Abstract
Introduction:
The use of β-blockers in the treatment of patients with coronary heart disease is associated with a decrease in the frequency of angina pectoris and mortality of patients. Due to the severity of the disease and previous cardiovascular interventions, many patients with coronary artery disease (CAD) use dual antiplatelet therapy to achieve greater inhibition of platelet aggregation. The influence of β-blockers on platelet aggregation in patients using antiplatelet therapy is not well understood.
Objective:
To examine the effect of different β-blockers on platelet aggregation in patients on dual antiplatelet therapy.
Methodology:
The study included 331 patients who were treated at the Department of Cardiology, Clinical Center Kragujevac during 2011. Patients were divided into 4 groups depending on the type of β-blockers that were used (bisoprolol, nebivolol, metoprolol, and carvedilol). Platelet aggregation was measured using the multiplate analyzer and expressed through the value of adenosine diphosphate (ADP) test (to assess the effect of clopidogrel), ASPI test (to assess the effect of acetyl salicylic acid), TRAP test (to assess baseline platelet aggregation), and the ratio of ADP/TRAP and ASPI/TRAP ASPI/TRAP (ASPI - aranchidonic acid induced aggregation, TRAP - thrombin receptor activating peptide) representing the degree of inhibition of platelet aggregation compared to the basal value. In consideration were taken the representation of demographic, clinical characteristics, laboratory parameters, and cardiovascular medications between the groups.
Results:
Patients who used nebivolol had a significantly lower value of the ratio of ADP/TRAP (0.39 ± 0.30) compared to patients who used bisoprolol (0.48 ± 0.26; P = .038), and trend toward the lower values of ADP test (328.0 ± 197.3 vs 403.7 ± 213.2; P = .059), while there was no statistically significant difference in values of other laboratory parameters of platelet function between other groups.
Conclusion:
Patients with CAD on dual antiplatelet therapy who used nebivolol had significantly lower levels of residual ADP-induced platelet aggregation compared to baseline than patients who used bisoprolol.
Keywords
Introduction
The adenosine diphosphate (ADP) receptor antagonist, clopidogrel, in addition to acetyl salicylic acid (ASA) reduces the incidence of cardiovascular events in acute coronary syndrome and after percutaneous coronary intervention with coronary stent implantation. 1,2
Measurements of platelet aggregability, after clopidogrel and ASA administration, have shown heterogeneous response. The variation in response to antiplatelet therapy could be attributed to many factors: genetic (polymorphism of the P2Y12 gene, polymorphism of the P-450 CYP 3A4 gene, polymorphism or mutation of cyclooxygenase [COX] 1, and overexpression of COX-2 gene), cellular (accelerated platelet turnover), or clinical (noncompliance, drug interaction, increased platelet reactivity, etc). 3 –7
Due to their antianginous effect, β-adrenergic blockers are often prescribed to the patients with manifested coronary artery disease (CAD). 8 Several trials and 1 meta-analysis 9 have reported antiaggregatory effect of some β-blockers, such as propranolol, 10 carvedilol, 11 and nebivolol, 12 but despite their extensive use in CAD, there isn’t much data on their antiaggregatory benefits in patients already receiving dual antiplatelet therapy. Except for their common effect on β-adrenoceptor antagonism, β-blockers are a heterogeneous group of drugs that differ in their β1 selectivity, intrinsic activation, liposolubility, and in specific drug properties such as induction of nitric oxide (NO) synthesis, α-receptor antagonism, membrane stabilization effect, and antioxidative effect which could potentially influence platelet function.
Aim
The aim of our study was to examine the influence of different types of β-blockers (bisoprolol, carvedilol, nebivolol, and metoprolol) on platelet aggregation induced by ADP, arachidonic acid, and thrombin receptor-activating protein in patients with CAD on dual antiplatelet therapy.
Methods
Our prospective, clinical study included 331 patients, of both sexes, with no age restriction, hospitalized for treatment and/or diagnostics of coronary heart disease at Cardiology department, Clinical Center Kragujevac, Serbia, during 2011.
Study included only the patients who had documented CAD except acute myocardial infarction, who were on antiplatelet therapy: ASA (100 mg daily) and clopidogrel (75 mg daily) for at least 1 week prior to study entry, and were using same β-blocker continuously for at least 1 month prior to study entry.
Patients who did not have continuity in the dual antiplatelet therapy (1 week prior to the study entry) or have stopped taking the β-blockers, or had replaced it with other β-blockers 1 month prior to the study entry, were excluded from our study. Patients who concomitantly used other antiplatelet medications such as dipyridamole, abciximab, tirofiban, eptifibatib, and prasugrel, also were not included.
Laboratory Analysis and Data Collected
After inclusion of the patients in the study, during their hospitalization, the blood samples were taken to determine the level of platelet aggregation and other laboratory parameters such as complete blood count, hemostasis, lipidogram, glycaemia, C-reactive protein, blood urea nitrogen, serum creatinin, and enzymes (creatinine kinase [CK], CK-MB, lactate dehydrogenase, aspartate aminotransferase, and alanine transaminase). We also collected data about patients’ demographics, risk factors for CAD, all medicaments patients were taking, indications for hospitalization (urgent hospitalization—within 1 month since CAD diagnosis, elective—more than 1 month since diagnosis), history of all previous diseases, hospitalizations, and interventions.
Platelet Function Monitoring
Platelet aggregability has been measured in heparinized whole blood samples by the method of impedance aggregometry using Multiplate analyzer (Dynabite, Munchen, Germany). The effect of antiplatelet therapy, ASA and clopidogrel, was assessed in 2 ways. First, by direct measurements of platelet aggregability after addition of agonist, ADP for the assessment of clopidogrel response (ADP test) and arachidonate for the assessment of ASA response (ASPI test), whereby higher values of these tests indicated a higher residual platelet aggregation and reduced antiplatelet effect of drugs. Second, by the ratio of ADP/TRAP or ASPI/TRAP (expressed as a percentage), which indicates percentage of residual platelet activity, compared to the basal value. The TRAP test is used to assess the effect of inhibitors of glycoprotein IIb/IIIa receptors on the platelet aggregability, and its value is not affected by treatment with ASA and the negligibly by clopidogrel, which is the reason why this test is used for measuring basal platelet aggregability if patient didn’t take glycoprotein IIb/IIIa antagonist.
Statistical Analysis
Data were analyzed by descriptive and analytical statistics. Depending on the obtained distributions, we used analysis of variance (ANOVA) or Kruskal-Wallis test for comparison of groups with different types of β-blockers in relation to the value of ADP, ASPI, and TRAP tests. In order to reduce family-wise error rate in multiple comparisons, we used Bonferroni correction, which is incorporated in post hoc analysis of ANOVA in statistical program SPSS. We also performed contrast tests for post hoc testing of ANOVA results of interest.
We also used chi-square (χ2) test, t test, and Mann-Whitney U test for analysis of influence of different demographic, clinical, and laboratory factors on the platelet aggregation test between the compared groups.
Results
Patients were divided into 4 groups depending on the type of β-blockers that were used, whereas the largest group was bisoprolol group and the smallest was metoprolol group. Patients’ demographics and clinical characteristics are shown in Table 1.
Patients’ Demographics and Clinical Characteristics.

Abbreviations: ANOVA, analysis of variance; CABG, coronary artery bypass graft surgery; PCI, percutaneous coronary intervention; χ2, chi-square.
aANOVA test.
bχ2 test.
cSignificant at P < .05.
There were no significant differences in demographic characteristics between groups. All risk factors, except hypertension, were equally represented among groups. The highest percentage of patients who were treated for hypertension was in the group, which used bisoprolol, and the smallest in the metoprolol group.
Between the groups, there was also no significant difference in the use of other cardiovascular drugs, as shown in Table 2. Nitrates, statins, ACE/AT inhibitors, ACE inhibitors/AT1 receptor blockers calcium (Ca)-channel blockers, trimetazidine, and inhibitors of proton pump were equally distributed among all groups.
Use of Other Cardiovascular Drugs Between the Groups.

Abbreviations: Ca, calcium; χ2, chi-square.
aχ2 test.
Values of laboratory parameters among the groups are shown in Table 3, while the values of hemostasis parameters are shown in Table 4. Values of laboratory parameters and hemostasis parameters, except for creatinine concentration, did not significantly differ between the groups that were compared.
Values of Laboratory Parameters Between the Groups.

Abbreviations: ANOVA, analysis of variance; AST, aspartate aminotransferase; ALT, alanine transaminase; CK, creatinine kinase; HDL, high-density lipoprotein; LDH, lactate dehydrogenase; LDL, low-density lipoprotein; SD, standard deviation; TGL, triglyceride; χ2, chi-square.
aχ2 test.
bSignificant at P < .05.
cANOVA test.
Values of Hemostasis Parameters Between the Groups.
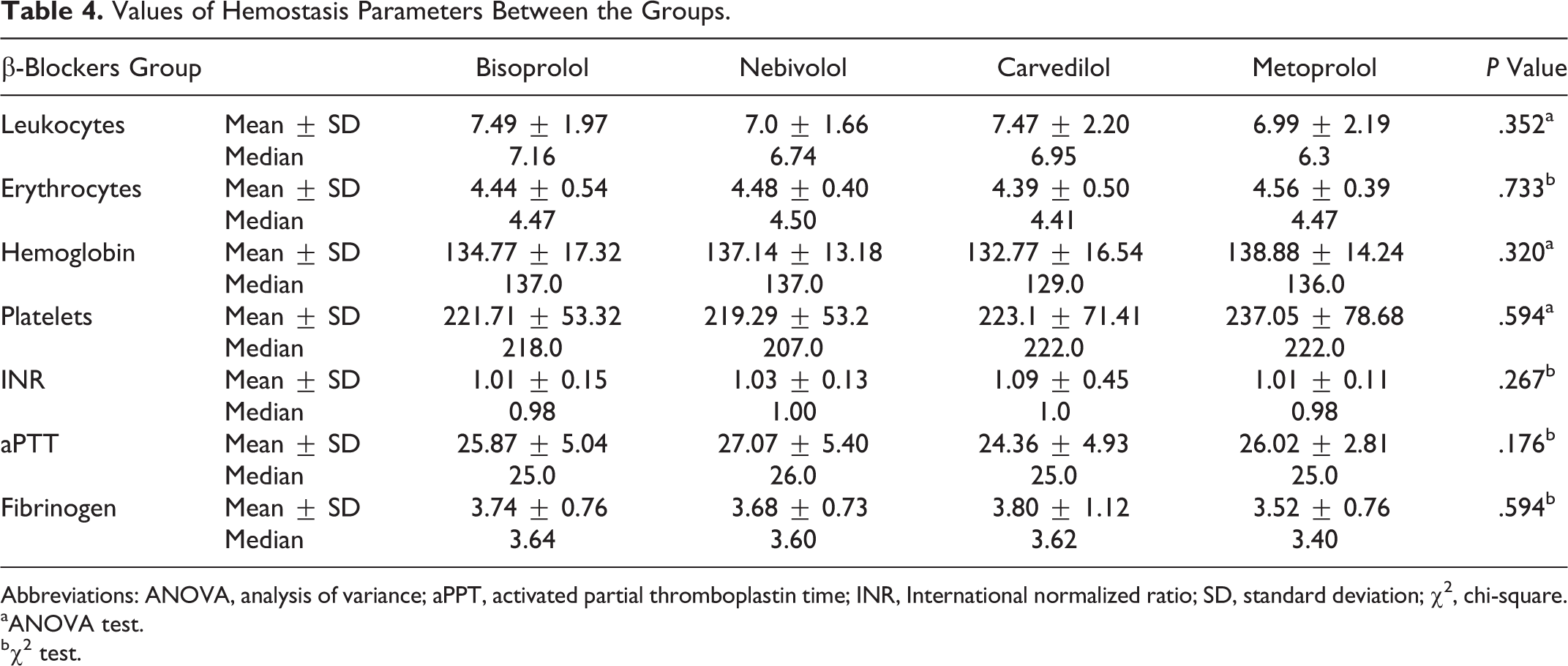
Abbreviations: ANOVA, analysis of variance; aPPT, activated partial thromboplastin time; INR, International normalized ratio; SD, standard deviation; χ2, chi-square.
aANOVA test.
bχ2 test.
Table 5 shows the mean, standard deviation, and median values for ADP, ASPI, and TRAP test and the ratio of ADP/TRAP and ASPI/TRAP between the test groups.
Differences in Platelet Aggregability Values Among the Groups.
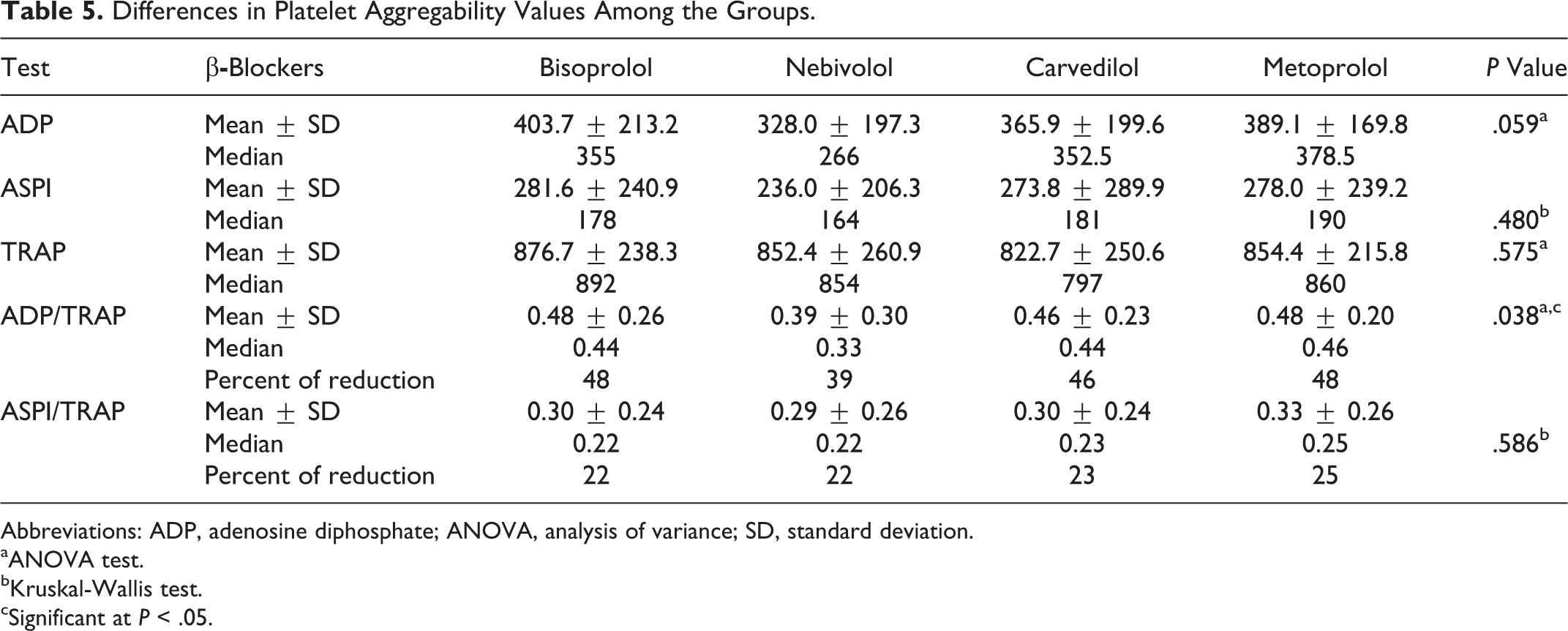
Abbreviations: ADP, adenosine diphosphate; ANOVA, analysis of variance; SD, standard deviation.
aANOVA test.
bKruskal-Wallis test.
cSignificant at P < .05.
The variables ADP, TRAP, and ADP/TRAP had normal distribution, so we used ANOVA in order to analyze the differences between 4 different β-blocker groups. For all variables, ADP, TRAP, and ADP/TRAP, Levene test indicated homogeneity of variances.
The ANOVA of the variable ADP indicated that differences between the 4 compared groups were not significant (F 3, 327 = 2.502, P = .059; Figure 1A). Because of the borderline level of significance, we have decided to make contrast analysis between the group with lowest ADP mean value, in contrast to all other groups combined, and, in second step, to compare it with the group with highest ADP mean value. Contrast analysis indicated significantly lower mean in nebivolol group, than rest of the 3 groups combined (t 327 = 2121, P = .035), and significantly lower mean, compared to bisoprolol group (t 327 = 2.679, P = .008), which could indicate that, despite nonsignificant finding in ANOVA test, significant difference between nebivolol and bisoprolol group could be present.

A, Distribution of values of ADP test between the groups. B, Distribution of values of ASPI test between the groups. C, Distribution of values of ADP/TRAP test between the groups. D, Distribution of values of ASPI/TRAP test between the groups. ADP indicates adenosine diphosphate.
The variable ASPI did not have normal distribution, so we performed Kruskal-Wallis test, which indicated there were no significant differences between the 4 different β-blocker groups (Figure 1B). The ANOVA of the variable TRAP did not find significant differences between the 4 compared groups.
There was significant difference in the ANOVA test of the variable ADP/TRAP between the compared groups for the 3 conditions (F 3,327 = 2.846, P = .038; Figure 1C). In order to identify between which groups the difference was significant, and to decrease the probability of type 1 error in multiple comparisons, we performed Bonferroni post hoc analysis, which indicated that the mean value of the ADP/TRAP in nebivolol group (0.39 ± 0.30) was significantly lower than the mean value of the ADP/TRAP in bisoprolol group (0.48 ± 0.26). Post hoc contrast analysis indicated that nebivolol group had significantly lower mean of the ADP/TRAP value, than the rest of the 3 groups combined (t 327 = 2,612, P = .009), and also significantly lower compared to bisoprolol group only (t 327 = 2.848, P = .005).
The variables ASPI/TRAP did not show normal distribution, so we performed Kruskal-Wallis test, which indicated there were no significant differences between the 4 different β-blocker groups (Figure 1D).
Discussion
Numerous studies have demonstrated a modulation of platelet function and increased ADP-induced platelet aggregability by circulating catecholamines. 13,14 Some authors have found increased ADP and thromboxane-induced platelet aggregation in conditions where the catecholamine level is elevated, such as in acute myocardial infarction 15 during early morning hours 16 or during physical 17 or mental stress, 18 although this was disputed by others. 19,20
On the surface of the platelets prevail adrenoceptors of α2A subtype and through their stimulation, epinephrine and norepinephrine potentiate the effects of other agonists and at higher concentrations, initiate platelet responses. Human platelets possess, in addition to α2 adrenoceptors, β2 adrenoceptors that could be of interest as potential target for β-blockers. 21
Beres et al examined influence of adrenergic system on the platelet function in patients who had angina pectoris and were on dual antiplatelet therapy (ASA, clopidogrel). Patients who had elevated adrenergic system activity (measured after administration of low doses of adrenaline and selective α2A adrenergic receptor inhibitor atipamezol) had higher levels of ADP and collagen-induced platelet aggregation and weaker inhibitory effect of thienopyridines measured with light transmission aggregometry (LTA). 22
We believe that in a state of elevated concentration of catecholamines use of β-blockers prevents excessive platelet activation by catecholamines. The effect of β-blockers on platelets should not be limited only to the antagonistic effect of catecholamines. It is known that certain drugs from the group of β-blockers have membrane-stabilizing effect as well as other various additional effects: blockade of α adrenoceptors and stimulation of NO production from the endothelium.
Antiplatelet activity of nonselective β-blocker, propranolol, has been reported in several trials in the 1980s and 1990s. 10,23 In the trial of Campbell et al, 23 treatment of patients with hypertension having high-dose propranolol, significantly inhibited thromboxane synthesis by platelets and platelet aggregation induced by thrombin or arachidonic acid. In vitro study of propranolol effect by Weksler et al 10 on platelet aggregation showed that propranolol inhibited ADP, epinephrine, collagen, and thrombin-induced platelet activation. This effect of the propranolol is explained by membrane-stabilizing effect, affecting Ca availability within the platelets and not by its β-blocking effect.
Several smaller studies did not show antiplatelet effect of β 1 selective blockers metoprolol and atenolol. 24 –26 The Scandinavian group of authors 24 examined the antiplatelet effect of metoprolol on platelet aggregation induced by ADP in patients who had myocardial infarction, 4 weeks before study entry. The researchers found no difference in ADP-induced platelet aggregation in 30 patients who took metoprolol compared to 33 patients who were on placebo. Some authors demonstrated more pronounced antiaggregatory effect of propranolol in comparison to metoprolol 25 and atenolol. 26
The antiaggregatory effect of nonselective β-blocker carvedilol, which is also α1 antagonist, has also been reported. Beside this, α adrenoceptor antagonizing effect, this β-blocker has membrane-stabilizing effect. Petrikova and colleagues examined the antiplatelet effect of carvedilol on platelet aggregation induced by thrombin, epinephrine, and ADP measured by LTA, as well as the production of thromboxane B2, measured by radioimmunoassay. These results were then compared with the antiplatelet effect of atenolol and propranolol. Carvedilol exerted a pronounced antiplatelet effect in reducing platelet activation and the formation of thromboxane B2 by propranolol, whereas atenolol did not show antiaggregational properties. 10 Antiplatelet effect of carvedilol is explained by the effect of the macromolecules of the cell membrane (phospholipids, ion channels, and enzymes). 27
Zakirova and colleagues compared the effect of carvedilol and metoprolol on platelet aggregation in patients after acute ST-segment elevation myocardial infarction using LTA. The carvedilol group consisted of 42 patients and the metoprolol group of 44 patients. After 4 weeks follow-up, carvedilol group had lower ADP and collagen-induced platelet aggregation compared to the metoprolol group. Researchers also noted a smaller mean platelet volume (MPV) in the carvedilol group, which is one of the predictors of outcome after myocardial infarction. 28
The most recent meta-analysis, which included 31 studies about antiplatelet effect of β-blockers, pointed out that clinically used doses of β-blockers reduce platelet aggregation, and this effect was more pronounced in nonselective, lipophilic β-blockers. 9
Nebivolol is a novel, highly selective β-blocker with additional vasodilatatory hemodynamic effects by acting on the
Celik and colleagues compared antiplatelet effect of nebivolol and metoprolol in 72 patients measuring the MPV, concentrations of P-selectin. After 6 months of follow-up, nebivolol group had a significantly lower MPV and P-selectin blood level, compared to the metoprolol group. 29
In examination of antiaggregatory effect of nebivolol and atenolol in Zucker-diabetic rats, authors have noted significant reduction in platelet aggregation and oxidative stress parameters in nebivolol group. Additionally, evaluation of expression of plasminogen activator inhibitor and cellular molecules such as vascular cell adhesion molecule 1 and platelet endothelial cell adhesion molecule 1 in the vascular wall of diabetic rats treated with nebivololol during 6 months showed favorable modification of these two prothrombotic markers compared to the control group with atenolol. 30
Group of Russian researchers examined effect of nebivolol on the microcirculation, blood viscosity, spontaneous, and ADP-induced platelet aggregation in 30 patients with moderate hypertension using laser-Doppler flowmetry. After 3 months of treatment, patients had significantly reduced spontaneous and ADP-induced platelet aggregation. 31
Antiplatelet effect of nebivol can be explained by the influence on the NO (
Reduced production of NO is one of the main causes of endothelial dysfunction, which is closely associated with development of atherosclerosis. Stimulation of β adrenergic receptors stimulates endothelial NO production, under the influence of eNOS, but paradoxically excessive stimulation of this mechanism inhibits the synthesis of NO by creation of excessive reactive oxygen species (ROS, such as superoxide anion). 34 Excessive activation of the sympathetic system and increased concentrations of catecholamines are one of the main factors for the development of endothelial dysfunction due to blockade of NO, increased production superoxide anion, and synthesis of inflammatory mediators. 35,36
In addition, NO plays a key role in maintenance of the normal function of the vascular endothelium. It reduces adhesion and aggregation of the platelets, inhibits thrombus formation, adhesion of monocytes to the vascular endothelium, and smooth muscle cell proliferation. 37 NO is also part of the protective mechanism by limiting inflammation of the endothelium and the production of tissue factor. 38
Antioxidant properties of nebivolol have been demonstrated in the laboratory and clinical trials. Through interaction with ROS, 39 nebivolol reduced concentration of peroxynitrite. 40 In two studies authors noted reduced NO inactivation by ROS in the group of patients with nebivolol, compared to the control group in patients with hypertension. 41,42 In experimental models of hypertension, hypertension of angiotensin II-treated rats, spontaneously hypertensive rats, and rats with transgenic overexpression of renin, nebivolol inhibited nicotinamide adenine dinucleotide phosphate oxidase and reduced oxidative stress. 43 –45
Induction of NO synthesis and neutralizing ROS together with inhibiton of β adrenergic receptors synergistically contribute to the preservation of endothelial function after administration of nebivolol and may explain more pronounced atiaggregatory effect of nebivolol in comparison to bisoprolol in our trial.
Process of platelet activation aggregation is influenced by numerous factors. During data collection and analysis, we considered all demographic, clinical, and laboratory factors that could affect aggregation test, but did not find significant difference in their distribution between the groups, with exception of hypertension, which was lowest in metoprolol (79.5%) and highest in bisoprolol and carvedilol group (93% and 94.4%, respectively). Values of laboratory parameters including hemostasis parameters and inflammation markers did not significantly differ between the compared groups. In our study, we observed different levels of reduction in ADP-induced platelet aggregation compared to baseline values (ADP/TRAP) and trend toward lower ADP-induced platelet aggregation between the groups of patients on dual antiplatelet therapy who used nebivolol and bisoprolol, which cannot be attributed to other factors than individual properties of those different β-blockers.
Metoprolol and carvedilol group had a similar level of ADP and arachidonate-induced platelet aggregation, as bisoprolol group, which indicate their similar effect on the platelet aggregation. Despite this, we didn’t find significant difference in ADP test value nor in the relationship ADP/TRAP test had between metoprolol and carvedilol groups, in comparison with nebivolol group. This could be explained by the smaller number of patients in those 2 groups (54 patient in carvedilol, 44 in metoprolol group) in comparison with bisoprolol group (157 patient), so observed differences in the values of aggregation tests could not manifest.
We acknowledge that smaller number of patients in the metoprolol and carvedilol groups as well as unequal group sizes among the groups could be one of the limitations of the of study. We can also assume that some of the observed differences in the values of aggregation test (especially regarding ADP test) could not manifest due to the smaller number of patients included in our study, so further research with larger number of patients is needed.
Conclusion
In patients with verified CAD on dual antiplatelet therapy we found significantly lower ADP-induced residual platelet aggregation compared to baseline values in the group of patients who used the nebivolol compared to the group of patients who used the bisoprolol.
There was no significant difference in neither ADP, nor arachidonic acid-induced platelet aggregation between the groups of patients who used different types of β-blockers (bisoprolol, nebivolol, carvedilol, and metoprolol). We have noticed a trend toward slower ADP-induced platelet aggregation in a group of patients who used the nebivolol in comparison to group who used bisoprolol. Therefore, we may conclude that nebivolol could be a better choice of β-blocker therapy, in patients with CAD, and on dual antiplatelet therapy, compared with other β-blockers (bisoprolol, carvedilol, and metoprolol).
Footnotes
Author Contribution
V. Ignjatovic, S. Pavlovic, and D. V. Ignjatovic contributed to conception or design; acquisition, analysis, or interpretation; drafted the article; critically revised the article; gave final approval; and agree to be accountable for all aspects of work ensuring integrity and accuracy. V. Miloradovic, G. Davidovic, P. Djurdjevic, and V. Iric-Cupic contributed to conception or design; acquisition, analysis, or interpretation; and gave final approval. N. Andjelkovic, R. Stolic, N. Petrovic, Z. Smiljanic, D. Jovanovic, and J. Nesic contributed to acquisition, analysis, or interpretation and gave final approval. I. Simic, V. Zdravkovic, and S. Simovic critically revised the article and gave final approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
