Abstract
Background:
Published data indicated that combination use of clopidogrel and proton pump inhibitors (PPIs) may increase the incidence of major adverse cardiovascular events (MACEs). This has been a highly controversial topic for years.
Design:
The present study was performed to evaluate whether combination therapy of clopidogrel and PPIs is associated with increased risk of MACEs than with clopidogrel alone in patients with coronary artery disease.
Methods:
A systematic search of MEDLINE, EMBASE, and the Cochrane Library was conducted for studies recording the occurrence of MACEs in patients with exposure to concomitant use of clopidogrel and PPIs up to February 2015. Odds ratios (ORs) were combined using a random-effects model.
Results:
Patients receiving combination therapy with PPIs and clopidogrel were at significantly increased risk of MACEs (OR: 1.42; 95% confidence interval [CI]: 1.30-1.55). Adding a PPI to clopidogrel treatment was associated with a higher rate of MACE occurrence in rapid metabolizers (RMs, *1/*1) of CYP2C19 (OR: 1.42; 95% CI: 1.12-1.81), but there was no obviously increased rate (OR: 1.43; 95% CI: 0.89-2.28) in decreased metabolizers (with 1 or 2 loss-of-function allele). The increased risk of MACEs was similar in 4 classes of PPIs (omeprazole, lansoprazole, esomeprazole, and pantoprazole), but rabeprazole (OR: 1.03; 95% CI: 0.55-1.95) wasn’t.
Conclusion:
The combination use of clopidogrel and certain types of PPIs (omeprazole, lansoprazole, esomeprazole, pantoprazole) increases the risk of MACE in patients with coronary artery disease. Only in the RMs of CYP2C19, PPIs were associated with significantly increased MACE in patients coadministered with clopidogrel.
Keywords
Introduction
As known, clopidogrel is an antiplatelet agent commonly used to reduce the incidence of cardiovascular events among patients with coronary artery disease. These patients often receive dual antiplatelet therapy to reduce the incidence of cardiovascular events, according to the recommendations of the American Heart Association (AHA) and the American College of Cardiology (ACC). 1 Unfortunately, the antiplatelet therapy often comes with the increased risk of gastrointestinal bleeding. Therefore, proton pump inhibitors (PPIs) are frequently administered to patients under the antiplatelet therapy to reduce the potential risk of gastrointestinal bleeding. 2 However, in recent years, some studies have showed that PPIs will reduce the antiplatelet effects of clopidogrel and increase the risk of major adverse cardiovascular events (MACEs). 3 -5 Both the US Food and Drug Administration (FDA) and the European Medicines Agency have published warnings against the coadministration of clopidogrel and PPIs. 6,7 So, there is a growing awareness of the risk of combination use of clopidogrel and PPIs in patients on antiplatelet therapy.
Four previous meta-analysis revealed that the concomitant use of PPIs and clopidogrel in patients who needed antiplatelet therapy was associated with an increased risk of adverse cardiovascular events. 8 -11 However, another 2 meta-analyses reached the conclusion that there was a lack of significant interactions between clopidogrel and PPIs therapy, 12,13 and 1 indicated that there were conflicting and inconsistent data regarding the adverse clopidogrel–PPI interaction. 14 Therefore, the cardiovascular risk of combining clopidogrel with PPIs in patients with coronary artery disease needs to be identified further. Meanwhile, in recent years, people are much more concerned with the influence of genotype on the antiplatelet effects of clopidogrel. Previous studies showed that carriers of the loss-of-function hepatic cytochrome P450 enzyme 2C19 (CYP2C19) allele displayed a reduced pharmacodynamic response to clopidogrel and reduced antiplatelet effects and thus resulted in a higher recurrence rate of cardiovascular events compared with normal CYP2C19 genotype groups. 15,16 Thus, the influence of CYP2C19 genotype to cardiovascular events in patients with the concomitant use of clopidogrel and PPIs needs to be studied in detail. Besides, according to Kwok et al, the combination use of clopidogrel and all kinds of PPIs did not increase the risk of adverse cardiovascular events. 13 But the FDA recommended avoiding omeprazole and esomeprazole in patients taking clopidogrel as these 2 PPIs can interact with clopidogrel and result in worse clinical outcomes. 7 So, whether the individual PPIs confer different risks for MACE in patients receiving clopidogrel has not been detailed yet.
Accordingly, we performed the present study to evaluate whether the combination therapy of clopidogrel and PPIs causes higher numbers of MACE in patients with coronary artery disease, and 3 subgroup analyses were performed based on the CYP2C19 genotype, the types of PPIs commonly used in clinics, and follow-up duration, respectively.
Methods
Trial Selection
Trials were selected from all published controlled clinical trials involving study groups administered with or without PPIs in addition to clopidogrel in patients with coronary artery disease. Both randomized controlled trials and nonrandomized controlled trials were included. Studies with articles reporting on the incidence of MACE in patients with coronary artery disease as primary or secondary end point were included. Studies that could not provide enough data for the meta-analysis even after statistical computing were excluded.
Search Strategy
We conducted a systematic search of MEDLINE, EMBASE, and the Cochrane Library for studies describing the occurrence of MACEs (cardiovascular death, nonfatal myocardial infarction, stroke, stent thrombosis, and revascularization) in patients with exposure to clopidogrel therapy and the concomitant use of PPI published before February 2015. The search themes are [proton pump inhibitor or PPI or omeprazole or esomeprazole or lansoprazole or pantoprazole or rabeprazole] AND [clopidogrel]. Meanwhile, we checked the references of the retrieved studies for additional studies. We considered reports on human studies published in any language. In addition, abstracts from the scientific sessions of the ACC, the AHA, the European Society of Cardiology, and the North American Society of Pacing and Electrophysiology were manually searched, and the articles from the bibliographies of retrieved trials were scanned.
Two of the authors (Q.N. and Z.W.) independently examined the titles and abstracts of all trials to eliminate irrelevant studies. Subsequently, the same authors examined the full texts of the remaining articles and the full-text reports of all potentially relevant trials and assessed them independently for eligibility on the basis of the defined inclusion criteria. Trials were excluded if there was no mention of combination use of clopidogrel and PPIs in the control group, the clopidogrel was used as a background intervention, the population were healthy volunteers or without coronary artery disease, and there was no mention of the occurrence of MACE. The nonrandomized trials were assessed for methodological quality by the Newcastle Ottawa Scale as recommended by the Cochrane Non-Randomized Studies Methods Working Group. Data from the included studies were extracted in duplicate. Authors of the published trial results, including abstracts, were contacted for required information when needed. Any discrepancies were resolved by discussion. To resolve disagreements, a final decision for trial eligibility and data extraction was made by the senior author (Y.H.).
Statistical Analysis
Statistical analysis was performed based on the intent-to-treat principle, with participants not completing the study considered to be free of the event. RevMan 5.3 (RevMan; The Nordic Cochrane Centre, Copenhagen, Denmark) was applied, and a pooled effect was calculated with a random effects model (inverse variance method) for pooled odds ratio (OR). We assumed the similarity between the OR and risk ratio because MACEs were uncommon events. Statistical heterogeneity was assessed using I 2 statistics, with I 2 values of 30% to 60% representing a moderate level of heterogeneity.
The risk of MACE with clopidogrel exposure, with or without the concomitant use of PPIs, was analyzed. Three subgroup analyses were performed: first to evaluate the influences of different hepatic cytochrome P450 enzyme 2C19 genotype groups (with or without variant allele of CYP2C19 genotype groups), second to assess the effects of different kinds of PPIs (omeprazole or lansoprazole or esomeprazole or pantoprazole or rabeprazole) on cardiovascular risk, and third to analyze the clinical outcomes with different follow-up time.
A sensitivity analysis was performed by selectively excluding studies based on the quality assessment to check the consistency of the overall effect estimate. Funnel plots were created to determine the possible influence of publication bias.
Results
Search Results
In total, 863 relevant articles were retrieved from MEDLINE, EMBASE, and Cochrane Controlled Trails Register (CCRT). After the selection of these studies, 21 studies reporting on the differences in terms of MACE between patients with and without the combination use of PPIs were deemed eligible for our meta-analysis (Figure 1). 17 -34 Of the 21 studies, omeprazole was studied in 7, lansoprazole in 4, esomeprazole in 2, pantoprazole in 6, and rabeprazole in 3. Besides, 4 studies reported on the differences in MACE in patients with certain CYP2C19 genotype between clopidogrel alone and combination use with PPIs. When taking follow-up time into consideration, 3 studies were discontinued with 1 month, 13 with 1 year, and 6 studies with a follow-up longer than 1 year.

Work flow of studies included in meta-analysis.
Study Characteristics
Of the 97 696 patients enrolled, 60 326 were assigned to receive clopidogrel alone and 37 310 received the combined use of clopidogrel and PPIs. The characteristics of each trial are shown in Table 1.
Characteristics of Included Studies.
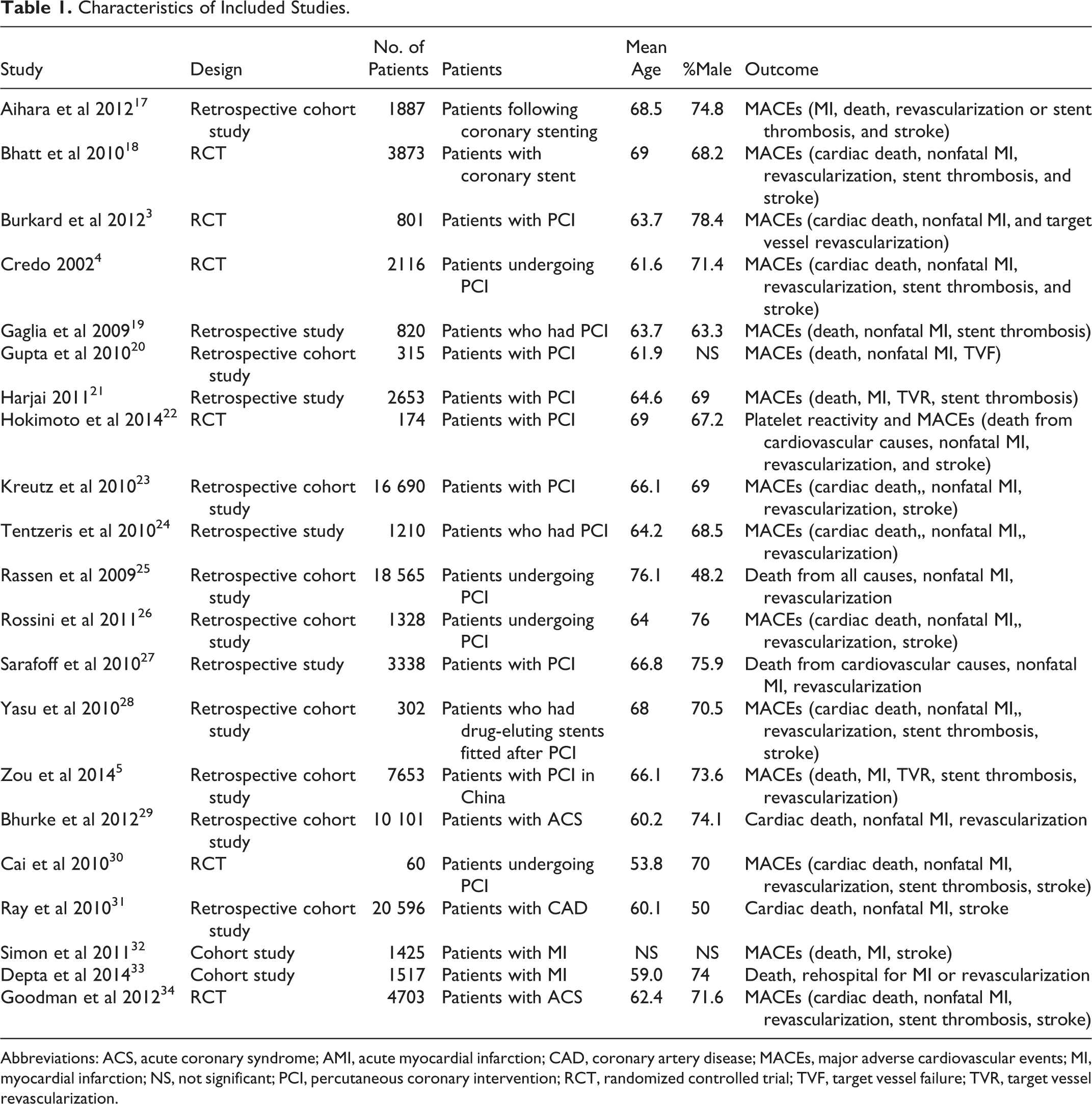
Abbreviations: ACS, acute coronary syndrome; AMI, acute myocardial infarction; CAD, coronary artery disease; MACEs, major adverse cardiovascular events; MI, myocardial infarction; NS, not significant; PCI, percutaneous coronary intervention; RCT, randomized controlled trial; TVF, target vessel failure; TVR, target vessel revascularization.
Meta-Analysis
The risk of MACE for the combination use of clopidogrel and PPIs is shown in Figure 2. The studies showed that patients receiving combination therapy with PPIs and clopidogrel were with significantly increased risk of MACE (OR: 1.42; 95% confidence interval [CI]: 1.30-1.55) compared to those with clopidogrel alone, with moderate heterogeneity (I 2 = 51%). When we consider stratifying by randomized and observational studies, the results were similar: patients receiving combination therapy with PPIs and clopidogrel were both with significantly increased risk of MACE in randomized studies (OR: 1.39; 95% CI: 1.17-1.66; I 2 = 25%) and observational studies (OR: 1.44; 95% CI: 1.32-1.57; I 2 = 64%). The MACE of the combination use of clopidogrel and PPIs in patients undergoing percutaneous coronary intervention (PCI) was studied in 15 of the 21 trials. Data were pooled and the results show significantly increased risk of MACE (OR: 1.47; 95% CI: 1.35-1.59) without substantial heterogeneity (I 2 = 14%; Figure 3).
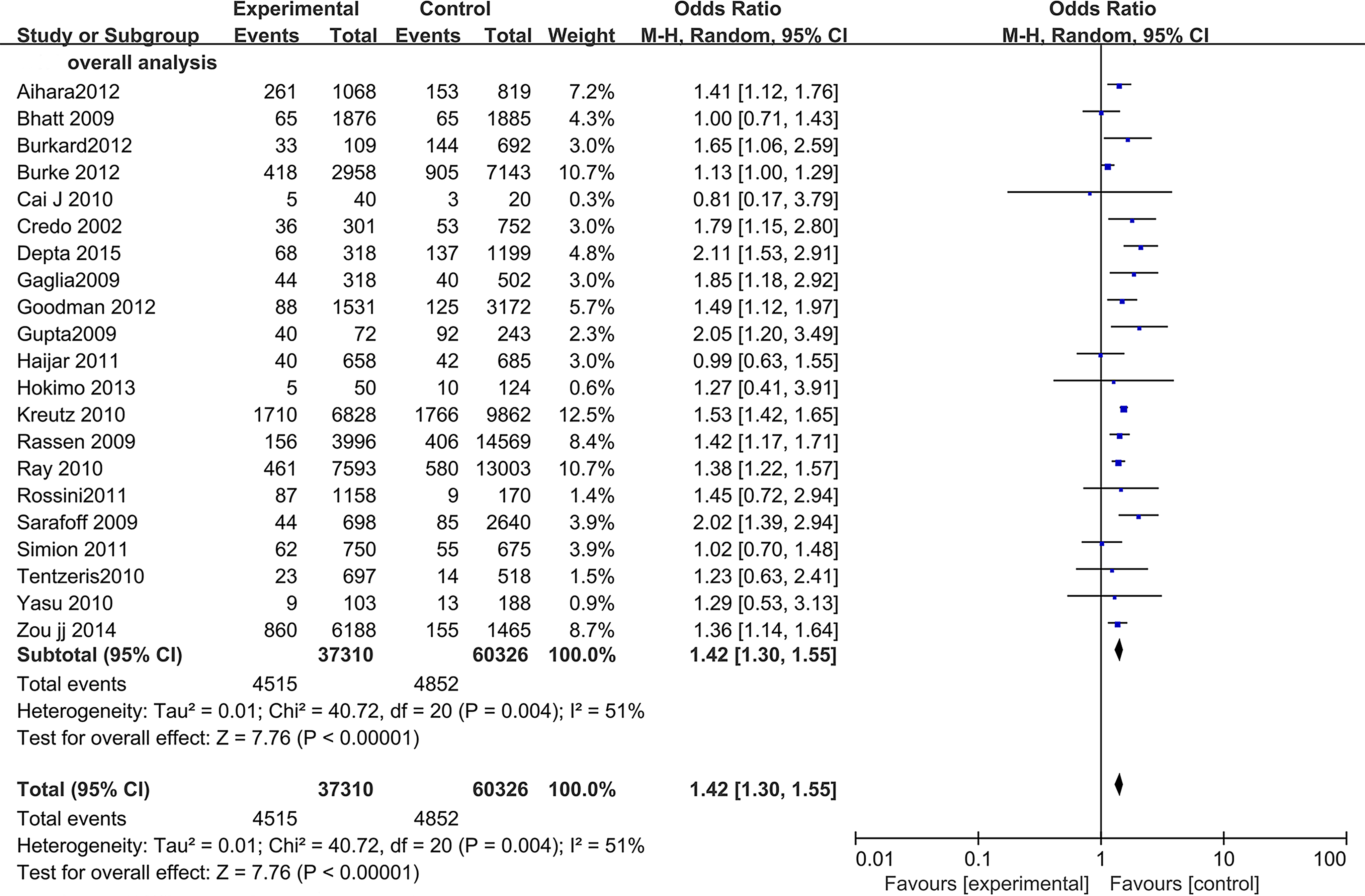
The risk of major adverse cardiac events in patients receiving clopidogrel–PPI therapy versus clopidogrel therapy. Experimental group received clopidogrel and PPIs treatment. Control group received clopidogrel alone. PPI indicates proton pump inhibitor.

The risk of major adverse cardiac events in patients undergoing PCI receiving clopidogrel–PPI therapy versus clopidogrel therapy. Experimental group received clopidogrel and PPIs treatment. Control group received clopidogrel alone. PCI indicates percutaneous coronary intervention; PPI, proton pump inhibitor.
A subgroup analysis assessed the impact of PPIs on the efficacy of clopidogrel in patients with or without variant allele of CYP2C19 genotype. The result is shown in Figure 4. This subgroup analysis showed an overall increased risk among patients without variant allele of CYP2C19 genotype groups (OR: 1.42; 95% CI: 1.12-1.81), and there is no substantial heterogeneity (I 2 = 0%). At the same time, among the patients with 1 or 2 variant allele of CYP2C19 genotype, there was no obviously increased cardiovascular risk (OR: 1.43; 95% CI: 0.89-2.28) and the heterogeneity was moderate (I 2 = 56%).

The risk of major adverse cardiac events in patients with confirmed CYP2C19 genotype receiving clopidogrel–PPIs therapy versus clopidogrel therapy. Experimental group received clopidogrel and PPIs treatment. Control group received clopidogrel alone. PPI indicates proton pump inhibitor.
Another subgroup analysis evaluating the risk of MACE for clopidogrel and individual PPIs is shown in Figure 5. Seven studies with omeprazole showed significantly increased risk of MACE (OR: 1.40; 95% CI: 1.15-1.70) with moderate heterogeneity (I 2 = 33%). Within 4 studies reporting on lansoprazole, 2 individual studies showed significant interaction between lansoprazole and clopidogrel. On average, the lansoprazole studies showed a significantly increased overall risk (OR: 1.51; 95% CI: 1.29-1.77) with no substantial heterogeneity (I 2 = 0%). Two studies reported on esomeprazole indicating an increased MACE risk (OR: 1.59; 95% CI: 1.29-1.95) with no substantial heterogeneity (I 2 = 23%). Furthermore, 6 studies reporting on pantoprazole yielded significantly increased risk of MACE (OR: 1.52; 95% CI: 1.13-2.05), limited by substantial heterogeneity (I 2 = 70%), whereas 3 studies reporting on rabeprazole yielded no significantly increased risk (OR: 1.03; 95% CI: 0.55-1.95) and no substantial heterogeneity (I 2 = 0%).
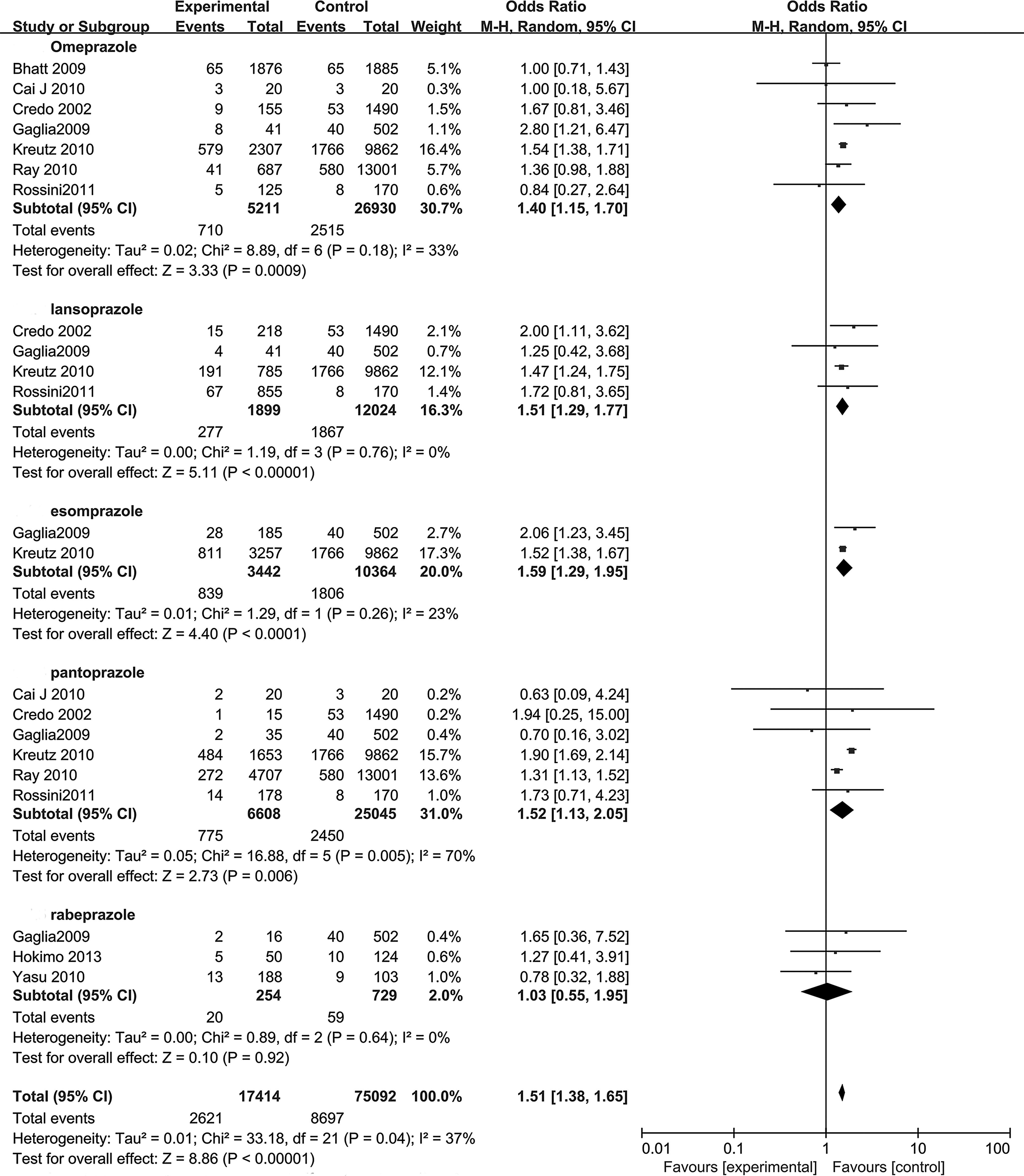
The risk of major adverse cardiac events in patients receiving different PPI versus without PPI. Experimental group received clopidogrel and PPIs treatment. Control group received clopidogrel alone. PPI indicates proton pump inhibitor.
Subgroup analysis was also performed in consideration of the follow-up duration (Figure 6). It was suggested that the risk of MACE was significantly increased in 1-month follow-up subgroup (OR: 1.90; 95% CI: 1.43-2.52), 1-year follow-up subgroup (OR: 1.43; 95% CI: 1.29-1.57), and >1-year follow-up subgroup (OR: 1.34; 95% CI: 1.14-1.58). There is no substantial heterogeneity (I 2 = 0%) in the 1-month follow-up subgroup, but the heterogeneity for >1-year (I 2 = 47%) and the 1-month to 1-year (I 2 = 40%) follow-up duration was moderate, respectively.

The risk of major adverse cardiac events in patients with different follow-up duration receiving clopidogrel–PPI therapy versus clopidogrel therapy. Experimental group received clopidogrel and PPIs treatment. Control group received clopidogrel alone. PPI indicates proton pump inhibitor.
Publication Bias
Funnel plots were visually symmetrical, suggesting no significant publication bias among the studies (Figure 7).

Funnel plots were visually symmetrical, suggesting no significant publication bias among the studies.
Discussion
Patients often receive antiplatelet therapy following coronary artery disease to reduce the incidence of MACE. Meanwhile, PPIs are frequently administered to patients under the antiplatelet therapy to reduce the risk of gastrointestinal bleeding. The combination use of clopidogrel and PPIs is frequently encountered in clinic. The aim of our meta-analysis is to estimate the association between the combination use of clopidogrel and PPIs and the risk of MACE. In our meta-analysis, we found that the combination use of clopidogrel and PPIs, compared with using clopidogrel alone, was associated with significantly increased risk of MACE in patients with coronary artery disease. Furthermore, the hazard of MACE in patients undergoing PCI is even more striking. Patients should need a long-term antiplatelet therapy after PCI, and the combination of clopidogrel and PPIs for these patients is most commonly used in clinical practice to reduce the incidence of cardiovascular events and gastrointestinal bleeding. So, we focus on the hazard in the certain group of these patients. And the result showed that the combination use of clopidogrel and PPIs significantly increases the risk of MACE in patients undergoing PCI. Four previous meta-analyses obtained a conclusion of visible influence of PPIs on the antiplatelet activities of clopidogrel, which is consistent with our results. 8 -11 And the reason could be that clopidogrel is a prodrug that requires transformation into an active metabolite by the hepatic cytochrome P450 enzyme 2C19 (CYP2C19) for its antiplatelet effect of irreversible binding to the platelet adenosine diphosphate receptor. 35 Meanwhile, PPIs convert to their active metabolites in the gastric parietal cell and undergo hepatic metabolism via cytochrome P450 enzymes, including CYP2C19. 7 So, there is a competition for CYP2C19 between clopidogrel and PPIs, and as a result, the active product and the antiplatelet effect of clopidogrel was considered to be reduced by the interaction of PPIs in coronary artery disease. As a consequence, we suggested that clinicians should pay attention to potential harm from the concomitant use of PPIs and clopidogrel and avoided the combination use of PPIs and clopidogrel unnecessarily.
CYP2C19 Genotype
In recent years, people are more and more concerned with the influence of genotype on the effect of drugs. We focus mainly on the influence of CYP2C19 genotype at this point. As known, the cytochrome variants CYP2C19*2 and CYP2C19*3, different from the normal CYP2C19*1 by 1 single nucleotide only, are considered as poor metabolizers characterized by a loss or severely decreased enzyme activity. The frequency for the most common loss-of-function variant CYP2C19*2 is <15% in Caucasians and Africans and is found more frequently in Asian populations (35%). The CYP2C19*3 allele is also more frequent in Asian populations (10%) compared with other racial groups (1%). 36 Individuals carrying at least 1 loss-of-function allele (either *2 or *3) of the CYP2C19 gene displayed a reduced pharmacodynamic response to clopidogrel and reduced antiplatelet effects and thus resulted in a higher recurrence rate of cardiovascular events compared with normal CYP2C19 genotype groups. However, previous data on the PPI use among clopidogrel-treated patients with CYP2C19 gene mutation were controversial and limited. A recent randomized crossover study of healthy *1 homozygotes demonstrated that all PPIs decreased the peak plasma concentration of clopidogrel active metabolite (omeprazole > esomeprazole > lansoprazole) and showed a corresponding order of potency for effects on maximal platelet aggregation and platelet response units. 37 Meanwhile, the conclusion of 3 recent studies that assessed the association between the PPIs use and the platelet function in people within certain CYP2C19 genotype groups indicated that in decreased metabolizers (DMs, carriers of *2 and/or *3) of CYP2C19, PPIs didn’t significantly attenuate the antiplatelet function of clopidogrel but did so in rapid metabolizers (RMs; *1/*1) of CYP2C19. 38 -40 And these studies provided evidence of the inhibition of the clopidogrel’s antiplatelet activities by PPIs, demonstrating increased platelet activities through various testing methods compared with using clopidogrel alone. Our second subgroup analysis explored the influence of PPIs on the antiplatelet effect of clopidogrel in people with certain CYP2C19 genotype. Through the subgroup analysis, we found that adding a PPI to the clopidogrel treatment is associated with a higher occurrence rate of MACE only in patients without variant allele of CYP2C19. But in the patients with 1 or 2 loss-of-function allele, there was no obviously increased rate of MACE. The mechanism of this may be that in the DMs of CYP2C19, efficacy of clopidogrel is decreased as a result of the reductive active metabolites in comparison with RMs. On the other hand, many PPIs are known to induce CYP1A2, which also plays an important role in metabolizing clopidogrel to the active metabolites in individuals with lower activity of CYP2C19. 35,41 The activation of CYP1A2 will make up the competition for CYP2C19 between clopidogrel and PPIs and result in insignificant differences between clopidogrel–PPIs combination use and clopidogrel therapy alone in DMs. While in RMs of CYP2C19, the metabolism of PPIs and clopidogrel is unaffected by genotype, arriving at the same conclusion with our overall analysis and indicating a somewhat more illuminating conclusion of interaction of PPIs and clopidogrel. Unfortunately, because of the limited number of trials, we didn’t distinguish among specific variant alleles such as *2 carrier, *3carrier, or *17 carrier. A recent study showed that no obvious difference in the adverse cardiovascular event incidence was observed between carriers of the CYP2C19*2 allele and normal CYP2C19 genotype, but the CYP2C19*17 carriers using PPIs have greater potential to significantly lower enzymatic activities, resulting in reduced clopidogrel-related platelet inhibition and worse clinical outcomes. 33 Overall, we suggest avoiding the combination use of clopidogrel and PPIs in patients without loss-of-function CYP2C19 alleles and instead choosing other appropriate therapeutic regimens as mentioned above. However, the number of involving trials was small, so further large-scale studies are needed to effectively guide therapeutic decisions.
Proton Pump Inhibitor Species
Our second subgroup analysis showed that omeprazole, lansoprazole, esomeprazole, and pantoprazole obviously increase the risk of MACE, but rabeprazole didn’t. A previous meta-analysis indicated that there was no consistent evidence of differential cardiovascular risk among PPIs when used with clopidogrel, 13 which was in discordance with the conclusion of our study. We have shown that omeprazole, esomeprazole, lansoprazole, and pantoprazole were metabolized mainly by CYP2C19 enzyme, however, the hepatic metabolism of rabeprazole involved both CYP-mediated and nonenzymatical metabolism, with the latter taking the dominant role. 42 As we have discovered, the increase in MACE risk was mainly contributed to the competition for CYP2C19 between clopidogrel and PPIs. Compared with the other 4 PPIs, rabeprazole is a weaker competitive inhibitor for CYP2C19 and have a negligible effect on the metabolism of clopidogrel. Consequently, the risk of MACE obviously increased in omeprazole, lansoprazole, esomeprazole, and pantoprazole subgroup but not in rabeprazole subgroup. Therefore, when patients need antiplatelet therapy and protection of gastric mucosa in clinic, we suggest the combination use of rabeprazole and clopidogrel or strategies to avoid the clopidogrel–PPIs interaction including the use of an H2 antagonist (H2RA) instead of PPIs or the use of ticagrelor or prasugrel instead of clopidogrel. Due to the small number of trials of esomeprazole and rabeprazole, more trials are needed in the future to support this conclusion.
Follow-Up Duration
Furthermore, we found that the combination use of clopidogrel and PPIs would increase the risk of MACE whether with 1-month, 1-year, or more than 1-year follow-up time. And there was no distinct discrepancy between different follow-up duration. The reason is inconclusive and more trials are needed in the future to support this conclusion.
Limitations
Our study has several limitations. Firstly, the presence of a significant statistical heterogeneity in this meta-analysis might indicate that the evidence is biased, confounded, or inconsistent. Secondly, definitions of MACE may have slight difference in each study, which may create bias too. Thirdly, the number of trials in some subgroup analysis are small, and further large-scale studies are needed. Fourthly, we used the adjusted OR/hazard ratio if provided, and it is most likely different from study to study.
Conclusion
In conclusion, the result of our meta-analysis supports the notion that the combination use of clopidogrel and PPIs will increase the risk of MACE in patients with coronary artery disease, which is in accordance with the pharmacokinetic and pharmacodynamic studies, and the same is true for patients undergoing PCI. Only in the RMs (*1/*1) of CYP2C19, PPIs were associated with significantly increased MACE in patients coadministered with clopidogrel. Rabeprazole is less likely to increase the risk of MACE compared with other PPIs.
Footnotes
Author Contributions
Qiang Niu and Zhongsu Wang contributed equally to this article, contributed to conception and design, acquisition, analysis, interpretation, drafted the manuscript, and critically revised the manuscript. Yong Zhang, Jiangrong Wang, Pei Zhang, Wangcong Yin, and Xiangcui Yin contributed to conception and analysis. Yinglong Hou contributed to conception, design, analysis, interpretation, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. All the authors read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Science Foundation of China (81270237), the Special Research Fund for the Doctoral Program of Higher Education (20110131110063), Science and Technology Development Planning of Shandong Province (2014GSF118180), and Shandong Taishan Scholarship (Yinglong Hou).
