Abstract
Background:
It is uncertain whether remote ischemic conditioning (RIC) has a protective effect on contrast-induced acute kidney injury (CI-AKI) after percutaneous coronary intervention (PCI)/coronary artery angiography (CAG). We performed a meta-analysis of randomized controlled trials (RCTs) to assess the effect of RIC on CI-AKI in such patients.
Methods:
PubMed, MEDLINE, EMBASE, ClinicalTrials.gov, and the Cochrane Central Register of Controlled Trials databases were searched for RCTs that assessed the effect of RIC on CI-AKI in patients undergoing PCI/CAG.
Results:
Ten RCTs with 1389 patients (RIC group, 757 and control, 632) were included. The RIC group significantly exerted a lower risk of CI-AKI compared to the controls (odds ratio [OR] = 0.52, 95% confidence interval [CI] = 0.34-0.77, P = .001), and they had the similar effect on major adverse cardiovascular events within 1 year (OR = 0.36, 95% CI = 0.20-0.66, P < .001). The RIC reduced the rates of death within 30 days, but this was not significant (OR = 0.16, 95% CI = 0.02-1.34, P = .091). The RIC was associated with a significantly lower incidence of CI-AKI in patients following elective PCI/CAG (OR = 0.54, 95% CI = 0.33-0.87, P = .011). The RIC before not after the intervention was effective in reducing the occurrence of CI-AKI (OR: 0.37 vs 1.05, P = .022). The RIC of the upper arm has statistically significant effect on protecting CI-AKI but not that of the lower limb (OR: 0.41 vs 1.41, P = .004). The effect of RIC on CI-AKI was similar between patients with a mean estimated glomerular filtration rate <60 mL/min/1.73 m2 and those with mean rates ≥60 (OR: 0.23 vs 0.41, P = .333).
Conclusion:
The RIC reduced the incidence of CI-AKI in those receiving PCI/CAG. And RIC of the upper arm significantly reduced the risk of CI-AKI but not RIC of the lower limb in patients undergoing PCI/CAG.
Keywords
Introduction
Contrast-induced acute kidney injury (CI-AKI) is a common complication of percutaneous coronary intervention (PCI)/coronary artery angiography (CAG), and it is associated with poor short- and long-term cardiovascular and renal outcomes. 1,2 The incidence of CI-AKI varies from 2% in the general population to ≥50% in high-risk groups. 3 However, apart from periprocedural hydration and limiting the amount of contrast medium (CM), few strategies have been approved as effective for preventing CI-AKI. Therefore, an increasing number of studies have been performed to determine timely and successful strategies for preventing and treating CI-AKI.
Remote ischemic conditioning (RIC), including remote ischemic preconditioning (RIPC) and remote ischemic postconditioning (RIPost), is a method that applies brief nonlethal episodes of ischemia and reperfusion to an organ or tissue that is remote from the target organ or tissue. 4 It has been proven to provide protection, particularly against experimental ischemic–reperfusion injuries of the heart, kidney, brain, and skeletal muscle, 5 –7 and it has a cardiac protective effect in patients undergoing cardiac and vascular surgery. 8,9 Unfortunately, although a number of well-designed and large-scale randomized controlled trials (RCTs) have been performed to assess the protective effect of RIC against CI-AKI, the findings remain controversial and contradictory. 10 –19 Yang et al 20 demonstrated that RIPC may be beneficial for preventing AKI after cardiac and vascular interventions. In contrast, Li et al 21 reported that they could not draw conclusions on the protective effect of RIPC against AKI. These meta-analyses focused on the inconsistent inclusion of patients who were treated with different interventions, such as cardiac surgery, vascular surgery, PCI, and so on. Although the precise pathophysiology of CI-AKI and postoperative AKI are not yet completely clear, it has been reported that 2 scenarios of AKI are somewhat different. 22 However, presently, few meta-analyses have evaluated the renoprotective effect of RIC in patients after CM exposure. Therefore, we aimed to perform a meta-analysis of RCTs on RIC against CI-AKI in patients undergoing PCI/CAG.
Methods
Literature Search Strategy
All studies relevant to RIC in patients undergoing PCI/CAG were identified by searching PubMed (1998 through December, 2014), MEDLINE, EMBASE (1998 through December, 2014), ClinicalTrials.gov, and the Cochrane Central Register of Controlled Trials databases. According to optimal search strategies, we used terms such as remote ischemic conditioning or remote conditioning or remote ischaemic conditioning or remote preconditioning or remote ischemic preconditioning or remote ischaemic preconditioning or remote postconditioning or remote ischemic postconditioning or remote ischaemic postconditioning or RIC. Abstracts presented at the American Heart Association, American College of Cardiology, and European Society of Congress from 2009 to 2014 (if available online) were also searched for additional unpublished data. The language was restricted to English.
Study Selection
Two reviewers (YH-L and WJ-B) independently scanned the selected studies based on their titles and abstracts. The full text of an article was reviewed carefully for inclusion if the topic was unclear after screening its title and abstract. 20 Any disagreements between the investigators were resolved through rereview and discussion with a third reviewer (K-W). Eligible trials were included if they met the following criteria: (1) RCTs of patients undergoing PCI/CAG; (2) RCTs with a control group as a comparison (without RIC), and the treatment method was RIC noninvasively before or after the interventions regardless of whether it involved the upper arms or lower limbs; and (3) RCTs that reported on the incidence of CI-AKI after interventional procedures in both the groups. Trials that did not report on the target populations or outcomes, nonrandomized studies, and duplicate articles were excluded. Moreover, if there were duplicate articles, we chose the article that included complete data.
The primary outcome was CI-AKI, which was defined as an absolute increase in the serum creatinine (SCr) ≥0.5 mg/dL or an increase of ≥25% from baseline to within 2 to 5 days after CM exposure. 23 The secondary outcomes were death within 30 days and major adverse cardiac events (MACEs) within 1 year.
Data Extraction and Assessment of the Risk of Bias
Two reviewers (YH-L and WJ-B) independently extracted data from the identified studies. The authors of the articles were contacted if any additional data were needed. The extracted data included the patients’ demographic characteristics (ie, the number of patients, mean age, and proportion of females and diabetes mellitus), inclusion criteria, contrast dose, protocol for RIC, baseline SCr levels, SCr and estimate glomerular filtration rate (eGFR) after the interventions, definition and incidence of CI-AKI, incidence of death within 30 days, and MACEs within 1 year.
The identified studies were independently evaluated for methodological quality by using the Jadad scoring system, 24 and a quality assessment was judged on the concealment of treatment allocation, similarity of study groups at baseline, eligibility criteria, any blinding procedures used, reporting on those lost to follow-up, and intention-to-treat analysis. 25 Disagreements were resolved by the third reviewer or by reaching a consensus.
Statistical Analysis
Data analysis was performed using R statistical software, version.3.1.2 (R Foundation for Statistical Computing, Vienna), 26 and the meta 27 package was used to perform the meta-analysis. The mean difference and 95% confidence intervals (CIs) were calculated for continuous outcome data, and the odds ratios (ORs) and 95% CIs were calculated and analyzed for dichotomous outcome data. The fixed effect model and the random effects model were used to pool studies. The Q test and I2 were used to evaluate statistical heterogeneity. I2 values of 25%, 50%, and 75% were considered as evidence for low, moderate, and severe statistical heterogeneity, respectively. 28 If the I2 value was >50% or the P value was <.1, we considered it heterogeneous and a random effects model was used for data synthesis. Otherwise, a fixed effect model was used. Additionally, a subgroup analysis was performed based on the types of conditioning (preconditioning or postconditioning), types of PCI (primary or elective), and definitions of CI-AKI. All the tests were 2 tailed, and a P value <.05 was considered statistically significant in the meta-analysis.
Results
Characteristics of the Included Studies and Interventions
We initially generated 739 relevant articles (Figure 1). After excluding 358 duplicates and 339 clearly irrelevant articles, 42 articles remained. The clearly irrelevant articles included nonrandomized studies, and no target outcomes or intervention procedures were described after carefully reading the titles and abstracts. Full-text assessment of the 42 potentially relevant articles finally identified 9 eligible trials. Consequently, 9 RCTs involving 1389 participants who had undergone PCI/CAG met the inclusion criteria and were included in the meta-analysis.

Flow diagram of the study selection.
All the included studies reported on the incidence of CI-AKI in 1389 patients who underwent intervention procedures. The RIC method was performed in 757 patients (treatment group) and 632 patients (control group). The RIC group used an inflatable tourniquet around the upper or lower limbs to result in intermittent ischemia or reperfusion through several cycles of inflations or deflations, while the control group used the same tourniquets but did not inflate to induce ischemia and reperfusion. Of all the studies, 7 performed RIC before the interventions (RIPC), while the other 2 performed after the interventions (RIPost). Elective PCI/CAG was performed in 7 studies, and primary PCI was performed in the other 2 studies. Moreover, 2 studies included all patients with diabetes mellitus 14,18 and 1 study included with moderate chronic kidney disease. 13 Several studies did not mention the details of the hydration protocols before or after CM exposure. 10,14,16,17 The mean baseline SCr level ranged from 0.82 to 1.63 mg/dL, and the mean CM dose ranged from 91.8 to 309 mL (Table 1).
Patients’ Baseline Characteristic and the Interventions of Included Studies.

Abbreviations: RPIC, remote ischemic preconditioning; pre, remote ischemic preconditioning; post, remote ischemic postconditioning; SCr, serum creatinine; eGFR, estimate glomerular filtration rate; PCI, percutaneous coronary intervention; DM, diabetes mellitus; CI-AKI, contrast induced acute kidney injury.
The criteria used to define CI-AKI varied among the individual studies. Two trials defined CI-AKI as a relative increase of >25% from baseline, 10,17 2 trials used the definition of an absolute increase in the SCr of >0.5 mg/dL (44.2 mmol/L) or a relative increase of >25% from baseline to within 16 hours after CM exposure, 12,14 1 trial employed the same within 24 hours, 11,14 and 3 trials within 48 hours. 11,13,15 Additionally, 1 trial defined CI-AKI as a 30% increase or an increase of 0.3 mg/dL in the SCr level. 18
Assessment of the Study Quality and Publication Bias
The quality of the included studies was assessed by 2 reviewers (YH-L and WJ-B) independently using the Jadad score criteria (Table 2). Only 5 studies provided the concealment of allocation, while the other 4 studies did not describe this in detail. Adequate blinding was performed in 7 studies. However, the other 2 studies did not mention it well. Almost all of the studies included patients with similar baseline characteristics, and they provided details about the eligibility criteria and completeness of follow-up and the intention-to-treat analysis. The details of the funnel plot are shown in Figure 2.
Quality of the Included Randomized Controlled Trials.a

Abbreviation: ITT, intention-to-treat.
aYes*: 1 score.
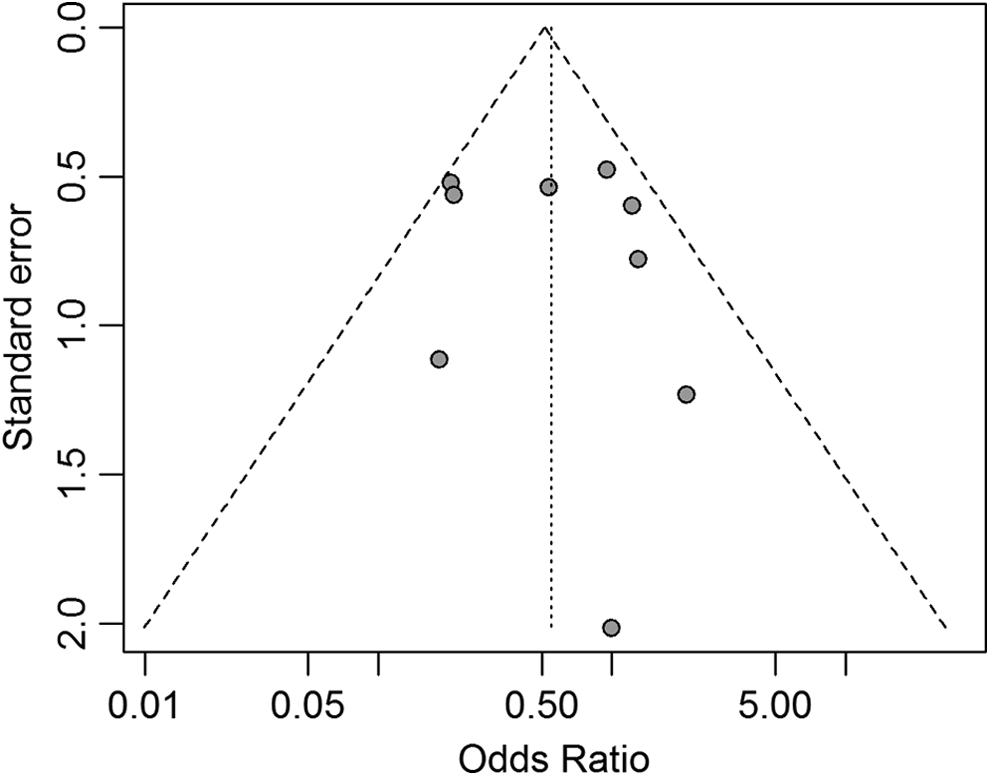
Funnel plot for the subjective assessment of bias among the included studies.
Study Outcomes
Incidence of CI-AKI
Almost all of the studies provided information on the incidence of CI-AKI. Although Igarashi et al 13 did not provide an accurate incidence of CI-AKI based on the SCr levels between the 2 groups, we contacted the author to request the results. In the overall population, RIC significantly reduced the risk of CI-AKI compared to the control group based on the fixed effect model (OR = 0.52, 95% CI = 0.34-0.77, P = .001). The statistical heterogeneity among the included trials was relatively moderate (I2 = 38.9%, P = .109; Figure 3A).
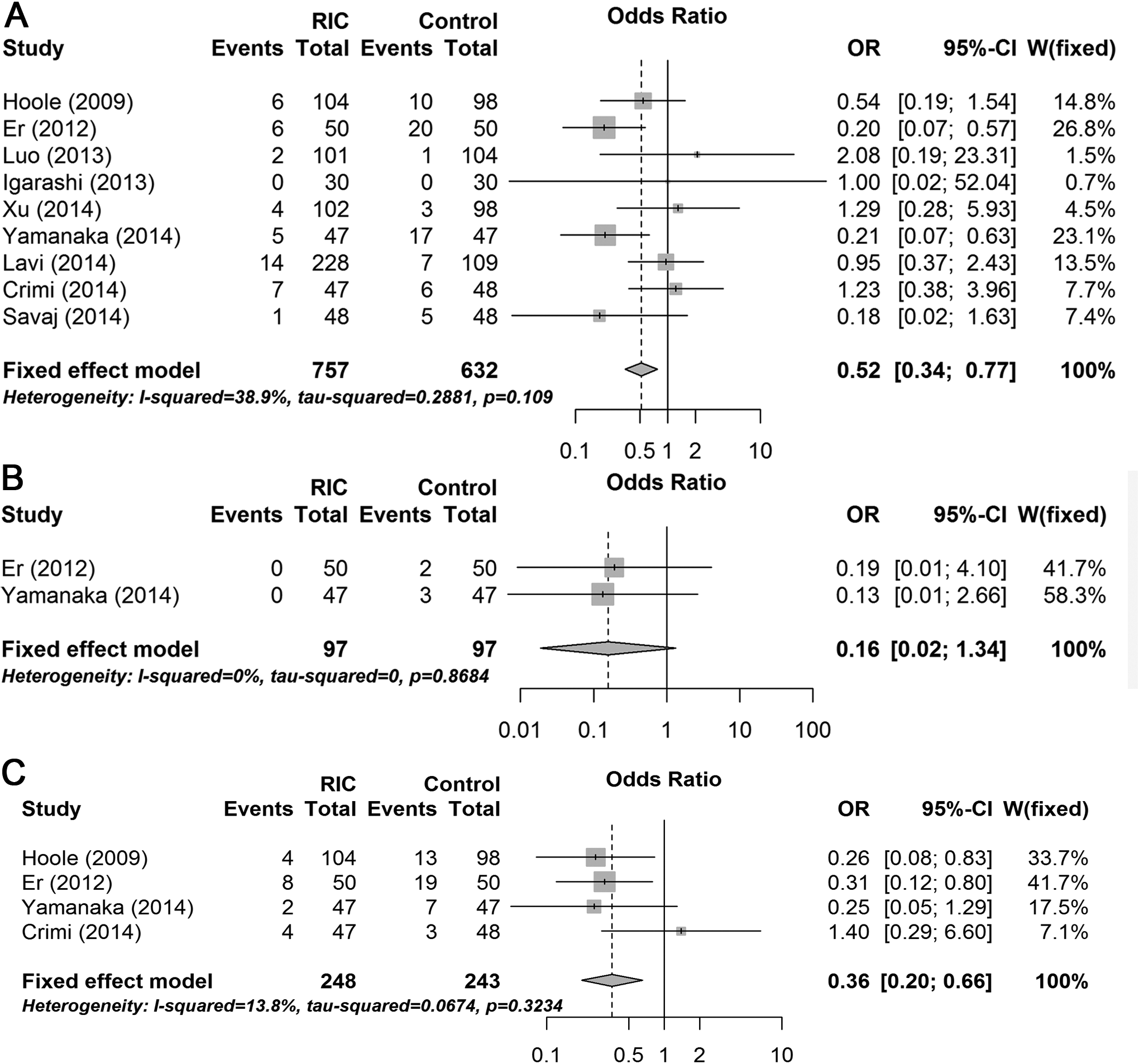
Forest plot of odds ratio (OR) and 95% confidence interval (CI) for contrast-induced acute kidney injury (A), death within 30 days (B), and major adverse cardiovascular events within 1 year (C) among patients assigned to the remote ischemic conditioning compared to the control group.
Death within 30 days and major adverse cardiovascular events within 1 year
There were only 2 trials 11,15 that reported on the incidence of death within 30 days and 5 patients died within 30 days. There was no significant difference between the 2 groups (OR = 0.16, 95% CI = 0.02-1.34, P = .091; Figure 3B). The incidence of MACEs was available in 4 trials, 10,11,15,19 and there were 18 (7.26%) patients in the RIC group and 42 (17.28%) in the control group who developed MACEs within 1 year. The RIC significantly decreased the incidence of MACEs according to the fixed effect model (OR = 0.36, 95% CI = 0.20-0.66, P < .001; Figure 3C).
Subgroup Analysis
We identified 7 trials that represented elective PCI/CAG, while the other 2 studies represented primary PCI. 15,17 According to a fixed effect model, the subgroup analysis demonstrated that RIC may statistically reduce the risk of CI-AKI in patients undergoing elective PCI/CAG (OR = 0.54, 95% CI = 0.33-0.87, P = .011). Because of the small sample size (47/48 and 47/47 patients), there was not enough power to draw conclusions about the effect of RIC on primary PCI.
In addition, since different types of remote conditioning may independently influence kidney function, we classified the trials according to whether they performed the measurements before or after the interventions. Among 9 studies, 7 used RIPC, while 2 used RIPost. 16,17 The RIPC effectively reduced incidence of CI-AKI, while RIPost was ineffective (OR = 0.37 vs 1.05, respectively, P = .022; Figure 4). Moreover, patients who received RIPC had a 63% lower risk of CI-AKI compared to patients in the control group (OR = 0.37, 95% CI = 0.22-0.61; P < .001).

Subgroup analysis of the forest plot of odds ratio (OR) and 95% confidence interval (CI) for contrast-induced acute kidney injury among patients assigned to the remote ischemic conditioning compared to the control according to the conditioning types (preconditioning vs postconditioning).
Furthermore, 2 studies involved the lower limbs, and we divided Lavi et als trial, 16 which included 3 arms (upper, lower, and control), into 2 studies (Lavi I and Lavi II) according to the different conditioning protocols. The RIC of the upper arm significantly prevented CI-AKI (OR = 0.41, 95% CI = 0.26-0.64, P < .001; Figure 5); however, RIC of the lower-limb in patients treated with PCI/CAG did not have the same effect (OR = 1.41, 95% CI = 0.70-2.86, P = .338).

Subgroup analysis of the forest plot of odds ratio (OR) and 95% confidence interval (CI) for contrast-induced acute kidney injury among patients assigned to the remote ischemic conditioning group compared to the control according to the different limbs (lower limbs vs upper arms).
Six trials reported the mean baseline eGFR, and we performed another subgroup analysis according to whether the mean eGFR was ≥60 mL/min/1.73 m2. The result indicated that the effect of RIC on CI-AKI was similar between patients with a mean eGFR <60 and those with eGFR ≥60 mL/min/1.73 m2 (OR: 0.23 vs 0.41, P = .333).
Ultimately, we evaluated the effect of RIC on CI-AKI using different definitions. Six studies defined CI-AKI as an absolute increase in the SCr level of >0.5 mg/dL or a relative increase of >25% from baseline to within 48 to 72 hours. Two other studies defined CI-AKI as a relative increase of >25% from baseline to within 48 to 72 hours. The risk of CI-AKI incidence was not significantly different under the different definitions of CI-AKI (OR: 0.53 vs 0.78, respectively, P = .523).
Sensitivity Analysis
A leave-one-out sensitivity analysis was performed and revealed that our results were sufficiently robust. The OR and 95% CI regarding the effect of RIC on CI-AKI prevention were still similar to the results obtained before excluding the low-quality studies (Jadad score <3) and removing the studies without an intention-to-treat analysis based on the fixed effect model (Figure 6).
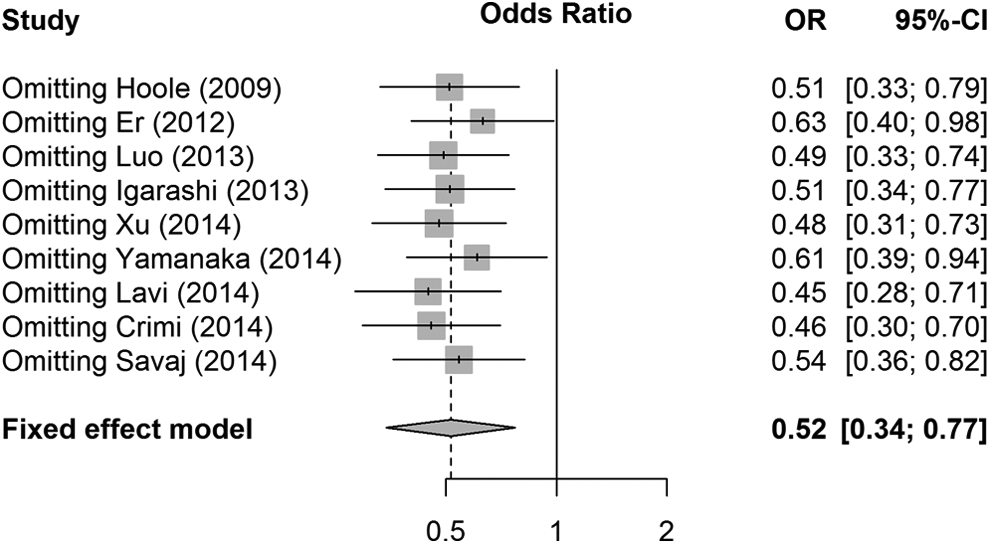
Leave-one-out sensitivity analysis.
Discussion
The present meta-analysis demonstrated that patients treated with RIC may have a significantly lower risk of CI-AKI than control participants in the entire cohort treated with PCI/CAG and in patients undergoing elective PCI. In addition, RIC significantly reduced the risk of MACEs within 1 year but not that of death within 30 days in patients undergoing PCI/CAG. Furthermore, the subgroup analysis indicated that RIPC effectively reduced the risk of CI-AKI. Additionally, RIC of the upper arm had significantly reduced the risk of CI-AKI, but RIC of the lower limb in patients undergoing PCI/CAG did not have the same effect. The protective effect of RIC on CI-AKI was similar between patients with mean eGFR <60 and those with eGFR ≥60 mL/min/1.73 m2. Moreover, the risk of CI-AKI occurrence was not significantly different according to the different definitions of CI-AKI.
The mechanisms of CI-AKI are still unclear. Mechanisms underlying CI-AKI included renal hemodynamics, damage to the tubular epithelial cells and vascular endothelium, medullary vasoconstriction, regional hypoxia, and the production of oxygen radicals, which scavenge nitric oxide (NO) and blunt NO activity. 29 The RIC was considered to play an important role in improving postischemic blood flow. 30 In addition, Gassanov et al showed that RIC may protect and prompt endothelial oxide synthase to enhance the production of NO, 31 which may be one of the medullary vasodilatory factors that reduce the incidence of regional hypoxia. Moreover, RIC may result in fewer productions of reactive oxygen species (ROS), which is an important factor in the late phase of reperfusion, as it reduces damage to the tubular epithelial cells and vascular endothelium. 7 However, low blood flow, less NO, and increased ROS have been proven to be important contributors of CI-AKI development. Therefore, researchers have hypothesized that RIC may reduce the risk of CI-AKI through the aforementioned mechanisms.
The RIC is an emerging treatment that provides intermittent stimulus on an organ (mostly a limb) to increase ischemic tolerance of a distant organ to the subsequent ischemic insult. 32 Previous studies have used different types of ischemic conditioning, which is performed by intermittently reinflating the stent balloon immediately after reperfusion during cardiac intervention. 33 After considering a more simple translation of RIC for clinical setting, we finally chose RCTs that used an applicable, harmess, cost-effective, and noninvasive procedure that simply involved inflating and deflating a blood pressure cuff placed on the upper arm or lower limb.
Although an increasing number of clinical trials have evaluated the effect of RIC on AKI in patients undergoing cardiac and vascular interventions or surgery, the findings are still conflicting and controversial. The precise pathophysiology of CI-AKI and postoperative AKI are not completely similar, and no meta-analysis has specially evaluated the more targeted renoprotection of RIC in patients undergoing PCI/CAG. Therefore, the present meta-analysis may be the first to systematically evaluate the effect of RIC in patients undergoing PCI/CAG. The findings showed that RIC, especially RIPC, significantly reduced the risk of CI-AKI in patients compared to that in the control group after CM exposure. Although Alreja et al 34 and Yang et al 20 performed meta-analyses of RCTs to confirm the protective effect of RIPC on renal injury following cardiac and vascular interventions, other studies have revealed the contradictory. The meta-analyses by Li et al 21 and Haji et al 35 indicated that RIPC had no protective effect on CI-AKI. However, these meta-analyses included participants who had undergone different interventions, such as cardiac and vascular surgery, complex valvular heart surgery, complex congenital heart disease surgery, or PCI. This may have resulted in the significant confounding factors and have greatly influenced the findings of the effect of RIC on AKI; thus, these findings should be discussed further.
Previous meta-analyses have shown similar findings regarding the effect of RIC on mortality rates 21,36 but not on the incidences of MACEs. 36 In addition, some previous studies have also been performed to evaluate the effect of RIC on MACEs in patients undergoing cardiac surgery. However, studies by Hausenloy 37 and Brevoord et al 36 failed to demonstrate statistically significant protective effect of RIC on MACEs within 1 year. This may be because high-risk patients having diabetes and hypertension, different causes of injury during surgery (coronary microembolization and inflammation), and perioperative myocardial injury were included in those studies. However, our present meta-analysis demonstrated that compared to the control group, the group receiving RIC showed a reduced risk of MACEs within 1 year, which was different from the findings of previous studies. The previous meta-analyses included such a broad population that underwent different interventions, instead of a small RCT (1 study), and high-risk patients were excluded, which weakened the reliability of the main findings. Conversely, our findings were sufficiently robust as these problems are not present.
Considering the effect of RIC on different types of PCI, our findings indicated there were no significant differences in CI-AKI between these 2 patient populations. However, compared to the control group, a statistically significant lower risk of CI-AKI was demonstrated in the RIC group that underwent elective PCI/CAG. This finding was consistent with those of Yang et al. 20 Moreover, only 2 studies 15,17 (one on RIPC and one on RIPost) with a limited number of participants were performed in patients treated with primary PCI, and the heterogeneity was severely high (I2 = 78.3%, P = .032). These data may influence our findings. Therefore, future studies are needed to confirm these data.
To date, no meta-analysis has been performed to estimate the effect of RIPost or the differences between RIPC and RIPost. We performed another subgroup analysis to reveal whether RIPC was better than RIPost in reducing the risk of CI-AKI. The findings indicated that RIPC was effective in reducing CI-AKI but not RIPost. However, currently, no large head-to-head studies have been performed to evaluate whether RIPC is similar or superior to RIPost in reducing the risk of CI-AKI. Our meta-analysis only included 2 trials 16,17 that analyzed patients who were treated with RIPost. Thus, caution should be exercised when interpreting these findings. Future research should compare RIPC to RIPost in order to determine whether they exhibit different capacities for reducing the risk of CI-AKI.
In addition, we may be the first to demonstrate the different effects of RIC on the upper arm compared to the lower limb with regard to CI-AKI. Our findings showed that RIC of the upper arm had a statistically significant effect on protecting CI-AKI; however, RIC of the lower limb in patients treated with PCI/CAG did not have this same effect. Some previous studies have demonstrated the protective effect of RIC on the lower limb, 19,38 which represents approximately 2 times the body surface and even more tissue volume than the upper limb to elicitate organ protection. The different mechanisms of RIC between the upper arm and the lower limb remain unclear. In addition, no large head-to-head studies have been performed to evaluate whether RIC of the upper arm is different from RIC of the lower limb for reducing the risk of CI-AKI. Therefore, future research should compare these two types of RIC to determine whether they exhibit different capacities for reducing the risk of CI-AKI.
Furthermore, previous study has demonstrated that chronic kidney disease (eGFR <60 mL/min/1.73 m2) is the most important risk factor for the development CI-AKI. 39 Our subgroup analysis indicated a similar effect of RIC on CI-AKI either in patients with a mean eGFR value of <60 mL/min/1.73 m2 or in those with an eGFR ≥60 mL/min/1.73 m2. The present meta-analysis may be the first to demonstrate this issue. However, we should consider that only 2 studies were included in the group of patients with a mean eGFR value of <60 mL/min/1.73 m2. In addition, no studies have included patients with a mean eGFR of <60 or ≥60 mL/min/1.73 m2. Therefore, we used the mean eGFR value to perform this subgroup analysis. This finding should be interpreted with caution, and more RCTs should be carried out in further research.
The incidence of CI-AKI varied widely according to the different definitions of CI-AKI, with a reported incidence ranging from 2% to 50%. The different incidence of CI-AKI may be an important influencing factor on the effect of RIC for CI-AKI. However, no studies or meta-analyses have performed a subgroup analysis using the different definitions of CI-AKI in order to evaluate the SCr values needed for predicting CI-AKI. The present meta-analysis may be the first to demonstrate that the risk of CI-AKI was not significantly different according to the different definitions of CI-AKI between the RIC and the control groups. Of note, only 2 trials used the single absolute increase in the SCr level to define CI-AKI; therefore, more RCTs should be performed to evaluate the effect of RIC against CI-AKI using a uniform definition.
Study Limitations
The present study has several limitations. First, few RCTs associated RIPost with primary PCI, which may make the relevant conclusions controversial. Second, the definition of CI-AKI varied among the individual studies, and this may influence the final incidence of CI-AKI. Third, several trials did not mention the hydration protocols, which plays an important role in the occurrence of CI-AKI, and we did not have access to data in order to estimate the impact. Fourth, despite the practical use of SCr in defining CI-AKI, there are limitations in its utility as a surrogate marker of renal function. Finally, the studies included in the present meta-analysis only used the SCr to define the CI-AKI, and they did not examine cystatin C, which is a more sensitive biomarker that increases faster than SCr after CI-AKI.
Conclusion
The present meta-analysis indicated that RIC may be associated with a significant lower incidence of CI-AKI compared to the control group in patients undergoing PCI/CAG, with the same incidence in MACEs within 1 year but not death within 30 days or the renal injury biomarkers. In addition, RIC may significantly develop a 54% lower risk against CI-AKI in patients undergoing elective PCI. Furthermore, RIPC, not RIPost, was effective for reducing CI-AKI. Additionally, RIC of the upper arm had a statistically significant effect on preventing CI-AKI, while RIC of the lower limb in patients undergoing PCI/CAG did not have this same effect. The effect of RIC on CI-AKI was similar between patients with a mean eGFR value <60 mL/min/1.73 m2 and those with an eGFR ≥60 mL/min/1.73 m2. Finally, no significant difference was observed based on the different definitions of CI-AKI. Thus, more studies based on a uniform definition of CI-AKI with similar intervention procedures will provide more evidence in the future.
Footnotes
Authors’ Contribution
W-J Bei and Y Liu contributed to acquisition, analysis, and interpretation; critically revised manuscript; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. C-Y Duan contributed to analysis, drafted the manuscript, critically revised manuscript, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. K Wang contributed to acquisition, critically revised manuscript, and agreed to be accountable for all aspects of work ensuring integrity and accuracy; J-Y Chen, Y-H Liu, and N Tan contributed to acquisition, analysis, and interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Authors’ Note
Wei-
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
