Abstract
Background:
Aspirin and clopidogrel dual antiplatelet therapy (DAT) reduce ischemic events in patients with cardiovascular disease. However, recurrent ischemic event occurrence during DAT remains a major concern. This systematic review assesses the efficacy and safety of adjunctive cilostazol to DAT in combination with DAT on reducing clinical adverse events.
Methods:
We searched randomized controlled trials (RCTs) in PubMed, Embase, Cochrane library, clinicaltrial.gov, and Chinese Biomedical Database through July 2011. Pooled risk ratio (RR) with 95% confidence intervals (CIs) was calculated. Two independent reviewers evaluated the included studies. The extracted data were analyzed by Review Manager 5.1.2 (The Cochrane Collaboration, Oxford, UK) and GRADEprofiler 3.6 (GRADE Working Group).
Results:
A total of 7 RCTs (4351 patients) were included in the analysis, with a follow-up period of 6 to 12 months. Pooled analysis showed that cilostazol was associated with a significant reduction in major adverse cardiac events (MACEs; pooled RR 0.69, 95% CI 0.52-0.91; P = .008) and repeat revascularization (RR 0.74, 95% CI 0.61-0.89; P = .002); however, cilostazol was not associated with a reduction in the risk of stent thrombosis (RR 1.00, 95% CI 0.41-2.45; P = 1.00). Cilostazol seems to be safe, with no significant increase in the risk of bleeding (RR 1.06, 95% CI 0.72-1.56; P = .77). The 4 outcomes were low-quality evidence for MACE, moderate-quality evidence for repeat revascularization, and high-quality evidence for bleeding and stent thrombosis.
Conclusions:
When compared to the currently recommended DAT, triple antiplatelet therapy with cilostazol can reduce repeat revascularization with no increase in the risk of bleeding.
Introduction
In patients undergoing percutaneous coronary intervention (PCI) with stent implantation, dual antiplatelet therapy (DAT) with aspirin and clopidogrel is the standard of care. 1 Adequate platelet inhibition by antiplatelet therapy is an important strategy for acute coronary syndrome (ACS) and PCI management; however, the response to aspirin and clopidogrel has substantial interpatient variability. Meta-analysis showed that the mean prevalence of clopidogrel nonresponsiveness was 25%, and the global relative risk ratio (RR) for recurrent ischemic events in clopidogrel nonresponders was 3.5. 2 High on-treatment platelet reactivity (HTPR) after standard DAT is significantly associated with the risk of major adverse cardiovascular events (MACEs). 3 Recurrent ischemic event occurrence during DAT, including stent thrombosis and in-stent restenosis, remains a major concern.
Several studies have aimed to lower the recurrent clinical events, especially in clopidogrel nonresponders by modifying therapy, such as using higher loading or maintenance doses of clopidogrel, adding cilostazol or glycoproteinIIb/IIIa inhibitors, and switching to potent alternative P2Y12 receptor blockers. The current therapeutic alternatives for treatment of clopidogrel resistance remain limited. Currently, one of the appealing strategies for reducing poor response to clopidogrel is adding cilostazol to standard DAT.
Triple antiplatelet therapy (TAT), aspirin and clopidogrel plus the cilostazol, is another treatment that has shown to be beneficial for the patients. Cilostazol is a potent oral antiplatelet agent with a rapid onset of action that selectively inhibits phosphodiesterase III (PDE-3), a mechanism different from aspirin and adenosine diphosphate receptor antagonists. 4 It can also reduce smooth muscle proliferation 5 and lower restenosis after percutaneous coronary balloon angioplasty. 6 A previous study has suggested that cilostazol has similar antiplatelet effects as clopidogrel, 7 and more importantly, clinical trials have shown the beneficial effect of adjunctive cilostazol. 8 –10 During our preliminary literature search, we came across a systematic meta-analysis on the same topic 11 ; however, some larger randomized controlled trials (RCTs) have been published to date. We therefore conducted an updated meta-analysis to compare the efficacy and safety of TAT with DAT on the incidence of MACE and bleeding in patients who underwent successful coronary artery stenting.
Methods
Search Strategy
We searched PubMed, Embase, Cochrane library, clinicaltrial.gov, and Chinese Biomedical Database for all relevant articles through July 2011. Literature was searched with the following keywords: cilostazol, clopidogrel, antiplatelet therapy, and triple antiplatelet therapy. A comprehensive search of reference lists of all review articles and original studies retrieved by this method was performed to identify additional studies. Furthermore, we also searched abstracts of conferences of American College of Cardiology, American Heart Association, Transcatheter Cardiovascular Therapeutics, and European Society of Cardiology.
Inclusion and Exclusion Criteria
Prospective RCTs of cilostazol in combination with aspirin and clopidogrel in patients who underwent PCI with stent implantation were included in our meta-analysis. We considered TAT as experimental and DAT as standard. Exclusion criteria were the following: (1) studies in which end points were not evaluated, (2) studies with ticlopidine therapy, (3) studies with follow-up <6 months, and (4) duplicate reports.
Data Extraction and Assessment of Study Quality
Characteristics of the study population, year of publication, country of origin, number of patients, stent type, dosage and duration of cilostazol administration, study outcomes, and duration of follow-up were obtained. The primary end point was MACE, including cardiac death, myocardial infarction (MI), stroke, and repeat revascularization. The secondary end points were ischemic-driven repeat revascularization (target lesion revascularization or target vessel revascularization), stent thrombosis, and thrombolysis in myocardial infarction (TIMI) bleeding. Two reviewers independently abstracted data and evaluated the quality of selected articles, with divergences resolved after consensus. The methodological quality of eligible studies was assessed by the Cochrane collaboration’s tool for assessing risk of bias including the following criteria: sequence generation, allocation concealment, blinding, incomplete outcome data, selective outcome reporting, and other issues.
Statistical Analysis
Dichotomous data were expressed as RR with 95% confidence intervals (CIs). Heterogeneity of effect size across the studies was tested using Q statistics at the P < .10 level of significance. We also calculated the I 2 statistic with a quantitative measure of inconsistency across the studies. The data were pooled by random effects model in case significant heterogeneity (Cochran test with P < .10 or I 2 > 50%) was found; otherwise, the fixed effects model was used. We conducted a sensitivity analysis to investigate the influence of a single study on the overall risk estimate by omitting 1 study in each turn, and we also conducted subgroup analyses according to geographic region and loading dose of cilostazol to assess the potential effect modification of these variables on outcomes. Potential publication bias was visually inspected by funnel plot. Meta-analysis was performed with the software of Cochrane Review Manager 5.1.2 (Cochrane Library Software, Oxford, UK).
Grading the Evidence of Meta-Analyses
Reliable and valid instruments for evaluating the quality of the evidence derived from systematic reviews and meta-analyses are essential for developing transparent and unbiased clinical recommendations and avoiding implicit subjectivity. For judgment of the strength of the meta-analyzed evidence for recommendations, we applied the GRADE methods, proposed by World Health Organization for producing practice guidelines. We constructed tables by using the GRADE system to summarize the findings and rate the overall quality of evidence. 12,13
Results
Study Characteristics
Figure 1 shows the selection of studies. From a total of 133 initial hits, 7 RCTs 8,14 –19 fulfilled the inclusion criteria and were included in the meta-analysis. Table 1 summarizes the characteristics of the selected studies. The patient characteristics of the 2 groups were well balanced in all the studies. Selected studies include 4351 participants in total, 2175 randomized to TAT and 2176 randomized to DAT. All patients included have stable angina or ACS, specially 3 studies enrolled patients with diabetes mellitus 19 or long lesions. 14,18 Patients of 2 studies 15,16 and 48% of the patients of 1 study 8 underwent bare mental stent placement, and all participants of the other studies underwent PCI with drug-eluting stent (DES) implantation. The methodological quality of the included studies was, in general, good as shown in Table 2.

Study flow diagram.
The Studies Included.
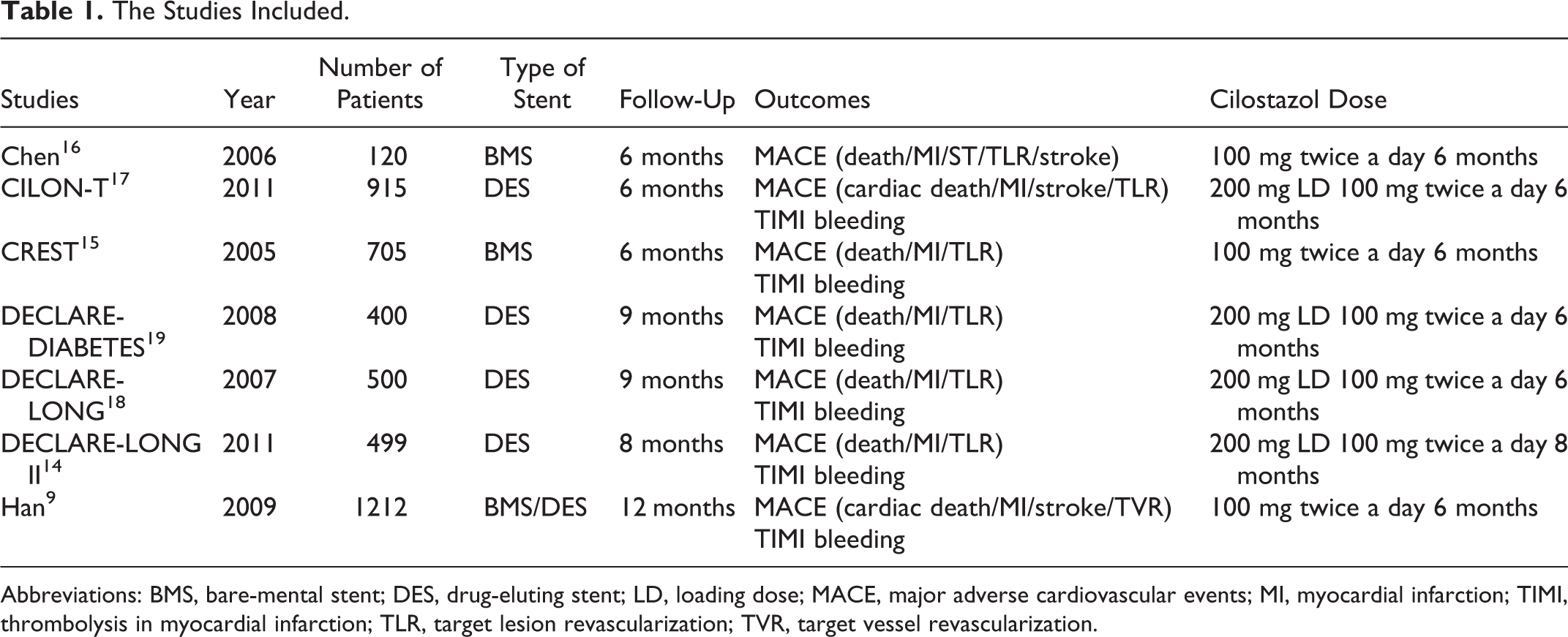
Abbreviations: BMS, bare-mental stent; DES, drug-eluting stent; LD, loading dose; MACE, major adverse cardiovascular events; MI, myocardial infarction; TIMI, thrombolysis in myocardial infarction; TLR, target lesion revascularization; TVR, target vessel revascularization.
Quality Assessment of Included Studies.

Main Outcomes Analysis
For MACE analysis, 7 studies including 4351 patients were included, 204 of the 2175 patients in the TAT group and 276 of the 2176 patients in the DAT group had MACE at the longest available follow-up. As shown in Figure 2, meta-analysis showed that TAT significantly reduced the incidence of MACE (pooled RR 0.69, 95% CI 0.52-0.91; P = .008). We analyzed the efficacy of cilostazol in patients with different stent types. Figure 2 showed that TAT significantly reduced the risk of MACE in the DES group (RR 0.62, 95% CI 0.40-0.94; P = .03), with no reduction of the risk in the bare mental stent (BMS) group (RR 0.67, 95% CI 0.24-1.84; P = .44). In the composition of BMS and DES groups, the benefit of cilostazol was also observed.

The effect of triple antiplatelet therapy on major adverse cardiovascular event (MACE).
Six studies reported bleeding outcomes as defined by the TIMI group. The RR for bleeding showed that there was no significant difference between the 2 groups (RR 1.06, 95% CI 0.72-1.56; P = .77; Figure 3).

The safety of triple antiplatelet therapy on bleeding.
We further evaluated the efficacy of cilostazol in the prevention of ischemic-driven repeat revascularization; Figure 4 showed that TAT significantly reduced the risk of repeat revascularization (RR 0.74, 95% CI 0.61-0.89; P = .002). However, TAT was not associated with a reduction in the risk of stent thrombosis (RR 1.00, 95% CI 0.41-2.45; P = 1.00; Figure 5), death (RR 0.79, 95% CI 0.49-1.28; P = .34), MI (RR 1.08, 95% CI 0.61-1.92; P = .78), and stroke (RR 0.88, 95% CI 0.43-1.80; P = .72; figures not shown).

The effect of triple antiplatelet therapy on repeat revascularization.

The effect of triple antiplatelet therapy on stent thrombosis.
Publication Bias
Funnel plot was constructed using the RR values obtained from MACE. The shapes of the funnel seemed asymmetrical, suggesting that the publication bias in the analysis could not be ruled out.
Sensitivity Analyses
There was no difference in the results between the fixed-effect model and the random-effect model for all outcomes. The sensitivity analysis that omitted 1 study at a time and calculated the combined RR for the remaining studies yielded consistent results with a RR range from 0.63 to 0.73. We conducted subgroup analysis according to geographic region, loading dose of cilostazol, and type of stent, and found that no evidence heterogeneity was observed in the subgroup of Asian patients (P = .22 and I 2 = 28%). The CREST study in the United States was the major contributor to heterogeneity test in the analysis for MACE; however, it could not affect the finding of statistical significance in favor of TAT.
Grading of Meta-Analyses
Following the GRADE algorithm, we downgraded the quality of the evidence of studies investigating MACE because of heterogeneity and publication bias, and the quality of the evidence for repeat revascularization was also downgraded due to publication bias.
Discussion
In recently published systematic reviews, Biondi-Zoccai 20 reviewed RCTs on the angiographic and clinical impact of cilostazol after PCI. They demonstrated that cilostazol was associated with significant reductions in binary angiographic restenosis and repeat revascularization, with no significant increase in the risk of stent thrombosis or bleeding. However, the randomized therapeutic regimens of the included studies were diverse. The benefit associated with triple therapy (aspirin, clopidogrel, and cilostazol) could not be conclusively tested because of the paucity of dedicated trials. Then, Singh 11 evaluated whether TAT has any advantage in efficacy compared to standard DAT in patients undergoing PCI with stent. They found that cilostazol seemed to be effective in reducing the rates of in-stent restenosis without any significant benefit for MACE. However, only 4 RCTs including 1457 patients were included in this analysis. Therefore, we retrieved 7 studies including 4351 patients and updated the data. Our meta-analysis results are more precise and close to the truth than previous studies because of the larger population trials. The added 3 trials, Han, CILON-T, and DECLARE-LONG II trials, included larger population, and the weight of the 3 trials in our meta-analysis was more than 50%. The heterogeneity in our meta-analysis was also lower than Singh’s study.
The results from 7 RCTs showed benefit of TAT in patients undergoing PCI with stent implantation. Our meta-analysis has demonstrated a significant (31%) relative risk reduction in the rate of MACE, especially in the DES group, and similar benefit (26% relative reduction) is also seen in the risk of repeat revascularization. However, cilostazol has no effect on the risk of death, stent thrombosis, MI, and stroke. Notably, cilostazol seems to be safe, with no significant increase in the risk of bleeding.
The MACE was reduced in TAT, mainly driven by reducing repeat revascularization. The relative risk reduction was similar between the 2 outcomes, probably because of the pharmacological profile of cilostazol. Cilostazol is a selective inhibitor of PDE-3 with therapeutic focus on increasing cyclic adenosine monophosphate (cAMP), which is related to an inhibition in platelet aggregation. In addition to its antiplatelet function, cilostazol was also noted for its antirestenosis effects. Cilostazol inhibits neointimal formation via several mechanisms. Cilostazol inhibited stent-induced P-selectin expression on platelets and upregulation of leukocyte Mac-1, which is associated with inhibition of neointimal hyperplasia and restenosis after stent implantation. 21 It also increases the cAMP level in the vascular smooth muscle cells, which upregulates antioncogenes p53 and p21 and hepatocyte growth factor, leading to an antiproliferative effect. 22 Stent thrombosis is of special concern, and cilostazol in our study was not shown to affect the incidence significantly. These data should be interpreted cautiously. First, the absolute risk of stent thrombosis was low (0.6% in both groups), and the follow-up period of the pooled studies was shorter than 12 months. The incidence was so low that potential benefit of cilostazol might be diluted by various influencing factors and insufficient statistical power to detect the difference.
Second, cilostazol, besides its antiplatelet function, was also reported for its antirestenosis effects by inhibiting neointimal proliferation and acting as a potent vasodilator. In this dosage, cilostazol reduced the repeat revascularization, with no influence in the risk of stent thrombosis and bleeding. Maybe, the dose of cilostazol was too low to demonstrate the stronger effect.
For 51% heterogeneity of MACE, we analyzed the possible causes and conducted subgroup analysis. First, there was different composition of patients among the different trials. Second, the stent used in different trials was also diverse. Third, a loading dose of cilostazol was adopted in 4 trials. Fourth, only the CREST trial was conducted in the United States, and others were in Asia. In our subgroup analysis among the above-mentioned diversities, the CREST trial was the major contributor to heterogeneity. The reason may be the difference of geographic region. The antiplatelet effect of clopidogrel is influenced by the genetic polymorphism of CYP2C19, which may be attributed to the high prevalence of CYP2C19 loss-of-function alleles that are associated with poor metabolic activation of clopidogrel in the Asian population.
Remarkably, previous clinical studies demonstrated that adjunctive cilostazol therapy resulted in an intensified inhibition of platelet aggregation, compared to high-maintenance dose clopidogrel in patients with HTPR. 23 Especially, TAT reduced the rate of HTPR compared to DAT even in high-maintenance dose clopidogrel therapy in patients with CYP2C19 loss-of-function allele but not in noncarriers. 24,25 Therefore, cilostazol in combination with clopidogrel can further improve the inhibition of platelet aggregation, especially in patients who have no response or low response to clopidogrel.
The safety profile of TAT has been repeatedly demonstrated among all the trials. No excessive risk of severe bleeding was reported after TAT. Therefore, cilostazol seems to be safe in our analysis with no increase in the rate of bleeding.
Within the GRADE evaluation process, RCTs are basically considered to present high quality of evidence. The following 5 factors may lower the quality of evidence: limitations of individual studies (risk of bias within a study), inconsistency (heterogeneity of results across studies), indirectness of evidence, imprecision, and publication bias. Ultimately, the quality of evidence for each outcome falls into 4 categories from high to very low. In assessing the outcome of MACE, studies are limited by heterogeneity and publication bias, providing low-quality evidence. The studies for assessing the outcome of repeat revascularization were also limited by publication bias and provide moderate-quality evidence. In our assessments for the outcomes of bleeding, stent thrombosis provide high-quality evidence.
Meta-analysis is a powerful tool to summarize the results of multiple studies. However, because of inherent limitations (such as publication, heterogeneity of enrolled studies, and different population), our meta-analysis had several limitations. First, the major studies of cilostazol were established in Asian populations; the patients in 6 of the selected studies were Chinese and Korean populations. The finding needs to be validated in larger patients in the worldwide population. Second, the number of clinical trials was not sufficient, and there is publication bias in our study. So the quality of evidence may have been limited.
In summary, our meta-analysis demonstrates that addition of cilostazol to DAT is a good strategy to attenuate the risk in patients undergoing PCI with stent implantation, especially suggesting that TAT can lower the risk of repeat revascularization with no increase in the outcome of bleeding. However, TAT does not have enough quality of evidence to be applied in practice routinely; high-quality RCTs in worldwide population are needed to confirm the results found in this meta-analysis.
Footnotes
Authors’ Note
The authors Xiao Liang Ding and Cheng Xie contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Nature Science Foundation of China (grant number: 81173132), the Jiangsu Province’s Key Provincial Talents Program (RC2007082), and the JSWSF (clinical pharmacy research grant, grant number: P200959).
