Abstract
Background:
Different antithrombotic treatments, from vitamin K antagonists to direct oral anticoagulants (DOACs), are available to reduce ischemic risks in patients with atrial fibrillation (AF) after percutaneous coronary intervention (PCI).
Methods:
A network meta-analysis and a stochastic multicriteria acceptability analysis (SMAA) were performed including randomized controlled trials (RCT) that evaluate antithrombotic treatments in adults with AF and PCI. Searches were conducted in PubMed and Scopus (updated November-2019). Outcomes compared included bleeding, stroke, and death (Prospero registration: CRD42019146813).
Results:
Five RCTs were included (11 532 patients). Vitamin K antagonists + dual antiplatelet therapy was associated with major bleeding (odds ratio: 0.52 [95% CI: 0.32-0.86]) compared to DOAC + P2Y12. No statistical differences were found among DOAC regimens for the main outcomes, including bleeding, stroke, and death. Surface under the cumulative ranking curve analysis (SUCRA) and SMAA demonstrated edoxaban 60 mg + P2Y12 inhibitor as the worst option (28%). Apixaban 5 mg + P2Y12 inhibitor was the safest alternative (63%) in all scenarios.
Conclusions:
Insufficient evidence on the clinical superiority among anticoagulant regimens exists, although apixaban slightly stands out. Edoxaban was associated with more adverse events. To strength this evidence, well-designed, low risk of bias clinical trials are needed. Cost-minimization analyses are required to provide further information for clinical decision-making.
Introduction
Atrial fibrillation (AF) is the most prevalent cardiac arrhythmia and one of main causes of heart failure, stroke, and cardiovascular morbidity. 1 Arrhythmia occurs in 20% to 40% patients after myocardial revascularization surgery. About 5% to 15% of these patients undergo stenting and antithrombotic therapy as the main therapeutic procedures. 2 In 2010, 33.5 million people worldwide suffered AF with 1.6 to 5 million requiring stenting and antithrombotic therapy. 3 By 2030, the number of AF patients in Europe is expected to be around 14 to 17 million, with an incidence of 120 000 to 215 000 new cases per year. 4
Antithrombotic therapy in patients with AF undergoing percutaneous coronary interventions (PCI) combines oral anticoagulant (OAC) and antiplatelet therapy (APT), aiming at reducing cardioembolic events and bleeding. Different APT alternatives (ie, aspirin, clopidogrel, prasugrel, ticagrelor) and OACs (ie, vitamin K antagonists [VKA] or new direct oral anticoagulants [DOACs]) exist, offering clinicians a wide range of therapeutic combinations. Nonetheless, this implies the challenge of selecting the best treatment for each scenario. Although the dual antiplatelet therapy (DAPT) with P2Y12 inhibitor + aspirin, or triple therapy that combines DAPT + OAC are commonly used in clinical practice, these treatments were associated to a significant increase in the risk of bleeding. 2,5,6
After the introduction of the DOAC, a large number of clinical trials were conducted to demonstrate the profile of these drugs in different clinical conditions. 7 -9 However, studies comparatively evaluating the effects of these drugs are limited in some specific patients’ groups. The available evidence of systematic reviews with conventional pairwise meta-analyses on the combination of anticoagulant and P2Y12 inhibitors (with and without aspirin) in patients following PCI has been conflicting. This may occur because of the different included studies and definitions of safety outcomes. 10 To date, network meta-analyses (NMA), combining both direct and indirect evidence, were published by Lopes et al including 4 randomized controlled trials (RCT), recently updated with a fifth study, and by Chua et al with 6 RCT. These studies evaluated antithrombotic strategies for post-PCI in high-risk patients with AF, and concluded that the combination DOACs plus a P2Y12 inhibitor is a preferable alternative for these patients. 10 -12 However, both NMAs avoided comparing between the different DOACs. Thus, we aimed to synthetize the evidence on the individual effect of different antithrombotic therapies for patients with AF undergoing PCI through a systematic review with NMA, and to quantitatively evaluate the benefit–risk ratio of each regimen by means of stochastic multicriteria acceptability analysis (SMAA).
Methods
This systematic review was registered on PROSPERO (International Prospective Register of Systematic Reviews - https://www.crd.york.ac.uk/prospero/) as CRD42019146813. The study was performed in accordance with the Preferred Report Items for Systematic Reviews and Meta-Analysis guidelines (PRISMA-NMA) extension 13,14 and The Cochrane Collaboration recommendations. 15 Two researchers performed all steps independently. In case of disagreement, a third researcher was consulted.
Search Strategy and Selection Criteria
We conducted systematic searches in PubMed and Scopus, without time of language restrictions (updated November 2019). A trial registration database (clinicaltrials.gov) and the reference lists of reviews were manually searched. The complete search strategy is available in the Supplemental material.
Two authors independently read titles and abstracts. Relevant articles were later fully appraised, and those that met inclusion criteria were included for data extraction and analyses. Inclusion criteria were as follows: (i) studies including adult patients (>18 years) diagnosed with AF undergoing PCI/stenting; (ii) evaluating DOACs in combination with the P2Y12 inhibitor (any dose or regimen); (iii) comparing with other classic double and triple antithrombotic therapies; (iv) evaluating major or clinically relevant nonmajor bleeding International Society for Thrombosis and Haemostasis (ISTH), death from cardiovascular causes, major bleeding Thrombolysis in Myocardial Infarction (TIMI), major or minor bleeding (TIMI), myocardial infarction, stent thrombosis, and stroke; and (v) studies designed as a RCT. Articles written in non-Roman characters were excluded.
Data Extraction and Synthesis
The following data were independently extracted by 2 researchers: (i) study characteristics (author names, year of publication, sample size, patient age, and comorbidities); (ii) evaluated treatments (dosage, regimens, duration of treatment); (iii) methodological aspects of the study; and (iv) clinical outcome data (efficacy and safety). The methodological quality of the included articles was assessed using both the Jadad scale and the Risk of Bias tool from the Cochrane Collaboration tool. 15,16
Network meta-analysis, combining both direct and indirect evidence, were performed for each outcome of interest using a Bayesian framework based on the Markov Chain Monte Carlo simulation method and assuming a common heterogeneity parameter for all comparisons. Noninformative priors for the analysis were used. Transitivity analyses were performed by comparing population, interventions, control, and outcome definitions among studies. A consistency model was built for each outcome and relative effect size for each treatment is reported as the odds ratio (OR) with 95% credibility intervals. The goodness-of-fit of the model was assessed using residual deviance. No adjustment for multiple comparisons was performed. Convergence was attained based on visual inspection of Brooks-Gelman-Rubin plots and potential scale reduction factor (PSRF; 1 < PSRF ≤1.05). Ranking probabilities were calculated via surface under the cumulative ranking curve analysis (SUCRA) for each outcome in order to increase the estimate precision of the relative effect sizes of comparisons. The geometry of the networks followed the complexity level of the primary studies. To ensure that there was no divergence between direct and indirect comparisons, and to estimate the robustness of the network, we performed inconsistency and node-splitting analyses (
Multicriteria decision analysis (MCDA) is a decision-making tool that estimates the benefit–risk ratio of health care technologies. Benefit is described as the effect that takes the patient from the disease condition to health. Conversely, risk is an effect that leads the patient from health to disease. Multicriteria decision analysis involves the simultaneous evaluation of multiple therapeutic efficacy and safety attributes finally providing a “rank” of the technologies, ranging from the worst to the best clinical option. 24,25 Stochastic multicriteria acceptability analysis were used as an extension of the MCDA approach to estimate the benefit–risk of the antithrombotic therapies in the treatment of patients with AF or PCI. This tool provides a holistic and quantitative assessment of the relative profile benefit–risk of treatments using evidence from a network of clinical trials with unknown or partially known preferences. 26,27 Three benefit criteria (ie, efficacy as myocardial infarction, stent thrombosis, and stroke) and 3 risk criteria (ie, safety as major or clinically relevant nonmajor bleeding [ISTH], major bleeding [TIMI], and major or minor bleeding [TIMI]) were initially considered. A model containing all therapies was constructed with missing preferences (ie, without a previously established order of importance for the 6 outcomes) to provide a brief overview of the evidence. Different scenarios considering preferred order for outcomes to occur were also built as part of the sensitivity analyses. Therapy VKA + DAPT was considered the baseline of the model. The models were performed using Monte Carlo iterations with measurements derived from the consistency models of the trials included in the NMA.
Results
This systematic search retrieved a total of 151 records after removing duplicates, and 122 were excluded based on title and abstracts. After full-text reading, 4 studies were included in the systematic review. One record was added after manual search, resulting in a total of 5 RCTs, namely: WOEST, PIONEER AF-PCI, RE-DUAL PCI, AUGUSTUS, ENTRUST AF-PCI 28 -32 (see Supplemental material Figure 1). These trials were multicenter (performed in more than 40 countries), published between 2013 and 2019, and included a total of 11 532 patients. Patients were predominantly male (73.7%), with a mean age of 70.0 (standard deviation: 7.9) years. Most studies had a follow-up of 12 months, except for the AUGUSTUS trial, 30 where patients were followed for 6 months. Most studies used the risk stratification schemas of thromboembolism events CHA2DS2-VASc (n = 4) and HAS-BLED (n = 3), reporting values ranging from 3.0 to 5.0 and 2.0 to 3.0, respectively. 33 The main characteristics of the 5 included trials are presented in Table 1.
Characteristics of the Studies Included in the Systematic Review.

Abbreviations: AF, atrial fibrillation; BM, bare metal stent; DAPT, dual antiplatelet therapy; DE, drug eluting stent; INR, International Normalized Ratio; NCT, ClinicalTrials.gov identifier; OL, open label; PCI, percutaneous coronary intervention; RCT, randomized controlled trial; TD, twice daily; VKA, vitamin K antagonists.
a In VKA treatments, the recommended dose was calculated from the INR, where the goal was to maintain the target INR (2.0-3.0). CHA2DS2-VASc scores reflect the risk of stroke in patients with atrial fibrillation. Values ranging from 0 to 9 and with higher scores indicating greater risk (>2 for men or >3 for women indicate the need of anticoagulant therapy). HAS-BLED scores reflect the risk of major bleeding. Values ranging from 0 to 9 and with higher scores indicating greater risk (score of 2 indicates 1.88 bleeding events/100 patients/year; score of 3 indicates 3.74 bleeding events/100 patients/year).
All studies evaluated the efficacy and safety of OACs associated with platelet APT, either dual platelet antiplatelet therapy—DAPT (P2Y12 inhibitor + aspirin) or mono platelet antiplatelet therapy (P2Y12 inhibitor). In 4 studies, researchers were allowed to choose the P2Y12 inhibitor. In the WOEST study, this therapy was limited to clopidogrel. Overall, clopidogrel (86%), ticagrelor (6.68%), and prasugrel (0.66%) were the most commonly evaluated drugs. New oral anticoagulants were represented by apixaban, rivaroxaban, dabigatran, and edoxaban (one study each) administered in different doses and dosage regimes. The VKA group (standard comparator) was warfarin used in fixed doses (International Normalized Ratio: 2-3).
Using the Cochrane risk of bias tool, 15 all studies were classified as having a high risk of bias for the domain of “blinding of participants and personnel assessment” because researchers were aware of the therapies used by patients, allowing them to choose the P2Y12 inhibitor (except in WOEST, which used clopidogrel as default). Also, all studies were classified as high risk of bias for the “blinding of outcome assessment” domain because they had no blinding end point evaluators. Most trials were funded by the pharmaceutical industry and presented conflict of interest, being judged as with high risk of bias for “other bias” domain. The WOEST and ENTRUST studies described the “allocation concealment” process as performed with sealed envelopes and through a computerized allocation sequence generated by an external programmer, respectively. The RE-DUAL and PIONEER trials were classified as unclear risk for this domain because the distribution of their samples was not reported, and the AUGUSTUS study was classified as with high risk because patients were stratified according to their weight. Finally, for the “incomplete outcome data” domain, the WOEST and ENTRUST studies were judged as having an unclear risk of bias because there was a slight variation in losses between groups, yet no data on the reasons for treatment discontinuation were available (see Supplemental material Figure 4). According to the Jadad scale, the scores obtained were 2, 3, 3, 4, and 4 for the PIONEER, RE-DUAL, AUGUSTUS, ENTRUST, and WOEST studies, respectively.
Networks were built for each of the 7 outcomes of interest: major or clinically relevant nonmajor bleeding (ISTH), major bleeding (TIMI), major or minor bleeding (TIMI), death from cardiovascular causes, myocardial infarction, stent thrombosis, including all the 5 studies whenever possible (Figure 1).
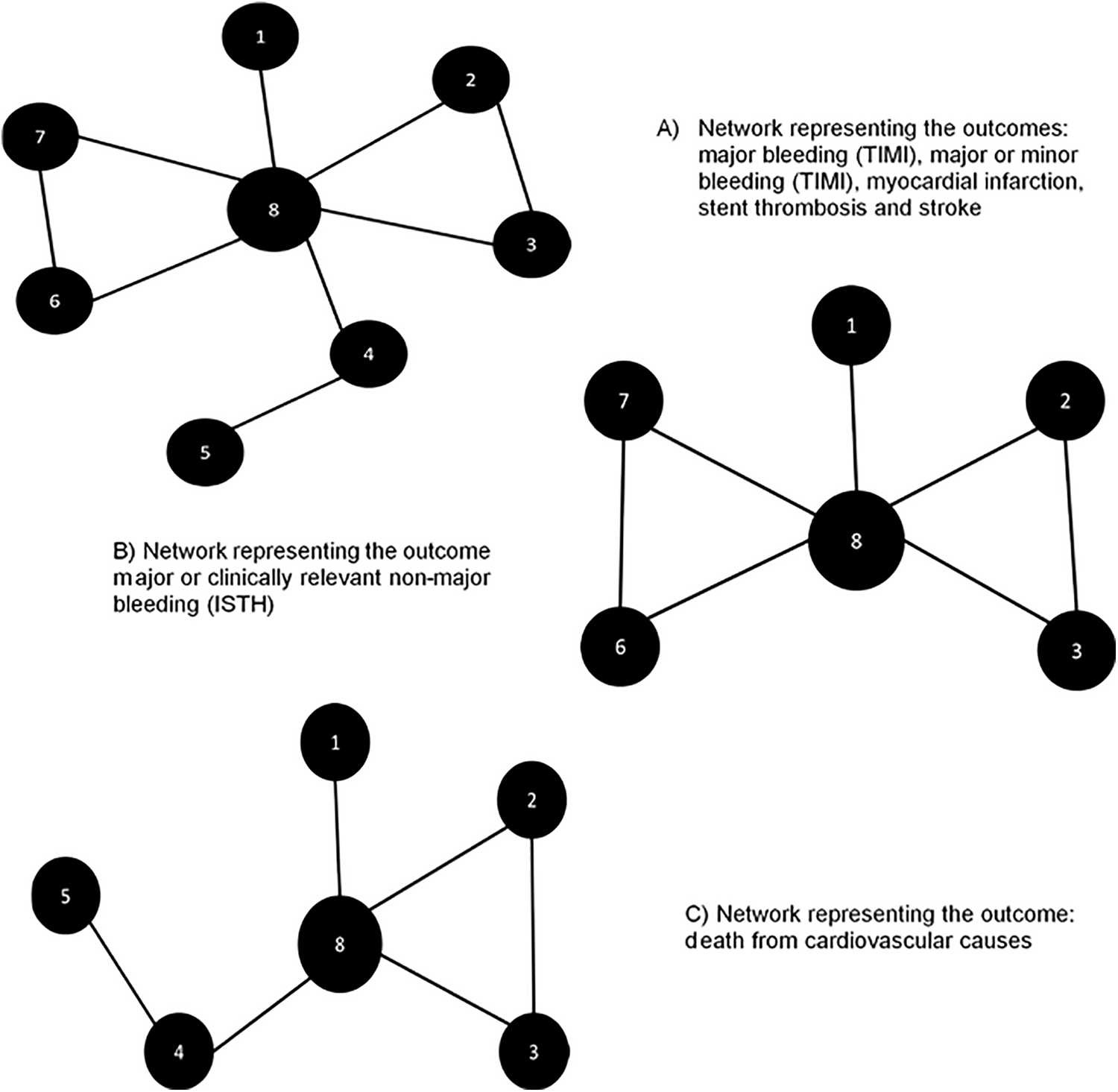
Networks of treatment comparisons for each outcome of interest.1 = Edoxab + P2Y12; 2 = Riva 2.5 mg + DAPT; 3 = Riva 15 mg + P2Y12; 4 = VKA + P2Y12; 5 = Apixab + P2Y12; 6 = Dabig 150 mg + P2Y12; 7 = Dabig 110 mg + P2Y12; 8 = VKA + DAPT. DAPT indicates dual antiplatelet therapy.
Consistency models were performed for all evaluated outcomes. No significant differences among the different antithrombotic therapies were obtained in any scenario (see Supplemental material Figure 5). However, when grouping all the DOACs regimens in one single node, results demonstrated significant difference for the major bleeding (TIMI) scenario, where the ORs were 0.52 (95% CI: 0.32-0.86) for VKA + DAPT compared to DOAC + P2Y12, and 0.63 (95% CI: 0.30-1.21) for VKA + DAPT compared to VKA + P2Y12. The comparison DOAC + P2Y12 versus VKA + P2Y12 had an OR of 1.20 (95% CI: 0.59-2.33) for this outcome (see the Supplemental material).
The SUCRA analyses of the networks evaluating individual regimens demonstrated VKA + DAPT with higher probabilities of being the less safe therapy for the outcomes major or clinically relevant nonmajor (ISTH; 80% chances) and major and minor bleeding (TIMI; 83% and 86%, respectively). Dabigatran 110 mg + P2Y12 was the safest option for the outcomes of major or clinically relevant nonmajor bleeding (TIMI; 16%). For the outcomes of death from cardiovascular causes, rivaroxaban 15 mg + P2Y12 had a 74% probability of being the worst option, while VKA + P2Y12 was considered a more effective alternative for preventing this outcome (18%). For myocardial infarction, stent thrombosis, and stroke, apixaban + P2Y12 was considered the safest option, with less chance of causing these outcomes (22%, 13%, and 6%, respectively; Figure 2).

Surface under the cumulative ranking curve analysis (SUCRA) is the inverse ratio of the cumulative frequency of probability ranking, where the larger the area below the curve, the greater the probability of the evaluated therapy generating a certain outcome. Apixab + P2Y12 = apixaban + inhibitor P2Y12; Riva 2.5 mg + DAPT = rivaroxaban 2.5 mg + aspirin + inhibitor P2Y12; VKA + DAPT = vitamin K antagonist + aspirin + inhibitor P2Y12; Dabig 110 mg + P2Y12 = dabigatran 110 mg + inhibitor P2Y12; Dabig 150 mg + P2Y12 = dabigatran 150 mg + inhibitor P2Y12; Edoxaban + P2Y12 = Edoxaban + inhibitor P2Y12; Riva 15 mg + P2Y12 = Rivaroxaban 15 mg + inhibitor P2Y12; VKA + P2Y12 = vitamin K antagonist + inhibitor P2Y12. DAPT indicates dual antiplatelet therapy.
The rank acceptability of the SMAA considering both efficacy and safety outcomes with missing preferences is depicted in Figure 3. The missing preferences scenario favored the use of apixaban + P2Y12 (61%), while edoxaban + P2Y12 (28%) and VKA + DAPT (26%) were considered the worst options. The results of SMAA in different scenarios are available in the Supplemental material (see Supplemental material Figures 6 and 7). When considering major bleeding as the most important outcome, followed by (in order) major or minor bleeding, myocardial infarction, stent thrombosis, and stroke, apixaban + P2Y12 continued to be the best alternative (50.5% chance) while VKA + P2Y12 and edoxaban + P2Y12 remained the worst options (35% and 30%, respectively).
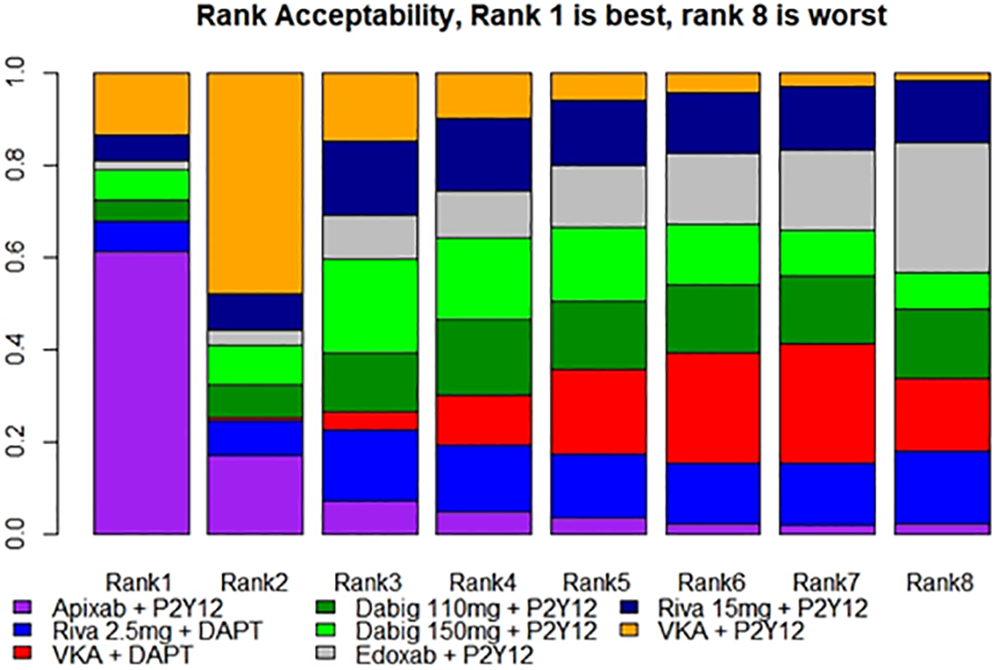
Rank acceptabilities from the stochastic multicriteria acceptability analysis. Each drug has a probability of being the best treatment (rank 1) or the worst treatment (rank 8) considering overall its benefits and risks.
Discussion
We were able to update and synthetize evidence on the efficacy and safety of 8 antithrombotic therapies through a systematic review with NMA including 5 RCTs. Additionally, we performed a MCDA via SMAA to weigh the risks and benefits of treatment using the evaluated regimens.
The use of anticoagulant and APT during the management of cardiovascular conditions has been associated with improvements in short and long-term clinical outcomes. However, when considering anticoagulant therapies for patients with AF, one must balance the reduction in risk of thromboembolism that these therapies offer against the risk of bleeding and stroke they may pose. 34 Unfortunately, no consensus on the best therapeutic approach exists. Clinical guidelines suggest the use of whether DAPT with clopidogrel and aspirin, or dual therapy combining an antiplatelet agent and an OAC, or even triple therapy (oral anticoagulation combining DAPT). 1,35
After 2013, the use of antiplatelet monotherapy (eg, aspirin or clopidogrel) was rarely recommended, grounded on the findings of the WOEST study that the first RCT to evaluate the safety of OAC plus P2Y12 inhibitor therapy (with and without aspirin) in post-PCI patients. The authors demonstrated that aspirin was responsible for increased bleeding and death rates. 31 Similarly, triple therapy in patients with AF is also associated with a more than 3-fold higher risk of nonfatal and fatal bleeding, compared to warfarin monotherapy. Besides, the frequency of anticoagulant interruption with triple therapy is high, with an estimated 250 000 patients undergoing temporary discontinuation every year in North America. 36,37 A recent study summarized the evidence of meta-analyses comparing the safety and efficacy of dual and triple antithrombotic therapies in high-risk patients. Although some differences among studies exist especially given the different included trials and selected outcomes, overall evidence demonstrates that dual therapy with an DOAC and P2Y12 inhibitor is superior to triple therapy. 10
The gradual introduction over the past decade of the DOACs to be used in different cardiovascular conditions aimed for a more convenient and fixed-dose regimen compared to previous therapies. New oral anticoagulants have many theoretical advantages over warfarin, including comparable or superior efficacy in trial populations (eg, in AF, reduced risk of stroke, thromboembolism, an all-cause mortality) and reduced monitoring requirements. 38 -42
Five main RCTs comparing the use of different regimens of DOAC plus P2Y12 inhibitor in patients with AF undergoing PCI have been performed, and were included in both NMAs published by Lopes et al and Chua et al. 10 -12 Additionally, Chua et al included the ISAR-TRILPE trial (2015) 43 which evaluates whether shortening the duration of clopidogrel therapy (from 6 months to 6 weeks) after stent implantation was associated with a superior clinical outcome in patients receiving concomitant aspirin and OAC. The results of this trial demonstrated no significant differences on the duration of triple therapy with respect to net clinical outcomes. Given the distinct design of the ISAR-TRILPE that do not meet the eligibility criteria of this review, this study was not included in our analyses.
The NMA methodological approach used by both Lopes et al and Chua et al considered that the interventions had a similar clinical profile, allowing them to be grouped into one single node (ie, DOACs). As a conclusion, the combination DOACs associated with a P2Y12 inhibitor was related with less hemorrhagic complications compared to VKA plus DAPT (OR: 0.49 [95% CI: 0.30-0.82] or hazard ratio 0.53 [95% CI: 0.35-0.78], respectively). This conclusion is based on the observed 46% reduction in major or minor TIMI bleeding with similar major adverse cardiovascular events. In our sensitivity analyses, when gathering the regimens in one group in the network, we obtained similar results (OR: 0.52 [95% CI: 0.32-0.86]). No differences among treatments were found for efficacy outcomes (eg, stroke, death). However, when individualized analyses were performed, SUCRA and SMAA demonstrated trends among drugs regimens that may contribute toward a more accurate decision-making process.
Network meta-analyses facilitates the comparison of all relevant drugs even when they were not directly compared with each other in clinical trials. The combination of this method with a decision analytic approach, such as MCDA (via SMAA), greatly improves the interpretability of existing evidence by making more explicit the difficult trade-offs between outcomes. Stochastic multicriteria acceptability analysis applies qualitative or quantitative preferences on different outcomes, allowing for a transparent judgment on their relative importance, even with incomplete or uncertain primary information. Another advantage of this technique is that the analysis can be rapidly updated when new trials are released or when preferences change, as demonstrated in our sensitivity analyses. Additionally, MCDA models can consider patients’ values and choices alongside the knowledge and expertise of prescribers, allowing for better shared decision-making in clinical practice. 26,44
In most of the evaluated outcomes, apixaban + P2Y12 showed a higher probability of being the best alternative. This was evident in the SMAA where this association presented the higher risk–benefit ratio. Our findings are consistent with those reported in the literature comparing the efficacy of apixaban versus warfarin in patients with AF. 41,45 In this study, apixaban led to 21% reduction in risk of stroke or systemic embolism, decreased in 31% the chances of major bleeding, and caused 11% less deaths compared to VKA. 45 The AUGUSTUS trial and a tradeoff analysis of risk (bleeding) and benefit (ischemic events) over time also demonstrated that apixaban + P2Y12 therapy is preferred over VKA. Use of aspirin, acutely and for up to 30 days, results in an equal tradeoff between an increase in severe bleeding and reduction in severe ischemic events. After 30 days, aspirin continued to increase bleeding without significantly reducing ischemic events. 30,46
In real-world settings, apixaban also demonstrated significantly lower risk of stroke/systemic embolism, major bleeding, and hospitalizations compared to rivaroxaban and warfarin, and a trend toward better outcomes compared to dabigatran in patients with nonvalvular AF, including elderly. 47 -50 The use of apixaban instead of VKA for stroke prevention was also associated to considerable reduction in cardiovascular events. 51 A cost-effectiveness analysis based on real-world data from a NMA (Dutch health care system) also showed apixaban as cost-effective over other anticoagulants including VKA and DOACS. 52
In our analyses, we found dabigatran 110 mg + P2Y12 also as a promising regimen after apixaban for the outcomes of stroke, myocardial infarction, and stent thrombosis. These results may be explained by dabigatran’s prolonged half-life. A twice a day regimen provides less variation in the anticoagulant activation, and the selective mechanism of thrombin inhibition may reduce the risk of bleeding. 53
Edoxaban + P2Y12 and VKA + DAPT were considered the worst options. In the ENTRUST trial, the association edoxaban + P2Y12 led to more cardiovascular deaths, stent thrombosis, and myocardial infarction, although the results were not statistically significant compared to VKA. For the main efficacy outcome (composite of cardiovascular death, stroke, systemic embolic event, myocardial infarction, or definitive stent thrombosis), the hazard ratio was 1.06 (95% CI: 0.71-1.69). 32 Other previous direct comparisons also demonstrated that edoxaban has a similar profile to warfarin with respect to preventing stroke or systemic embolism in patients with AF, but less risk for major bleeding for bleeding and hemorrhagic stroke. 54 Nevertheless, caution during the prescription of edoxaban is needed, especially considering patients at high risk of adverse events.
Although we were able to demonstrate slight clinical differences among the assessed antithrombotic regimens, direct comparisons among DOAC are limited. Further well-designed RCT directly comparing these therapies, especially apixaban and dabigatran, are needed to strengthen the evidence and support recommendations for their use in clinical practice. Additionally, real-world studies, cost-effectiveness, and convenience analyses should be performed to improve AF clinical guidelines. Cost-minimization analyses are the most recommended evaluation to establish the least costly alternative among interventions that are assumed to produce equivalent health outcomes. These are important given the high cost of the DOACs, which may hinder their use on a routine basis. While the conventional therapy with warfarin has an average cost per patient per month of US$ 21.87, rivaroxaban 15 mg costs around US$337.68, and regimens with apixaban costs US$90.45 per patient per month (https://www.va.gov/).
Our study has limitations. Although NMA is a robust method for evidence synthesis when direct comparisons in the literature are absent, our study was limited to 5 RCTs. These trials are not completely homogeneous (eg, some variations in population characteristics and trial follow-up), requiring attention to data generalization. Nevertheless, our networks were found to be robust and sensitivity analyses showed no significant differences from the original results. No further analyses on patient’s risk stratification schemas of thromboembolism events were possible given the absence of standard reporting of results. Treatment rankings should not be interpreted separated from the relative treatment effect measures obtained by the NMA. We applied an SMAA decision model based on additive value functions. Although this model is widely applied and reasonably easy to understand, we acknowledge that other approaches are possible. 26,27 Stochastic multicriteria acceptability analysis requires that decision-makers preferences about the criteria are determine unambiguously, which is not always possible. We opted to perform different SMAA scenarios to avoid inconsistencies or selective bias, and to potentially increase the informative value of existing evidence for prescribing decisions. Nevertheless, to conduct MCDA, a panel of experts is recommended for the selection of evaluated alternatives and decision on the importance of each criterion. Finally, our model is based only on criteria that were measured in clinical trials, which is appropriate in the context of health decision-making. However, further criteria such as costs in reimbursement decisions could be considered.
Conclusion
Apixaban + P2Y12 showed a slightly better clinical profile for the treatment of patients with AF undergoing PCI. Edoxaban + P2Y12 is probably the less safe alternative. Cost-minimization analyses should be performed to strengthen the evidence. Meanwhile, the therapeutic choice of antithrombotic regimen should consider, among others, the risks of thromboembolism, bleeding and stroke, patient preferences (eg, treatment convenience, adherence), access, and treatment costs.
Supplemental Material
Supplemental Material, Supplementary_material - Safety and Efficacy of Oral Anticoagulants Therapies in Patients With Atrial Fibrillation Undergoing Percutaneous Coronary Intervention: A Network Meta-Analysis
Supplemental Material, Supplementary_material for Safety and Efficacy of Oral Anticoagulants Therapies in Patients With Atrial Fibrillation Undergoing Percutaneous Coronary Intervention: A Network Meta-Analysis by Felipe F. Mainka, Vinicius L. Ferreira, Antonio M. Mendes, Gustavo L. Marques, Fernando Fernandez-Llimos, Fernanda S. Tonin and Roberto Pontarolo in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Author Contributions
FFM, FFL, FST and RP designed the study and wrote the protocol. FFM, VLF, AMM and GLM screened and abstracted publications. FFM and VLF evaluated methodological quality. FFM FST and FFL statistically analysed data. FFM and FST wrote the manuscript, with editorial contributions from VLF, AMM, GLM, FLL and RP. All authors reviewed the manuscript for accuracy and scientific content.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001. The funding sources had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
