Abstract
Background
The effect of combined nicorandil and beta-adrenergic receptor blockers (BBs) compared with that of BBs alone on long-term clinical outcomes in patients with coronary artery disease (CAD) remains undetermined.
Methods
A multicenter retrospective cohort study was performed. Adult patients who had been hospitalized for CAD and treated for angina with a combination of nicorandil and BBs or BBs alone were included. The effect of different treatments on the cumulative incidence of major adverse cardiovascular event (MACE) and their components within a follow-up duration of 2.5 years were analyzed using Kaplan–Meier survival curves. An inverse probability of treatment weighting (IPTW) method was used to adjust for the possible effect of confounding factors.
Results
A total of 137,714 patients were screened, of whom 16,912 individuals (mean age: 61.5 years, men: 67.1%) were successfully enrolled. Among the enrolled participants, 4669 received the combined treatment of nicorandil and BBs, while 12,243 received BBs alone. After IPTW, the results demonstrated that the combined treatment was associated with a significantly reduced incidence of MACE (hazard ratio [HR] 0.79, 95% conidence interval [CI] 0.72-0.87) and stroke (HR 0.48, 95% CI 0.42-0.54) but not of MI (HR 1.03, 95% CI 0.92-1.15) or all-cause mortality (HR 0.93, 95% CI 0.64-1.37). Sensitivity analyses revealed similar results.
Conclusions
A combined antiangina treatment of nicorandil and BBs may be more effective than treatment of BBs alone in reducing the long-term incidence of MACE in patients with CAD.
Keywords
Introduction
Despite rapid advances in the diagnosis and treatment of coronary artery disease (CAD) in recent decades, CAD is still a leading killer worldwide. 1 According to the Report on Cardiovascular Health and Diseases in China 2021, there are approximately 11.39 million patients with CAD in China, and CAD has become a severe threat to the health of Chinese population. 2 Clinically, stable angina pectoris is the most common type of angina pectoris. According to the current guidelines, for symptomatic angina, beta-adrenergic receptor blockers (BBs), calcium channel blockers (CCBs), and short-acting nitrates can be used as first-line drugs.3,4 In addition, other medications, such as ivabradine, nicorandil, ranolazine, and trimetazidine, are recommended for patients unable to tolerate or effectively respond to the priority agents. 5
BBs are first-line antianginal drugs for the treatment of CAD. 5 Nicorandil, which functions as a nitrate-moiety nicotinamide ester and adenosine-sensitive potassium channel opener, has been recommended for use worldwide as an alternative or combined treatment for angina with other first-line drugs. Nicorandil has a similar effect to nitrates, which are effective for improving stenotic coronary arteries and coronary microcirculation. 5 Nicorandil is widely used in the clinical treatment of patients with refractory angina pectoris. However, in real clinical practice, whether the use of BBs in combination with nicorandil has a greater benefit than the use of BBs alone in the treatment of patients with CAD remains undetermined. In the present study, we performed a real-world observational and multicenter retrospective cohort study to assess the long-term potential of nicorandil combined with BBs in Chinese patients with CAD.
Methods
The study was approved by the Ethics Committee at Tongji Hospital affiliated with Huazhong University of Science and Technology Tongji Medical College (approval No. TJ-IRB201909112). The study was conducted in accordance with the most recent version of the Declaration of Helsinki and the Guidelines for Good Epidemiology Practices. This study was registered at the Chinese clinical trial registry (ChiCTR1900027812).
Population and Study Design
This was a multicenter observational retrospective cohort study. The medical records of the patients were collected for the period between August 2002 and March 2020 from the hospital information systems and other electronic medical records of two tertiary healthcare institutions in Wuhan, China. Eligible patients fulfilled the following criteria: patients aged at least 18 years, were hospitalized for CAD, myocardial infarction (MI), stable angina, or acute coronary syndrome (ACS), received treatment with nicorandil and/or beta-blockers at discharge, and had at least one clinic medical record visit after discharge more than seven days. Patients with any of the following clinical conditions were excluded: Vasospastic angina, variant angina, coronary spasm, latent CAD, asymptomatic myocardial ischemia, restrictive cardiomyopathy, hypertrophic cardiomyopathy, dilated cardiomyopathy, cardiac amyloidosis, congenital heart disease, or key demographic or medical data were missing or erroneous. The “treatment” in this study was defined as a record of the prescription of BBs with or without nicorandil at the time of hospital discharge and the medical records of outpatient clinics. Based on the medical records, the included patients were categorized as a combined treatment group with BBs and nicorandil and a monotherapy group with BBs alone.
Data Collection
Data collection was based on a methodology reported previously. 6 Data on demographics, medical history, treatment course, and outcomes were collected. Demographics included age, sex, history of smoking and alcohol drinking, and medical history of revascularization (percutaneous coronary intervention [PCI] and/or coronary artery bypass graft). Comorbidities included diabetes mellitus, hypertension, hyperlipidemia, ACS, types of angina (stable or unstable), MI, and heart failure. Moreover, use of concurrent cardiovascular medications, including antiplatelet drugs, anticoagulants, statins, CCBs, angiotensin-converting enzyme inhibitors (ACEIs), angiotensin II blockers (ARBs), BBs, nitrates, nicorandil, and mineralocorticoid receptor antagonists (MRAs), were also recorded. This information was primarily extracted from the stored medical and prescription databases. Using medical records from outpatients, the discharge diagnoses of patients with CAD were collected, and follow-up data were collected from inpatient and outpatient databases. Data processing consisted of three stages: data curation, data transformation, and data cleaning. In addition to extracting patients’ basic demographic information, the data curation process encoded the diagnosis of CAD because different staff members may have assigned different categories to CAD terms. During the data cleaning process, special symbols (eg, commas and periods), missing or invalid data, and empty tabs were removed.
Outcome Measures
The primary outcome was the cumulative incidence of major adverse cardiovascular event (MACE) within a follow-up period of 2.5 years, which was defined as the composite outcomes of MI, stroke, and all-cause mortality. The secondary outcomes were cumulative incidence of the MACE components at the 2.5-year follow-up.
Statistical Analyses
Continuous data are presented as mean (standard deviation [SD]) and compared using the two-sample Student t-test or Wilcoxson test. Categorical data are shown as frequency (percentage) and compared using the chi-squared test or Fisher's exact probability test, as appropriate. The cumulative incidence of the primary and secondary outcomes was compared between the two groups of patients, those administered the combined treatment of nicorandil and BBs and those administered BBs alone, through an analysis using the Kaplan–Meir survival curves and log-rank test. The hazard ratios (HRs) and corresponding 95% confidence interval (CI) were calculated using the Cox proportional hazards regression method to indicate the relative risk of the primary and secondary outcomes in patients with the combined treatment compared with those taking BBs alone. Patients with missing data were excluded from the analysis. The incidence density of MACE and their components (per 1000 person-years) was calculated as the number of events divided by the number of years of follow-up, with 95% CIs estimated using exact Poisson limits.
To minimize the potential influence of confounding factors, an inverse probability of treatment weights (IPTW) method, using the calculated propensity score, was applied to adjust the confounding factors between the two comparison groups. 7 Baseline covariates used to calculate the propensity scores were age, sex, smoking and alcohol history, history of revascularization, comorbidities (diabetes mellitus, hypertension, hyperlipidemia, ACS, stable angina, unstable angina, and heart failure), and concomitant medications, including antiplatelet drugs, statins, CCBs, ACEI/ARBs, nitrates, trimetazidine, and MRAs. Standardized differences were calculated for the IPTW-weighted covariates and generally considered representative of acceptable balance is equal to or below 0.10. 8
In addition, sensitivity analyses using IPTW with trimming or limited to patients admitted after January 1, 2012, were performed to evaluate the stability of the findings. First, to reduce any unmeasurable confounding resulting from weakness, the sensitivity analysis was conducted after trimming the stable weight score. Specifically, the two tails of the stable weight distribution are trimmed (<fifth percentile and >95th percentile), as proposed by Stürmer et al. 9 Moreover, because nicorandil has been available in China since 2012, a sensitivity analysis limited to patients admitted after January 1, 2012, was also performed. The E-values were calculated to assess the potential effects of unmeasured confounding factors. 10 Furthermore, subgroup analyses were performed for the association between the combined therapy and incidence of MACE according to the predefined variables: age, sex, smoking and alcohol history, stable angina, unstable angina, previous revascularization, PCI, and histories of diabetes, hypertension, and dyslipidemia. The statistical analyses were performed using SAS 9.4 software (SAS Institute Inc), and P < .05 reflects statistical significance.
Results
Study Population
The flowchart of patient selection and grouping is presented in Figure 1. A total of 137,714 patients were screened, of whom 16,912 individuals (mean age: 61.5 years, men: 67.1%) were successfully enrolled. Among the enrolled participants, 4669 received the combined treatment of nicorandil and BBs, while 12,243 received BBs alone. The median follow-up durations were 10.4 months (interquartile range [IQR]: 2.5-8.0) and 9.7 months (IQR: 2.0-18.0), respectively.

Flowchart of patient inclusion.
Baseline Characteristics
The clinical characteristics of the included patients before and after IPTW are presented in Table 1. Overall, the mean age of the included patients was 61.5 (SD 11.6) years, and 63% of them were ≤65 years. In addition, 67.1% of them were male, 24.4% had a smoking history, and 18.6% had a history of alcohol drinking. Before IPTW, the percentages of smokers and alcohol drinkers were higher in patients receiving the combined treatment than in those receiving BBs alone (both, P < .001). In addition, the proportions of patients with diabetes, hypertension, hyperlipidemia, stable angina, and heart failure were significantly lower in patients with the combined treatment than in those with BBs alone, whereas the proportions of patients with ACS and unstable angina were significantly higher (all, P < .001). The concomitant medications were also unbalanced between the two groups. After IPTW adjustment, the standardized differences for all baseline characteristics were <0.1, suggesting that the patient characteristics were well balanced between the two cohorts.
Baseline Characteristics.

*including percutaneous coronary intervention and coronary artery bypass grafting. Abbreviations: N, sample size before IPTW; n, pseudo sample size after IPTW; ACEI/ARBs, angiotensin-converting enzyme inhibitor or angiotensin II receptor blockers; ACS, acute coronary syndrome; MRAs, mineralocorticoid receptor antagonists; BBs, beta-adrenergic receptor blockers; CCBs, calcium channel blockers; SD, standardized deviation; IPTW, inverse probability of treatment weighting.
Effect of the Combined Treatment on major Adverse Cardiovascular Events and Their Components During Follow-up
The crude cumulative MACE-free survival rate was significantly higher in patients with combined nicorandil and BBs than in those with BBs alone at the 2.5-year follow-up (HR 0.80, estimated 95% CI 0.73-0.87, P < 0.0001; Figure 2A), and the incidence density of MACE was lower in the combined group than in the group with BBs alone (risk ratio [RR] = 0.80, 95% CI 0.73-0.87, P < .0001; Table 2). After IPTW adjustment, the cumulative MACE-free survival rate was still higher in patients with the combined treatment than in those with BBs alone (HR 0.79, 95% CI 0.72-0.87, P = .0042; Figure 2B), suggesting that the combined treatment with nicorandil and BBs was associated with a reduced risk of MACE in patients with CAD during follow-up.
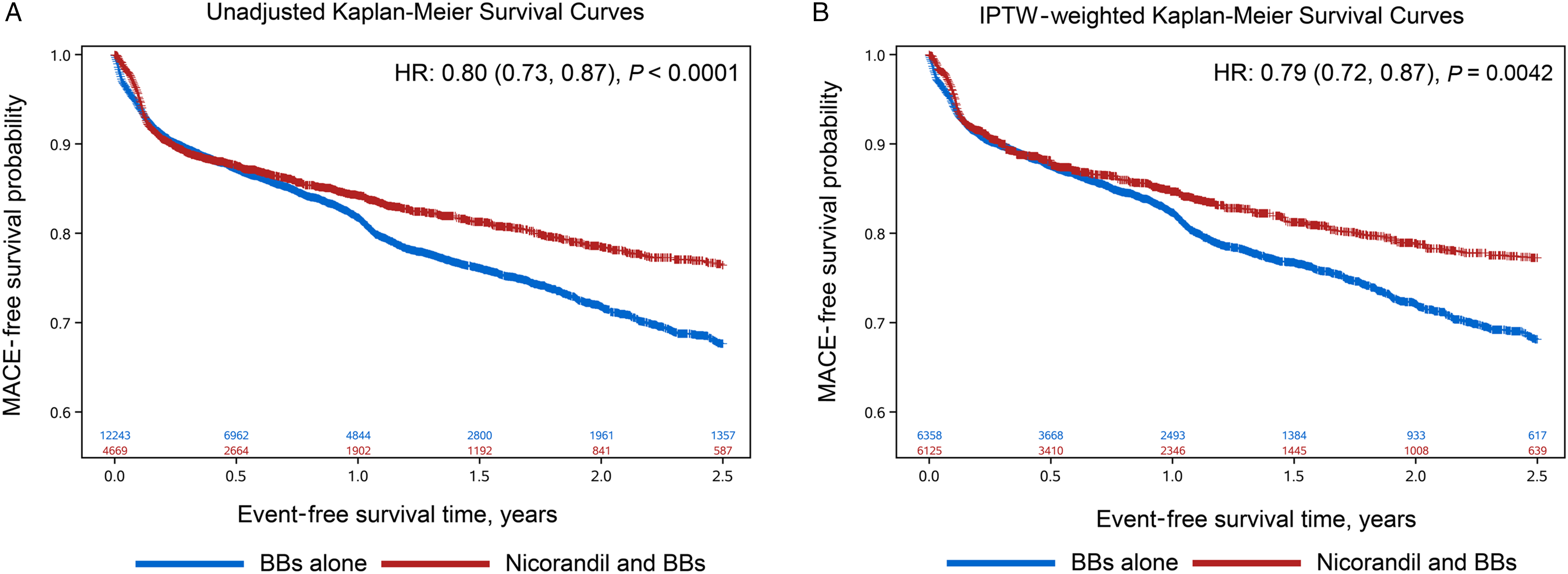
Kaplan–Meier survival curves for the estimation of the accumulative incidence of MACE. (A) Unadjusted Kaplan–Meier survival curves for the incidence of MACE; (B) IPTW-weighted Kaplan–Meier survival curves for the incidence of MACE. Abbreviations: IPTW, inverse probability of treatment weighting; MACE, major adverse cardiovascular event.
IDRs of MACE, MI, Stroke, and all-Cause Mortality of Patients at 2.5-Year Follow-up.

Abbreviations: IDR, incidence density ratio; P-Y, person-years; BBs, beta-adrenergic receptor blockers; MACE, major adverse cardiovascular event; MI, myocardial infarction.
The 2.5-year cumulative stroke-free survival rate was also significantly higher in patients with combined treatment in both the unadjusted (HR 0.43, estimated 95% CI 0.37-0.49, P < .0001; Figure 3A) and IPTW-weighted models (HR 0.48, 95% CI 0.42-0.54, P < .0001; Figure 3B), with a lower incidence density in the combination group (P < 0.0001; Table 2). These results associated the combined treatment of nicorandil and BBs with a reduced risk of stroke in patients with CAD during follow-up. The unadjusted and IPTW-adjusted MI-free survival curves are presented in Figure 3C and 3D, both of which indicate a similar MI-free survival in patients in the two groups (unadjusted HR 1.08, 95% CI 0.97-1.20, P = .1659; IPTW-adjusted HR 1.03, 95% CI 0.92-1.15, P = .6098). The incidence density of MI between patients with the combined treatment and BBs alone was also not significantly different (P = .1494; Table 2). In addition, although the unadjusted overall survival curves showed that the survival rate was higher in patients with the combined treatment than in those with BBs alone (unadjusted HR 0.62, 95% CI 0.43-0.88, P = .0084; Figure 3E), with a similar result observed in the incidence density of all-cause mortality (P = .0144; Table 2), overall survival was not statistically different between patients in the two groups after IPTW adjustment (HR 0.93, 95% CI 0.64-1.37, P = .7259; Figure 3F).

Kaplan–Meier survival curves for the estimation of the accumulative incidence of stroke, MI, and all-cause mortality. (A) Unadjusted Kaplan–Meier survival curves for the incidence of stroke; (B) IPTW-weighted Kaplan–Meier survival curves for the incidence of stroke; (C) unadjusted Kaplan–Meier survival curves for the incidence of MI; (D) IPTW-weighted Kaplan–Meier survival curves for the incidence of MI; (E) Unadjusted Kaplan–Meier survival curves for the incidence of all-cause mortality; (F) IPTW-weighted Kaplan–Meier survival curves for the incidence of all-cause mortality. Abbreviations: IPTW, inverse probability of treatment weighting; MI, myocardial infarction
Sensitivity Analysis and Subgroup Analysis for the Incidence of major Adverse Cardiovascular Events
The sensitivity analyses based on IPTW with trimming and limited to patients admitted after January 1, 2012, are presented in Table 3. They reveal that the combined treatment with nicorandil and BBs was associated with reduced incidence of MACE and stroke (both P < .0001) compared with the treatment of BBs alone. The E-values in the multivariate analysis of 2.5-year MACE-free, stroke-free, MI-free, and overall survival rates were 1.81, 1.17, 3.67, and 1.70, respectively, for the sensitivity analysis with trimming and 1.85, 1.19, 3.63, and 1.37, respectively, for the sensitivity analysis limited to patients admitted after January 1, 2012, indicating the robustness of the results. The results of the sensitivity analyses were consistent with those of the main analyses after IPTW adjustment.
Sensitivity Analyses.

Abbreviations: CI, confidence interval; HR, hazard ratio; IPTW, inverse probability of treatment weighting; LL, lower limit; MACE, major adverse cardiovascular event; MI, myocardial infarction.
Multiple predefined subgroup analyses were also performed to evaluate the association between the combined treatment and incidence of MACE according to multiple patient characteristics (Figure 4). The results revealed that the benefits of nicorandil combined with BBs in patients with CAD were generally consistent for MACE across the stratified subgroup analyses.

Subgroup analyses for the effect of combined nicorandil and beta-adrenergic receptor blockers (BBs) compared with BBs alone on the incidence of major adverse cardiovascular events.
Discussion
In this real-world observational study, we found that a combined treatment of nicorandil and BBs was associated with a significantly reduced incidence of MACE and stroke in patients with CAD with angina compared with patients that received BBs alone, although the risk of MI or all-cause mortality was similar. The robustness of the findings was further validated through multiple sensitivity and subgroup analyses. Taken together, the results revealed that a combined antiangina treatment of nicorandil and BBs may be more effective than treatment of BBs alone in reducing the long-term incidence of MACE in patients with CAD.
The antiangina effect of nicorandil has been validated in many clinical trials, including those with Chinese patients with stable angina. 11 As evidenced in the Impact of Nicorandil in Angina (IONA) study, nicorandil may also have cardioprotective properties. 12 Oral nicorandil could reduce the risk of composite adverse outcomes (all-cause mortality death, non-fatal MI, or unplanned hospital admission for cardiac chest pain) by 17% in patients with stable angina. 12 However, all these results were obtained in randomized controlled trials, and real-world studies evaluating the potential influences of nicorandil on clinical outcomes in patients with CAD with angina are limited. In our previous real-world retrospective study, combined nicorandil and trimetazidine significantly reduced the risk of MACE compared with trimetazidine monotherapy by 15% in patients with CAD. 6 The present study demonstrated that a combination of nicorandil and BBs could benefit patients with CAD by reducing the risk of MACE. The use of BBs has been well established in multiple clinical scenarios with respect to their potential benefits for adverse cardiovascular events, such as in patients with stable angina, ACS, after PCI, or with heart failure.13–17 In pharmacology, nicorandil reduces ventricular preload and afterload by exerting a nitrate-like effect. 18 Thus, nicorandil alleviates angina and protects against myocardial ischemia, which translates into benefits in the composite adverse outcomes. 18 Within a 2.5-year postbaseline follow-up, our study showed that a combined therapy of nicorandil and BBs reduced the risk of MACE by 20% in patients with CAD compared with BBs alone. This result is consistent with the IONA study, which indicated that the additional use of nicorandil decreased the risk of the primary composite endpoint by 17% in British patients. 14 The effectiveness of nicorandil combined with BBs is also robustly supported by the consistent results of the two sensitivity analyses in our study.
The additional benefits of nicorandil combined with BBs were primarily observed in the potential reduction in the risk of stroke in patients with CAD but not in MI or all-cause mortality. These findings are also consistent with the results of the IONA study, in which nicorandil was demonstrated to confer no additional benefits on these two secondary outcomes. 12 However, in a large prospective observational cohort study of patients with CAD (JACD study), added nicorandil reduced all-cause mortality by 35% and decreased other outcomes such as cardiac death (56%), fatal MI (56%), cerebral or vascular death (71%), and congestive heart failure (33%). 19 This difference in results might be because of variations in patients’ pre-existing conditions; in the JACD study, all patients were diagnosed with documented significant stenosis in at least one coronary artery on coronary angiography. Another possible contributing factor was the difference in background medications; only 20% of the JACD patients were prescribed BBs because of concern over vasospastic angina, whereas 100% of patients were prescribed BBs in our study. 20
Our subgroup analyses revealed the benefits of added nicorandil in most subpopulations with respect to MACE, except for smokers, alcohol drinkers, and patients without histories of hypertension or hyperlipidemia. However, the subgroup analyses of the IONA study produced no evidence of significant heterogeneity regarding the benefits of nicorandil across all subgroups studied, with the risk of MACE reduced by between 15.5% and 13.1%. 21 Again, this difference might be explained by the potential variation in the baseline characteristics of the patients as well as their concurrent medications. Although patients in the IONA study had been recently diagnosed with angina, the symptoms seemed to be less severe, and accordingly, BBs were prescribed less frequently (57% and 56% for the nicorandil group and placebo group, respectively). According to the results of the current study, the favorable effects of combining nicorandil with BBs in patients with CAD were primarily attributed to their potential ability to reduce the risk of stroke. The precise mechanisms underlying the beneficial outcomes of the combined use of nicorandil and BBs on MACE and stroke in CAD patients are not yet fully understood. However, some previous preclinical studies have indicated the potential neuroprotective efficacy of nicorandil. For instance, in a rat model of subacute ischemic stroke, nicorandil demonstrated improvements in neurobehavioral and motor function. 22 Additionally, nicorandil has exhibited antineuroinflammatory effects during cerebral ischemic injury.23–25 Further research is necessary to elucidate the underlying mechanisms behind the benefits of combining nicorandil and BBs in patients with CAD. Studies are warranted to further determine the mechanisms underlying the benefits of the combined use of nicorandil and BBs in patients with CAD.
Two sensitivity analyses were performed in this study to evaluate the stability of the findings. Furthermore, the E-value was calculated for each outcome to further indicate the robustness of the results. In general, the E-value method was utilized to determine the strengths of unmeasured confounding factors that could invalidate the results of this observational study. Regarding stroke-free survival, in this study, the HRs of the combination group (vs BB monotherapy) were 0.47 and 0.48 in the sensitivity analyses with trimming and limited to patients admitted after January 1, 2012, respectively, and the corresponding E-values were 3.67 and 3.63, respectively. The results showed that the E-value was relatively larger than the inverse of the HR for the outcome of stroke, demonstrating the robustness of the findings of this study.
This study has some limitations. First, the study design was retrospective, and we were unable to establish a causative link between the real-world treatment and the incidence of MACE. However, this study contributes to the real-world evidence of MACE outcomes in patients medicated with combined nicorandil and BBs, strongly supporting future clinical trials. Second, the baseline data were not consistent among the medical record systems, which might lead to baseline characteristics that were not fully matched between the two groups. Third, the treatment group was determined based on the medications prescribed at the time of discharge, which might not reflect actual drug use. Additionally, we were unable to thoroughly investigate any modifications made to the medication regimen during the follow-up period. Consequently, the potential correlation between medication compliance, changes in medication, and clinical outcomes could not be assessed in this study. However, a statistical trend indicating the superiority of a combined treatment of nicorandil and BBs in the cumulative incidence of MACE and stroke over BB monotherapy was robustly supported by multiple statistical analyses. Finally, the selectivity, pharmacological mechanisms, and indications varied significantly among different individual drugs of BBs. 26 However, an explicit analysis considering all of these potential differences in types, doses, and durations of BB treatments is extremely difficult to conduct because it would be underpowered in our study.
In conclusion, although the results need to be validated in large-scale clinical trials, the present study suggests that a combined treatment of nicorandil and BBs may be more effective than the treatment of BBs alone in reducing the incidence of MACE in patients with CAD, particularly in relation to incidence of stroke.
Footnotes
Acknowledgments
The authors acknowledge Chugai Pharma China Co., Ltd for providing financial support for editing and proofreading services rendered by Medjaden Inc.
Authorship
All named authors meet the International Committee of Medical Journal Editors criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for the publication of this version.
Authors’ Contributions
J.C. and L.Q. made substantial contributions to drafting the manuscript. Z.Z., N.L., and H.S. contributed to the acquisition of clinical data. Z.X. and N.Z. contributed to the study conception, design, and interpretation of data. All authors approved the final manuscript.
Compliance with Ethics Guidelines
The study was approved by the Ethics Committee of Tongji Hospital (Ethics Committee approval number TJ-IRB201909112) in accordance with the most recent version of the Declaration of Helsinki and the Guidelines for Good Epidemiology Practices.
Data Availability Statement
The datasets generated and/or analyzed during the current study are not publicly available due to data confidentiality but are available from the corresponding author on reasonable request.
