Abstract
Background:
Antiplatelet (AP) therapy is well established for the secondary prevention of acute coronary events. However, patients may discontinue treatment, often owing to gastrointestinal (GI) complications, leaving them at elevated risk of recurrent cardiovascular events.
Objectives:
This descriptive retrospective study assessed trends in prescription of AP agents and coprescription of gastroprotective therapy, after an acute coronary event. Discontinuation of AP therapy within 2 years of an event and factors predicting discontinuation were investigated.
Methods:
The study was conducted in a UK primary care setting from 2000 to 2008; a total of 27 351 patients aged 50 to 84 years were included in the analysis. Main outcome measures were exposures to low-dose acetylsalicylic acid (ASA), clopidogrel, and proton pump inhibitors (PPIs).
Results:
At 90 days after an acute coronary event, 85.9% of patients had been prescribed some form of AP therapy and 33.6% of patients who were issued at least 1 ASA prescription in this period were also issued a PPI prescription. The use of dual antiplatelet therapy (DAT) 90 days after an event increased from 2% in 2000 to over 50% in 2008. An estimated 15.1% of patients on ASA monotherapy and 37.5% on DAT discontinued treatment within 1 year. A bleeding event during follow-up, including upper GI bleeding or hemorrhagic stroke, was the strongest predictor of discontinuation.
Conclusion:
Although most patients were prescribed AP therapy in the 90 days following an acute coronary event, a substantial proportion discontinued DAT or ASA monotherapy within 1 year. It is essential that physicians consider strategies to reduce the risk of discontinuation of AP therapy.
Keywords
Introduction
Antiplatelet (AP) therapies are well established for the secondary prevention of acute coronary events. Current guidelines recommend the use of dual antiplatelet therapy (DAT), specifying low-dose acetylsalicylic acid (ASA) in combination with a P2Y12 inhibitor, such as clopidogrel, for 12 months and ASA continued in the long term. 1,2 However, patients may discontinue such treatments, often owing to gastrointestinal (GI) complications, 3 –6 leaving them at elevated risk of recurrent cardiovascular (CV) events. 7 –9
The aims of this study were to assess the use of low-dose ASA and other AP agents 90 days after an acute coronary event in routine clinical practice, and to estimate the rate of discontinuation of AP monotherapies and DAT 1 and 2 years after an event. We also investigated patient characteristics that predicted an increased risk of discontinuation. In addition, coprescription of gastroprotective therapy with ASA during the study period (2000-2008) was analyzed.
Methods and Materials
Data Source
A descriptive retrospective cohort study was performed using data from The Health Improvement Network (THIN), a computerized primary care database containing anonymized records for approximately 4 million individuals currently registered with participating primary care practices in the United Kingdom. Data include patient demographics, details of consultations with primary care physicians, information about consultant referrals and hospitalizations, laboratory test results, diagnoses, and prescriptions.
Study Population
A previous study identified patients aged 50 to 84 years who had been hospitalized for an acute coronary event (acute myocardial infarction [MI], revascularization of coronary arteries, or unstable angina) between 2000 and 2008; 10 this population constituted the study cohort.
Data Collection and Analysis
Exposure to low-dose ASA, clopidogrel, dipyridamole, warfarin, and proton pump inhibitors (PPIs) after an acute coronary event was evaluated. Exposure at the date of the qualifying event was classified as current use (when the supply of the most recent prescription lasted until the date of the qualifying event or ended in the previous 30 days) or as nonuse (when the supply of the most recent prescription ended more than 31 days before the date of the qualifying event or there was no recorded use at any time).
To explore treatment patterns immediately after an acute coronary event, prescriptions were analyzed between the date of the qualifying event and 90 days after the event. Individuals were classified into 4 mutually exclusive categories based on these data: nonusers (not current users at the date of the qualifying event and received no prescription in the 90 days after the event); initiators (not current users at the date of the qualifying event but were issued at least 1 prescription in the 90 days after the event); continuers (current users at the date of the qualifying event and were issued at least 1 prescription in the 90 days after the event); and discontinuers (current users at the date of the qualifying event but were not issued any prescription in the 90 days after the event).
Determinants of Type of AP Therapy
To investigate determinants of AP therapy use 90 days after an acute coronary event, 5 cohorts were defined within the overall study population. The ASA monotherapy cohort included patients who were issued a prescription for ASA in the 90 days after their acute coronary event but were not issued a prescription for clopidogrel in the 90 days after the first ASA prescription. Patients in the clopidogrel monotherapy cohort comprised those who were issued a prescription for clopidogrel in the 90 days after their acute coronary event but were not issued a prescription for ASA in the 90 days after the first clopidogrel prescription. The DAT cohort included individuals who were issued prescriptions for ASA and clopidogrel in the 90 days after the acute coronary event, with the first prescription of each drug occurring no more than 30 days apart and a second prescription of each drug occurring no more than 90 days after the end of the supply of the respective first prescription. The other AP therapy cohort comprised patients who were issued ASA and/or clopidogrel prescriptions in the 90 days after their acute coronary event but who did not meet the criteria for any of the first 3 cohorts. The fifth cohort (nonuse of AP therapy) contained individuals who were not prescribed either ASA or clopidogrel in the 90 days after their event.
Odds ratios (ORs) associated with the use of different AP therapy regimens and their 95% confidence intervals (CIs) were calculated using unconditional logistic regression analysis models, adjusted (when appropriate) for sex, age, year of qualifying event, comorbidities (diabetes mellitus, hypertension, stroke, or peptic ulcer), whether or not the qualifying event was a recurrent event, type of event (MI, unstable angina, or coronary artery bypass graft/revascularization), smoking status, alcohol use, body mass index, hospitalizations in the year before the qualifying event, Townsend deprivation index (a measure of material deprivation within a population that takes into account 4 main variables: unemployment rate, car ownership, home ownership, and household overcrowding), 11 and use of ASA, clopidogrel, and warfarin at the date of the qualifying event.
Discontinuation of AP Therapies
In order to estimate the time to discontinuation of AP therapies, individuals in the ASA monotherapy, clopidogrel monotherapy, and DAT cohorts were followed up from the date of the first prescription (monotherapy cohorts) or the date of prescription of ASA or clopidogrel (whichever was prescribed second; DAT cohort) after the qualifying event until drug discontinuation, death, or the end of the study (June 30, 2012). Discontinuation was defined as a period of more than 90 days with no use of ASA or clopidogrel in the respective monotherapy cohorts and with no use of ASA, no use of clopidogrel, or no use of either drug in the DAT cohort. Kaplan-Meier survival analyses provided estimates of the rate of continuation of treatment at 1 and 2 years after initiation of treatment. Cox proportional hazards models were employed to assess determinants of treatment discontinuation. Those models were adjusted for all the variables used in the unconditional logistic regression models, PPI use, and a time-dependent variable indicating the occurrence of bleeding events (upper GI bleeding and/or hemorrhagic stroke) during follow-up.
Proportions of patients switching between AP monotherapies or initiating DAT were analyzed in the ASA monotherapy and clopidogrel monotherapy cohorts. Individuals were considered to have switched if their first prescription of the new AP agent occurred in the period between 30 days before the start and 30 days after the end of the supply of the last consecutive prescription of the previous AP agent. The date of the first prescription of the new AP agent was considered the date of switching. Individuals were considered to have initiated DAT if there were at least 2 consecutive prescriptions of the additional AP agent that occurred before the date of the last consecutive prescription of the original AP agent. The date of the second prescription of the additional AP agent was considered the date of switching to DAT.
All statistical analyses were carried out using Stata SE (version 12.0; StataCorp, College Station, Texas). The analysis was approved by the Multicenter Research Ethics Committee (REC reference 09/H0305/90).
Results
In total, 27 351 patients in the study cohort (N = 27 707) had follow-up data for at least 90 days after the date of their qualifying acute coronary event. The qualifying event was a first acute coronary event in 20 730 patients and a recurrent event in 6621 patients. Among patients with a recurrent episode, 33% had had their previous event less than 1 year before the qualifying event and 27% had had their previous event between 1 and 3 years before the qualifying event. Overall, the median time from the most recent previous event to the qualifying event was 714 days (1.96 years).
Use of AP Therapy 90 Days After Hospitalization for an Acute Coronary Event
At 90 days after an acute coronary event, 85.9% of patients were receiving some form of AP therapy (sum of initiators and continuers; Table 1); proportions were similar for patients for whom the event was a first or recurrent acute coronary event (86.8% and 82.8%, respectively). Overall, 78.5% of patients were receiving ASA therapy at 90 days, and the proportion of ASA use was similar in patients with a first acute coronary event and in those with a recurrent event (79.6% and 75.2%, respectively). As can be expected, the proportion of ASA initiators in patients with a first acute coronary event was more than twice that in patients who had experienced a recurrent event (52.2% and 24.2%, respectively). Among patients with a recurrent event, 6.4% discontinued ASA within the 90-day period following their event, compared with 4.0% of patients who had experienced their first event.
Patterns of Use of AP Therapy 90 Days After an Acute Coronary Event, Including Use of ASA in Combination With a PPI.a

Abbreviations: AP, antiplatelet; ASA, acetylsalicylic acid; DAT, dual antiplatelet therapy; PPI, proton pump inhibitor.
aData are n (%).
Overall, 37.5% of patients were receiving clopidogrel 90 days after their qualifying event, most (81.1%) with ASA as DAT (30.4% of the total study cohort). Prescription of DAT 90 days after an acute coronary event increased by more than 20-fold between 2000 and 2008, with similar values reported for patients experiencing their first or a recurrent episode (Figure 1A). This trend was statistically significant after adjustment for age and sex (P trend < .001). Over one-quarter of patients receiving ASA monotherapy (2499 of 9477 [26.4%]) or clopidogrel monotherapy (230 of 790 [29.1%]) at the date of the qualifying event had switched to DAT by 90 days after their event (Figure 1B). Among patients receiving DAT at the date of the qualifying event, over two-thirds (570 of 827 [68.9%]) continued this treatment to at least 90 days after their event, but the remaining 31.1% had discontinued DAT by this time. A minority (8.2%) of patients were prescribed warfarin, alone or in combination with AP agents, in the 90 days after their qualifying acute coronary event, but 10.2% of patients had no recorded prescription for any form of antithrombotic treatment (neither anticoagulant nor AP).
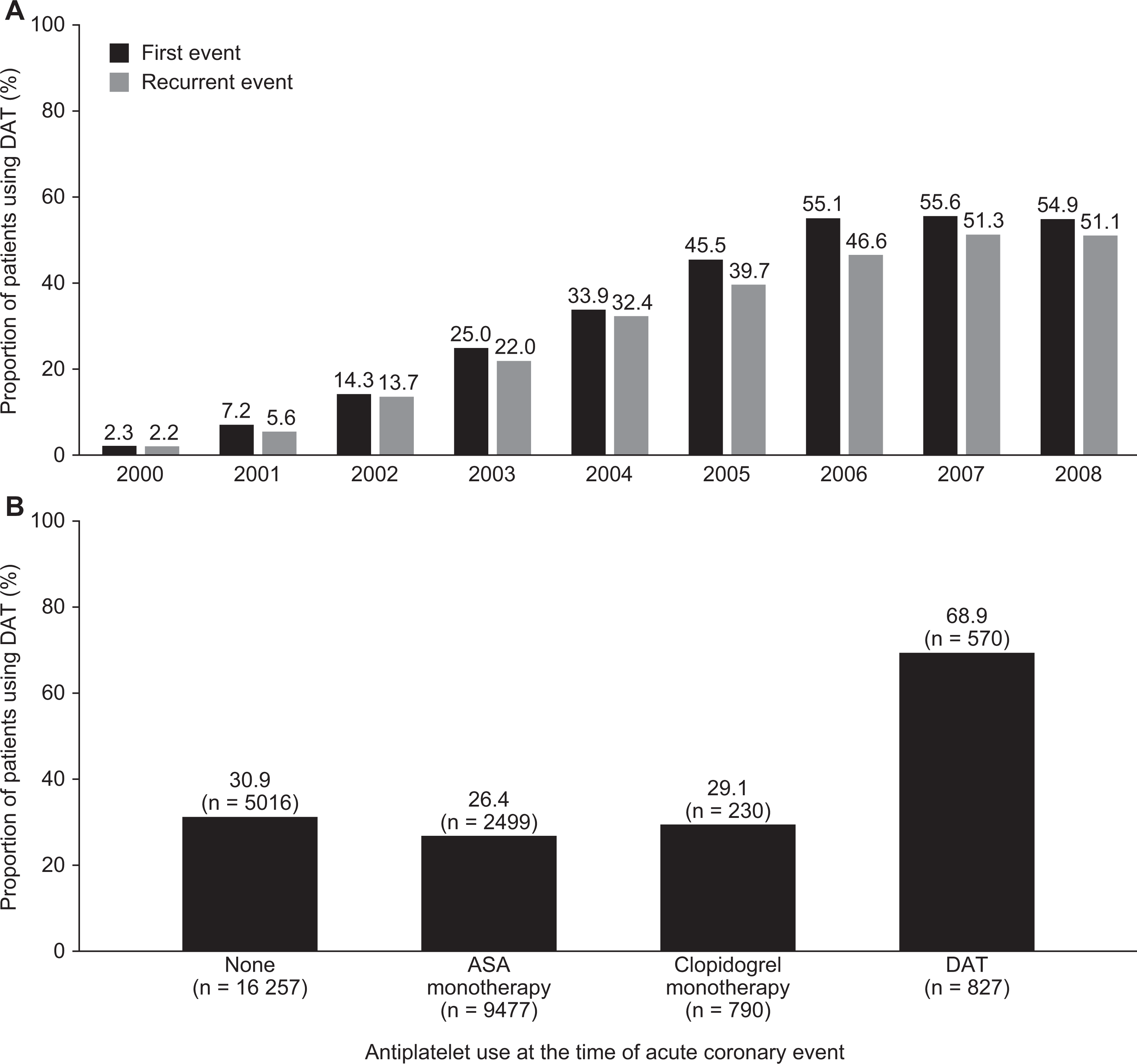
Use of DAT (ASA plus clopidogrel) 90 days after an acute coronary event, stratified by year (A) and antiplatelet use at the time of the event (B). ASA indicates acetylsalicylic acid; DAT, dual antiplatelet therapy.
Coprescription of Gastroprotective Therapy With AP Agents
At 90 days after their qualifying acute coronary event, about one-third (33.6%) of patients who were issued at least 1 ASA prescription in those 90 days were also issued a PPI prescription (26.4% of the study cohort; sum of initiators and continuers; Table 1). Most (76.7%) of these patients were initiators of ASA/PPI coprescription (Table 1). A similar proportion of nonusers of ASA (1493 of 4625 [32.3%]) were also receiving PPI therapy 90 days after their qualifying acute coronary event; these included nonusers of AP therapy and individuals receiving another AP agent. The proportion of users of ASA with a PPI was similar in initiators and continuers of ASA therapy (4230 of 12 422 [34.1%] and 2987 of 9048 [33.0%], respectively). The proportion of patients who were coprescribed ASA and PPI increased from approximately 16% in 2000 to 39% in 2008, irrespective of whether the qualifying event was their first or a recurrent event (Supporting Information Figure S1).
Predictors of Use of AP Therapy
The likelihood of receiving ASA monotherapy 90 days after an acute coronary event, rather than another or no AP therapy, decreased by 86% from 2000 to 2008 (Supporting Information Table S1). The probability of receiving clopidogrel monotherapy increased in the early years of the study but decreased again from 2005 (Supporting Information Table S2). The likelihood of receiving DAT compared with other therapy options increased by 200-fold over the study period (Supporting Information Table S3).
Patients with a history of uncomplicated (1595 of 27 351 [5.8%]) or complicated (993 of 27 351 [3.6%]) peptic ulcer had a significantly reduced probability of receiving ASA monotherapy compared with those without such a history (adjusted OR: 0.85, 95% CI: 0.76-0.95 and adjusted OR: 0.71, 95% CI: 0.61-0.82, respectively; Supporting Information Table S1). Patients with a history of complicated peptic ulcer were also significantly less likely to be prescribed DAT than those without such a history (adjusted OR: 0.59, 95% CI: 0.49-0.71; Supporting Information Table S3). Conversely, a history of peptic ulcer disease (PUD) significantly increased the probability of receiving clopidogrel monotherapy (Supporting Information Table S2).
The DAT was more frequently prescribed in the 90 days after the qualifying event in younger patients than in patients aged 70 years or older and was significantly more likely to be prescribed after an MI than following admission for unstable angina or a revascularization procedure (Supporting Information Table S3). In addition, having diabetes mellitus or a history of stroke significantly reduced the likelihood of receiving DAT rather than ASA monotherapy, with a history of hemorrhagic stroke reducing it by half (Supporting Information Table S4).
Discontinuation of AP Therapy
Patients in the ASA monotherapy cohort (N = 12 999; 55% initiators; 74% with a first acute coronary event) were followed for a mean of 4.9 years. An estimated 15.1% (95% CI: 14.5%-15.7%) of patients in the cohort discontinued ASA within 1 year, and 24.1% (95% CI: 22.4%-24.9%) discontinued ASA within 2 years (Figure 2A). During follow-up, 3.3% of the cohort switched to clopidogrel; thus, a small proportion of the ASA discontinuers continued to be prescribed an AP agent. Furthermore, most (72.3%) discontinuers reinitiated ASA at some point after discontinuation. Patients in the clopidogrel monotherapy cohort (N = 1776; 74% initiators; 76% with a first acute coronary event) were followed for a mean of 3.4 years. After 1 and 2 years of follow-up, an estimated 22.3% (95% CI: 20.4%-24.3%) and 42.4% (95% CI: 40.0%-44.8%) of patients had discontinued clopidogrel, respectively (Figure 2B). During follow-up, 13.1% of the cohort switched to ASA. The proportion of discontinuers that reinitiate clopidogrel at some point after discontinuation was 30.3%. The DAT cohort (N = 6545; 93% initiators; 82% with a first acute coronary event) was followed for a mean of 1.4 years; 37.5% (95% CI: 36.3%-38.7%) of patients were estimated to discontinue DAT before completing 1 year of treatment and 83.1% (95% CI: 82.1%-84.0%) before completing 2 years of treatment (Figure 2C). Among patients who discontinued DAT before completing 1 year of treatment, 73% discontinued clopidogrel and remained on ASA, 14% discontinued ASA and remained on clopidogrel, and 13% discontinued simultaneously ASA and clopidogrel.

Kaplan-Meier estimates of the rate of continuation of ASA (A) and clopidogrel (B) in the respective monotherapy cohorts, and DAT (C), during the 2 years following an acute coronary event. ASA indicates acetylsalicylic acid; DAT, dual antiplatelet therapy.
Patients who were receiving ASA at the date of their qualifying event had a significantly reduced risk of discontinuing ASA monotherapy during follow-up compared with patients who started ASA later (Table 2). Similarly, use of clopidogrel at the date of the qualifying event reduced the risk of discontinuing clopidogrel or DAT (Table 2). Compared with patients who had experienced a recurrent acute coronary event, patients with a first event were less likely to discontinue ASA but more likely to discontinue DAT (Table 2). A history of stroke was associated with an increased risk of discontinuing ASA but did not significantly affect the risk of discontinuation of DAT or clopidogrel monotherapy (Table 2). Although a history of PUD influenced the prescription pattern of AP agents, it did not significantly increase the risk of discontinuation of any agent. A bleeding event during follow-up (either an upper GI bleed or a hemorrhagic stroke) occurred in 2.2% of patients (286 of 12 999) in the ASA monotherapy cohort and was the strongest determinant of ASA discontinuation, resulting in a 5-fold increase in risk of discontinuing (Table 2). The proportions of patients experiencing a bleeding event in the clopidogrel monotherapy and DAT cohorts were 3.7% (66 of 1766) and 1.0% (67 of 6545), respectively; such events increased the risk of discontinuing either clopidogrel or DAT by approximately 2-fold (Table 2).
Factors Significantly Affecting the Risk of Discontinuing ASA or Clopidogrel Monotherapy or DAT Following an Acute Coronary Event.a

Abbreviations: ASA, acetylsalicylic acid; CPU, complicated peptic ulcer; DAT, dual antiplatelet therapy; PPI, proton pump inhibitor; PUD, peptic ulcer disease; TIA, transient ischemic attack; UPU, uncomplicated peptic ulcer.
aData are hazard ratios (95% confidence intervals).
bHistory of disease at start date.
cBleeding events occurring during follow-up.
Discussion
This large-scale, descriptive retrospective study in UK primary care indicates that, in 2000 to 2008, almost 9 of the 10 patients were prescribed AP therapy during the 90 days after hospitalization for an acute coronary event, regardless of whether their qualifying event was their first or a recurrent event; almost 8 of the 10 patients were prescribed ASA.
The use of DAT 90 days after an acute coronary event increased throughout the study period, from 2% in 2000 to over 50% in 2008. However, among patients prescribed AP therapy in the 90 days after an acute coronary event, over one-third (37.5%) who received DAT were estimated to discontinue 1 or both AP agents before completing 1 year of treatment, and 15% and 22% of patients who received ASA or clopidogrel monotherapy, respectively, discontinued within 1 year. Experiencing a bleeding episode (an upper GI bleed or a hemorrhagic stroke) during follow-up was the strongest predictor of discontinuing AP therapy, increasing the risk of discontinuation by 5-fold for ASA and by 2-fold for clopidogrel or DAT.
About halfway through the study period, in 2004, the National Institute for Health and Care Excellence in the UK published guidance recommending the use of DAT (clopidogrel plus ASA) for at least 12 months after a non-ST-segment- elevation acute coronary syndrome. 12 The results of the present descriptive study indicate a shift in clinical practice from use of ASA monotherapy to use of DAT over the period 2000 to 2008. Uptake of clopidogrel monotherapy increased early in the study but then reduced in 2005 and later, presumably being replaced by DAT. About one-quarter of patients receiving ASA or clopidogrel monotherapy at the date of the qualifying event had switched to DAT by 90 days after their acute coronary event.
Factors that reduced the likelihood of receiving DAT compared with ASA monotherapy may reflect physician concerns over an increased bleeding risk with DAT compared with AP monotherapy. 13,14 Patients who received DAT were younger and less likely to have comorbidities such as diabetes mellitus or a history of stroke, particularly hemorrhagic stroke, than those who received ASA monotherapy.
AP therapy has been shown to be associated with an increased risk of upper GI complications, including peptic ulcer bleeding; 15,16 indeed, a history of PUD reduced the likelihood of receiving DAT. Patients with a history of PUD were also less likely to be prescribed ASA monotherapy and more likely to be prescribed clopidogrel monotherapy, compared with patients without such a history. This may reflect a perception during the study period that use of clopidogrel was an appropriate approach to reducing the risk of peptic ulcer bleeding. However, expert consensus guidelines published in 2008 17 recommended against the use of clopidogrel monotherapy and in favor of coprescription of a PPI with ASA to reduce the risk of recurrence of peptic ulcer bleeding. 18,19 The present study analyzed events occurring in an 8-year period before this guidance was published and observed that about one-third of patients who received ASA in the 90 days after an acute coronary event also received PPI therapy; most of these patients had not been receiving a PPI at the date of their qualifying event. This may reflect both the use of PPIs to treat emergent GI symptoms and their prophylactic use to prevent GI complications associated with ASA therapy. Current international guidelines suggest that PPI therapy may be considered for patients receiving AP therapy who are at high risk of GI complications but advise that physicians should carefully evaluate the risks and benefits of starting PPI therapy in patients receiving clopidogrel. 20
In the present study, a bleeding event during follow-up, including upper GI bleeding or hemorrhagic stroke, was the strongest predictor of discontinuation of AP therapy. Bleeding events were found to predict discontinuation of clopidogrel, or of either component of DAT, in the year after an acute coronary event in other observational studies. 6,21 GI problems or symptoms have also been reported to affect the risk of ASA discontinuation and the rate of adherence to ASA. 3 –5
Withdrawal of ASA has been demonstrated to be associated with increased risk of adverse vascular outcomes in patients with acute coronary syndrome. 7,22 –24 In patients receiving ASA for secondary CV prevention, we have previously demonstrated a 43% increase in the risk of nonfatal MI or coronary death and a 40% increase in the risk of ischemic stroke in patients who have recently discontinued ASA, compared with those who continued treatment. 8,9 Other recent observational studies in patients with acute coronary syndrome have reported that discontinuation of clopidogrel after less than 12 months of therapy significantly increases the risks of recurrent CV events, CV death, and hospitalization for an acute coronary event. 6,21,25,26 The rates of discontinuation in the present study reflect previous reports that up to 30% of patients requiring long-term ASA therapy for secondary CV prevention may discontinue their medication, 3,4,27 and results from another recent study in UK primary care that indicate that the probability of a patient still being prescribed clopidogrel 12 months after an MI is only about 53%. 6
Strengths and Limitations
Strengths of this study include the large sample size of over 27 000 patients and the external validity of the source data: THIN has been validated for use in pharmacoepidemiological studies, 28 and patients included in the database are representative of the general UK population with respect to age, sex, and geographical region. 29 As with any observational study, it is not possible to control prospectively for factors that may influence the outcome of interest. However, we have attempted to minimize confounding by adjusting for multiple factors when analyzing predictors of drug use.
Limitations include the fact that it was not possible to assess patient adherence directly and that the databases used record issuing, rather than dispensing, of prescriptions. In addition, hospital prescribing of AP therapy is not routinely recorded in THIN. In order to capture data for all patients receiving AP therapy after an acute coronary event, we analyzed a 90-day window, which should allow for the fact that some patients had a prescription at the time of the event and some will have received one on discharge from hospital. It should also be noted that individuals were not followed up after they had discontinued their AP therapy; some individuals classified as discontinuers might have reinitiated AP treatment more than 90 days after the end of their last AP prescription. Additionally, THIN does not report use of over-the-counter (OTC) medications. However, in a previous study, we analyzed free-text comments in a sample of records in THIN and estimated that approximately 10% of individuals aged between 40 and 89 years with CV antecedents were likely to be using OTC ASA, 30 which should not greatly affect our study results. Prescription medications are free for patients aged 60 years or older in the United Kingdom, and health care is easily accessed, which is likely to encourage prescription rather than OTC use. Finally, therapy for acute coronary syndrome has changed since the study period (2000-2008) and, therefore, our results may not be directly applicable to the current practice.
Conclusions and Clinical Implications
Although this descriptive study indicates that, from 2000 to 2008 in the United Kingdom, most patients received AP therapy in the 90 days after an acute coronary event, over one-third of patients discontinued DAT and about 15% of patients discontinued ASA monotherapy in the year after their event. Given the increased risk of adverse CV outcomes associated with early discontinuation of DAT or any discontinuation of long-term therapy with ASA, 6,8,9,21,25,26 our observations indicate that it is essential that physicians consider strategies to reduce the risk of discontinuation of AP therapy.
Footnotes
Author Contributions
M. E. Sáez, A. González-Pérez, and L. A. García Rodríguez contributed to the conception and design of the study, contributed to the acquisition of the data, and performed the statistical analysis. S. Johansson and P. Nagy contributed to the design of the study. All the authors contributed to the analysis and interpretation of the data. All the authors reviewed the intellectual content of the article, approved the final version and are accountable for all aspects of the work.
Authors’ Note
Medical writing support was provided by Dr Carolyn Brechin and Dr Stéphane Pintat of Oxford PharmaGenesis Ltd, Oxford, UK.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M. E. Sáez, L. A. García Rodríguez, and A. González-Pérez work for CEIFE, which has received research funding from AstraZeneca R&D, Mölndal, Sweden and Bayer Pharma AG, Berlin, Germany. L. A. García Rodríguez has also received honoraria for serving on scientific advisory boards for AstraZeneca and Bayer. S. Johansson and P. Nagy are employees of AstraZeneca R&D, Mölndal, Sweden.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the study was funded with financial research support from AstraZeneca R&D, Mölndal, Sweden.
