Abstract
Introduction:
Generic clopidogrel recently became available in the United States and was rapidly adopted as a cost-effective alternative to the brand name formulation. However, unlike other medications, subtle differences in clopidogrel bioavailability may lead to acute consequences including stent thrombosis (ST).
Materials and Methods:
We studied the incidence of acute and subacute ST during the initial period of generic clopidogrel use (June 18, 2012-September 6, 2012 [80 days]) at a single percutaneous coronary intervention (PCI) center. There were 4 definite ST cases within 30 days of successful PCI in patients receiving generic clopidogrel, which were compared to historic control ST cases from 80 days prior to generic clopidogrel use and for 3 years from June 18, 2009 to June 17, 2012.
Results:
During generic clopidogrel administration, 1054 PCIs were performed, giving a definite 30-day ST incidence of 0.38% (4 of 1054) among these patients. By comparison, there were 2 episodes of definite 30-day ST during the 80 days immediately preceding generic clopidogrel use (2 of 1114), while 3-year historic data indicated a definite 30-day ST incidence of 0.14% (20 of 14 432), representing a 2.7-fold increase in definite 30-day ST with generic clopidogrel use (P = .076). Exclusion of 3 historic controls with a defined reason for ST (noncompliance, marked thrombocytosis) gave a 3.2-fold increase in 30-day ST with generic clopidogrel (P = .050). An ST-predictive algorithm revealed no difference in the likelihood of ST between patients receiving generic clopidogrel and historic controls.
Conclusions:
We observed an unexpected >2-fold increase in ST coincident with generic clopidogrel use. Although we cannot ascribe causality, this observation warrants increased vigilance and close monitoring of patients receiving generic clopidogrel.
Introduction
Stent thrombosis (ST) following percutaneous coronary intervention (PCI) is a devastating clinical event and is associated with increased myocardial infarction and death. 1 Many factors predispose to ST. These include patient comorbidities, genetic factors and drug metabolism, 2 –5 stent properties, 6 stent apposition, and lesion characteristics. 2,7,8 Furthermore, it is critical that patients with recently implanted intracoronary stents receive an appropriate antiplatelet regimen. 9 –12 In this regard, patient nonadherence to antiplatelet therapy has been the subject of intense recent study and is likely to be a major cause of ST. 9 , 10 However, as an alternate possibility, it has long been suspected that inferior preparations of clopidogrel, which are associated with reduced plasma levels of active clopidogrel metabolites, might also be associated with ST. This concern has led to a number of small and often underpowered studies comparing either pharmacological or clinical end points in patients taking generic versus brand name clopidogrel—the latter best known as Plavix (Bristol-Myers Squibb/Sanofi-Aventis). 13 –19 Despite the fact that most studies found no differences between generic versus brand name clopidogrel, 13 –19 specific concerns have been raised 20 with occasional studies suggesting important potential differences. 21,22
On May 17, 2012, the US Food and Drug Administration (FDA) approved the introduction of generic clopidogrel into the US market. 23 Of relevance with respect to differences between preparations and product stability, the FDA regulations stipulate that although generic drugs are required to have the same active ingredient, strength, dosage form, and route of administration as the brand name product, they do not need to contain the same inactive ingredients. 24 Furthermore, it is not practical for the FDA to assess and check the human bioactivity of every lot and batch of these generic formulations, many of which are manufactured outside the United States.
Following the FDA announcement, on June 18, 2012, our institution switched to the exclusive use and prescription of generic forms of clopidogrel. This coincided with the rapid and widespread availability of these formulations in pharmacies throughout the United States. We observed an unexpected spike in the incidence of acute and subacute ST coincident with the use of generic clopidogrel. These cases are reported here and are compared to historical ST data from our interventional PCI registry.
Methods
Data Source, Patient Population, and Definitions
Patients presented here were drawn from a single center, institutional review board approved prospective interventional cardiology registry. All patients undergoing PCI are entered into this registry within 24 hours of the index intervention. Details of this registry have been published previously. 10 , 25 Further to this, additional and specific local institutional review board approval was sought and obtained for the purposes of data collection and compiling as relevant to this case series.
Percutaneous coronary intervention was performed according to best clinical practice and using bivalirudin for anticoagulation. All technical decisions were at the discretion of the operators. Glycoprotein IIb/IIIa inhibitors were only administered as a bolus dose, and no patient received an infusion of these medications. At the conclusion of the PCI, clopidogrel-naive patients who did not receive a glycoprotein IIb/IIIa inhibitor received a 1-hour infusion of bivalirudin.
After discharge, patients are routinely contacted and undergo 30-day and 12-month follow-up. Major adverse cardiac events (MACEs) and mortality are ascertained via phone contact with the patient, family, or primary physician. In addition, hospital records and the social security death index are queried for MACE and mortality.
Stent thrombosis was defined according to the Academic Research Consortium definitions. 26 Acute occlusion was defined to occur at <24 hours, while subacute occlusion was defined to occur at >24 hours to ≤ 30 days after the study procedure. 26 All cases of ST reported here were independently reviewed and verified by a clinical events committee. Original source documentation and/or angiograms were obtained for ST cases that were managed at outside hospitals. We are not aware of any cases for which we were unable to obtain appropriate documentation for a suspected case of definite ST that occurred at an outside institution.
Antiplatelet Therapy and Clopidogrel Administration
All patients routinely received 325 mg aspirin >90 minutes prior to angiography. In clopidogrel-naive patients, clopidogrel 600 mg was administered “on table” immediately prior to PCI, while patients already taking clopidogrel received an additional 300 mg dose. For historic control patients who received prasugrel at index PCI, the on-table loading dose was 60 mg for prasugrel-naive patients and 30 mg for those already receiving this therapy.
Generic clopidogrel, rather than Plavix, was exclusively administered and prescribed at our institution during discharge from June 18, 2012, to September 6, 2012 (inclusive). All the 4 patients with definite ST described herein received generic clopidogrel at on-table loading and at discharge. For the historic control patients (June 18, 2009, to June 17, 2012) with definite ST, 17 received Plavix while 3 received prasugrel. No ST patient reported here received ticagrelor, ticlopidine, or any other alternate agent following index PCI. Thus, following index PCI, all patients described here as having ST were discharged taking aspirin ≥81 mg daily and either generic clopidogrel 75 mg daily (n = 4), Plavix 75 mg daily (n = 17), or prasugrel 5 or 10 mg daily (n = 3).
Genetic Testing for Clopidogrel Responsiveness
Genetic testing for CYP2C19 and ABCB1 genotypes was performed using whole blood with the Clopidogrel Genetic Absorption Activation Panel (Transgenomic Inc, New Haven, Connecticut). Testing was performed with patient consent and as a guide to decision making in the clinical care of these patients.
Statistical Analysis
Comparisons of means between the 2 groups were performed by 2-tailed Student t test (parametric data) or Fisher Exact test (nonparametric data). Continuous variables are presented as mean ± standard deviation. For assessment of ST risk, we applied a recently described clinical algorithm: score = (0.6859 × proton pump inhibitor [PPI] use) + (0.6203 × diabetes) + (0.9250 × left ventricular ejection fraction <40%) + (0.8970 × American College of Cardiology /American Heart Association type C lesion) + (1.1197 × acute setting) − (0.3089 × clopidogrel loading dose) − 2.0957. 5 For clopidogrel loading dose: <150 mg = 0; 150 to 299 mg = 1; 300 to 600 mg = 2; >600 mg = 3; 5 as a minor modification, the loading dose of prasugrel was scored as <30 mg = 1, 30 to 60 mg = 2. Differences were deemed significant if P < .5. Statistical analyses were performed using IBM SPSS/PASW Statistics 18 (SPSS Inc. Chicago, Illinois).
Results
We observed 4 cases of definite acute or subacute ST that occurred between June 18, 2012, and September 6, 2012, in patients receiving generic clopidogrel and are reported subsequently.
Case 1
A 47-year-old female with hypertension, obesity, insulin-requiring diabetes, and hyperlipidemia presented with Canadian Class Society (CCS) III angina. The patient had a history of prior PCI to the right posterior descending coronary artery (RPDA) and balloon angioplasty (percutaneous transluminal coronary angioplasty [PTCA]) to the high lateral (HL). Angiography revealed a patent RPDA stent, 80% to 90% stenosis of the obtuse marginal (OM) branch, and subtotal occlusion of the HL. PCI was performed at the same procedure, with both the OM and the HL lesions treated with successful drug-eluting stent (DES) implantation (Figure 1A). A single 20 mg bolus of eptifibatide was given at the end of the procedure. The patient had an uneventful recovery and was discharged with medications including aspirin, generic clopidogrel, and esomeprazole. It was confirmed that the patient received generic clopidogrel (not Plavix) from their pharmacy after discharge. Four days later, the patient presented to another institution with a non-ST-segment elevation acute myocardial infarction (NSTEMI). Urgent angiography revealed subacute ST in the HL and OM branches (Figure 1E). Mechanical thrombectomy and PTCA were performed with successful restoration of Thrombolysis In Myocardial Infarction (TIMI) III flow. Platelet reactivity testing was not performed, but the patient reported full compliance with antiplatelet therapy. Genetic testing indicted that the patient was likely to be at increased risk of ST (Table 1). The patient was subsequently discharged home receiving prasugrel and aspirin.
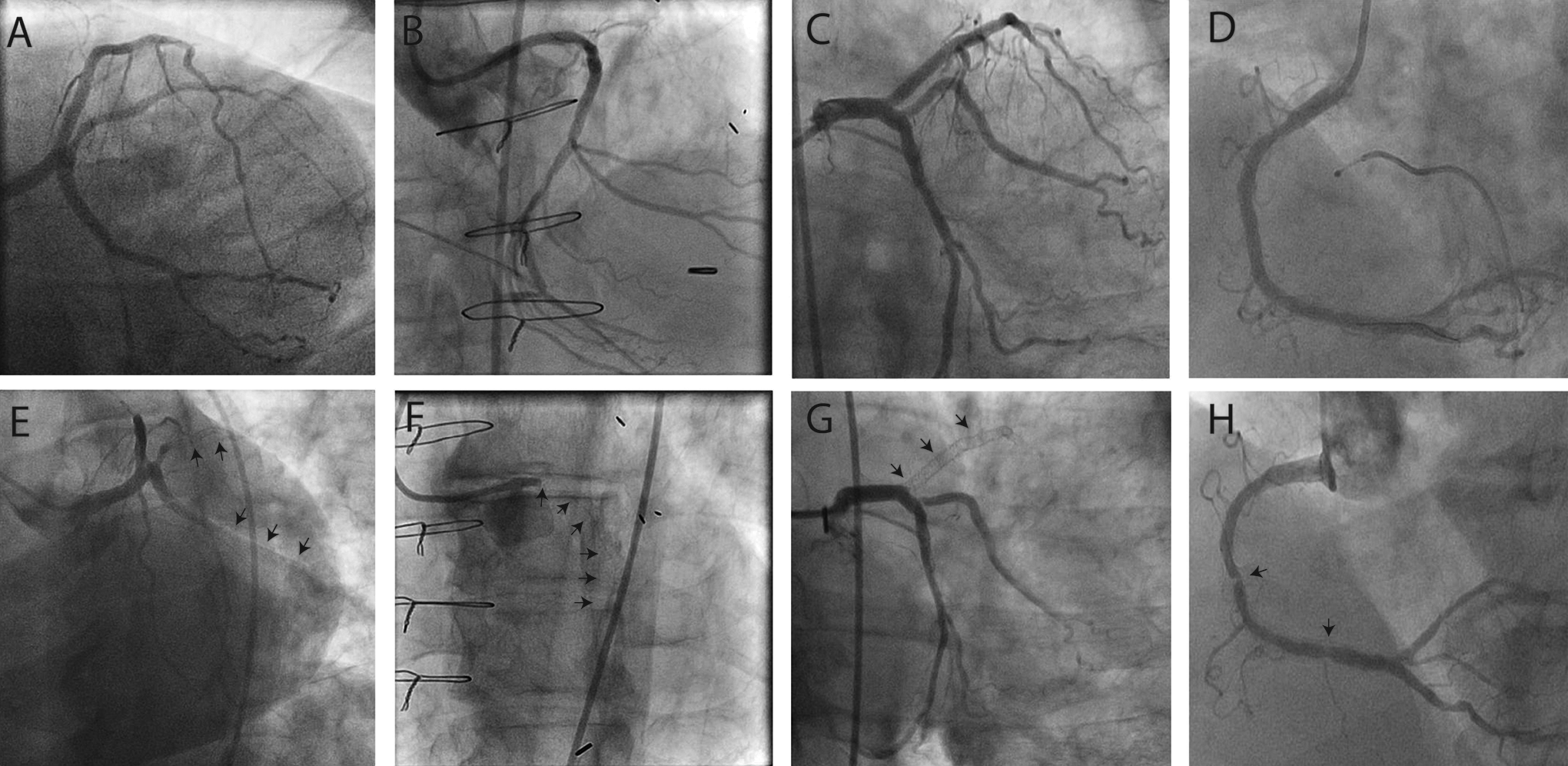
Four cases of definite acute or subacute ST coincident with the use of generic clopidogrel. A-D, Final angiograms for cases 1 to 4, respectively, following successful index PCI. E-H, Corresponding initial angiographic images for cases 1 to 4, respectively, on subsequent presentation with ST. Arrows indicate sites of ST.
Genetic Testing for CYP2C19 and ABCB1 Genotype and Clinical Risk for Patients having Definite Acute or Subacute ST During Generic Clopidogrel Use at Our Institution. 5

Abbreviation: ST, stent thrombosis.
Case 2
A 75-year-old male with hypertension, diabetes, hyperlipidemia, and prior coronary artery bypass surgery presented with CCS III angina and was found to have a positive myocardial perfusion scan. During August of 2012, he underwent PCI of the (protected) distal left main coronary artery, left circumflex, and left posterolateral branches (Figure 1B). He was discharged taking aspirin and generic clopidogrel (without PPI). The patient represented 13 days later with severe chest pain at rest and rapidly decompensated requiring intubation. An intra-aortic balloon pump was placed, and urgent angiography revealed ST with occlusion of the stented segments (Figure 1F). Abciximab was administered, and the patient underwent successful PCI with restoration of TIMI III flow. However, the patient sustained a large myocardial infarction (peak creatine kinase-MB > 300 ng/mL) and died 7 days later with multiorgan failure. Due to abciximab administration, platelet reactivity testing could not be performed. Although it was not possible to verify that generic clopidogrel (and not Plavix) was dispensed by their pharmacy after initial discharge, the patient’s family members affirmed that the patient had been compliant with antiplatelet therapy. Genetic testing indicated that the patient was at low risk of ST (Table 1).
Case 3
An 83-year-old male with hypertension, diabetes, hyperlipidemia, and prior PCI of the proximal left anterior descending coronary artery (LAD) presented with CCS IV angina. During August of 2012, he underwent angiography revealing in-stent restenosis of the prior LAD stent and a denovo mid LAD lesion. The PCI to the proximal and mid LAD with successful DES placement was performed at the same procedure (Figure 1C). The patient received aspirin and generic clopidogrel and was also taking lansoprazole. Approximately 18 hours later, while still in hospital, the patient had an anterior STEMI. Urgent angiography revealed acute ST (Figure 1G), which was successfully managed by further PCI. Platelet reactivity testing (VerifyNow, Accumetrics, San Diego, California) revealed a P2Y12 reaction unit value of 378—indicating a failure of platelet inhibition; however, genetic testing revealed that the patient had only a modest predisposition toward a poor response to clopidogrel (Table 1). The patient was discharged home uneventfully taking aspirin and ticagrelor.
Case 4
A 57-year-old female with hypertension, diabetes, hyperlipidemia, current smoking, obesity, and prior PCI to the distal right coronary artery (RCA) and RPDA presented with CCS III angina. During August of 2012, angiography revealed a denovo mid-RCA lesion and in-stent restenosis in the distal RCA, with successful PCI then performed with PTCA and placement of a single DES (Figure 1D). The patient was discharged receiving aspirin and generic clopidogrel (without PPI). It was confirmed that the patient received generic clopidogrel (not Plavix) from their pharmacy after discharge. Twelve days later (September 4, 2012), the patient presented to another hospital with severe chest pain and STEMI. Urgent angiography demonstrated ST within the mid-RCA stent (Figure 1H), which was successfully treated by PCI. Platelet reactivity testing was not performed but genetic testing indicted that the patient was likely to be responsive to clopidogrel (Table 1) and the patient confirmed compliance with antiplatelet therapy. The patient was later discharged home taking aspirin and Plavix.
Historical Incidence of Definite ST and Statistical Comparison
During the period that generic clopidogrel was dispensed at our institution (June 18, 2012-September 6, 2012, inclusive [80 days]), a total of 1054 PCIs were performed, giving a 30-day definite ST incidence of 0.38% (4 of 1054) among these patients. Seven core interventional cardiologists performed 77.1% of all PCIs during this time, including the index PCI for all 4 cases of definite ST reported here.
We initially performed a retrospective analysis to identify all historic cases of definite 30-day ST occurring for PCIs performed during an identical length time window (80 days) immediately prior to the introduction of generic clopidogrel (March 29, 2012-June 17, 2012, inclusive). During this period, 1114 PCIs were performed at our institution, with 75.8% of these PCIs performed by the same 7 core interventionalists. There were only 2 cases of definite 30-day ST identified following these procedures, giving a definite ST incidence of 0.18%.
A more extensive retrospective case–control analysis spanning 3 years was then performed to identify all cases of definite ST occurring within 30 days of PCI for stents implanted from June 18, 2009, to June 17, 2012, at our institution (inclusive). During this 3-year time window, 14 432 PCIs were performed with 83.8% of these being by the same 7 core interventionalists. The clinical characteristics of all control patients undergoing PCI during this time are presented in Table 2, with no consistent differences seen when compared with patients having PCI during the 80-day period of generic clopidogrel administration. A total of 20 patients had definite 30-day ST during this 3-year period, giving a historic background definite 30-day ST incidence of 0.14% (20 of 14 432). These 20 patients were generally at increased risk of ST (Table 3) and also included 5 patients with proven clopidogrel hypo- or nonresponsiveness by platelet reactivity testing, 1 patient with marked thrombocytosis (platelet count > 1000 × 103/µL), 2 patients who were known to be noncompliant with clopidogrel therapy, and 1 patient with prior cardiac transplant. Of these 20 historic controls, 3 were receiving aspirin and prasugrel (rather than clopidogrel) at the time of ST. By comparing these historic controls (0.14% definite ST incidence) to the 4 cases of ST observed from June 18, 2012, to September 6, 2012 (0.38% definite ST incidence), we observed a 2.7-fold increase in the occurrence of definite 30-day ST coincident with the use of generic clopidogrel at our institution (P = .076). A recently described clinical algorithm revealed no difference in the likelihood of ST between the 4 cases who received generic clopidogrel and the 20 historic controls (−0.85 ± 0.40 vs −1.06 ± 0.84, P = .63).
Clinical Characteristics and Index PCI Details of All Patients Undergoing PCI During the Period of Generic Clopidogrel Use (June 18, 2012, to September 6, 2012) and Background Historical Data Spanning 3 Years Prior to the Use of Generic Clopidogrel (June 18, 2009, to June 17, 2012).a

Abbreviations: ACC, American College of Cardiology; AHA, American Heart Association; BMI, body mass index; BSA, body surface area; CABG, coronary artery bypass graft surgery; CKD, chronic kidney disease; eGFR, estimated glomerular filtration rate (by modification of diet in renal disease formula); LVEF, left ventricular ejection fraction; PCI, percutaneous coronary intervention; ST, stent thrombosis; SD, standard deviation.
a Data presented are % or mean ± SD (range) and are reported on a per-PCI basis.
Clinical Characteristics and Index PCI Details of Patients Having Definite 30-day ST Comparing the Period of Generic Clopidogrel Use (June 18, 2012, to September 6, 2012) to Background Historical ST Events in the 3-Year Period Prior to the Use of Generic Clopidogrel (June 18, 2009, to June 17, 2012).a

Abbreviations: ACC, American College of Cardiology; AHA, American Heart Association; BMI, body mass index; BSA, body surface area; CABG, coronary artery bypass graft surgery; CKD, chronic kidney disease; eGFR, estimated glomerular filtration rate; LVEF, left ventricular ejection fraction; PCI, percutaneous coronary intervention; ST, stent thrombosis.
a Data presented are % (n) or mean ± SD (range).
Exclusion of the 3 historic control patients who had clear provoking reasons for ST (noncompliance with clopidogrel therapy or marked thrombocytosis) gave a historic background definite 30-day ST incidence of 0.12% (17 of 14 432). When this was taken as the historic control incidence, the 4 of 1054 definite 30-day ST cases that were observed during generic clopidogrel administration represented a 3.2-fold increase in acute or subacute ST coincident with generic clopidogrel use (P = .050).
Discussion
Generic formulations of commonly administered medications have been available for decades, arguably even centuries. 27 This includes most types of cardiovascular medications, such as statins, β-blockers, diuretics, angiotensin-converting enzyme inhibitors, calcium channel blockers, and anti-arrhythmics. Currently, generic drugs represent 70% of the total prescriptions dispensed in the United States. They resulted in an estimated saving of US$53 for every prescription sold and saved American consumers US$824 billion in the last decade. 24 Importantly, minor differences in the biologic efficacy of many of these generic medications, for example diuretics, antihypertensives, or statins, would be unlikely to have any discernible clinical effect. Somewhat unique to the situation of antiplatelet agents in the immediate post-PCI setting, where therapeutic efficacy is critical, even a minor decrement in bioavailability of clopidogrel active metabolites could result in dramatic clinical consequences. Here, we report 4 cases of definite acute or subacute ST that occurred with generic clopidogrel use at our institution, representing an unexpected >2-fold increase in ST coincident with the use of this drug formulation as compared to Plavix.
Although the patients reported here were generally at increased risk of ST compared to many patients with PCI, their clinical risk profile was similar to 20 historic control patients with definite ST (Tables 2 and 3). Although we did not have genetic testing results on control patients, the genetic pattern of the CYP2C19 and ABCB1 genes was such that only 1 of the 4 definite ST cases was at significantly increased risk of ST based on these results. Furthermore, the use of genetic testing for CYP2C19 alleles is controversial and, as was the case here, is often only recommended on a case-by-case basis after a recurrent acute coronary syndrome event. 28 Current data do not support the use of CYP2C19 genotype data to assess clopidogrel responsiveness or guide treatment in other scenarios. 29 With respect to the technical adequacy of stent deployment, the same 7 core interventional cardiologists performed >75% of all PCIs at our institution during all examined time periods, including all 4 index PCIs that later resulted in ST during the period of generic clopidogrel use. Figure 1 also demonstrates that at least angiographically, there were no obvious concerns with respect to the adequacy of stent deployment. Of the 4 ST cases, 2 (50%) were receiving generic clopidogrel with a PPI at the time of ST. Although this is higher than the background use of PPI’s in control patients who had ST (20%, Table 3), the clinical relevance of PPI use with respect to clopidogrel activity and ST is unclear. In contrast to earlier reports, recent randomized studies 30,31 and meta-analysis data 32,33 have indicated that the use of PPIs with clopidogrel is not associated with an increase in adverse clinical events. In addition, a further study has shown that the use of esomeprazole (taken by Case 1) has no effect on the action of clopidogrel. 34
It is highly improbable that all generic clopidogrel formulations are inferior to Plavix. Indeed, it is not possible to ascribe causality to the use of generic clopidogrel and the 4 cases of definite ST described here. We speculate that, at worst, only certain brands or even certain batches or lots may have inferior bioactivity compared to Plavix. Nevertheless, our data are concerning and prompted us to abandon generic clopidogrel and revert to using Plavix. From June 18, 2012, to September 6, 2012, our institution stocked 2 brands of generic clopidogrel; however, the majority of our stocks were traced to 2 to 3 lots of 1 particular brand. Given that the 4 patients who had ST during generic clopidogrel use all underwent PCI during August of 2012, it is even possible that a single lot or batch of generic clopidogrel was responsible for our observations. Importantly, we verified that of the 3 patients who were discharged from hospital between the index PCI and the ST, 2 definitely received generic clopidogrel (and not Plavix) from their outside pharmacy. An additional patient had ST while still in hospital, therefore in this latter case there is almost complete certainly that only generic clopidogrel was administered. As an additional consideration, it is plausible that these ST cases associated with the use of generic clopidogrel were particularly evident at our institution due to our high PCI volume. Although in lower volume institutions a single case of ST may go unnoticed, the 4 cases reported here stand out from our historic incidence of ST.
Relevant to our observations, Gomez et al 22 compared 18 preparations of generic clopidogrel against Plavix and found that most generic formulations were inferior in that there were more impurities, the content of clopidogrel was lower, and the dissolution profiles were different. Furthermore, after 3 months under stressful storage conditions in the original packaging, the results for the generic versus Plavix comparisons were significantly different in most cases. 22 We are not aware of any unusual storage or handling conditions at our institution. However, the generic clopidogrel that we dispensed was manufactured in India and we are not aware of the specific shipping and handling details involved in the importation of these tablets to the United States.
Limitations
These data were obtained from a single center and it remains possible that our observations may be due to random chance. Although the incidence of definite ST reported here for our historic controls is consistent with numerous prior reports, 35 –39 it is possible we may have underestimated the true background rate of ST. Genetic testing was not performed on control patients with definite ST who received Plavix, therefore comparisons based on genetic predisposition to ST are not possible. Finally, a multitude of factors may result in ST including patient compliance/noncompliance, suboptimal stent expansion, undiagnosed hypercoagulable conditions, so on. Therefore, we are unable to ascribe causality to the use of generic clopidogrel and ST.
Conclusions
All generic drugs undergo extensive evaluation by the FDA prior to their approval for use in the Unites States. Nevertheless, it is not practical to account for all potential differences between generic and brand name formulations, particularly as the inactive components are permitted to differ. Coincident with the introduction of generic clopidogrel at our institution, we observed a concerning spike in the incidence of definite acute and subacute ST after successful PCI. Although we are unable to prove causality, this hypothesis-generating observation warrants increased vigilance and close monitoring of all patients receiving generic clopidogrel.
Footnotes
Acknowledgments
Genetic testing for responsiveness to clopidogrel was provided free of cost by Transgenomic Inc.
Declaration of Conflicting Interests
The following authors have no conflicts of interest with respect to this publication: Jason Kovacic, Joseph Sweeney, Jennifer Li, Pedro Moreno, Usman Baber, Prakash Krishnan, Juan Badimon, Jean-Sebastien Hulot, Annapoorna Kini and Samin Sharma.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: Roxana Mehran reports institutional research grant support from The Medicines Company, Bristol-Myers Squibb/ Sanofi-Aventis, and Lilly/ Daiichi Sankyo; Consulting for Abbott Vascular, AstraZeneca, Boston Scientific, Covidien, CSL Behring, Janssen Pharmaceuticals, Maya Medical, Merck, Regado Biosciences, and Sanofi-Aventis; and serves on advisory boards for Covidien, Janssen Pharmaceuticals, and Sanofi-Aventis.
