Abstract
Background:
Previous meta-analyses have shown that statins may cause incident diabetes. This article reviews randomized controlled trials using proprotein convertase subtilisin/kexin 9 inhibitors (PCSK9i) or ezetimibe on the risk of new-onset diabetes.
Methods:
Eight trials involving PCSK9i and 3 trials of ezetimibe were selected for review. PubMed, Cochrane Central Register of Controlled Trials, and Clinicaltrials.gov were thoroughly searched for relevant trials. Inclusion criteria included at least 100 patients per treatment arm, follow-up of at least 52 weeks, and at least double-blinded study design. Exclusion criteria included patients with previously diagnosed diabetes, nonrandomized, placebo-controlled, open-label, and crossover trials. The primary outcome was the number of incident diabetes cases. A random effects model was used. Heterogeneity in effect sizes was measured with I 2 parameter and the Q statistic was used to test for excessive between-study heterogeneity.
Results:
A total of 52 214 participants for the PCSK9i and a total of 20 084 for the ezetimibe meta-analyses were included. Participants randomized to PCSK9i did not differ from the control patients in diabetes incidence (risk ratio [RR] = 0.99, P = .87, 95% CI = 0.92-1.07). Participants randomized to ezetimibe did not differ from the control patients in diabetes incidence (RR = 1.05, P = .37, 95% CI = 0.95-1.15).
Discussion:
The use of PCSK9i and ezetimibe does not appear to impact the risk of incident diabetes mellitus when added to guideline-directed medical therapy.
Introduction
Statins are commonly used in clinical practice as they have been shown to effectively lower low-density lipoproteins (LDL), which are known to be a modifiable risk factor for atherosclerotic cardiovascular disease. 1,2 Proprotein convertase subtilisin/kexin 9 inhibitors (PCSK9i) such as alirocumab and evolocumab are Food and Drug Administration (FDA)–approved drugs that have been found to significantly lower LDL and other harmful cardiovascular risk factors. 3 -6 Ezetimibe lowers levels of circulating cholesterol by blocking intestinal absorption via Niemann-Pick C1-Like 1 and is usually prescribed as an adjunct to a statin. 7,8
Several meta-analyses regarding large clinical trials with statins have shown an increased risk of incident diabetes. 9 -11 Specifically, Sattar et al reported a 9% increased risk. 9 Preiss et al noted that intensive-strength statin therapy was associated with the increased risk of incident diabetes compared to moderate strength. 10 Carter et al determined that treatment with higher potency statins (ie, atorvastatin and simvastatin) may be associated with a higher risk of new-onset diabetes mellitus. 11 One meta-analysis by Coleman et al demonstrated that statins as a class do not significantly increase the chances of having new-onset diabetes; however, when pravastatin was excluded in the analysis, there was a statistically significant increase in risk. 12 Most studies investigating PCSK9i have stated that there is no significant risk of causing new-onset diabetes. 13 -16 However, one review states that there is a risk of increased plasma glycemia. 17 Moreover, there are few meta-analyses regarding ezetimibe in conjunction with statins on whether it may cause incident diabetes. 18-19 In this study, we conducted a systematic review and meta-analysis of large, double-blinded, randomized controlled trials involving PCSK9i and ezetimibe to evaluate their risk of causing new-onset diabetes mellitus. In particular, we analyzed trials that had at least a 52-week usage of medications, of which a few large statin trials used this criterion to assess for longer-term impact of medication use. 9,10 We hypothesized that PCSK9i and ezetimibe would increase the risk of incident diabetes.
Methods
Search Strategy
Two independent investigators (S.W.C and C.M.P.) performed the screening process according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines (Figure 1). PubMed, Cochrane Central Register of Controlled Trials, and Clinicaltrials.gov were thoroughly searched for relevant trials. Boolean operators and MeSH terms were used for optimal searching (Appendix 1). Any duplicates found were removed. No date restrictions were applied, and only human participants were considered in the search. Only articles in English were selected. This study was registered with PROSPERO (registration number CRD42019129626; URL https://www.crd.york.ac.uk/PROSPERO/display_record.php?%20RecordID=129626).

Study selection according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. CENTRAL indicates Cochrane Central Register of Controlled Trials; DM, diabetes mellitus.
Study Selection
To have good statistical power with low bias in study design, we aimed to create inclusion and exclusion criteria similar to meta-analyses involving large clinical trials. The inclusion criteria included at least 100 participants per treatment arm, follow-up of at least 52 weeks, and at least a double-blinded study. Exclusion criteria included trials that solely had type 1 or 2 diabetic patients, nonrandomized controlled trials, placebo-controlled trials, open-label trials, and crossover trials. Studies that did not include published results on either new cases of incident diabetes mellitus, baseline hemoglobin A1c (HbA1c), or fasting plasma glucose (FPG) were excluded.
Data Extraction and Quality Assessment
Data extraction was executed by 2 independent investigators (S.W.C and C.M.P.) and any discrepancies were discussed with a third investigator (S.C.). If there were situations in which the data were inconsistent between 2 articles, then the data from the original article was chosen. Previous systematic reviews and meta-analyses that included more recent data from the original trials were used if the information was more accurate or if the original article did not conspicuously publish these. The investigators and authors of the original clinical trials were not contacted for clarification of data.
The data were separated by treatment arm, that is, PCSK9i group versus control or ezetimibe group versus control (Table 1). Collected data included patient demographics such as mean age, sex, ethnicity (eg, black, Asian), and country. Mean body mass index and baseline values of HbA1c and/or FPG, total cholesterol, LDL, high-density lipoprotein, and triglycerides were extracted. The change of LDL from baseline to end point was also noted.
Trial Characteristics.

Abbreviations: CHD, coronary heart disease; CVD, cardiovascular disease; Q2W, every 2weeks.
a Notes which country/continent contributed the most trial participants.
b Both treatment and control groups received background therapy and control group participants received matching placebo; information not stated from studies and supplementary information.
c Background therapy: diet alone, atorvastatin only, or atorvastatin with ezetimibe.
d Background therapy: maximally tolerated statin with or without other lipid-lowering therapy.
e Background therapy: moderate- or high-intensity statin.
f Background therapy: moderate- or high-intensity statin (at least atorvastatin 20 mg) with or without ezetimibe.
g Background therapy: high-dose statin or maximally tolerated statin.
h Although stated, it was a 104-week trial, there is no separate article describing results from this end point to date.
The number of new-onset diabetes mellitus cases was collected as the primary outcome for this study. The definition of incident diabetes mellitus is stated in each trial and includes the patient having had 2 consecutive FPGs of ≥126 mg/dL (7.0 mmol/L) and HbA1c ≥ 7.0% or requiring an antidiabetic agent. Two reviewers (S.W.C. and C.M.P.) independently performed bias risk assessment for each trial using the Cochrane Collaboration tool (Appendix 2).
Data Synthesis and Analysis
The effect size measure used was the risk ratio (RR) for diabetes incidence. Risk ratios were calculated using the sample sizes and number of reported occurrences of each of the aforementioned outcomes. For outcomes where none of the patients in one of the trial arms experienced the event of interest, the Haldane−Anscombe correction 20 was applied to estimate the RR.
A random effects model was used for the primary outcome. Heterogeneity in effect sizes was measured with I 2 and the Q statistic was used to test for excessive between-study heterogeneity. Forest plots were generated to display the effect sizes of each study along with the weighted aggregate effect size. Publication bias was assessed using Egger’s regression test. A sensitivity analysis was conducted where the model was rerun with each study removed one at a time. Analyses were performed using the “metan” package in Stata v14.2 (StatCorp).
Results
Table 1 lists the characteristics of each trial that qualified per inclusion and exclusion criteria (Appendix 3 for reference). Tables 2 and 3 present the baseline characteristics of the PCSK9i and ezetimibe trials, respectively. Almost all trials (9/10) reported results within the last 5 years. All trials were multisite with 4 trials covering different continents. Eight trials used the PCSK9i and 3 used ezetimibe (ODYSSEY COMBO I included both; a total of 10 trials were analyzed). Dosage level for the PCSK9i ranged from 75 mg every 2 weeks to 150 mg every 2 weeks for alirocumab and 140 mg every 2 weeks to 420 mg every 4 weeks for evolocumab (Table 1). The dosage for ezetimibe was 10 mg daily, which was either given alone (ODYSSEY COMBO II, NCT01644188), combined with simvastatin 20 mg into one pill form (Vytorin) with or without 2 g of niacin (trial 0653A-091, NCT00271817), or with simvastatin 40 mg daily in 1 pill (IMPROVE-IT trial, NCT00202878). For the PCSK9i trials, a total of 52 214 participants were included, of whom 26 887 received a PCSK9i (25 327 assigned control treatment). The ezetimibe trials included 20 084 participants, of whom 10 256 received a regimen that included ezetimibe (9828 assigned control). Depending on the trial, control treatment groups received either diet alone, a statin alone, a statin with ezetimibe, or niacin.
Baseline Characteristics for PCSK9i Trials.

Abbreviations: AA, African Americans; BMI, body mass index; FPG, fasting plasma glucose; HbA1c, hemoglobin A1c; HDL, high-density lipoprotein; LDL, low-density lipoprotein; N, number; NA, information not available from original article or supplemental material; PCSK9i, proprotein convertase subtilisin/kexin 9 inhibitor; TC, total cholesterol; TG, triglycerides.
a Characteristics of the nondiabetic group were selected.
Baseline Characteristics for Ezetimibe Trials.

Abbreviations: AA, African Americans; BMI, body mass index; E/S, ezetimibe/statin group; E/S/N, ezetimibe/statin/niacin group; FPG, fasting plasma glucose; HbA1c, hemoglobin A1c; HDL, high-density lipoprotein; LDL, low-density lipoprotein; N, number; NA, information not available from original article or supplemental material; TC, total cholesterol; TG, triglycerides.
For all trials, the risk of bias was determined to be low. For the 7 categories of bias listed in the Cochrane assessment tool, every category that could be judged was considered low for all trials. The results of the Egger regression revealed no evidence of publication bias (P values > .10 for all outcomes).
PCSK9i
Of the 26 887 patients receiving a PCSK9i, 1399 patients developed diabetes. There were 1365 cases of incident diabetes in the control group, giving a total of 2764 cases of new-onset diabetes overall. Figure 2 shows the forest plot for the outcome diabetes mellitus incidence. None of the individual trials showed a significant difference between the treatment and comparison arms, and there was no evidence of study heterogeneity (I 2 = 0.0, P = .78). The combined overall effect was near one (RR = 0.99, 95% CI = 0.92-1.07, P = .87) indicating PCSK9i do not increase the risk of diabetes. The sensitivity analysis did not change the results; the CI consistently contained 1 for each trial removed from the model. The largest change occurred when the ODYSSEY OUTCOME trial was excluded (RR = 1.03, 95% CI = 0.94-1.15, P = .50).
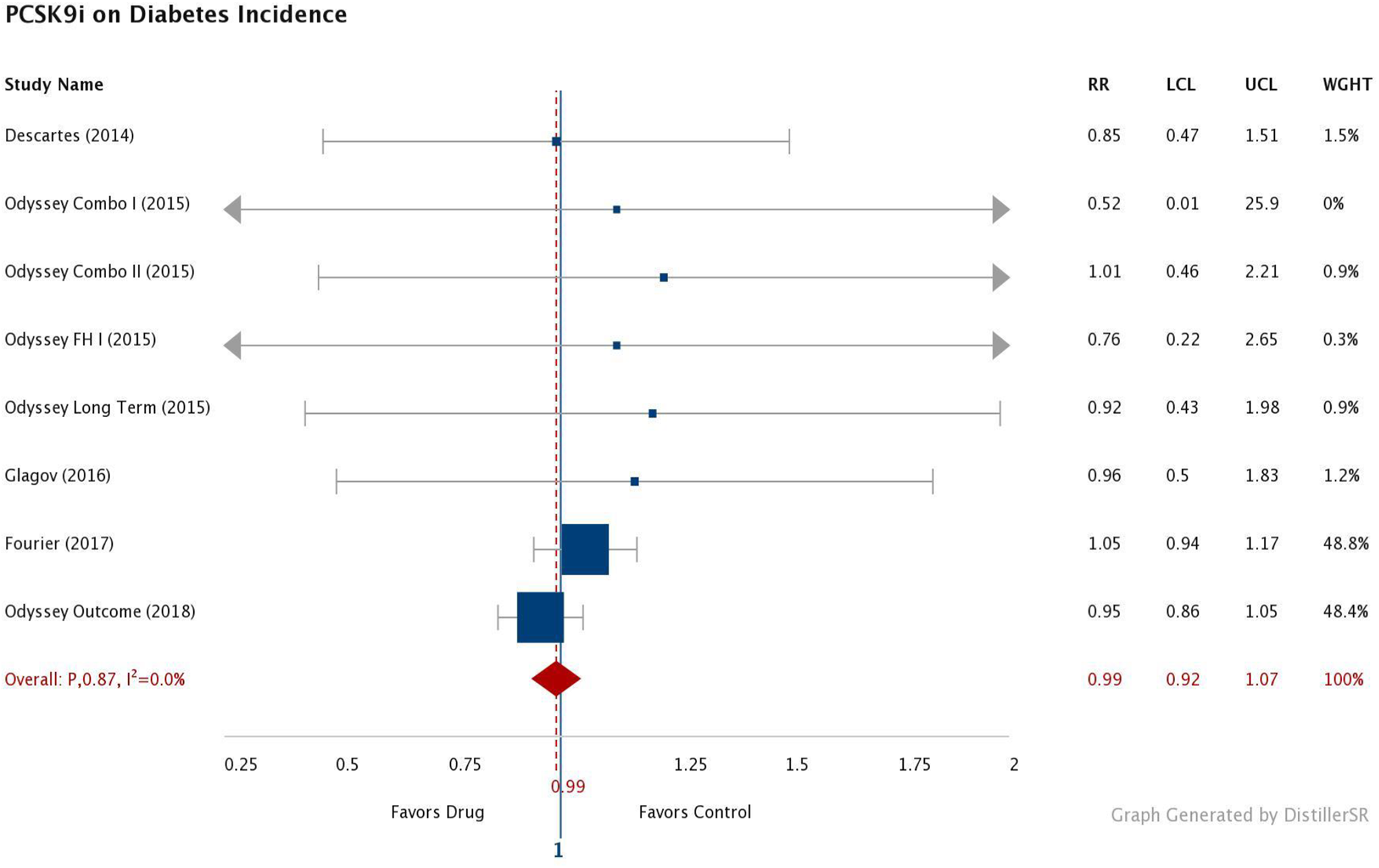
Incident diabetes mellitus in PCSK9i trials. LCL indicates lower confidence level; PCSK9i, proprotein convertase subtilisin/kexin 9 inhibitor; RR, relative risk; UCL, upper confidence level; Wt, weight of each study.
Ezetimibe
There were 764 cases of new diabetes mellitus out of the 10 256 patients assigned to ezetimibe. Overall, 717 assigned to the control group developed diabetes mellitus; a total of 1481 patients had new diabetes mellitus. Figure 3 shows the forest plot for diabetes incidence. There was no evidence of study heterogeneity (I 2 = 0.0, P = .38) and the IMPROVE-IT trial overshadowed the other 2 trials, where it was weighted over 97% in the analysis. The combined overall effect was near one (RR = 1.05, 95% CI = 0.95-1.15, P = .37) indicating ezetimibe does not increase the risk of diabetes. The sensitivity analysis did not change the results. Removal of the IMPROVE-IT study increased the RR slightly, but the CI was much wider and still contained one (RR = 1.33, 95% CI = 0.73-2.43, P = .35).
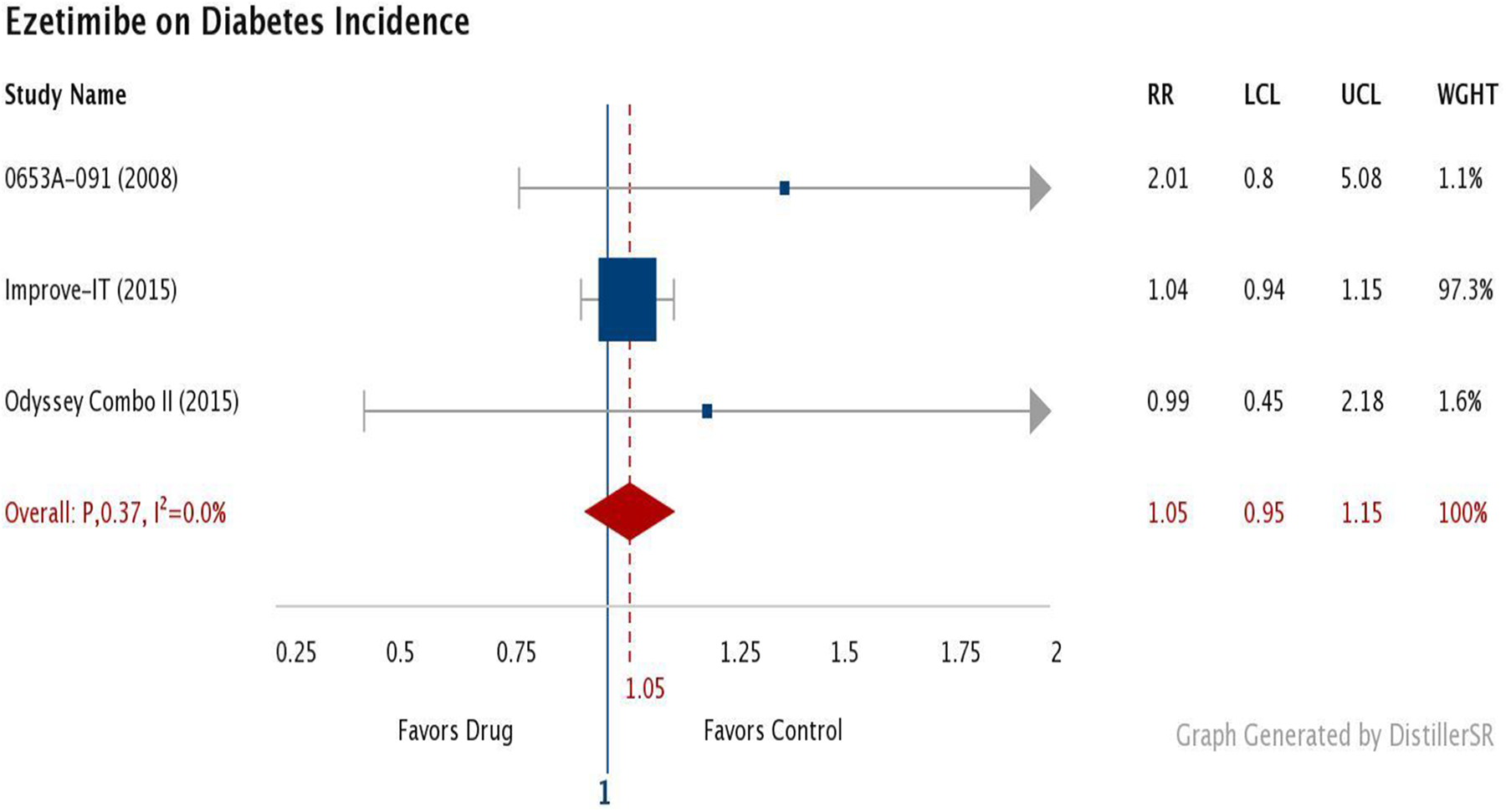
Incident diabetes mellitus in ezetimibe trials. LCL indicates lower confidence level; RR, relative risk; UCL, upper confidence level; Wt, weight of each study.
Discussion
This meta-analysis involving the usage of PCSK9i over 1 year demonstrates that this class of medications does not significantly impact the risk of new-onset diabetes compared to the control group. Similarly, ezetimibe had no associated risk of new-onset diabetes. Our meta-analysis had relative risk estimates close to 1 (0.99 for PCSK9i and 1.05 for ezetimibe) with CIs near ± 0.10.
Proprotein Convertase Subtilisin/Kexin 9 Inhibitor
There are many published clinical studies involving PCSK9i; however, many had a short follow-up time of 8 to 24 weeks. Other studies were not included in this meta-analysis because of study design; either they did not provide measurements of glycemic levels (ie, HbA1c, FPG) in their publications or were not randomized, double-blinded trials. Three meta-analyses that included these studies determined that PCSK9i alirocumab and evolocumab do not result in increased incidence of diabetes mellitus or elevate glycemic levels. 3,13,14 One recent meta-analysis by Chen et al reported that PCKS9i are generally not associated with new-onset diabetes mellitus. However, they also noted a few interesting findings: (1) that alirocumab and evolocumab appear to have a lower risk of incident diabetes compared to ezetimibe (which was used as the lipid-modifying drug in the control group) and (2) that there appeared to be an increase in incident diabetes in alirocumab and evolocumab when the use of statins was the same between the experimental and active comparator arms. 16 One meta-analysis determined that PCSK9i administration did not increase the risk of incident diabetes, yet it significantly increased FPG and HbA1c in the setting of a mean follow-up time of 1.5 years. 17 Another study evaluating patients already diagnosed with diabetes mellitus concluded that PCSK9i do not affect glucose metabolism. 21
Interestingly, there have been mixed findings regarding genetic studies. One study investigating the loss-of-function PCSK9 activity has shown that these subjects have reduced LDL but higher fasting blood glucose and thus are at risk of diabetes. 22 This difference seen between genetic PCSK9 variants and PCSK9i may be due to the fact that genetic variants affect both intracellular and extracellular PCSK9, while PCSK9i only affect the extracellular form. 21 However, another study analyzing the effects of variants in PCSK9 and 3-hydroxy-3-methylglutaryl-coenzyme A reductase (HMGCR which is the target of statins) demonstrated that the increased risk of incident diabetes occurred in patients with impaired fasting glucose levels. Moreover, this study emphasized that although there is potentially an association for new-onset diabetes, the proportional reduction in cardiovascular risk outweighed the increased risk of diabetes. 23
Although we declare that PCSK9i do not significantly impact the risk of incident diabetes mellitus, it is imperative to involve the patient in clinical decision-making, especially if the patient is already at risk of acquiring diabetes mellitus, to decide whether to initiate PCSK9i therapy. Having diabetes mellitus increases the risk of stroke and myocardial infarction. However, given that PCSK9i are known to decrease the risk of major cardiovascular adverse events, stroke, and myocardial infarction, administering these medications may provide more benefit than harm (eg, low possibility of acquiring diabetes mellitus). PCSK9i should be considered if the patient develops a true intolerance to statins or is refractory to statin therapy. One major downside is the high cost of PCSK9i.
Ezetimibe
Ezetimibe inhibits Niemann-Pick C1-Like 1 (NPC1L1), an LDL cholesterol transporter, thus effectively lowering lipid levels when administered with statins. 7,8,24 A meta-analysis determined that genetic variants in or near the NPC1L1 gene were associated with a higher risk of type 2 diabetes mellitus. 25 One retrospective study in Greece determined that high-intensity statins cause an increased risk of diabetes in prediabetics, yet the addition of ezetimibe to statins has an overall neutral effect on glucose. 26 Results from Wu et al demonstrated that ezetimibe did not increase glycemic levels, and in fact, ezetimibe with low-dose statin therapy for more than 3 months may even improve glycemic control. 18 A pooled data analysis determined that adding ezetimibe to statin monotherapy did not increase FPG levels for up to 96 weeks of treatment. 27 In a recent frequentist network meta-analysis (which essentially illustrates the probability of events by testing significance with P value, CI; in contrast to the Bayesian model which uses subjective prior knowledge for statistical inference), the researchers concluded that statins were associated with an increase of incident diabetes while PCSK9i and ezetimibe were not. 19
Limitations
There were several limitations in this meta-analysis. First, this meta-analysis was performed without access to individual patient data. Thus, we were not able to execute subgroup analyses or elucidate the extent of variations within patient groups if they existed. Second, most of the trials analyzed had mostly white males as patients. The results may not be generalizable to other populations such as females and other ethnicities (ie, African Americans and Asians). Third, the results of ezetimibe must be considered with concomitant use of a statin as it is unusual for patients to take only ezetimibe for lipid-lowering purposes. Fourth, the mechanisms of how PCSK9i and ezetimibe affect glycemic levels need to be elucidated. Fifth, although we used the inclusion criteria of 52-week follow-up, this is still considered a short period. This was the best time frame to our knowledge based on the availability of studies. For instance, the Sattar et al meta-analysis on statins was a mean follow-up time of approximately 4 years. 9 This can be explained by the fact that statins have been used for many years (the first being lovastatin which was approved by the FDA in 1987), while the PCSK9i alirocumab and evolocumab were recently approved in 2015. In addition, our study used stricter inclusion and exclusion criteria compared to other studies such as requiring such a time frame of at least 1 year, an adequate sample size of at least 100 participants per treatment arm and excluding nonrandomized controlled trials, placebo-controlled, open-label, and crossover trials. Thus, even though our study had a shorter time frame than the statin trials, our meta-analysis is the only one to date that required a longer time frame and sample size for better quality analysis. Furthermore, a few trials—specifically FOURIER, ODYSSEY OUTCOMES, and IMPROVE-IT—contributed the most weight to the studies as shown by the forest plots in Figures 2 and 3. Finally, there were only 3 studies eligible for the ezetimibe analysis per our inclusion and exclusion criteria. The ezetimibe meta-analysis could be considered underpowered and the upper limit of the CI (1.15) encompassed the significant effect size found by Sattar et al (odds ratio = 1.09). However, any potential clinical risk, if exists, would be low by either estimate. For ezetimibe, the number needed to harm is over 650 patients, and in Sattar, it is near 300. 9
In this systematic review and meta-analysis of large, randomized controlled trials with a follow-up of at least 1 year, we conclude that PCSK9i and ezetimibe do not affect the risk of incident diabetes mellitus.
Supplemental Material
Supplemental Material, Appendix_1_(2) - Proprotein Convertase Subtilisin/Kexin Type 9 Inhibitors and Ezetimibe on Risk of New-Onset Diabetes: A Systematic Review and Meta-Analysis of Large, Double-Blinded Randomized Controlled Trials
Supplemental Material, Appendix_1_(2) for Proprotein Convertase Subtilisin/Kexin Type 9 Inhibitors and Ezetimibe on Risk of New-Onset Diabetes: A Systematic Review and Meta-Analysis of Large, Double-Blinded Randomized Controlled Trials by Sarah W. Chiu, Cristina M. Pratt, Richard Feinn and Saurav Chatterjee in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Authors’ Notes
This work was performed at the Frank H. Netter MD School of Medicine at Quinnipiac University in North Haven, Connecticut, USA.
Author Contributors
S.W.C. was involved in study design, data collection, and writing the manuscript; C.M.P. was involved in data collection; R.F. was involved in data analysis and manuscript revision; and S.C. was involved in study design, data analysis, and manuscript revision.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
