Abstract
Keywords
Introduction
Cardiovascular diseases are the leading cause of death worldwide. According to the latest global burden of ischemic heart disease (coronary heart disease) report, there were an estimated 197 million people with coronary heart disease in 2019, among which 9.14 million died. 1 Percutaneous coronary intervention (PCI) has been proved to be the most effective way to quickly restore adequate flow in the affected coronary artery in patients with acute myocardial infarction (AMI). 2 All PCI requires contrast agent in normal practice, which may lead to complications after the operation, such as contrast-induced acute kidney injury (CIAKI). Previous studies have suggested that the incidence of CIAKI in the general population after coronary angiography is generally less than 5%, but the incidence is significantly higher in patients with renal insufficiency, AMI, shock, and diabetes.3-5 CIAKI can increase the need for renal replacement therapy, the incidence of major adverse cardiovascular events (MACE), prolong the average length of hospital stay, and increase the cost of hospitalization. 6 Therefore, the prevention and treatment of CIAKI are extremely important. Volume expansion with sodium chloride or bicarbonate or both, administration of statins, N-acetyl cysteine, hemofiltration or hemodialysis, and reducing the volume of contrast media administered are some of the preventive measures for CIAKI; hydration or drug prevention are commonly used for patients with elective PCI to improve acute kidney injury and clinical prognosis. However, ST-segment elevation myocardial infarction (STEMI) patients need emergency PCI to restore blood flow in the occluded segment, and these known common preventive measures are not readily available to STEMI patients. Therefore, the effective prevention measures for CIAKI need to be explored.
Nicorandil is adenosine triphosphate (ATP) sensitive potassium channel opener agent that promotes vasodilation, antagonizes ischemia-reperfusion injury, and protects the myocardium. 7 It is commonly given to patients with coronary heart disease. Considering that ATP-sensitive potassium channels are also found in other organs besides the heart, including the kidney, 8 nicorandil may have a beneficial effect on the kidney. Nawa et al 9 found that continuous intravenous infusion of nicorandil 4 h before PCI and 24 h after PCI could prevent contrast-induced nephropathy in patients with renal dysfunction. Similarly, Fanz et al 10 discovered that oral nicorandil was superior to hydration alone in preventing contrast-induced nephropathy and could reduce adverse events (for 1 year after operation) in patients with chronic renal insufficiency who received PCI. However, the PRINCIPLE study 11 found that hydration combined with intravenous administration of low doses of nicorandil did not reduce the incidence of CIAKI after coronary angiography in patients with estimated glomerular filtration rate (eGFR) < 60 mL/min/1.73 m2. Thus, the effect of nicorandil on CIAKI in those who undergo PCI needs to be further investigated. This single-center, retrospective controlled study examined whether nicorandil has a protective effect on STEMI patients with CIAKI after emergency PCI.
Methods
Statement of Ethics
The study was approved by the Ethics Committee of the Second Affiliated Hospital of Nanchang University. All participants provided their written informed consent upon admission. The study was conducted in accordance with the Declaration of Helsinki.
Study Population
STEMI patients admitted to the Second Affiliated Hospital of Nanchang University, Jiangxi Province, China between October 1, 2017 and February 28, 2019 were included in this single-center, retrospective control study. STEMI was diagnosed based on the 2017 guidelines for the management of patients with ESC ST-elevation myocardial infarction. 2 Inclusion criteria were: emergency PCI within 24 h of onset. Exclusion criteria were the following: patients with severe renal insufficiency (eGFR < 30 mL/min/1.73 m2) or long-term hemodialysis, cardiogenic shock, hypotension (systolic blood pressure < 80mm Hg for at least 1 h) or use of vasoactive drugs, remedial hydration after PCI or exposure to contrast agent 7 days before PCI; severely incomplete clinical data, and contraindication to nicorandil.
Study Protocol
Patients were divided into nicorandil and control groups according to whether nicorandil was used. Patients in the nicorandil group were treated with AMI standard therapy (double antiplatelet, statin, and other drugs) and 72 mg nicorandil that was intravenously pumped (Trade name: Recoxi, Specification: 12 mg/dose, usage: 6 mg/h for 12 h) after PCI. The control group received standard AMI therapy after PCI. Non-ionic hypotonic contrast agent iodoplomide injection (Trade name: Youweixian) was used for all patients as part of PCI. The interventional procedure was performed using the radial or femoral artery approach.
Clinical baseline data, including age, gender, previous medical history, personal history, Killip grade, infarction site, drug use, surgical data (such as TIMI grade before PCI, number of vascular lesions, contrast agent dosage, etc), and laboratory examination indicators were collected from all patients. Blood samples were collected at admission and 24/48/72 h and 5 to 7 days after PCI to detect serum creatinine (Scr), eGFR, uric acid, and β2-microglobulin levels. In addition, creatine kinase isoenzymes (CK-MB) were detected 24 h after admission and 1/3/5 days after PCI to evaluate the difference in the peak value of CK-MB between the 2 groups.
Endpoints
The incidence of CIAKI, defined as an increase of >25% or absolute values > 44.2 μmol/L in Scr from baseline within 72 h of exposure to contrast agent after exclusion of other causes, was the primary endpoint. Secondary endpoints included: (1) changes in Scr, eGFR, uric acid, and β2-microglobulin at 24/48/72 h and 5 to 7 days after PCI; (2) peak value difference of CK-MB after PCI; (3) adverse events within 6 months after PCI, including hospital hemodialysis rate, all-cause death, cardiac death, recurrent angina, acute heart failure or nonfatal myocardial infarction readmission.
Statistical Analysis
The measurement data following normal distribution are expressed as mean ± standard deviation (x ± s) and compared by t-test. Non-normally distributed measurement data are presented as median (interquartile spacing) [M (QR)] and are compared by the rank-sum test. The counting data are presented as numbers or percentages, and the Chi-square test was used for comparison. Two-way analysis of variance test was performed to evaluate differences in renal function parameters and CK-MB between nicorandil and control group. Univariate and multiple logistic regression analysis were used to determine the factors of CIAKI. SPSS 24.0 was used for statistical analysis. P < .05 is considered to be statistically significant.
Results
Baseline Characteristics
A total of 156 STEMI patients, including 128 males (82%) and 28 females (18%), with an average age of 61.53 ± 13.13 years, were included in the study. Among the 156 patients, 55 were in the nicorandil group and 101 in the control group. Baseline characteristics were similar between the 2 groups (all P > .05, shown in Table 1). There were no significant differences between the 2 groups in age, gender, hypertension, diabetes, smoking history, Killip grade, TIMI blood flow grade, number of vascular lesions, infarct site, drug treatment, and laboratory examination (all P > .05). The contrast dosage was 90 (80,120) mL in the nicorandil group and 90 (80,100) mL in the control group (P = .076).
Baseline Clinical Characteristics of the Patients.

Data are the mean ± SD or median (interquartile spacing), or number (percentage).
Abbreviations: ACEI: angiotensin-converting enzyme inhibitors; ARB: angiotensin receptor blockers; Hb: hemoglobin; BNP: B-type natriuretic peptide; TC: total cholesterol; LDL-C: low-density lipoprotein-cholesterol.
Incidence of CIKAI
The overall incidence of CIAKI was 21.8% (34/156). Also, the incidence of CIAKI in the nicorandil group was significantly lower [12.7% (7/55)] than in the control group [26.7% (27/101)] (P = .043, shown in Figure 1).

Incidence of CIAKI between the nicorandil group and control group. Among the 156 patients, 55 were in the nicorandil group and 101 in the control group. The Chi-square test was used to compare the incidence of CIAKI between the 2 groups. The result showed that the incidence of CIAKI in the nicorandil group was significantly lower [12.7% (7/55)] than in the control group [26.7% (27/101)] (P = .043).
Comparison of Renal Function Parameters at Baseline and After PCI
The changes in Scr, eGFR, uric acid, and β2-microglobulin were compared between the 2 groups (shown in Figure 2). Baseline Scr between nicorandil and control groups (79.90 ± 24.15 vs 86.19 ± 23.07 μmol/L, P = .112), eGFR (94.96 ± 25.04 vs 87.48 ± 41.04 mL/min/1.73 m2, P = .220), uric acid (378.31 ± 82.26 vs 404.08 ± 112.63 μmol/L, P = .138), and β2-microglobulin (1.95 ± 0.71 vs 1.95 ± 0.89 mg/L, P = .973) levels were not significantly different in the 2 groups. However, Scr, uric acid, and β2-microglobulin in both groups increased at 24 h after surgery, peaked at 72 h, and gradually decreased after that; still, these levels were significantly lower in the nicorandil group than those in the control group at 48 h, 72 h, and 5 to 7 days after PCI (P < .05). Moreover, eGFR significantly decreased in both groups, reaching a minimum level at 72 h, after which it gradually increased. Compared with the control group, eGFR levels in the nicorandil group were significantly higher at 48 h, 72 h, and 5 to 7 days after PCI (P < .05).
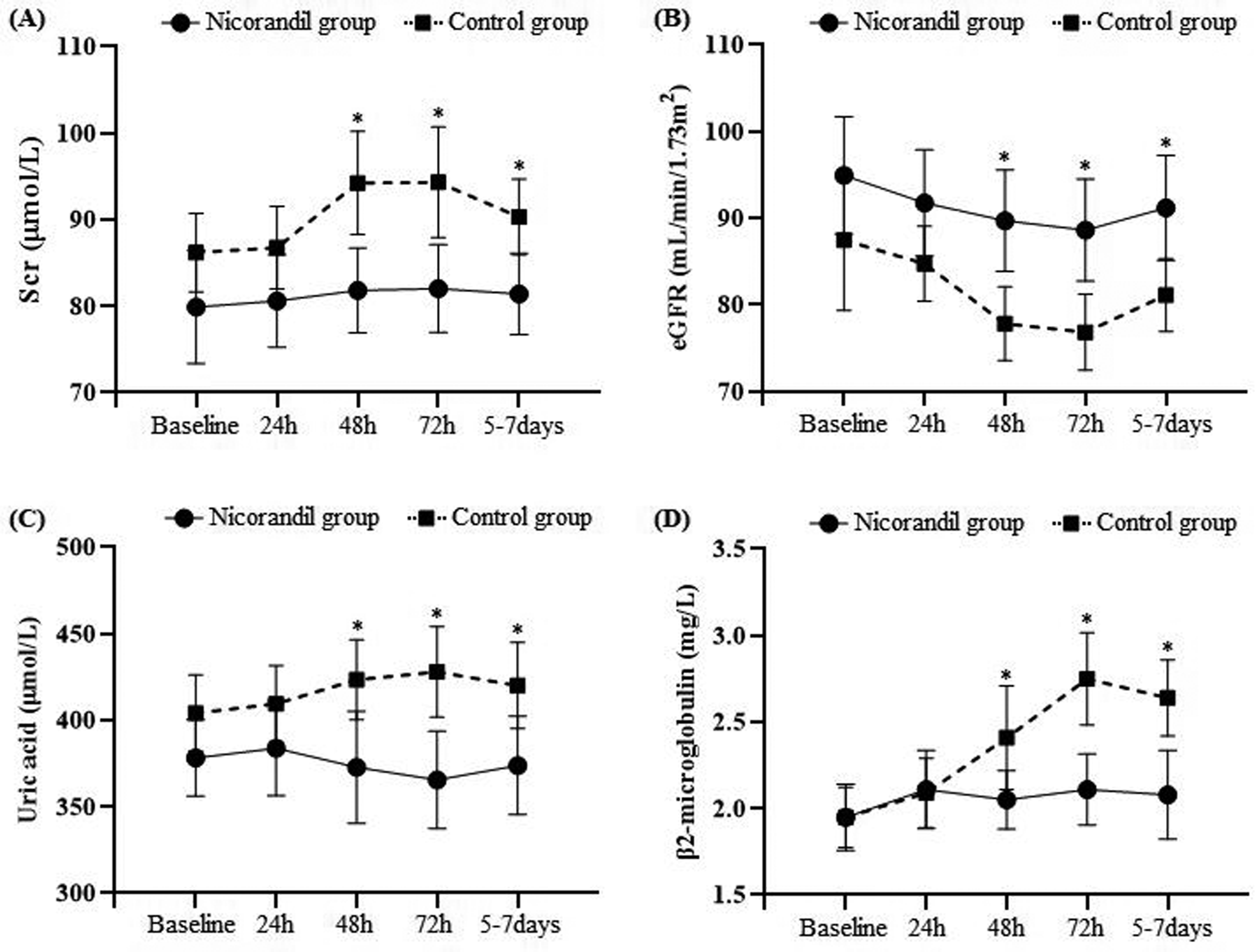
The changes of SCr, eGFR, Uric acidand β2-microglobulin between the nicorandil group and control group. The baseline level of Scr, eGFR, uric acid, and β2-microglobulin are not significantly different in the 2 groups. Scr, uric acid, and β2-microglobulin in both groups increased at 24 h after PCI, peaked at 72 h, and gradually decreased after that. The levels of these indicators are significantly lower in the nicorandil group than those in the control group at 48 h, 72 h, and 5 to 7 days after PCI (P < .05). eGFR significantly decreased in both groups, reaching a minimum level at 72 h, after which it gradually increased. Compared with the control group, eGFR levels in the nicorandil group were significantly higher at 48 h, 72 h, and 5 to 7 days after PCI (P < .05) (*P < .05 nicorandil group vs control group).
Comparison of the CK-MB Peak Value
Compared with the control group, the peak value of CK-MB in the nicorandil group was significantly reduced [105.30 (56.61,232.04) vs178.00 (77.08,271.91) U/L, P = .042] (shown in Table 2).
The Peak Value of CK-MB.

Abbreviation: CK-MB: creatine kinase isoenzymes.
Adverse Events Within 6 Months After PCI
During the follow-up period, there were 2 cardiac deaths in the nicorandil group, including one death from end-stage heart failure and one another from fatal myocardial infarction. Moreover, 2 cardiac deaths were observed in the control group; one patient died in hospital from malignant arrhythmia, and one had a fatal myocardial infarction. There were no hospital hemodialysis events in any of the groups (shown in Table 3).
Adverse Events Within 6 Months After PCI.

Abbreviation: PCI: percutaneous coronary intervention.
Univariate and Multiple Logistic Regression Analysis
According to the occurrence of CIAKI, 156 STEMI patients were divided into CIAKI group (n = 34) and non-CIAKI group (n = 122). Baseline characteristics showed that the proportion of hypertension and diabetes in the CIAKI group was significantly higher than that in the non-CIAKI group (P < .05), and there were no significant differences in other baseline characteristics between the 2 groups (P > 0.05) (shown in Supplemental Table). With the incidence of CIAKI as the dependent variable, hypertension, diabetes, and nicorandil treatment were included in univariate and binary logistic regression analysis. The results of univariate analysis showed that hypertension and diabetes were positively correlated with CIAKI, and nicorandil treatment was negatively correlated with CIAKI (shown in Table 4). The results of binary logistic regression analysis showed that hypertension and diabetes were independent risk factors for CIAKI, while nicorandil treatment was a protective factor (shown in Table 5).
Univariate Analyses on the Risk Factors for CIAKI in Patients.

Abbreviation: CIAKI: contrast-induced acute kidney injury.
Logistic Regression Analyses on the Risk Factors for CIAKI in Patients.

CIAKI: contrast-induced acute kidney injury; SD: standard deviation.
Discussion
In this retrospective study, we observed that nicorandil could significantly reduce the incidence of CIAKI in STEMI patients receiving emergency PCI. Moreover, multiple logistic regression analysis showed that hypertension and diabetes history were independent risk factors for CIAKI, while nicorandil was a protective factor.
In this study, the overall incidence of CIAKI was as high as 21.8%, which was higher than the incidence reported in previous studies, mainly because the STEMI patients were included in this study. Liu et al 12 showed that the incidence of CIAKI in STEMI patients undergoing primary PCI was 21.8% in aggressive hydration group and 31.1% in general hydration group. There is evidence that patients with acute coronary syndrome have hemodynamic instability, activation of the neuroendocrine system, and a strong inflammatory response.13,14 Moreover, STEMI is one of the most important factors in the increased incidence of CIAKI and hemodialysis rate. 15 However, the patients are unable to undergo proven prophylactic measures to attenuate CIAKI due to the emergent nature of the STEMI. Therefore, STEMI patients may have a higher risk of developing CIAKI and require active prevention and management of CIAKI. Although the specific pathological mechanism of CIAKI remains unclear, direct cytotoxic effects and hemodynamic changes, which lead to renal hypoperfusion and, in turn, reduce glomerular filtration rate, have been suggested to be the key mechanism in the pathophysiology of CIAKI. 16 Contrast agents can cause the injury of the endothelial cells of renal blood vessels and renal tubular epithelial cell by increasing the level of inflammatory mediator, inducing oxidative stress and increasing blood viscosity.16-20 In addition, the oxygen partial pressure in peripheral renal medulla further reduced after administration of contrast agent. 16 Coupled with enhanced metabolic requirements, the medulla is thus particularly vulnerable to the effect of contrast agent on renal hemodynamics.16,21 Previous studies have also found that the onset of CIAKI usually starts gradually, within 24 h after PCI, it then peaks between 48 and 72 h and recovers about 7 days after PCI. 22 The trend of renal function parameters in this study was generally consistent with previous studies.
The risk factors for CIAKI after PCI include age, hypertension, diabetes, primary renal insufficiency, heart failure, transarterial injection of contrast agent, and high-dose contrast agent application, 23 and the incidence of CIAKI increases in patients with multiple risk factors. 24 In this study, hypertension and diabetes were independent risk factors for CIAKI after emergency PCI. Other common risk factors were not indicated in this study due to the following reasons: (1) Patients with hypotension and cardiogenic shock were excluded from this study, so the proportion of patients with heart failure and the implantation rate of left ventricular auxiliary devices were both low. (2) The median amount of contrast agent used in 156 patients included in this study was 90 mL, making it difficult to determine the relationship between high-dose contrast agent and CIAKI. (3) Only 7.69% of the included patients had eGFR < 60 mL/min/1.73 m2, which may lead to the effect of renal insufficiency on the incidence of CIAKI being covered by hypertension and diabetes factors.
Nicorandil is an ATP-sensitive potassium channel (KATP) opening agent synthesized by nitriester and nicotinic acid that has a nitrate-like effect. Several studies have confirmed that PCI combined with nicorandil can reduce the size of myocardial infarction and the incidence of MACE in patients.25,26 Based on its unique dual pharmacological mechanism and cardioprotective efficacy, nicorandil has often been used to treat microvascular angina and AMI. The results of this study showed that the peak value of CK-MB after PCI in the nicorandil group was significantly lower than that in the control group, which further confirmed that nicorandil could improve cardiac function in patients with STEMI. However, KATP is not only distributed in the myocardium but also present in the kidney, pancreas, skeletal muscle, and other tissues. 8 One study showed that the activation of KATP reduces the accumulation of ROS in renal tissue caused by ischemia-reperfusion injury, thereby alleviating renal injury. 27 When acute kidney injury occurs, the injury of renal vascular endothelial cells and tubular epithelial cells leads to a decrease in glomerular filtration rate, resulting in a significant increase in Scr, uric acid and β2-microglobulin. Studies have shown that nicorandil can improve renal hemodynamics through NO-mediated vasodilation, inhibition of ROS formation and reduction of inflammatory mediators, so as to improve glomerular filtration rate and play a protective role in the kidney.9,27 These results suggest that nicorandil may be an effective strategy for treating CIAKI.
So far, several clinical studies have reported the effect of nicorandil on patients with CIAKI. However, the results are contradictory. Nawa et al 9 included 213 patients with elevated serum cystatin that were randomly divided into a normal saline group (n = 107) and a nicorandil group (n = 106, continuous infusion 4 h before PCI and 24 h after PCI). The results showed that the incidence of CIAKI was significantly lower in the nicorandil group than in the normal saline group (2.0% vs 10.7%, P < .02). However, the PRINCIPLE study 11 failed to produce beneficial results for nicorandil. The study involved 166 patients with eGFR < 60 mL/min/1.73 m2 randomly divided into nicorandil and control groups and found that hydration combined with intravenous administration of low-dose nicorandil does not reduce the incidence of CIAKI (6.8% vs 6.6%, P = .79) after coronary angiography. The results of that study did not show an effect of nicorandil on CIAKI and we speculate 2 possible reasons. On the one hand, the dose of nicorandil in the PRINCIPLE study is only 12 mg, which was different from the 72 mg in present study. The relatively low dose of the nicorandil maybe resulted in insignificant renal protection. On the other hand, nicorandil used for only 30 min before coronary angiography may not prevent contrast nephropathy. Studies have shown that the average elimination half-life of nicorandil is only about 1 h, while the average elimination half-life of contrast agents is up to 2 h, and even up to 4 h in people with impaired renal function.28,29 Therefore, continued use of nicorandil after PCI can better prevent the occurrence of CIAKI. In addition, oral nicorandil can also reduce the incidence of CIAKI. Fan et al 10 found that oral nicorandil has a protective effect on CIAKI in patients with chronic renal insufficiency who undergo elective coronary surgery and can reduce the incidence of adverse events within 1 year after surgery and was superior to hydration alone. Iranirad et al 24 similarly suggested a protective effect of oral nicorandil against CIAKI in high-risk patients undergoing cardiac catheterization. However, the results of the meta-analysis showed that compared with the intravenous route, oral nicorandil had greater efficacy and independently reduced the incidence of CIAKI. 30 We hypothesized that the high heterogeneity of the effects of intravenous and oral administration was mainly due to the dosing regimen, and that continuous administration after PCI could significantly reduce the incidence of CIAKI. Our findings also suggest that intravenous nicorandil after PCI can reduce the incidence of CIAKI. At present, there is a lack of relevant studies comparing intravenous and oral nicorandil for the prevention of CIAKI. In the future, it is necessary to conduct randomized controlled trials to compare the effects of different administration methods and administration time of nicorandil on the prevention of CIAKI.
This study has a few limitations. First, failure to use nicorandil before the opening of infarct-related vessels may increase the incidence of CIAKI. Ishii et al 25 found that intravenous nicorandil administered before reperfusion in STEMI patients resulted in beneficial clinical outcomes and prevention of long-term cardiovascular events and death. Second, the majority of the patients have a normal renal function before the start of the case. In future studies, we could consider selecting patients based on their risk of CIAKI (ie, Mehran score). Third, the concentration of Scr is susceptible to various factors such as age, gender, race, muscle activity, dietary meat intake, wasting diseases, and drugs, and the kidney has a strong compensatory ability. Therefore, the change of Scr after a renal injury is relatively lagging, which is less suitable for detecting mild renal injury. Last, this study is a single-center retrospective study with a small sample size.
Conclusion
This study suggest that intravenous nicorandil after emergency PCI has a protective effect on the occurrence of CIAKI in STEMI patients. Nicorandil may be used in high-risk patients undergoing emergency PCI to reduce the incidence of CIAKI.
Supplemental Material
sj-doc-1-cpt-10.1177_10742484231174296 - Supplemental material for Protective Effect of Nicorandil on Contrast-Induced Acute Kidney Injury After Emergency Percutaneous Coronary Intervention
Supplemental material, sj-doc-1-cpt-10.1177_10742484231174296 for Protective Effect of Nicorandil on Contrast-Induced Acute Kidney Injury After Emergency Percutaneous Coronary Intervention by Zuo-zhong Yu, Zheng-xiong Xu, Yue-hua Ruan, Long-long Hu, Ming-hua Wen, Tie-qiu Huang, Zhi-gang You, Yan-qing Wu and Ren-qiang Yang in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Acknowledgments
The authors would like to thank all the reviewers who participated in the review.
Author Contributions
Zuozhong Yu and Zhengxiong Xu performed the data collection, reviewed articles, and wrote the manuscript. Longlong Hu, Yuehua Ruan, Minghua Wen, Tieqiu Huang, Zhigang You, and Yanqing Wu completed the data analysis and provided the second views during the manuscript preparation. Renqiang Yang designed the study and revised the manuscript. All the authors read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 81960081) and the “5511” Innovative Drivers for Talent Teams of Jiangxi Province (No. 20171BCB18004).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
