from experimental and clinical studies 31 –33 ; however, the precise mechanism for such relationship is still poorly understood. One possibility might be attributed to upregulation of LDL receptor–related protein 5 (Lrp5) as a consequence to PCSK9-induced LDLRs endocytosis. 34 Supportively, Awan et al have reported abundant expressions of Lrp5 and Wnt proteins, along with intimal calcification in aortic tissue of Western diet-fed transgenic Pcsk9 mice. 8
Lrp5 is a well-known coreceptor for LDLRs and frizzled receptors (FZDs). The latter plays a crucial role in activating Wnt/β-catenin pathway. Upon coupling of Wnt proteins, particularly Wnt3a, to FZD receptors/Lrp5 coreceptor, they trigger cascade of downstream signaling events, resulting in dephosphorylation, stabilization, and consequently accumulation of β-catenin in the cytosol. This can lead to translocation of β-catenin into the nucleus, where it initiates the transcription of target genes. 35 Upregulation of Wnt3a has been associated with increased expression and activity of ALP, a specific marker of mineralization. 36,37 Meanwhile, it has been shown that Wnt3a can also activate BMP-2 signaling pathway, another osteogenic factor, which promotes the differentiation of osteoblasts, upregulates OCN and OPN expressions, and ultimately induces matrix biomineralization. 38 Interestingly, some reports have demonstrated that both Wnt and BMP-2 signaling pathways are tightly integrated such that each one can regulate the other, offering synergistic effects on osteogenesis and calcification. 39
In parallel with the previous findings, we found significant increases in Wnt3a/BMP-2 gene expressions in aortic tissue of dyslipidemic rabbits and their downstream proteins including ALP, OCN, and OPN. Moreover, our data indicated positive correlations between serum PCSK9 levels and all studied calcification markers that further emphasized the modulatory role of PCSK9 on aortic calcification. Concurring with these results, a previous study has shown that injection of mice with a mutant PCSK9 adeno-associated virus vector induces aortic calcification along with elevated levels of ALP and OCN. 32 These findings raised an intriguing question: whether PCSK9 could be a biomarker for prognosis or diagnosis of aortic calcification. Alonso et al. have shown that PCSK9 level can be a relevant predictor of coronary artery calcification in patients with asymptomatic FH. 40 Additional studies are warranted in this trend to address the predictive and/or diagnostic value of PCSK9 in the setting of aortic calcification. On the other hand, inhibition of PCSK9 with the subsequent suppression of Wnt3a/BMP-2 signaling pathways by POL therapy might represent a putative mechanism for its anticalcifying effect. The excellent safety profile and tolerability of POL provide additional advantages over the currently available PCSK9 inhibitors.
Several studies have demonstrated that TNF-α is critically involved in the pathogenesis of aortic calcification, 15,16 which prompts investigating the significance of combination of PTX as anti-TNF-α with POL on aortic calcification. As expected, we found that PTX markedly enhanced the anticalcifying effect of POL in dyslipidemic rabbits. In addition, our data for the first time indicated that PTX therapy could attenuate serum levels of PCSK9 presumably via its inhibitory effect on TNF-α. Recently, TNF-α has been shown to induce PCSK9 gene and protein expressions in hepatic HepG2 cell lines in a dose-dependent manner. 41 Consistently, our findings indicated that TNF-α was positively correlated with PCSK9 levels, which might further support the regulatory effect of TNF-α on PCSK9.
Inhibition of PCSK9 appeared to be involved in the modulatory effect of PTX on lipid profile of dyslipidemic rabbits seen in this study and might partly underlie its anticalcifying effect, as well. Moreover, PTX therapy induced downregulation of Wnt3a/BMP-2 signaling pathways and calcification markers, including ALP, OCN, and OPN. A previous study has shown that transgenic mice overexpressing TNF-α exclusively in vascular smooth muscle cells under SM22 promoter display increased gene expression of BMP-2, Msx2, Wnt3a, and Wnt7a in their aortic tissue. 42 In addition, upregulation of BMP-2 has been reported upon exposure of coronary artery endothelial cells to TNF-α. 43 Accordingly, inhibition of TNF-α seemed to be implicated in PTX-induced suppression of Wnt3a/BMP-2 pathways and their downstream signaling proteins. Whether this effect was a direct consequence to TNF-α inhibition or mediated through PCSK9 modulation is still an unresolved issue and would warrant further mechanistic studies.
Conclusion
Our findings demonstrated a therapeutic role of POL against aortic calcification which might be related to inhibition of PCSK9. Since POL markedly attenuated serum TC and LDL-C levels, therefore, we could not exclude the contribution of improving the dyslipidemia in this setting.
Furthermore, inhibition of TNF-α by PTX enhanced the anticalcifying effect of POL probably via inducing more inhibition of PCSK9 and further suppression of Wnt3a/BMP-2 signaling pathways. Surely, further experimental and clinical studies are required to certify these results.
Study Limitations
One limitation of the present study that we could not separate the causal roles of PCSK9 and TC levels, particularly LDL-C, in aortic calcification. Unfortunately, the experimental design lacked a group of cholesterol-rich diet–fed rabbits treated with pharmacologic interventions, which has been reported to decrease TC and LDL-C, but elevate PCSK9 levels, such as statins. This group would help to investigate the distinctive pathologic roles of PCSK9 and lipids independently in aortic calcification.
Footnotes
Author’s Note
M. Elseweidy contributed to conception and design, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. H. Mohamed contributed to conception and design, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. R. Elrashidy contributed to acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. H. Atteia contributed to acquisition, analysis, interpretation, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. G. Elnagar contributed to acquisition and analysis, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Acknowledgments
The authors appreciate Dr Naif A. Al-gabri, Pathology Department, Faculty of Veterinary Medicine, Zagazig University, for his assistance in the histological analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
