Abstract
Background:
Statins have been demonstrated to prevent the development of contrast-induced nephropathy (CIN). Nevertheless, clinical research has indicated conflicting results. We performed a meta-analysis of randomized controlled trials (RCTs) to evaluate the protective effects of statins on CIN and the requirement of renal replacement therapy (RRT) in patients undergoing coronary angiography/percutaneous interventions.
Methods:
PubMed, MEDLINE, Web of Science, EMBASE, ClinicalTrials.gov, and the Cochrane Central RCTs were searched for RCTs from inception to February 2014 to compare statins with placebo treatment for preventing CIN in patients undergoing coronary angiography/percutaneous interventions.
Results:
Nine RCTs were identified and analyzed in a total of 5143 patients involving 2560 patients with statin pretreatment and 2583 patients as control. Patients who received statin therapy had a 53% lower risk of CIN with different definitions (within 48 or 72 hours) compared to the control group based on a fixed effect model (risk ratio = 0.47, 95% confidence interval = 0.37-0.60,
Conclusion:
This updated meta-analysis demonstrated that preprocedural statin treatment could reduce the risk of CIN and the need for RRT in patients undergoing coronary angiography/percutaneous interventions. Moreover, statin therapy would be helpful in reducing the incidence of CIN in high-risk patients with preexisting renal dysfunction or diabetes mellitus. Additionally, rosuvastatin and atorvastatin had similar efficacies in preventing CIN development.
Keywords
Introduction
Contrast-induced nephropathy (CIN) is increasingly common and significantly contributes to excess health care costs, long-term morbidity, and mortality in patients undergoing coronary angiography/percutaneous interventions. Several significant risk factors (eg, chronic kidney disease [CKD] and diabetes mellitus [DM]) have been demonstrated to be strongly associated with the development of CIN. 1,2 The risk of CIN is dramatically higher in patients with CKD, with a reported incidence as high as 20%. 3 Other than prophylactic intravenous hydration, use of low- or iso-osmolar contrast media (CM), and reduced CM dose, few strategies have been approved or shown to be effective for preventing CIN. 4,5 Therefore, it is urgent to develop new strategies to decrease CIN occurrence, particularly in high-risk patients with CKD or DM.
Previous studies have suggested that statins exhibit pleiotropic effects, including improving endothelial function, increasing nitric oxide production, and having a rapid onset of antioxidant efficacy after the initiation of treatment, 6,7 which are considered as the most important contributing factors to CIN progression, although the pathophysiological mechanisms of CIN are still unclear. 8 -10 However, randomized clinical trials (RCTs) and meta-analyses assessing the effect of statins on reducing CIN risk have published conflicting results. 11 -16 There is a need for evidence to clarify this issue. Furthermore, a number of well-designed and large-scale RCTs have been published since those meta-analyses were conducted. 17 -20 Consequently, we performed a meta-analysis of RCTs to evaluate the effects of statins on CIN prevention and requirement of renal replacement therapy (RRT) compared with placebo, especially in high-risk patients with CKD or DM undergoing coronary angiography/percutaneous interventions.
Methods
Literature Search Strategy
We performed a literature search in PubMed, MEDLINE, Web of Science, EMBASE, ClinicalTrials.gov, and the Cochrane Central Register of Controlled Trials databases from the date of inception until February, 2014. The following search formula was used: (statins OR atorvastatin OR rosuvastatin OR simvastatin OR pravastatin OR lovastatin) AND (contrast OR radiocontrast OR contrast media OR contrast medium OR radio-contrast media OR radiocontrast medium OR contrast agent) AND (nephropathy OR kidney injury OR renal injury OR renal failure) AND (coronary angiography or percutaneous interventions). We also searched published abstracts presented at the meetings of the American Heart Association, the American College of Cardiology, and the European Society of Congress from 1998 to 2014. We also searched reference lists in relevant publications and abstracts. Language was restricted in English or Chinese.
Study Selection
Two independent reviewers (YHL and YL) scanned the titles and abstracts of all identified articles. Only studies that were clearly irrelevant were excluded at this stage, and the same 2 reviewers independently assessed article eligibility. A third reviewer (CYD) resolved any disagreements between the other 2 reviewers with discussion. Studies were included in our review if they met the following criteria: (1) RCTs investigating the efficacy of statins in preventing CIN; (2) compared with placebo, the treatment groups received statins before the contrast exposure at any dose, for any length of time; and (3) studies that provided oral or intravenous N-acetylcysteine (NAC) preparations were only included if both arms received NAC. Trials directly comparing 2 different doses of statins were excluded as were nonrandomized trials and duplicate publications.
The primary outcome was CIN, defined as an absolute increase in serum creatinine (SCr) ≥0.5 mg/dL or an increase ≥25% from baseline to within 2 to 5 days after CM exposure. 4 The secondary outcomes were requirement for RRT and all-cause death.
Data Extraction and Assessment of Risk of Bias
Data extraction was performed by 2 independent reviewers. One reviewer (YHL) extracted relevant data from the included studies, which was then checked by a second reviewer (YL). The extracted data included patient characteristics (number of patients, mean age, proportion with DM, and baseline SCr), the incidence of patients with renal dysfunction (defined as estimate glomerular filtration [eGFR] ≤60 mL/min/1.73 m 2 or creatinine clearance [CrCl] ≤60 mL/min), inclusion and exclusion criteria, hydration protocol, CM type and dose, statins type and dose, specific definition of CIN, and clinical outcomes.
Two reviewers (YHL and YL) independently evaluated the methodological quality of the included studies. Disagreements were resolved by discussion and adjudicated by a third reviewer (CYD). We employed the Jadad scoring system to assess study quality, 21 and quality assessment was judged on concealment of treatment allocation, similarity of study groups at baseline, eligibility criteria, use of a placebo, use of any blinding procedure, reporting of losses to follow-up, and intention-to-treat analysis. 22
Statistical Analysis
Data were analyzed using Review Manager Version 5.2. (The Nordic Cochrane Centre, The Cochrane Collaboration, 2012) and STATA version 11 (Stata Corp, College Station, Texas). The effect of statins on CIN development was shown as risk ratio (RR) with 95% confidence interval (CI). The Q statistic was calculated, and heterogeneity was quantified using the I
2
statistic. We regarded I
2
≤ 25%, 25% to 50%, and >50% as low, moderate, and high heterogeneity, respectively.
23
Random effect models were performed when I
2
> 50%, otherwise, a fixed effect model was employed. A funnel plot was conducted to assess publication bias. We also performed several subgroup analyses to identify potential differences in treatment across the trials. Subgroup analysis was conducted based on different definition of CIN (within 48 or 72 hours), patients with renal dysfunction, statins plus NAC or NAC only, and different types of statins (atorvastatin or rosuvastatin). All tests were 2 tailed and
Results
Characteristics of the Included Studies
The flowchart of search strategy is provided in Figure 1. Our initial search identified 347 potentially relevant articles. After adjusting for duplication, 195 articles remained. Of these, 163 articles were discarded after independently reviewing the abstracts and titles. The most common reasons for exclusion was that the articles were review articles, basic research, or studies that assessed the effect of statins on acute kidney injury after cardiac surgery. Ultimately, 32 relevant studies were assessed in full text. A total of 23 studies were further excluded after reviewing the full text. In all, 3 studies were excluded for comparing 2 different doses of statins to each other; 13 articles for retrospective research and 2 articles for protective observational research were excluded; 2 studies that evaluated the effect of different types of statins (rosuvastatin vs atorvastatin and simvastatin vs pravastatin) on CIN were also excluded; 2 studies which were conducted to compare the effect of statins plus alprostadil or probucol with statins only were excluded; and 1 study published in Chinese was excluded because it did not report the incidence of CIN. 24 Although 1 study 25 did not report the incidence of CIN, we requested it by directly contacting the author. Consequently, 9 RCTs involving 5143 patients undergoing coronary angiography/percutaneous interventions met the inclusion criteria and were included in the meta-analysis, and none of them were sponsored by industry.

Flow diagram of study selection.
Study Characteristics and Interventions
All included studies had SCr levels measured between 24 hours and 5 days and reported the incidence of CIN (except for 25 ) in 5143 patients undergoing coronary angiography/percutaneous interventions. Statins were given to 2560 patients and 2583 patients served as controls. The mean patient ages ranged from 54 ± 10 to 76 ± 7 years. The mean baseline SCr level ranged from 0.84 to 1.32 mg/dL. Patients with DM were included in 9 studies, with prevalence rates varying from 15% to 44% in statin groups and 22% to 38.5% in control groups. And study by Han et al 20 only assessed patients with DM. Of 9 trials, 3 evaluated the effect of statins on CIN in patients with eGFR ≤60 mL/min/1.73m 2 or CrCl ≤ 60 mL/min, and 2 trials evaluated patients with eGFR ≥ 60 mL/min/1.73m 2 . One trial included patients with 30 ≤ eGFR ≤ 90 mL/min/1.73m 2 . Two trials did not report renal function levels but excluded patients with SCr > 3 mg/dL. One trial did not record precise renal function data and only demonstrated patients without renal dysfunction or dialytic treatment. 17
The criteria used to define CIN varied among the individual studies. Four trials used the definition of an absolute increase in SCr ≥ 0.5 mg/dL or an increase ≥25% from baseline to within 48 hours after CM exposure, and 3 trials employed the same within 72 hours, and 1 trial was within 5 days. One trial defined CIN as an increase in serum cystatin C (sCyC) concentration 10% above the baseline value at 24 hours after CM exposure 18 and another did not report CIN definition. 25
Percutaneous coronary intervention (PCI) was performed in almost all patients in studies by Li et al 17 (100%) or Patti et al 26 (98%-99%). However, studies by Acikel et al 25 and Ozhan et al 27 included all patients who underwent coronary artery angiography but not PCI. In another 5 studies, the incidence of patients treated with PCI in the statins group ranged from 30.6% to 67.9% 18,19,20,28,29 .
In addition to the study by Quintavalle et al, 18 which hydrated with sodium bicarbonate at a rate of 3 mL/kg/h for 1 hour before CM injection and 1 mL/kg/h during and after the procedure for 6 hours, the hydration protocol in other studies was isotonic saline at a rate of 1 mL/kg/h before and after the procedure. The duration of hydration ranged from 4 to 12 hours before and 6 to 24 hours after CM exposure. All patients in the statin and control groups received low- or iso-osmolar nonionic CM, but the dose varied widely. The highest and lowest average doses of CM reported were 213 ± 13 and 93 ± 6mL, respectively (Table 1).
Baseline Characteristic of Patients and Interventions of Included Studies.
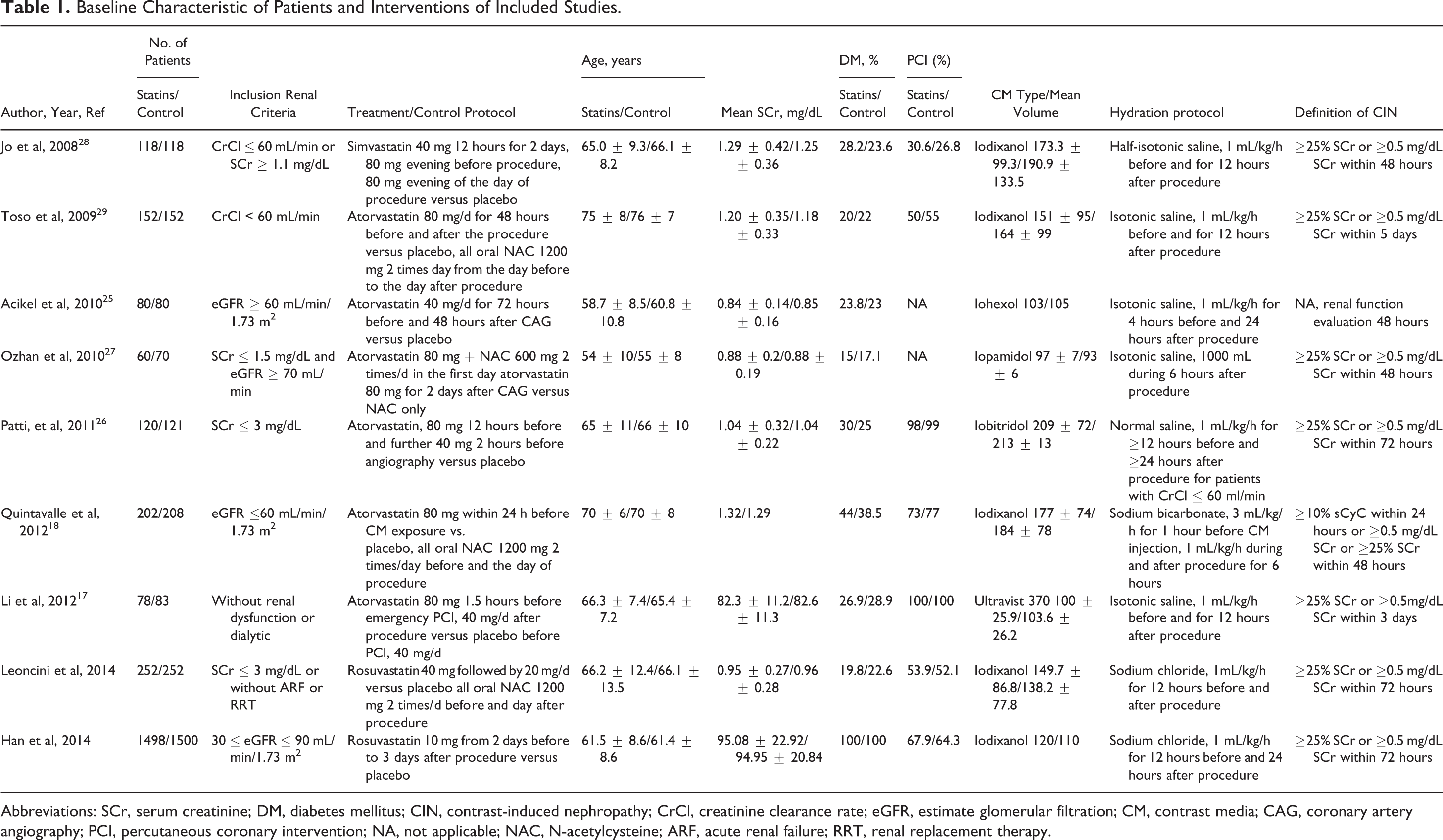
Abbreviations: SCr, serum creatinine; DM, diabetes mellitus; CIN, contrast-induced nephropathy; CrCl, creatinine clearance rate; eGFR, estimate glomerular filtration; CM, contrast media; CAG, coronary artery angiography; PCI, percutaneous coronary intervention; NA, not applicable; NAC, N-acetylcysteine; ARF, acute renal failure; RRT, renal replacement therapy.
Assessment of Study Quality and Publication Bias
The quality of the included trials was moderate (Table 2). Only 5 of 9 studies provided the concealment of allocation. Almost all of the studies included patients with similar baseline characteristics and provided details about the eligibility criteria and completeness of follow-up. Of the 9 studies, 7 described the appropriate methods of randomization and 2 studies did not provide details to evaluate the appropriateness of randomization. Of the 9 studies, 5 reported blinding of both patients and providers to treatment assignment. The intention-to-treat analysis was only provided in 4 research studies.
The funnel plot was relatively symmetrical (Figure 2), and the result of Egger test confirmed the absence of obvious publication bias among the included trials for primary or secondary outcomes (CIN or RRT;

Funnel plot with 95% confidence interval (CI) for subjective assessment of bias among the included studies.
Quality of Included RCTs.

Abbreviations: RCTs, randomized controlled trials; ITT, intention to treat; NS, not significant.
Outcomes
Patients who received statins had a 53% lower risk of CIN compared with the control group based on a fixed effect model (RR = 0.47, 95% CI = 0.37-0.60,
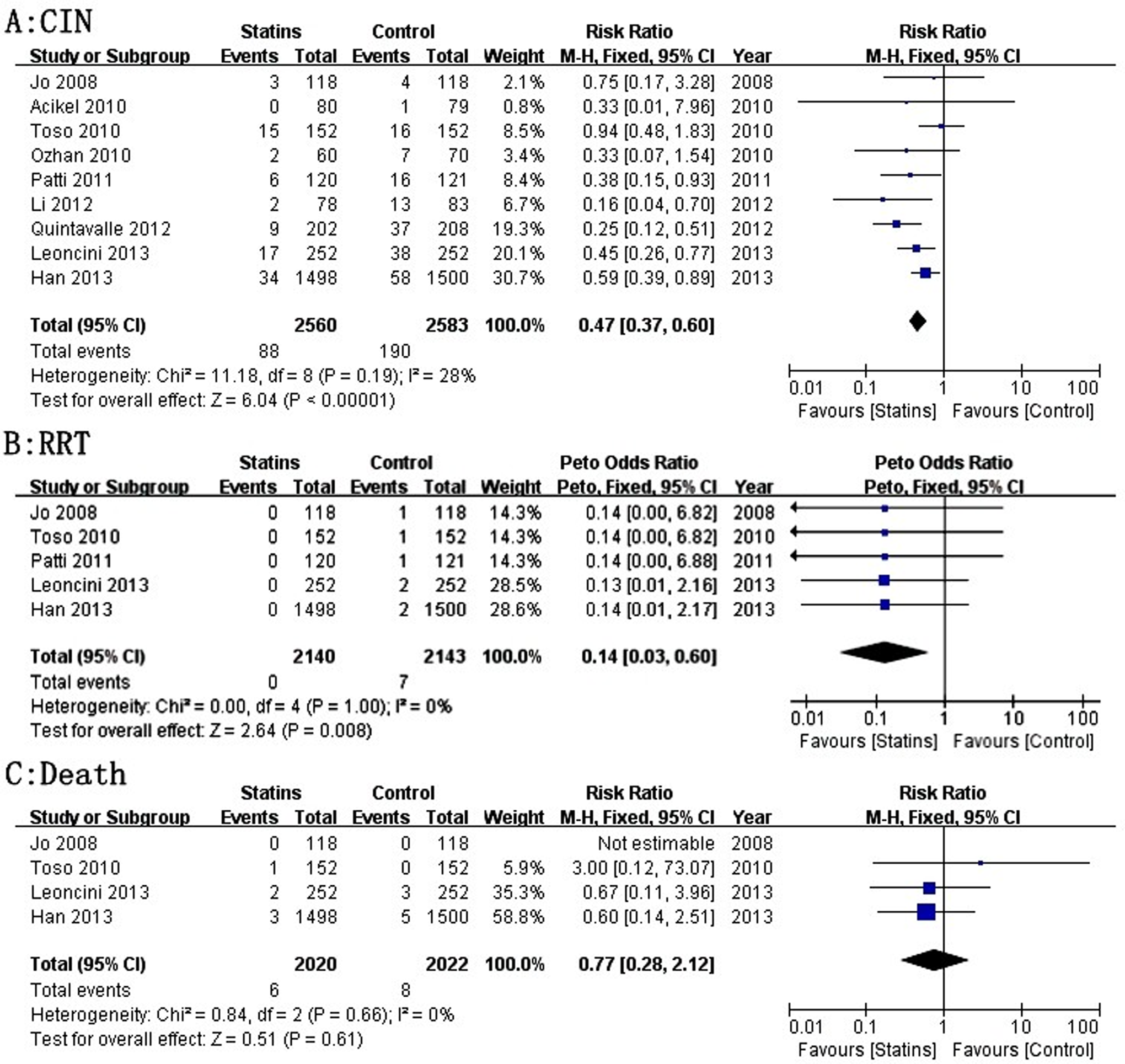
Forest plot of RR and 95% CI for CIN (A) and the requirement of RRT (B), and all-cause death (C) among patients assigned to statins versus placebo therapy. RR, risk ratio; CI, confidence interval; CIN, contrast-induced nephropathy; RRT, renal replacement therapy.
Whether RRT was required was described in 5 studies. All of which reported that no patient in the statin group required RRT. The overall results based on Peto fixed effect analysis showed that the requirement of RRT was significantly lower in patients with statin treatment than that in the control groups (I
2
= 0%,
The incidences of all-cause death were only reported for 4 trials. One trial reported that no patients died, and the other demonstrated that 14 patients died (6 and 8 patients in statins and placebo groups, respectively). Nevertheless, there was no significant difference between the statins and the placebo treatment groups (I
2
= 0%, RR = 0.77, 95% CI = 0.28-2.12,
Sensitivity Analysis
We performed a sensitivity analysis by removing low-quality studies (Jadad score ≤ 3). However, the I
2
, RR, 95% CI, and
Subgroup analysis
We evaluated the effect of statin treatment on different definitions of CIN, which is defined as an absolute increase in SCr ≥ 0.5 mg/dL (or an increase ≥25%) from baseline to within 48 hours (3 trials) or 72 hours (4 trials) after CM exposure. The risk of CIN was significantly decreased regardless of the definitions of CIN (Figure 4).

Subgroup analysis of the forest plot of RR and 95% CI for CIN incidence among patients assigned to statins versus placebo therapy according to different definitions of CIN. RR, risk ratio; CI, confidence interval; CIN, contrast-induced nephropathy; RRT, renal replacement therapy.
Among the 9 RCTs, 4 trials compared the efficacy of statins plus NAC with NAC only. We found that the risk of CIN was significantly lower in patients who received statins plus NAC than in patients who only received NAC using the randomized effect. A reduced risk of CIN was also demonstrated in patients treated with statins only compared to control without NAC (Figure 5).

Subgroup analysis of forest plot of RR and 95% CI for CIN among patients assigned to statins plus NAC versus NAC therapy only. RR, risk ratio; CI, confidence interval; CIN, contrast-induced nephropathy; NAC, N-acetylcysteine.
Next, we classified studies according to whether they included patients with renal dysfunction. The result indicated that either patients with or without renal dysfunction had a significant less risk of CIN, although studies that included patients with renal dysfunction showed moderate heterogeneity, but it was not significant. (χ2 = 8.86, I
2
= 44%,

Subgroup analysis of forest plot of RR and 95% CI for CIN among patients assigned to statins versus placebo therapy according to the presence of renal dysfunction. RR, risk ratio; CI, confidence interval; CIN, contrast-induced nephropathy.
In addition, we showed that use of preprocedural statins in patient with DM had 40% less risk of developing CIN than control group (Figure 7).

Subgroup analysis of forest plot of RR and 95% CI for CIN among patients assigned to statins versus placebo therapy in patients with DM. RR, risk ratio; CI, confidence interval; CIN, contrast-induced nephropathy; DM, diabetes mellitus.
Subgroup analysis according to statin type indicated that compared with placebo, rosuvastatin treatment showed the same effect as atorvastatin treatment on reducing CIN incidence (Figure 8).

Subgroup analysis of forest plot of RR and 95% CI for CIN among patients assigned to statins versus placebo therapy according to statins types (atorvastatin versus rosuvastatin). RR, risk ratio; CI, confidence interval; CIN, contrast-induced nephropathy.
Discussion
Our results suggested that statin treatment was strongly related to a significantly lower incidence of CIN with different definitions (within 48 or 72 hours) and lesser need for RRT in patients undergoing coronary angiography/percutaneous interventions, but it did not decrease the significant occurrence of all-cause death. Furthermore, the reduced risk of CIN due to statin therapy was also observed for patients with preexisting renal dysfunction or DM who are at high risk of CIN. In addition, we might be the first to report that rosuvastatin and atorvastatin similarly reduced the risk of CIN.
Although the pathogenesis of CIN remains ill defined, multiple mechanisms may be involved. Reactive oxygen species (ROS), mainly generated by renal medulla hypoxia during CM administration, have been implicated as an important contributing factor. 10 These ROS have important effects on renal microcirculation, and they induce increases in angiotensin II-, endothelin 1-, and thromboxane A2-induced vasoconstriction. Moreover, ROS can rapidly scavenge nitric oxide (NO) and blunt NO activity in renal vasodilatation, 9 resulting in renal ischemic injury and CIN. In addition to their traditional effects on blood cholesterol levels, statins also decrease expression of angiotensin receptors and synthesis of endothelin. These changes may prevent CIN by decreasing the period of renal hypoperfusion and ischemia by increasing vascular smooth muscle relaxation, resulting in vasodilation. 30 Endothelial dysfunction and inflammation are also involved in CIN pathogenesis. Endothelial dysfunction can be caused by an imbalance between NO and endothelin 1 after CM exposure. Previous studies have demonstrated that statins may help correct this imbalance by increasing NO production and reducing endothelin 1 synthesis. 31 C-reactive protein (CRP), as a marker of systemic inflammation, is associated with CIN, and patients with high periprocedural CRP levels are at high risk of developing CIN. 26,32,33 Renal protection due to statins’ anti-inflammatory effects during CM exposure is considered to play a major role in preventing CIN. 20,26,34
Based on the existing evidence, clinical trials have been conducted with an objective to evaluate the effects of short-term statins use on CIN development. However, results from RCTs and meta-analyses regarding the use of statins to prevent CIN are conflicting and inconclusive. Previously published meta-analyses of RCTs performed by Zhang et al 11 (included 6 RCTs), Zhang et al 12 (4 RCTs), and Pappy et al 13 (only 3 RCTs) suggested that statins pretreatment had no effect on the incidence of CIN or the need for RRT. In fact, a major limitation of these meta-analyses is the lack of large randomized trials adequately powered to address this clinically important issue. Moreover, 2 of the previous meta-analyses by Zhang et al 11 and Pappy R et al 13 et al included both randomized and nonrandomized trials; the latter could lead to potential bias because unknown confounding factors could not be completely controlled, even with statistical adjustments. Therefore, the results from previous meta-analyses could not determine the real effects of statins pretreatment on CIN prevention.
In the current meta-analysis, the results showed that statin therapy before CM exposure could reduce the risk of CIN but not all-cause death. These results were in agreement with recent meta-analyses. 14 -16 However, almost all of the previous RCTs had small sample sizes, and the previous meta-analyses did not include 4 recent large-scale and high-quality studies with Jadad scores ≥3. In addition, the recent meta-analysis performed by Li et al 15 reported that statin therapy was only shown to prevent CIN in lower quality studies (Jadad score ≤ 3), but it was not found to be effective in high-quality studies. This finding would weaken the evidence of statins’ ability to prevent CIN. However, in our meta-analysis, sensitivity analysis with high-quality studies (Jadad score > 3) demonstrated that statins remain effective in preventing CIN, indicating that low-quality studies did not bias our results.
Additionally, it was observed that there was a 86% reduction in the risk of RRT in patients undergoing coronary angiography/percutaneous interventions, which was different from findings reported in previous studies. 15 This effect would additionally contribute to the beneficial effect of statin therapy on postoperative mortality. Recent observational studies and meta-analysis suggested that patients undergoing coronary artery bypass grafting benefitted from preoperative statin therapy in terms of reduced need for postoperative RRT. 35 In addition, this study also demonstrated that preoperative statin therapy was associated with lower short-term mortality in patients undergoing cardiac surgery. However, our recent meta-analysis did not demonstrate the effect of statins on lowering mortality in patients who underwent coronary angiography/percutaneous interventions. 15 Nevertheless, few well-designed, multicenter randomized studies have been designed to investigate the effects of statins on mortality or the need for RRT in patients undergoing coronary angiography/percutaneous interventions. In this regard, caution is advised when interpreting this result.
The timing of SCr measurement after the procedure remained debatable. Previous studies found that SCr concentration typically peaks 3 to 5 days after CM administration and returns to baseline or near baseline within 1 to 3 weeks. 36 A single 24-hour measurement would have missed 58.2% of the CIN cases that were detected by the 48-hour measurement. 37,38 Therefore, the different timing of SCr measurement might affect the real incidence of CIN. And these would affect the ability of statins to prevent CIN to some degree. However, our subgroup analysis demonstrated that statin pretreatment reduced the risk of CIN with different definitions (within 48 or 72 hours).
Previous analyses reported that the difference between patients treated with statins plus NAC and patients who only received NAC was not significant. 15 However, our subgroup analysis by the use of randomized effect indicated that statins plus NAC were more effective in preventing CIN than NAC only. We may consider that statins would exert additive renal protection to NAC for CIN prevention, although both statins and NAC exert anti-inflammatory and antioxidant effects. However, our pooled RR should be interpreted with caution, and the result should be confirmed in additional studies comparing statins with NAC.
In accordance with a previous analysis,
15
we found that statin therapy prevented CIN among patients without preexisting renal dysfunction. However, previous meta-analyses have demonstrated that statin pretreatment had no preventive effect on CIN in patients with preexisting renal dysfunction.
14,15
These authors argued that chronic renal dysfunction may have a different pathogenetic mechanism from acute renal dysfunction, such as enhanced inflammatory status, a high prevalence of atherosclerosis involving multiple vascular beds, more complications, and impaired response to statin agents, contributing to the failure of statins to prevent CIN. However, we found that statins prevented CIN development in patients with preexisting renal dysfunction; although the fixed effect model showed moderate heterogeneity, it was not significant (χ2 = 8.86, I
2
= 44%,
DM is an additional important risk factor for CIN.
39
Our subgroup analysis suggested that DM patients pretreated with statins had a 40% lower risk of developing CIN than that of controls, which was in contrast to a subgroup analysis in a previous meta-analysis (OR = 0.76, 95% CI = 0.28-2.12,
In addition, although rosuvastatin was suggested to be a more potent statin relative to atorvastatin with respect to its anti-inflammatory efficacy, 40 ,41,42 in our subgroup analysis, we might be the first to demonstrate that rosuvastatin exerts the same effect as atorvastatin treatment on reducing the risk of CIN in patients undergoing coronary angiography/percutaneous interventions. The result was consistent with that from a recent RCT performed in patients with ST-segment elevation myocardial infarction undergoing primary coronary angioplasty (atorvastatin 80 mg, n = 98 or rosuvastatin 40 mg, n = 94). 43 Nevertheless, we should consider the fact that a number of different factors are involved in the development of CIN and different pathophysiological mechanisms coexist. Moreover, no large head-to-head studies have assessed whether rosuvastatin is superior to other statins in reducing the risk of CIN. Therefore, future research should compare hydrophilic and lipophilic statins to determine whether they exhibit different abilities in reducing CIN risk.
Study Limitations
This study had several limitations. First, the studies in our meta-analysis evaluated the efficacy of statins with different dose regimens for varied periods of time. It is possible that dose and duration may influence the preventive effect of statins on CIN. Second, publication bias is always a potential limitation. In other words, neutral or negative studies may not be published in a peer-reviewed journal, whereas positive studies are more likely to be published. Third, the different hydration protocols among included studies influence the occurrence of CIN. However, we did not have access to data that would allow us to estimate the impact of hydration protocols on CIN. Despite these limitations, the present review provides, to date, the most comprehensive review of benefits of statins in the management of CIN.
Conclusion
The present meta-analysis supported the use of periprocedure statin treatment to reduce the risk of CIN with different definitions (within 48 or 72 hours) and the need for RRT in patients undergoing coronary angiography/percutaneous interventions. The subgroup analysis demonstrated that statin pretreatment would be helpful in reducing the incidence of CIN in patients with or without preexisting renal dysfunction or DM. We also found that rosuvastatin and atorvastatin had similar preventive effects against CIN. However, future research should attempt to determine which stages of renal dysfunction benefit most by statin pretreatment and if various statins exert different preventive effects.
Footnotes
Author Contribution
Y-H. Liu contributed to acquisition, analysis, and interpretation, drafted manuscript, and agrees to be accountable for all aspects of work ensuring itegrity and accuracy; Y. Liu contributed to design, critically revised manuscript, and agrees to be accountable for all aspects of work ensuring itegrity and accuracy; C-Y. Duan contributed to design, critically revised manuscript, and agrees to be accountable for all aspects of work ensuring itegrity and accuracy; N. Tan contributed to conception and design, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring itegrity and accuracy; J-Y. Chen contributed to design, critically revised manuscript, and agrees to be accountable for all aspects of work ensuring itegrity and accuracy; Y-L. Zhou contributed to design, critically revised manuscript, and agrees to be accountable for all aspects of work ensuring itegrity and accuracy; L-W. Li contributed to design, critically revised manuscript, and agrees to be accountable for all aspects of work ensuring itegrity and accuracy; P-C. He contributed to design, critically revised manuscript, and agrees to be accountable for all aspects of work ensuring itegrity and accuracy. All authors were involved in the manuscript preparation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grant from the National Natural Science Foundation of China (grant no. 81270286). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. And the work was not funded by any industry sponsors.
