Abstract
This review examines the rationale for using remote ischemic conditioning (RIC) in elective percutaneous coronary intervention (PCI) to prevent procedure-related ischemia–reperfusion injury and justifies the importance of periprocedural biomarker elevation following elective PCI as a valid target for RIC. We review the evidence for the use of RIC as a treatment in this setting and document the salutary rules that must be followed to successfully translate RIC for clinical benefit.
Keywords
Introduction
Ischemic preconditioning (IPC) is a protective response to prolonged periods of ischemia and subsequent infarction, which is elicited by brief periods of repeated tissue ischemia followed by reperfusion. 1 Ischemic preconditioning was first discovered by Murry et al in a canine model where transient coronary artery occlusion, followed by a period of reperfusion, resulted in an impressive reduction in infarct size after a more prolonged but still, importantly, transient ischemic episode. This protection was independent of collateral flow. It was subsequently demonstrated that this effect remained, even when the transient ischemia occurred in a remote vascular bed. Przyklenk et al found that occlusion followed by reperfusion in 1 coronary territory led to protection from infarction during prolonged ischemia in another territory. This phenomenon was termed remote IPC (RIPC). 2 Crucially for safe translation, RIPC can be induced in a wide range of vascular beds including skeletal muscle, which is accessible and far less vulnerable to the deleterious effect of transient ischemia required to elicit RIPC. In humans, skeletal muscle ischemia can be produced by successive rounds of inflation of a sphygmomanometer cuff around a limb; this noninvasive approach has facilitated translational research into the potential benefits of RIPC to limit the deleterious clinical consequences of ischemia–reperfusion (IR) injury.
The signal for RIPC is mediated through both neural and humoral mechanisms, which are reviewed elsewhere. 3,4 Remote IPC only protects against IR injury, and although the protective effect is potent, fixed occlusion of the coronary artery will result in infarction and cell death irrespective of RIPC. Protection is maximal beyond a threshold of stimulation, and classical (early) conditioning begins to wane after 1 to 2 hours. The required threshold varies depending upon patient characteristics such as biological age 5 and diabetes mellitus, 6 and in the case of the latter, a greater stimulus (further rounds of transient IR) may be needed to activate the all or nothing protective effect. The second window of protection occurs 24 to 72 hours after the original RIPC stimulus as a result of changes to gene transcription and other intracellular processes. 7
The unpredictability of the onset of ischemia presents a challenge to the use of RIPC to protect the myocardium against IR injury. Myocardial infarction (MI) is usually unheralded, and therefore, RIPC (or similar perconditioning protocols) may not reach the threshold required for protection prior to reperfusion. This will negate any benefit of RIPC, as the majority of reperfusion injury occurs within 5 minutes of opening the infarct-related artery. However, elective percutaneous coronary intervention (PCI) and coronary artery bypass grafting (CABG) are situations in which ischemia is predictable and transient, and RIPC may have a therapeutic effect. Disappointingly, recent trials assessing RIPC in CABG (ERRICA and RIPHeart) have been negative. 8,9 However, in surgical revascularization, there are confounders that may explain the lack of RIPC efficacy: propofol was frequently used as the general anesthetic agent, which may interfere with successful RIPC signal transduction and the drugs, cooling and transient ischemia required to establish a patient on cardiac bypass may replicate cardioprotection in the control group. Neither of these confounders exist during elective PCI and this may be a clearer model to translate the clinical benefits of RIPC.
Consequences of MI During Elective PCI
Figure 1 illustrates the potential targets of IR injury during elective PCI that may be potentially abrogated by RIPC. Inflation of balloon-mounted coronary stents obstruct flow, combined with the loss of “jailed” side branches behind the stent, and the embolization of “plaque gruel” and vasoactive substances into the distal microvasculature 10,11 are all mechanisms of lethal and nonlethal myocardial injury after elective PCI. Iodinated contrast can also cause an IR injury to the kidney.
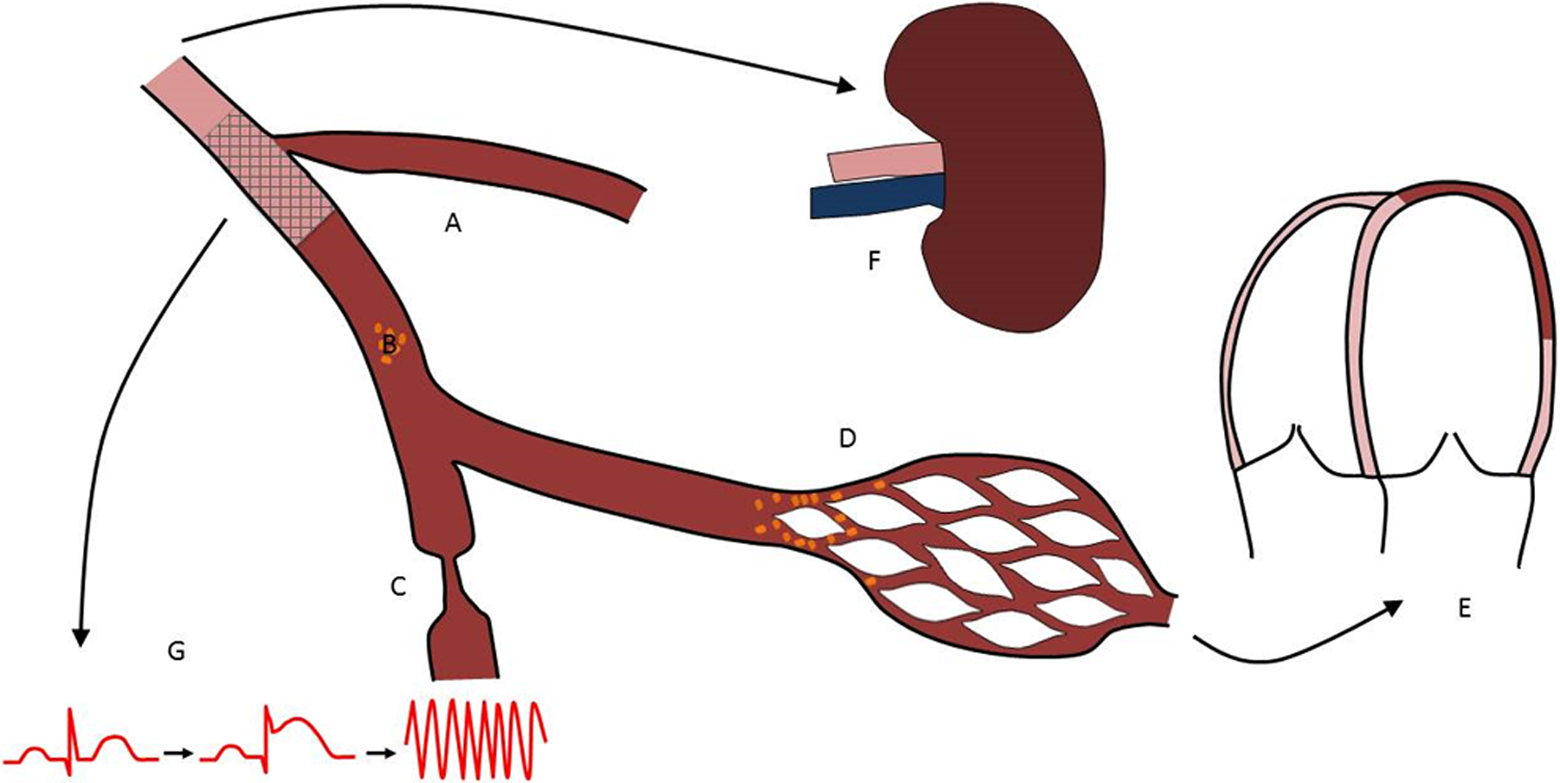
Ischemia–reperfusion injury during elective PCI. Elective PCI risks ischemia–reperfusion injury through a number of mechanisms. The potential loss of coronary side branches (A) directly contributes to ischemia during the procedure. Flow reduction may be transient or fixed. Debris released from the plaque during angioplasty (B) may embolize into the distal vessel and microcirculation, whereas vasoactive substances (C) may cause (transient) vasoconstriction of the coronary vasculature. The combination of vasoconstriction, loss of side branches, and microemboli may obliterate flow in the microcirculation (D). This injury may result in transient or permanent ventricular dysfunction (E). Contrast-induced afferent arteriolar vasoconstriction may contribute to contrast-induced nephropathy (F) that may be exacerbated by ischemic ventricular dysfunction and hypotension. Furthermore, ongoing ischemia may result in electrical changes leading ventricular arrhythmia (G). PCI indicates percutaneous coronary intervention.
A number of procedural factors including stent length and procedure complexity have been associated with PCI-induced lethal injury. 12 The degree of myonecrosis during elective PCI can be assessed using a number of different end points. The most recent third universal definition of MI defines a PCI-associated MI (MI4a) as a rise in high-sensitivity cardiac troponin (cTn) to greater than 5× the 99th centile of the reference range. 13 Cardiac magnetic resonance imaging has confirmed that cTn rise correlates with late gadolinium enhancement volume both adjacent to the stent (side-branch loss) and in the down-stream distribution of the target vessel (distal embolization). 14 The prognostic value of biomarker evidence of myonecrosis after elective PCI has been extensively debated. 15 While postprocedural troponin rise has been shown to be predictive of major adverse cardiovascular events (MACEs), particularly when levels exceeded the third universal definition for MI4a, 16 other studies have refuted a strong association between smaller post-PCI troponin elevations and prognosis. 17 ∓19 A meta-analysis of 20 studies confirmed a weakly positive association between troponin elevation and long-term mortality. 20 Most recently, a large study of 5626 patients found no relationship between postprocedural troponin T above the upper reference limit and mortality at 3-year follow-up. It was noted by the authors that there was a trend toward interaction with mortality when the troponin rise was greater than 7× the 99th centile of the reference range. 21 The prognostic importance of post-PCI troponin is accentuated in those with preprocedural left ventricular (LV) dysfunction and chronic renal impairment. 22,23 Studies enriched for high-risk complex PCIs have a reported MI4a rate between 25% and 50%. 24,25 Studies targeting high-risk individuals undergoing complex PCI are warranted on clinical grounds, and potentially, this will maximize the chance of demonstrating a clinically important protective effect of RIPC.
Other nonlethal PCI-induced myocardial ischemic injuries include ventricular arrhythmias such as ventricular tachycardia or fibrillation, microcirculatory dysfunction (no-reflow), and LV stunning. All exacerbate myocardial ischemia after PCI. Ischemic LV dysfunction that persists despite successful restoration of myocardial blood flow is termed stunning. 26 Cumulative ischemic LV dysfunction can occur following multiple coronary balloon occlusions. It may be particularly important in patients who are vulnerable to periprocedural acute heart failure, such as those with existing LV dysfunction.
There is evidence that contrast-induced nephropathy (CIN) is an acute kidney injury (AKI) caused by contrast-induced afferent arteriolar vasoconstriction and medullary hypoxia as well as direct nephrotoxicity. Contrast-induced nephropathy is traditionally defined as a 25% increase in serum creatinine from baseline or 0.5 mg/dL (44 µmol/L) increase in absolute value, within 48 to 72 hours of intravenous contrast administration, although calculated estimated glomerular filtration rate and cystatin C (a protein secreted by nucleated cells and freely filtered by the kidney) may be more accurate. Neutrophil gelatinase-associated lipocalin also represents an early predictive troponin-like biomarker for AKI. 27 Those with preexisting renal impairment, diabetes mellitus, and receiving larger volumes of contrast who are elderly individuals and hemodynamically unstable are particularly at risk. Contrast-induced nephropathy is associated with increased mortality. 23,28,29
Remote IPC Protection During Elective PCI
Remote IPC has been used in many clinical studies to attempt to protect against IR injury. Usually, inflation and deflation of a pneumatic sphygmomanometer cuff around a limb are used to induce brief IR prior to PCI. The precise protocol varies between studies and, as discussed below, is a contributor to some of the heterogeneity in clinical trial outcomes of RIPC.
The largest study of RIPC during elective PCI to date (the Cardiac Remote Ischemic Preconditioning in Coronary Stenting [CRISP]-stent trial) randomized 242 patients stratified for diabetes mellitus to receive either RIPC (three 5-minute upper-arm sphygmomanometer cuff inflations to 200 mm Hg with 5-minute intervals of reperfusion) or a sham procedure (deflated upper-arm cuff applied). 30 This study demonstrated reduction in median postprocedural troponin rise at 24 hours compared to control. There was also an increase in the number of participants with undetectable troponin levels at this time point. Periprocedural measures of myocardial ischemia such as chest pain score and ST-segment deviation on electrocardiogram (EKG) were also reduced. Intriguingly, the MACE (death, MI, stroke) rate at 6-month follow-up was significantly reduced in the RIPC group compared to control. Long-term follow-up at 6 years confirmed that the reduction of MACE rate in the RIPC group was maintained. 31 Although the CRISP-stent trial was not powered to detect a difference in MACE, it was hypothesized that the diminished post-PCI troponin levels translated to a meaningful clinical benefit in the RIPC group with a number needed to treat with RIPC to prevent a MACE at 6 of 8 years.
These findings were encouraging but other studies have produced varying results. Table 1 summarizes all published studies of RIPC applied to elective PCI to date. These are characterized by small, single-center studies with nonstandardized protocols. Negative studies have included very low numbers of participants with inadequate power to detect an effect of RIPC 32 or have used a subthreshold dose of RIPC that would not have elicited a cardioprotective signal. 34 In another negative study, RIPC was assessed in 200 elderly patients with diabetes mellitus. This is a group known to be resistant to the standard dose of RIPC, but further cycles of RIPC were not performed to compensate for the elevated threshold for protection. 24 Dissemination of the biological rules of RIPC to inform trial design may help to reduce the heterogeneity of trial results in this field—see below. 38
Randomized Trials of RIPC During Elective PCI to Reduce Cardiac Troponin Elevation.

Abbreviations: BP, blood pressure; CRP, C-reactive protein; MACE, major adverse cardiovascular event; MI, myocardial infarction; PCI, percutaneous coronary intervention; RIPC, remote ischemic preconditioning.
Few studies have examined the effect of RIPC on LV dysfunction in isolation. Reports of improved ventricular function after RIPC have not been able to distinguish whether the effect is a result of diminished infarct size or a direct effect on ventricular contractility. 39,40 In human models of supply (transient coronary balloon occlusion) and demand (dobutamine-stress) ischemia, assessing nonlethal ischemic LV dysfunction by conductance catheter and tissue Doppler echocardiography, respectively, RIPC did not prevent myocardial stunning or cumulative ischemic dysfunction compared to control. 41
Contrast-induced nephropathy is a frequently observed (incidence 2% to 5%) complication of coronary angiography and PCI. It is associated with increased hospital stay, as well as long-term morbidity and mortality. Of particular importance is the progression to dialysis in those patients with preexisting renal impairment. The effect of RIPC on CIN after elective PCI has been assessed either as a primary of secondary end point. One study of patients treated for non–ST-segment elevation myocardial infarction reported RIPC-induced protection from AKI. 42 Patients with moderate preexisting renal impairment have been shown to benefit from RIPC before both PCI and diagnostic angiography. 43,44 Er et al showed that 4 cycles of 5-minute sphygmomanometer cuff inflation prior to diagnostic angiography in a population with preexisting renal impairment reduced the incidence of CIN from 40% to 12% (odds ratio, 0.21; 95% confidence interval, 0.07-0.57; P = .002). 41 A recent meta-analysis of all randomized studies of RIPC reporting AKI demonstrated that it was beneficial following PCI. 45
Challenges and Future Directions
While assessing troponin release after elective PCI has historically been a useful translational model to demonstrate the beneficial effects of RIPC, MI4a has been less common in recent years. Unpublished data from our own institution have confirmed falling rates of postprocedural troponin elevation after elective PCI from a high of 49.5% in 2006 to 27% in 2011 and now 8.2% in 2016. This has occurred despite lowering the threshold definition of MI4a and utilizing high-sensitivity troponin assays. Evolving PCI techniques, simplifying previously complex procedures (eg single stent strategy for bifurcation treatment 46 ), and improvements in stent technology, for example, thinner struts, appear to have made elective PCI safer. The diminishing MI4a rate and continued doubt about the prognostic importance of post-PCI troponin elevations may explain the reticence to adopt RIPC into mainstream clinical practice. Appreciation of the lower MI4a rate is crucial in interpreting contemporaneous literature; RIPC only works when cardioprotection is required. The ability of small-scale, single-center studies to demonstrate the benefits of RIPC by assessing troponin elevations is over. Large multicenter trials assessing mortality are now required and are justified, 31 but even these may be too large to be feasibly accomplished in elective PCI, although emergent primary PCI 47 may be achievable and are ongoing (ERIC-PPCI: NCT02342522 and CONDI-2: NCT01857414).
The published literature of translational research into cardioprotection is littered with negative studies; protocols that breach the biological rules of conditioning and are poorly designed are partly to blame. Table 2 outlines a manifesto for the optimal design of future translational studies of conditioning and cardioprotection in elective PCI. These human trials of RIPC need to be conducted on a larger scale, with a focus on high-risk patients to demonstrate prognostic benefit from RIPC. High-risk groups that may deserve attention include complex lesions, including those requiring rotational atherectomy or bifurcation treatment, multivessel disease, PCI to saphenous vein grafts, or large territories needing revascularization. Patients at high risk of CIN are also an important focus for further investigation.
A Manifesto for Trial Design to Investigate RIPC in Elective PCI.
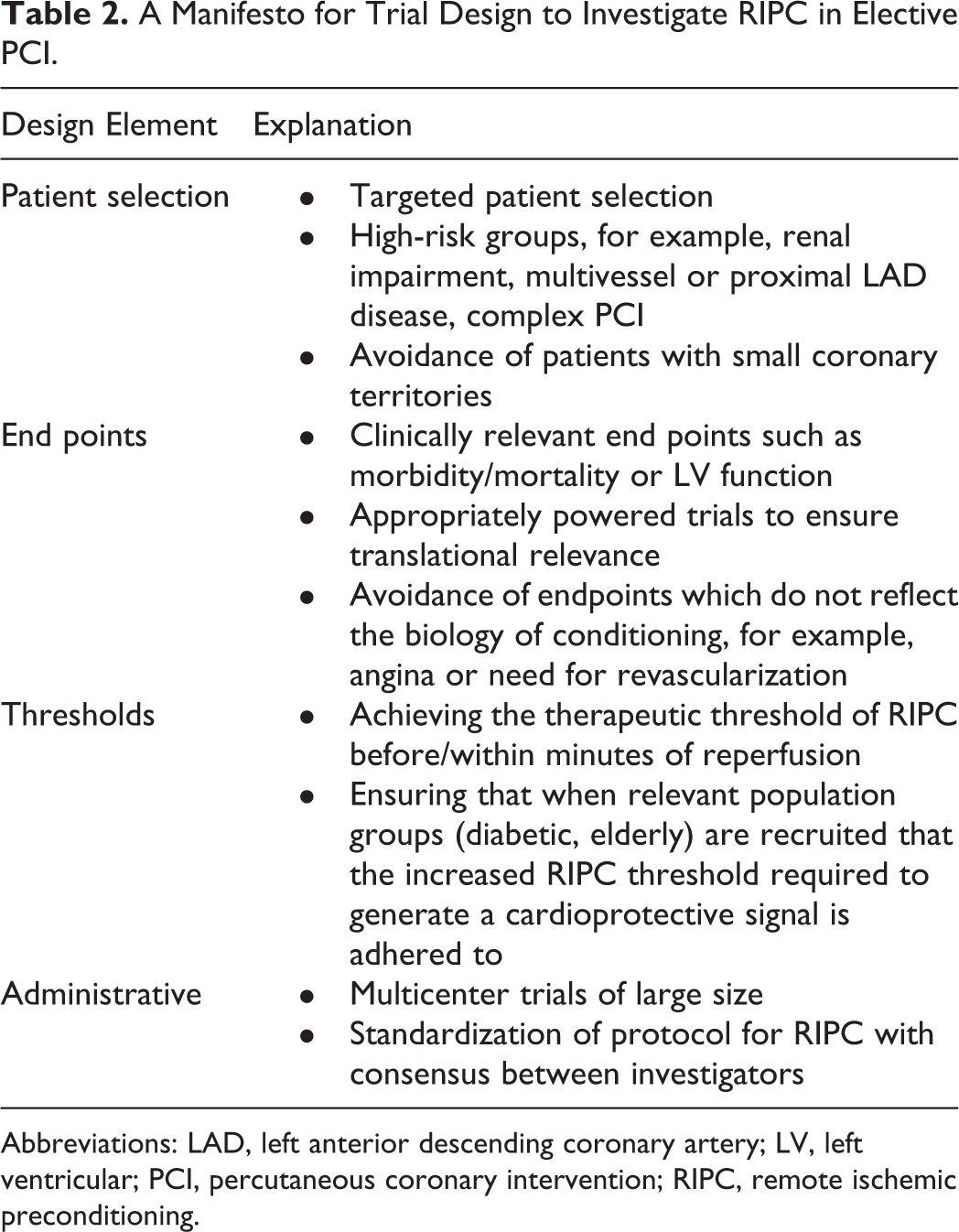
Abbreviations: LAD, left anterior descending coronary artery; LV, left ventricular; PCI, percutaneous coronary intervention; RIPC, remote ischemic preconditioning.
Conclusion
Elective PCI can be complicated by the risk of myocardial and renal IR injury. In high-risk groups, the clinical consequence of these injuries may be clinically important. Remote IPC is a safe and inexpensive treatment that remains a promising potential treatment for IR injury during elective PCI. Unlocking its possible benefits will require close scrutiny of trial design and the engagement of large numbers of investigators in producing bigger studies powered for hard clinical end points to justify its continued translation as a clinically meaningful treatment.
Footnotes
Authors’ Contributions
Hoole drafted manuscript, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring itegrity and accuracy. Giblett drafted manuscript and critically revised manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
