Abstract
Background:
Proprotein convertase subtilisin/kexin type 9 (PCSK9) is expressed in multiple tissues, including the small intestine. The effect of PCSK9 inhibition on cholesterol absorption is not known.
Objectives:
Measure serum cholesterol absorption markers before and after initiation of PCSK9 inhibitors.
Methods:
Single-center retrospective cohort of patients administered evolocumab and alirocumab between July 2015 and January 2017. Paired t tests were used to compare mean serum cholesterol marker concentrations, and ratios to total cholesterol, before and after PCSK9 inhibitor initiation. Analyses were repeated for those taking and not taking statins and taking or not taking ezetimibe at both initiation and follow-up, for each PCSK9 inhibitor, and based on follow-up time (<60, 60-120, and >120 days).
Results:
There were 62 possible participants, 34 were excluded for lack of data or unknown PCSK9 inhibitor initiation date. Average follow-up was 92.5 days. Mean campesterol (before 3.14 μg/mL, 95% CI: 2.79-4.38 μg/mL; after 2.09 μg/mL, 95% CI: 1.87-2.31 μg/mL; P < .0001), sitosterol (before 2.46 μg/mL, 95% CI: 2.23-2.70 μg/mL; after 1.62 μg/mL, 95% CI: 1.48-1.75 μg/mL; P < .0001), and cholestanol (before 3.25 μg/mL, 95% CI: 3.04-3.47 μg/mL; after 2.08 μg/mL, 95% CI: 1.96-2.21 μg/mL; P < .0001) all significantly decreased at follow-up. There was no significant change in absorption marker to total cholesterol ratios. Findings were not influenced by statin or ezetimibe use or nonuse, which PCSK9 inhibitor was prescribed, or time to follow-up.
Conclusion:
Proprotein convertase subtilisin/kexin type 9 inhibition was associated with decreased cholesterol absorption markers.
Keywords
Introduction
Proprotein convertase subtilisin/kexin type 9 (PCSK9) is an important regulator of low-density lipoprotein cholesterol (LDL-C) via mediation of LDL receptor (LDL-R) availability. The mechanism of action occurs primarily via its activity within the liver where it binds to LDL-R and promotes its lysosomal degradation. 1 However, it is known that PCSK9 is also expressed in other tissue beds, including adipose tissue, heart, muscle, kidney, central nervous system, pancreas, and the small intestine. 1,2 The role of PCSK9 and its inhibition in these other tissue beds are not well understood.
Within the small intestine, PCSK9 activity is thought to increase intestinal production of triglyceride (TG)-rich B apolipoproteins (B48), 1,3 contribute to postprandial TG enrichment of chylomicrons, 1 and decrease transintestinal cholesterol excretion (TICE). 1,4 –6 These changes occur through LDL-R-dependent and -independent mechanisms. 3,5,7
Studies on human enterocyte cells found PCSK9 to increase cholesterol uptake by influencing Niemann-Pick C1-like 1 (NPC1L1) protein, cluster differentiation protein 36, and the activity of acyl-coenzyme A: cholesterol acyltransferase 2 (ACAT2). 7,8 The ACAT2 has a preferential affinity for cholesterol compared to noncholesterol sterols. The effect of PCSK9 on NPC1L1 protein and ACAT2 was also found to be independent of LDL-R activity. Microsomal TG transfer protein is also enhanced by PCSK9 in a mechanism independent of LDL-R. 3 Inhibition of PCSK9 activity within enterocytes decreases the aforementioned pathways. 3,7 The TICE is hypothesized to be regulated by PCSK9 in a study demonstrating PCSK9 knockout mice have increased cholesterol excretion compared to wild-type mice in a LDL-R-dependent mechanism. 5
The aforementioned effects are known to occur via the basolateral membrane. 5,7,9 Inhibition of PCSK9 may significantly alter intestinal regulation of cholesterol and TG. Understanding this process may provide additional insight into how PCSK9 inhibition alters total body cholesterol homeostasis. Since increased cholesterol absorption is associated with the development of coronary artery disease and genetic-related reduction in cholesterol absorption is associated with reduced coronary artery disease (CAD) risk, it is important to understand the role of PCSK9 in this process. 10 –12 We sought to investigate the role of PCSK9 inhibition on intestinal cholesterol absorption by measuring the concentrations of validated cholesterol absorption markers (campesterol, sitosterol, and cholestanol) 13 –15 in a cohort of clinic patients prescribed evolocumab or alirocumab.
Methods
Patients were retrospectively identified who were prescribed evolocumab or alirocumab at the University of Chicago from July 2015 through January 2017. Patients were not randomly assigned to evolocumab or alirocumab. The medication was determined by the clinician and the insurance provider’s prescription drug formulary.
Inclusion and Exclusion Criteria
We included all patients who were 18 years or older and met Food and Drug Association qualifications for use of PCSK9 inhibitors. Patients were excluded from analyses if they had no cholesterol absorption biomarker data prior to and/or following PCSK9 inhibitor initiation or if the temporal relation of those biomarkers was not available.
Laboratory studies were done at True Health Diagnostics (Richmond, VA). The comprehensive lipid/lipoprotein panel included traditional biomarkers (total cholesterol [TC], directly measured LDL-C, high-density lipoprotein-cholesterol [HDL-C], TG, and non-HDL-C), apolipoproteins B and A1, lipoprotein (a) [Lp(a)] particle number, cholesterol absorption markers (campesterol, sitosterol, cholestanol, and their ratios to TC), a cholesterol synthesis marker (desmosterol and its ratio to TC), an inflammatory marker (high-sensitivity C-reactive protein), and other routine nonlipid tests.
The lipid/lipoprotein markers are reported to ensure that any findings related to the cholesterol absorption markers occur in the setting of what would have been previously predicted based on prior PCSK9 evaluations in the literature. Noncholesterol sterols and stanols were assayed using liquid chromatography with tandem mass spectrometry.
Note that multiple tests reached a threshold that was below the lowest limit of detection. In the aforementioned tests, these include Lp (a) particle number (lowest reported value = 50 nmol/L) and all sterol markers (lowest reported value = 0.5 µg/mL). In the case that a biomarker was below the detection limit, values were assumed to be equivalent to the lowest detection limit. Given that most values falling below the lowest reported value were those measured at follow-up, this served to not bias results away from the null hypothesis. Given that a large portion of measured desmosterol values fell below the detection limit (n = 14), we were unable to reliably estimate ratios of campesterol, sitosterol, or cholestanol to desmosterol.
Paired t tests were used to compare mean absolute values before and after PCSK9 inhibitor initiation. Analyses were repeated individually for each PCSK9 inhibitor (evolocumab or alirocumab). In addition, it is known that both statins and ezetimibe may alter sterol absorption markers and cholesterol concentrations. Therefore, analyses were repeated for those taking and not taking statins at both initiation and follow-up and for those taking and not taking ezetimibe at both initiation and follow-up. Patients were excluded if the statin or ezetimibe was discontinued at time of follow-up or it was not able to be determined whether they were taking the statin or ezetimibe. In addition, it is conceivable that sterol absorption response to PCSK9 inhibition may change with time, so results were also analyzed by time to follow-up, categorized as <60, 60-120, and >120 days.
The relationship between changes in LDL-C and sterol absorption markers was evaluated by plotting percentage change in follow-up values compared to baseline values then applying a linear regression trend line. Correlation was estimated by R2 coefficient.
All analyses were performed within Microsoft Excel version 15.32. This study was approved by institutional review boards of the University of Chicago. This analysis used deidentified clinical data, so participant consent was not required. Data analysis was conducted from October 2016 through May 2017.
Results
There were 63 patients identified as being on a PCSK9 inhibitor between July 2015 and January 2017. Thirty-four patients were excluded from the analyses. Thirty-three of these patients were excluded because of a lack of comprehensive lipoprotein panel data before, after, or both before and after PCSK9 inhibitor initiation. One patient was excluded because it was unclear when they started the PCSK9 inhibitor in relation to the comprehensive lipoprotein panel data. See Figure 1 for schematic of study selection process.

Schematic of the study selection process.
There were 29 patients included in the analysis. The mean age was 58.6 years. A minority of patients were female (n = 9; 31.0%). The indications for initiation of PCSK9 inhibitor included those using maximally tolerated statin with or without ezetimibe who were not at desired LDL-C goal with dyslipidemia (n = 9; 31.0%), statin intolerance (n = 15; 51.7%), and familial hypercholesterolemia (n = 19; 65.5%). Mean follow-up time was 92.5 days after initiation of PCSK9 inhibitor. See Table 1 for further population characteristics.
Characteristics of the Study Population.a

Abbreviations: BMI, body mass index; SD, standard deviation.
a Results are reports as total patients with the characteristics with percentage of study population in parenthesis, unless otherwise indicated.
After PCSK9 inhibitor initiation, there were drastic changes in lipid and lipoprotein measurements. There were decreases in TC, LDL-C, non-HDL-C, TG, apolipoprotein B, and lipoprotein (a) levels (Table 2). Apolipoprotein A1 levels increased. Both HDL-C and high-sensitivity C-reactive protein remained unchanged.
Lipoprotein Profiles Before Versus After Initiation of PCSK9 Inhibitor.a
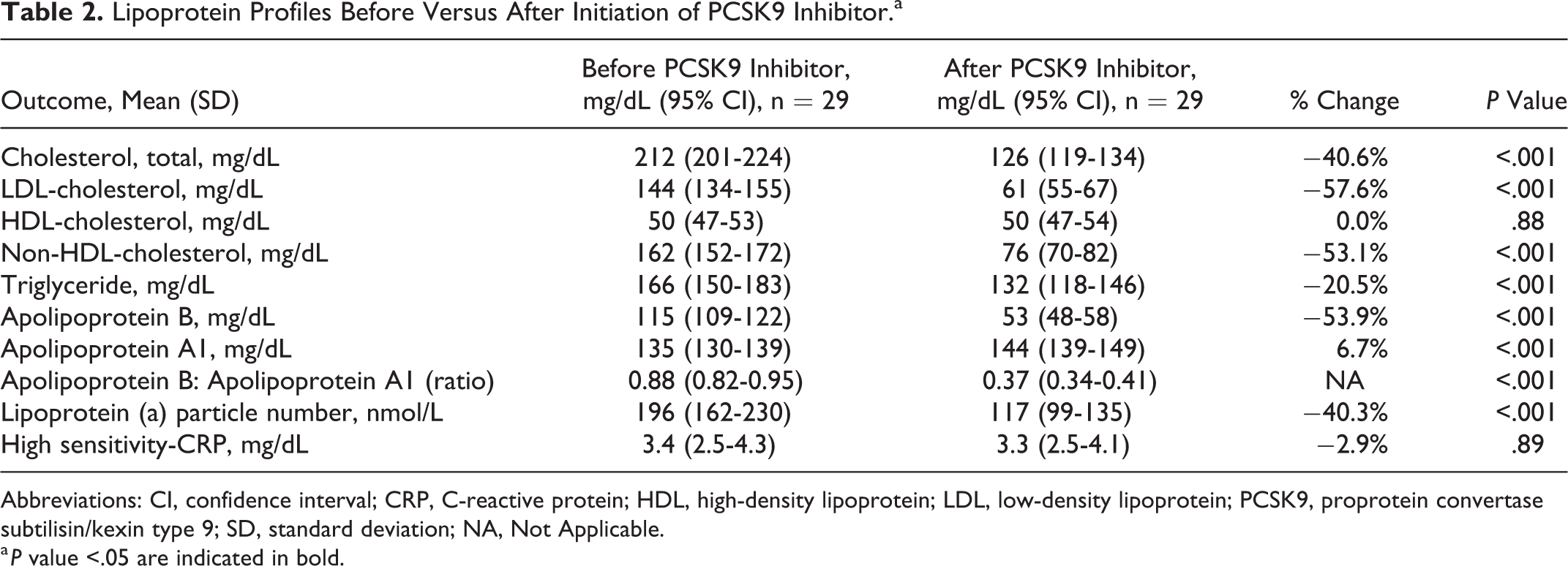
Abbreviations: CI, confidence interval; CRP, C-reactive protein; HDL, high-density lipoprotein; LDL, low-density lipoprotein; PCSK9, proprotein convertase subtilisin/kexin type 9; SD, standard deviation; NA, Not Applicable.
a P value <.05 are indicated in bold.
There were significant declines in serum cholesterol absorption markers after initiation of PCSK9 inhibitors (Table 3). Campesterol values declined by 33.3% from 3.14 µg/mL (95% CI: 2.78-3.49 µg/mL) to 2.09 µg/mL (95% CI: 1.87-2.32 µg/mL; P < .001). Sitosterol values declined by 34.3% from 2.46 µg/mL (95% CI: 2.22-2.70 µg/mL) to 1.62 µg/mL (95% CI: 1.48-1.75 µg/mL; P < .001). Cholestanol decreased by 35.9% from 3.25 µg/mL (95% CI: 3.03-3.48 µg/mL) to 2.08 µg/mL (95% CI: 1.96-2.21 µg/mL; P < .001). When evaluating ratios of cholesterol absorption marker to cholesterol, there was no significant differences between values at initiation compared to follow-up (Table 3). In addition, we evaluated the relationship between changes in LDL-C and changes in cholesterol absorption markers. We found that the changes in campesterol (R2 = .009), sitosterol (R2 = .004), and cholestanol (R2 = .340) all poorly correlated with changes in LDL-C.
Comparison of Cholesterol Absorption Markers Before Versus After Initiation of PCSK9 Inhibitors.a
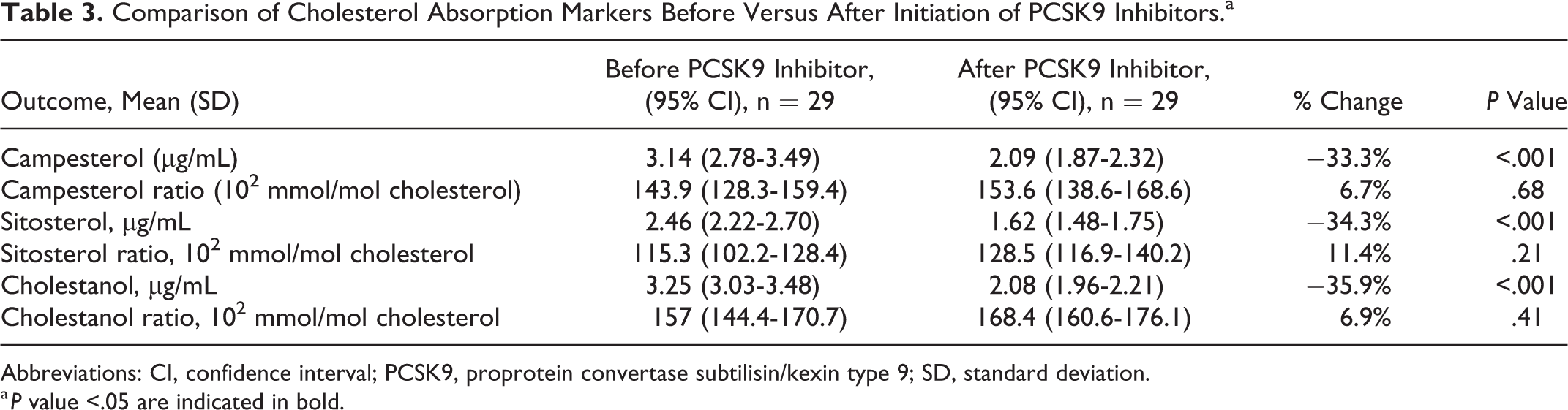
Abbreviations: CI, confidence interval; PCSK9, proprotein convertase subtilisin/kexin type 9; SD, standard deviation.
a P value <.05 are indicated in bold.
There were similar number of patients on evolocumab (n = 15) and alirocumab (n = 14). Regardless of PCSK9 inhibitor, there were significant declines in all sterol absorption markers (Table 4).
Comparison of Cholesterol Absorption Markers Before Versus After Initiation of PCSK9 Inhibitors for Patients on Evolocumab or Alirocumab.a
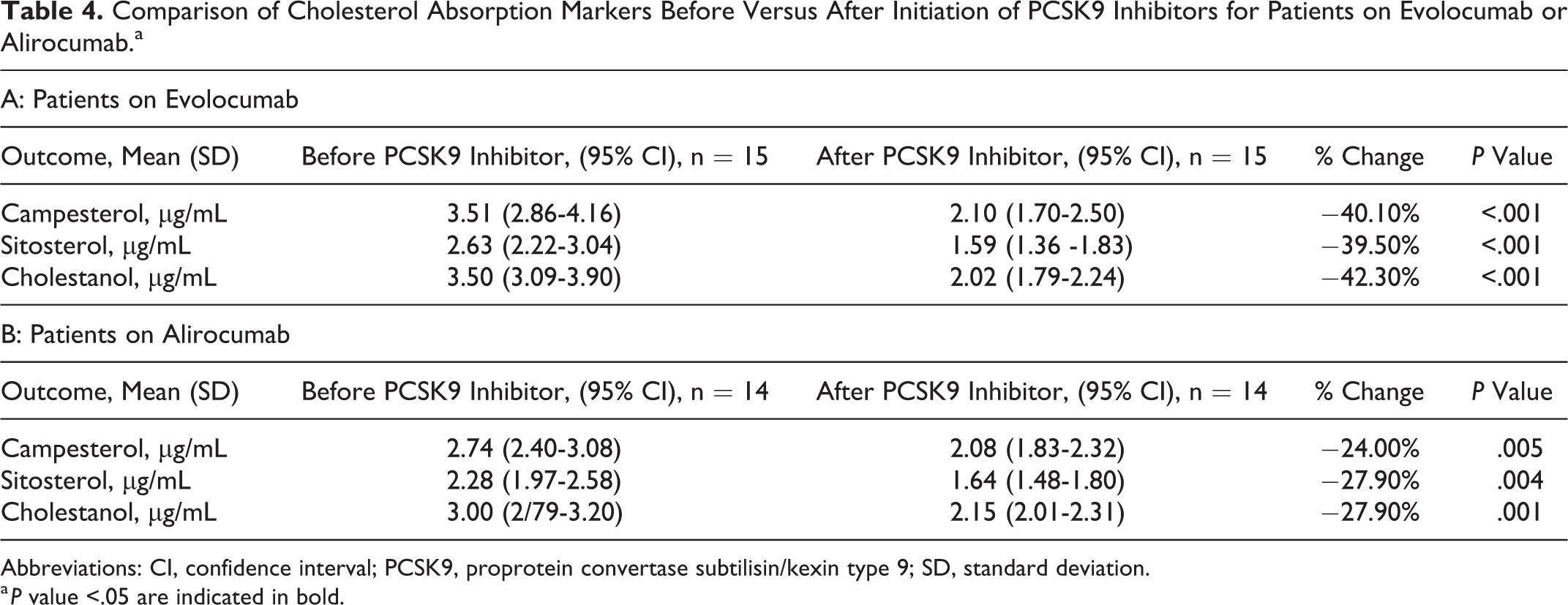
Abbreviations: CI, confidence interval; PCSK9, proprotein convertase subtilisin/kexin type 9; SD, standard deviation.
a P value <.05 are indicated in bold.
There were more patients taking statins (n = 17) than not taking statins (n = 11) at both initiation and follow-up. Regardless of whether statin was prescribed, both groups experienced a decline in sterol absorption markers (Table 5).
Comparison of Cholesterol Absorption Markers Before Versus After Initiation of PCSK9 Inhibitors for Patients Taking Versus Not Taking Statins.a

Abbreviations: CI, confidence interval; PCSK9, proprotein convertase subtilisin/kexin type 9; SD, standard deviation.
a P value <.05 are indicated in bold.
There were more patients not taking ezetimibe (n = 16) than taking ezetimibe (n = 10) at both initiation and follow-up. Although baseline values were slightly lower for patients taking ezetimibe, both patients taking and not taking ezetimibe had a decline in sterol absorption markers (Table 6).
Comparison of Cholesterol Absorption Markers Before Versus After Initiation of PCSK9 Inhibitors for Patients Taking Versus Not Taking Ezetimibe.a
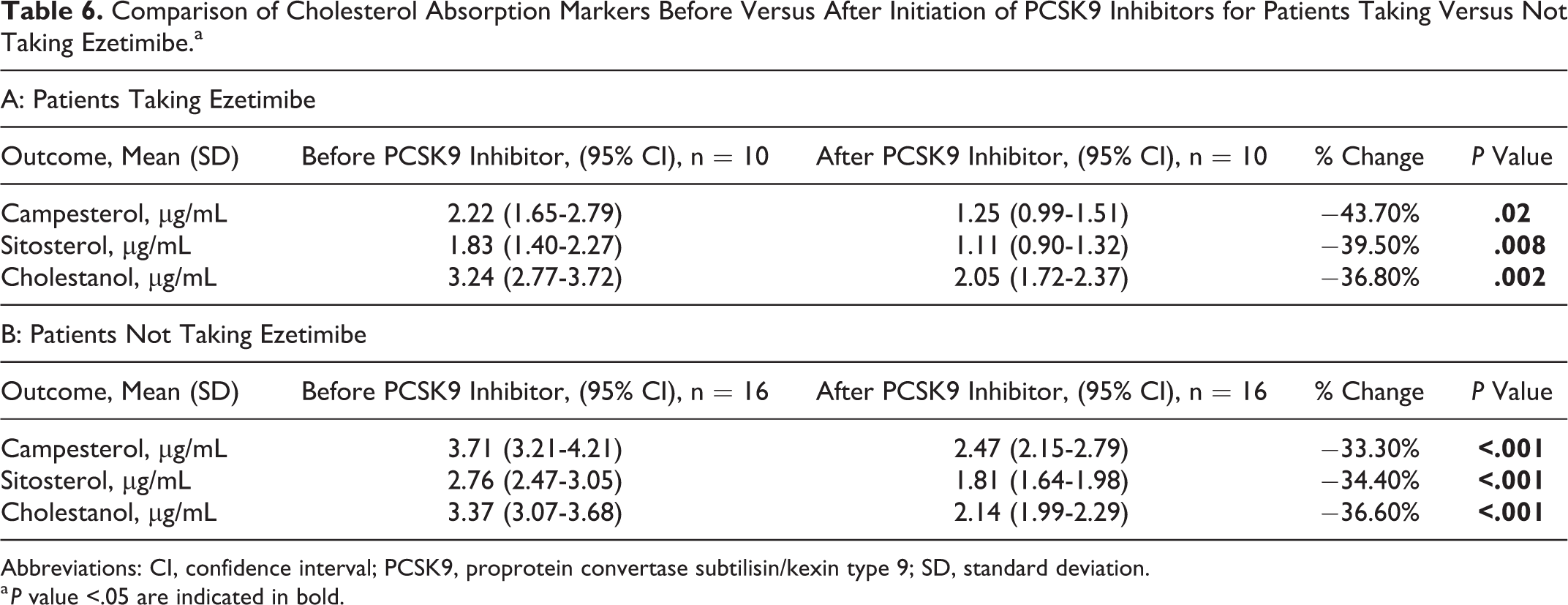
Abbreviations: CI, confidence interval; PCSK9, proprotein convertase subtilisin/kexin type 9; SD, standard deviation.
a P value <.05 are indicated in bold.
Finally, there were significant declines in all sterol absorption markers for patients who had followed up at <60 days (n = 9) and between 60 and 120 days (n = 15). For the cohort of patients with follow-up at >120 days (n = 5), there was significant decline in cholestanol (2.47 µg/mL, 95% CI: 1.97-2.98 µg/mL to 1.70 µg/mL, 95% CI: 1.29-2.12 µg/mL; P = .002) but no significant change in campesterol or sitosterol (Table 7).
Comparison of Cholesterol Absorption Markers Based on Length of Follow-Up After Initiation of PCSK9 Inhibitors.a
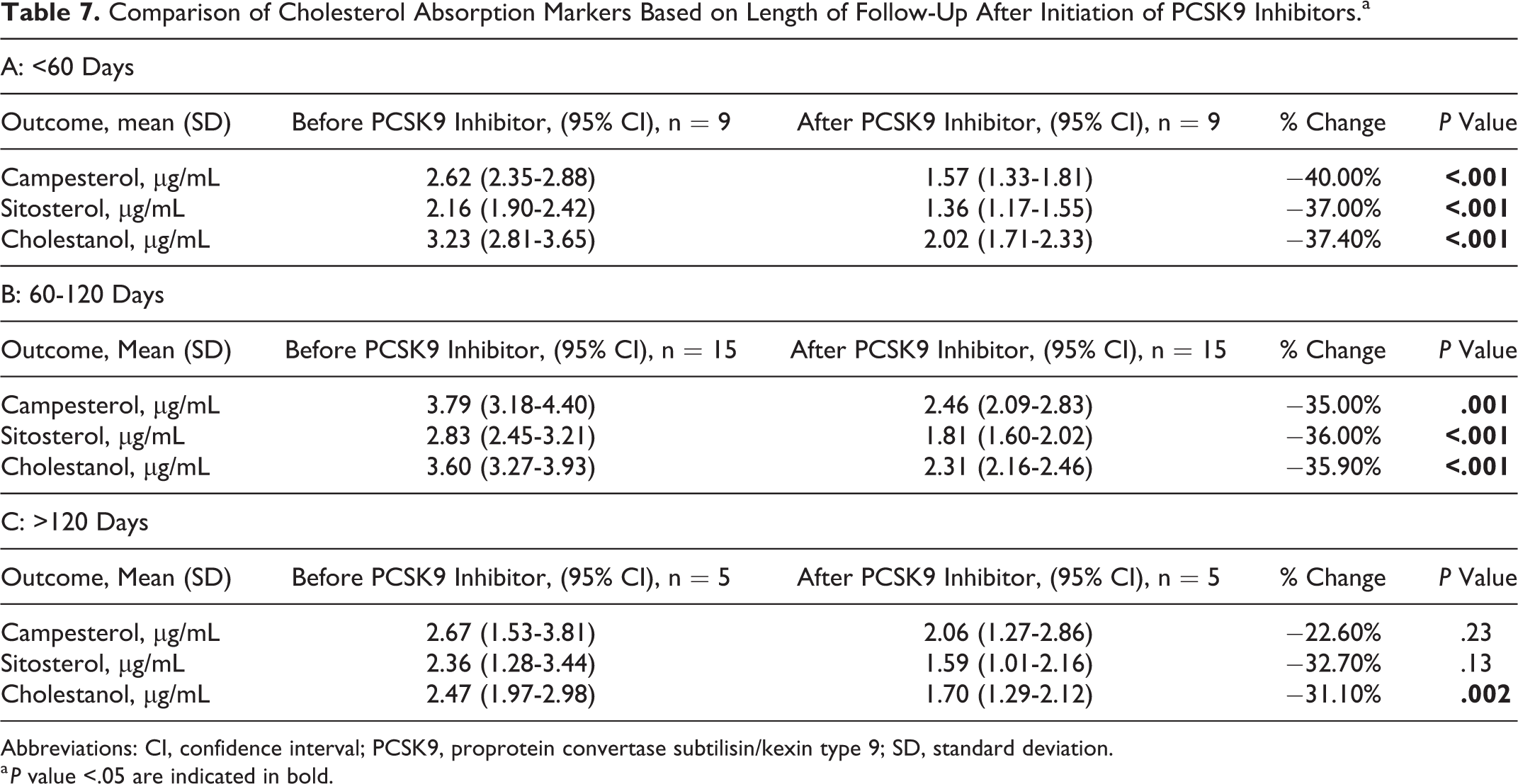
Abbreviations: CI, confidence interval; PCSK9, proprotein convertase subtilisin/kexin type 9; SD, standard deviation.
a P value <.05 are indicated in bold.
Discussion
Prior studies have found that PCSK9 is expressed in multiple tissue beds. In the small intestine, PCSK9 influences cholesterol absorption and excretion via multiple LDL-R-dependent and LDL-R-independent mechanisms. 3,5,7 In this retrospective cohort of patients prescribed PCSK9 inhibitors, we found patients had decreased levels of cholesterol absorption markers after initiation of a PCSK9 inhibitor. When these absorption markers were evaluated as ratios to TC, there were no significant differences. To our knowledge, this is the first study examining the association between PCSK9 inhibition and cholesterol absorption markers.
The full understanding of PCSK9 mechanisms and inhibition of such mechanisms in the small intestine remains incompletely elucidated. Our data support a mechanism by which PCSK9 is active in the small intestine and its activity is modulated by PCSK9 inhibition with monoclonal antibodies. Proprotein convertase subtilisin/kexin type 9 is known to be present and active in serum and basolateral intestinal membrane. 5,7,9 Multiple studies have elucidated potential mechanisms by which inhibition of PCSK9 activity could modulate intestinal cholesterol absorption and excretion, including effects on NPC1L1 protein, cluster differentiation protein 36, ACAT2, microsomal TG transfer protein, and TICE. 3,5,7 –9 Inhibition of PCSK9 alters all or some of these mechanisms, but additional studies will be needed. 16 For the most part sitosterol, campesterol, and cholestanol are cleared via ABCG5/G8 efflux from hepatocytes/enterocytes into bile/gut lumen with the enterocyte route termed TICE. 5,17 –19 A minor pathway for unesterified phytosterols to exit hepatocytes or enterocytes is efflux via ABCA1 into unlipidated or minimally lipidated apolipoprotein A-I. 20,21 Another minor path for phytosterols that may occur in enterocytes or hepatocytes is their potential esterification, via ACAT2, to phytosterol esters (cholesterol is the preferred substrate); these can be incorporated into the cores of chylomicrons or very low density lipoproteins (VLDL), respectively, and subsequently secreted into the circulation. 22 –24 Finally, core sterol esters can be internalized by tissue expressing scavenger receptor B1, such as within hepatocytes and enterocytes. 25,26 There is no current data on how PCSK9 inhibitors might affect these pathways of sterol homeostasis.
Studies have shown that medications, including statins and ezetimibe, may alter cholesterol absorption. The effect of statins on absorption markers is unpredictable in individual patients but may be influenced by specific statin, the dose employed, and total time the patient has taken the drug. 27 Over time, cholesterol absorption increases for most patients taking statins. 28 –30 This is important because cholesterol absorption may play an important role in risk reduction in relation to statins, as it has been shown that those in the highest quartile of sterol absorption did not have a reduction in coronary events from simvastatin risk reduction in the Finnish arm of the 4S trial. 31 Our finding that cholesterol absorption markers decreased in response to PCSK9 inhibition is important in this context. If there is residual risk in patients related to their cholesterol absorption, our findings of decreased cholesterol absorption markers would suggest that this absorption related risk could conceivably be avoided in patients on PCSK9 inhibitors, although more data are needed.
Ezetimibe acts at NPC1L1 protein, an intestinal cholesterol transmembrane transporter. 32,33 Patients taking ezetimibe were found to have reduced absolute campesterol (−48%) and sitosterol (−41%) levels, as well as their ratios to TC (−41% and −34%, respectively). 34 In the case of patients taking ezetimibe and simultaneously on a statin, there were also decreased campesterol and sitosterol absolute values and ratios to TC. 35 The primary mechanism of ezetimibe is thought to be via inhibition of cholesterol absorption, and the recent Improved Reduction of Outcomes: Vytorin Efficacy International Trial (IMPROVE-IT) showed that addition of ezetimibe to simvastatin lead to a reduction in recurrent cardiovascular events. 36 However, there are no data yet available as to whether this was due to its effect on cholesterol absorption. The possibility of there being multiple mechanism for PCSK9 activity in the intestine is further support by our observation of absorption markers in patients already taking ezetimibe. In patients on ezetimibe, there were measurable levels of absorption markers in the blood which were further reduced after PCSK9 inhibition. This suggests either an additive effect of PCSK9 inhibition on absorption via NPC1L1 or an alternative mechanism (as previously discussed).
Campesterol, sitosterol, and cholestanol serve as biomarkers of cholesterol absorption. 13 –15 Campesterol and sitosterol are phytosterols, and cholestanol is a stanol metabolite of cholesterol. Serum noncholesterol sterols and their ratios have long been accepted as markers for cholesterol absorption. 13 –15 Some have also suggested using ratio of cholesterol absorption to synthesis biomarkers, such as campesterol–lathosterol ratio, to evaluate absorption. 13,37 –39 These data were not available for our study due to low levels of synthesis markers that frequently fell below the level of detection and that lathosterol was not measured in our study. Utilization of phytosterol serum values as markers for cholesterol absorption have recently been called into question by Jakulj et al, 40 although the accompanying editorial commentary had concerns for methodologic issues. 37 It seems that the ideal noninvasive method for accurately measuring cholesterol absorption remains debatable, although various methods have supportive data. Serum sterols and stanols continue to be used because they can be obtained noninvasively through a readily available assay.
In our study, we measured absolute serum cholesterol absorption markers and their ratio to TC. We took care to analyze the markers and their ratios to TC with paired t tests to increase the power of the study. Noncholesterol sterols and stanols were assayed using liquid chromatography with tandem mass spectrometry. However, the assay that measures TC cannot differentiate specific sterols from cholesterol and therefore includes all of the sterols within lipoproteins, which limits interpretation of the ratios. Furthermore, although absorption markers to TC ratios have been commonly reported in epidemiologic studies for measuring changes in absorption, absorption may not be best measured in this manner. When evaluating individual patients during periods of large variations in LDL-C and TC, such as when initiating a PCSK9 inhibitor, statin, or ezetimibe, there may be large changes in TC even without a change in noncholesterol sterol levels. Van Himbergen et al have identified various advantages and disadvantages for using absolute levels or their ratios to total depending on the study being performed. They have advised using absorption markers to TC ratios to demonstrate differences between groups but using absolute levels when investigating relationships in individual patients such as we have done in our study. 28 This avoids error in normalizing or adjusting for changes in TC. Given that many factors may alter marker estimations, using multiple noncholesterol sterol markers, such as the 3 we have measured in our study, may decrease the likelihood for error in interpretations. 13
Our study has multiple strengths. First, we measured multiple cholesterol absorption markers, the results for which reached a high level of significance that suggests a low likelihood for error. Next, in our patients, there were similar changes in lipid and lipoprotein concentrations that were seen in larger clinical trials, including FOURIER and ODYSSEY. 41,42 This suggests that patients were adherent to the PCSK9 inhibitors. Finally, we performed multiple sensitivity analyses to ensure that other medications that are known to alter cholesterol absorption were not interfering with our results.
Our study has multiple limitations. First, this is a single-center trial that was performed in retrospective fashion. There was no control group for comparison. In addition, there were no studies performed on serum samples to verify adherence to therapy. Furthermore, we were unable to estimate ratios of absorption to synthesis markers, which would have provided further evidence for PCSK9 inhibition altering cholesterol absorption. Campesterol to lathosterol ratios would have been ideal for this measurement, but lathosterol markers were not available from this laboratory panel. There were no data for longer term follow-up. Only a few patients (n = 5) had followed up beyond 120 days, so it is unclear whether these changes continue beyond our study period and will need further investigation to understand long-term relationships. Furthermore, sterols may also be incorporated into LDL or HDL particles; these lipoproteins carrying the sterols may then be internalized. This study is unable to quantitate the amount by which these alternative mechanisms account for our findings. Continuing, medication adherence was based on chart review at the time of the visit, and there could be discrepancies between the charted medications and actual patient adherence. Finally, although our study supports mechanisms of PCSK9 activity in the intestine, understanding the specific mechanisms by which differing effects are mediated by the liver and intestine remain limited. In the future, studying the differences between siRNA PCSK9 inhibitors and PCSK9 monoclonal antibody inhibitors may help to better elucidate the mechanisms by which PCSK9 is active within the intestine.
In conclusion, inhibition of PCSK9 with evolocumab or alirocumab was associated with decreased serum cholesterol absorption markers. Proprotein convertase subtilisin/kexin type 9 activity in multiple tissue beds beyond the liver may be responsible for changes in lipid profiles observed in patients on PCSK9 inhibitors.
Footnotes
Authors’ Contribution
E.J. Brandt contributed to conception and design; acquisition, analysis and interpretation; drafted manuscript; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. L.B. Benes contributed to design; acquisition, analysis, and interpretation. L. Lee contributed to acquisition and interpretation. T.D. Dayspring contributed to interpretation. M. Sorrentino contributed to conception and design. M. Davidson contributed to conception and design; acquisition, analysis, and interpretation; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. All authors crically revised the manuscript and gave final approval. All of the authors have read and approved of the final manuscript as well as the data being presented in the manuscript. These data are original, have not been previously published, and are not under consideration by any other journal.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
