Abstract
Purpose:
The impact of antiplatelet therapy with availability of CYP2C19 genotyping on bleeding in a real-world setting has not been extensively studied.
Methods:
Prospective, single-center, cohort study conducted between December 2015 and October 2019 with 1-year follow-up. Patients underwent percutaneous coronary intervention (PCI), CYP2C19 genotyping, and received P2Y12 inhibitor therapy. The primary outcome was time to first bleed of any severity using Bleeding Academic Research Consortium criteria. Secondary outcomes included time to first major bleed and rates of antiplatelet switching.
Results:
The primary outcome occurred in 697 of 2091 (33%) participants at a median of 15 days. Major bleeding occurred in 176 (8%) of patients. Compared to clopidogrel, treatment with ticagrelor or prasugrel was associated with increased risk of any bleeding (adjusted HR [aHR] 2.04, 95% CI 1.69-2.46). For patients without CYP2C19 no function alleles, treatment with prasugrel or ticagrelor was associated with increased risk of any bleeding (aHR 2.31, 95% CI 1.83-2.90). Similar associations were observed for major bleeding. No difference in ischemic events was observed. Among patients discharged on ticagrelor or prasugrel, 199 (36%) were de-escalated to clopidogrel within 1 year. De-escalation was more likely after a bleed if patients did not have a no function allele (35.9% vs 19.1%; P = .02).
Conclusion:
Bleeding is common in post-PCI patients on antiplatelet therapy. Patients on high potency agents had higher bleeding risk in the population at-large and in non-carriers of CYP2C19 no function alleles. Genotype-guided antiplatelet de-escalation should be further explored in prospective studies.
Keywords
Introduction
Clopidogrel remains the most commonly prescribed P2Y12 inhibitor, despite interindividual variability leading to high on-treatment platelet reactivity for some patients. 1,2 Several factors contribute to this risk, notably genetic polymorphisms in the hepatic cytochrome P450 2C19 (CYP2C19) enzyme. 3 Clopidogrel is a pro-drug that requires conversion by the liver primarily through the polymorphic CYP2C19 enzyme to an active metabolite. 4 Prospective studies have demonstrated that patients carrying a CYP2C19 no function (NF) allele who received alternative therapy (ALT; ticagrelor or prasugrel) had reduced rates of major adverse cardiovascular events (MACE) compared to those who received clopidogrel. 5,6 However, these agents are associated with an increased risk of bleeding. 7,8 Bleeding itself is associated with worse clinical outcomes, including mortality, thrombotic complications, and antiplatelet therapy non-adherence. 9 -13
Studies of guided antiplatelet therapy with genetic testing have been recently conducted and primarily addressed escalation from clopidogrel to ALT therapy. 5,6,14,15 In those with NF alleles, ischemic benefit was apparent with use of ALT treatment versus clopidogrel. However, evidence is more limited regarding de-escalation (ALT to clopidogrel) in those without NF alleles (“normal function”) and impact on bleeding reduction. 14,16 Therefore, the primary aim of this study was to evaluate the impact of antiplatelet therapy with availability of genetic testing on the incidence of any bleeding between ALT and clopidogrel using the Bleeding Academic Research Consortium (BARC) definition. 13 The secondary aims were to evaluate major bleeding events (BARC 3 or 5) between groups, and to describe the relationship between BARC bleeding, CYP2C19 metabolizer status, and changes in P2Y12 inhibitor therapy post-percutaneous coronary intervention (PCI).
Methods
Study Design and Participants
Consecutive adult patients who underwent PCI between December 2015 and October 2019 were evaluated in this prospective, single center, observational cohort study with 1 year follow-up. Included patients underwent intracoronary stenting and had a CYP2C19 genotype test result reported in the electronic health record (EHR). Patients were excluded from the study if they received liver or allogeneic stem cell transplant before or during the study period, or if the antiplatelet treatment at the time of the primary outcome was unknown. We conducted this study as part of our PreCISE-Rx (Pharmacogenomics-guided Care to Improve the Safety and Effectiveness of Medications) program. The study complied with the principles of the Declaration of Helsinki and was approved by the institutional review board of the University of Pittsburgh with waiver of informed consent (study 19060266).
Pharmacogenomics Service
The hospital’s PCI order set includes CYP2C19 genotype by default to assist with antiplatelet medication selection. The assay (GenMark Diagnostics, Carlsbad, CA) detects CYP2C19 *2, *3, *4, *5, *6, *7, *8, and *17 and returns the diplotype and the interpretation as a genetically predicted phenotype (e.g., CYP2C19 poor metabolizer) to the electronic health record. A clinical pharmacist provides a consultation regarding antiplatelet selection that considers both genetic and non-genetic patient-specific factors. In general, ALT therapy is recommended for patients with a NF allele in accordance with the Clinical Pharmacogenetics Implementation Consortium guidelines. 4 The ultimate prescribing decision, however, was left to prescriber discretion.
Data Collection
Study data were collected retrospectively and managed using a secure, web-based software that supports data capture for studies with audit trails for data tracking, automated export procedures to other programs, and interoperability access with external sources [Research Electronic Data Capture (REDCap®)]. 17,18 A trained research nurse collected baseline and follow-up data for each patient, with a goal of 365 days of post-PCI follow-up for each patient.
Patient data collected at baseline included: demographic, genetic, clinical, procedural, and laboratory variables. We recorded the P2Y12 inhibitor used (if any) prior to admission, during PCI, at discharge, and during every clinic or inpatient hospital visits during the follow-up period. Follow-up visits were defined as inpatient or outpatient encounters at which the patient was evaluated by a medical professional, and thus could report clinical outcomes (bleeding) and medication changes.
Clinical Assessment
The primary outcome of our study was time to first bleed of any severity within 1 year of the index PCI. Time to first major bleed, defined by BARC 3a, 3b, 3c, or 5 criteria, was a secondary outcome. BARC 1 includes non-actionable bleeding that does not cause the patient to seek medical care for the bleed. BARC 2 is actionable bleeding that causes non-surgical intervention, hospitalization, or further evaluation. BARC 3a, 3b, 3c and 5 encompassed major bleeding. BARC 3a includes a hemoglobin drop between 3 and <5 g/dL or need for transfusion. BARC 3b includes any surgical intervention to control bleeding, use of vasoactive agents, or a hemoglobin drop of >5 g/dL. BARC 3c includes intracranial hemorrhage or intraocular bleed compromising vision. BARC 5 is death due to bleeding. BARC 4, coronary artery bypass graft-related bleeding, was excluded from our study analysis. 13 Additionally, bleeding events secondary to trauma were excluded from our analysis. Other secondary outcomes included treatment switches (from ALT to clopidogrel and vice-versa) and discontinuation within 1 year of the index PCI.
For bleeding outcomes, patients were censored when lost to follow-up or if they discontinued P2Y12 inhibitor treatment. Treatment assignment was based on P2Y12 inhibitor use on the event or censor date. CYP2C19 genotypes were grouped into patients who were NF carriers (CYP2C19 poor or intermediate metabolizers) and those who were not NF carriers (CYP2C19 normal, rapid, and ultra-rapid metabolizers).
We examined MACE as a secondary outcome. MACE was a composite endpoint consisting of all-cause mortality, myocardial infarction, or ischemic stroke. Treatment was assigned as described in the primary analysis.
Statistical Analysis
Descriptive statistics were used to summarize baseline characteristics, length-of-stay, and length of follow-up. Univariate analyses of associations of baseline variables and the primary outcome were performed using Chi-square tests and unpaired t tests. A Kaplan-Meier failure function and multivariable Cox proportional hazards model were used for unadjusted and covariate-adjusted time-to-event analyses, respectively. Covariates considered for the Cox model included: age, race, sex, ACS presentation at index PCI, history of gastrointestinal (GI) bleeding, concomitant anticoagulation therapy prior to index PCI, serum creatinine >1 mg/dL, thrombocytopenia (defined as platelet <100 × 109/L), and anemia (defined as hemoglobin <11 g/dL) at baseline. Stepwise forward and backward regression were performed to determine covariates in the multivariable model, using the occurrence of any BARC bleeding event as the dependent variable with a threshold of P < .05. A log-rank test was used to test equality of survivor functions between treatment groups in unadjusted analyses. A univariate Cox regression was used to generate unadjusted hazard ratios (HRs). Adjusted hazard ratios (aHRs) were reported for differences between treatment groups in the multivariable Cox proportional hazards model. Differences in discontinuation rates were compared using Chi-square or Fisher exact tests, as appropriate. Analyses were performed with Stata software, version 17.0 (StataCorp LLC).
Results
Characteristics of the Study Population
A total of 2091 patients were included in the study (Figure 1). Patient demographics, baseline clinical variables, and medication use are displayed in Table 1. Most of the cohort was represented by middle-age Caucasian males. The majority of patients presented with ACS and received drug-eluting stents. The median length-of-stay was 1 day (interquartile range [IQR] 1-3 days). The median length of follow-up was 365 days (IQR 207-365 days).

Study flowchart. PCI, percutaneous coronary intervention; CYP2C19, cytochrome P450 hepatic 2C19 isoenzyme.
Baseline Demographic and Clinical Characteristics.a

Abbreviations: CAD, coronary artery disease; GI, gastrointestinal; NSTE-ACS, non-ST-segment elevation myocardial infarction acute coronary syndrome; PCI, percutaneous coronary intervention; STEMI, ST-segment-elevation myocardial infarction; TIA, transient ischemic attack.
a SI conversion factors: To convert serum creatinine to milligrams per liter, multiply by 10; hemoglobin to grams per liter, multiply by 10.
Antiplatelet Medication Use at Discharge and Genotype
Following the index PCI, 1446 patients (72%) were discharged on clopidogrel, 520 (26%) on ticagrelor, and 41 (2%) on prasugrel. A total of 605 patients (29%) had a NF allele. ALT was prescribed as the discharge P2Y12 inhibitor in 192 patients with NF alleles (33%) and 369 patients without NF alleles (26%) at discharge (P = .001).
Bleeding and Ischemic Outcomes
The primary outcome of bleeding of any severity occurred in 697 patients (33%). Among patients with a bleeding event, the severity of the first bleeding event was BARC 1 for 313 patients (45%), BARC 2 for 249 (36%), BARC 3 for 128 (18%), and unknown for 7 patients (1%). No patients had BARC 5 as their first bleeding event. The median time to first bleed of any severity was 15 days (IQR 1-100 days). Major bleeding (BARC 3 or 5) occurred in 176 patients (8%). The median time to first major bleed was 17 days (IQR 1-121 days).
Stepwise forward and backward regression generated identical Cox proportional hazards model of covariates: age (HR 1.01, 95% CI 1.01-1.02 per year), anticoagulation at admission (HR 1.43, 95% CI 1.15-1.78), anemia (HR 1.74, 95% CI 1.44-2.11), and history of GI bleed (HR 1.42, 95% CI 1.04-1.94). Results of the crude and adjusted survival analyses are listed in Table 2. ALT therapy was associated with greater risk of any bleeding (Figure 2A) and major bleeding (Figure 3A). NF allele carrier status was not associated with risk of any bleeding or major bleeding. Among patients treated with clopidogrel, NF allele carrier status was not associated with risk of bleed of any severity or major bleeding. In patients carrying NF alleles, ALT therapy was associated with increased risk of bleed of any severity and major bleeding in adjusted analyses. In patients without NF alleles, ALT therapy was associated with increased risk of bleed of any severity (Figure 2B) and major bleeding (Figure 3B).
Crude and Adjusted Analyses of Bleeding Outcomes.a

Abbreviations: ALT, alternative treatment; HR, hazard ratio; aHR, adjusted hazard ratio; NF, no function allele carriers; non-NF, patients who did not carry no function alleles (CYP2C19 normal, rapid, and ultrarapid metabolizers).
a Statistically significant associations are italicized.
b Univariate Cox regression.
c Cox regression adjusted for age, anticoagulation, anemia, and history of gastrointestinal bleed.
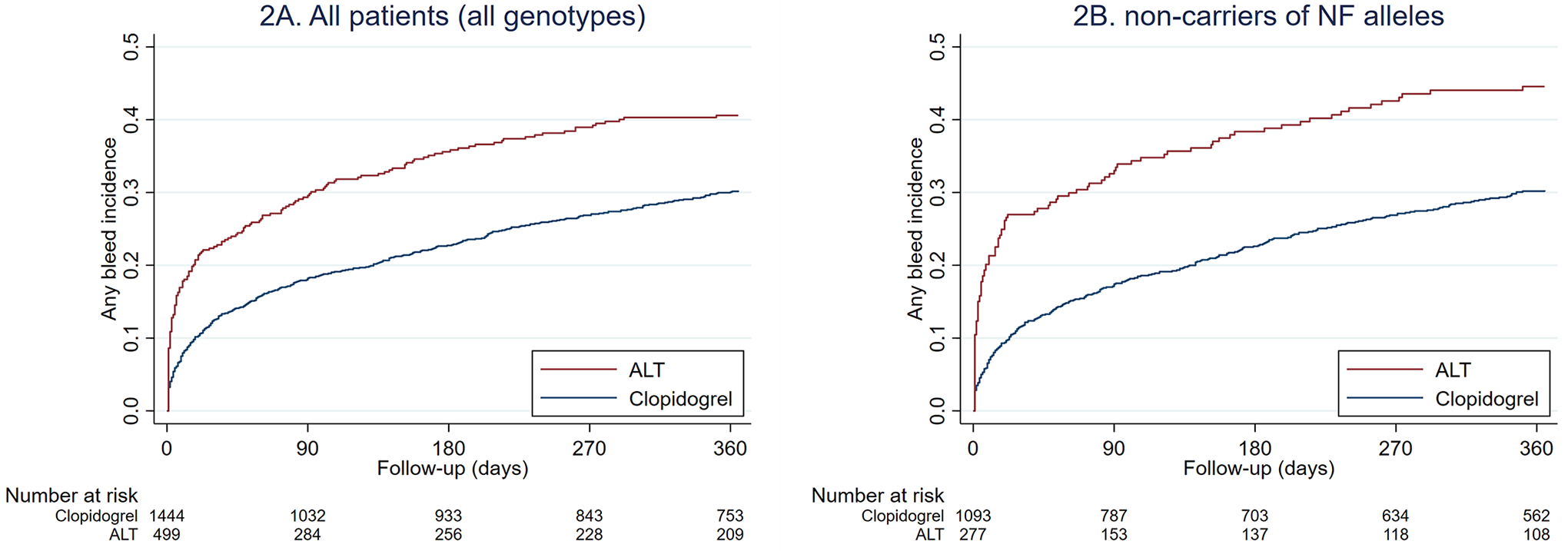
Kaplan-Meier failure function for bleed of any severity. ALT, alternative therapy (prasugrel or ticagrelor); NF, no function allele.

Kaplan-Meier failure function for major bleeding events. ALT, alternative therapy (prasugrel or ticagrelor); NF, no function allele.
Two-hundred and thirty patients died within 1 year of the index PCI (11%). MACE occurred in 367/2079 patients during follow-up (17.7%). In patients without NF alleles, 1-year MACE incidence did not significantly differ by treatment (clopidogrel: 198/1161 [17.1%] vs ALT: 65/317 [20.5%], P = .16).
Antiplatelet Medication Use at Follow-Up
Of 1533 patients who had a full year of follow-up, 1122 (73.2%) were on uninterrupted DAPT for at least 1 year. Of patients who survived to discharge, 284/2013 (14.1%) were on triple therapy (DAPT plus any anticoagulant). In these patients, the median duration of uninterrupted triple therapy was 44.5 days (IQR 28-115 days). In the overall study population, P2Y12 inhibitor treatment was discontinued before 1 year in 215 (10.3%) of patients. Patients who had a bleed of any severity were more likely to discontinue within 1 year than patients without any bleeding event (15.8% vs 7.53%, P < .001). This was also true of patients with a major bleeding event (23.3% vs 9.1%, P < .001).
Among 561 patients discharged on ALT therapy, 199 (36%) were de-escalated to clopidogrel within 1 year of follow-up. Conversely, 102 (7%) of the 1446 patients discharged on clopidogrel were switched to ALT therapy within 1 year. Patients who had bleeding of any severity were more likely to de-escalate compared to those without bleeding (42% vs 32%, P = .02). De-escalation was numerically more common in patients with major bleeds compared to those without major bleeding events, however this was not statistically significant (41% vs 35%, P = .45). Prescribers were more likely to de-escalate to clopidogrel for patients with a bleed of any severity if patients did not have NF alleles (36% vs 19%, P = .02). Similarly, prescribers were numerically more likely to de-escalate to clopidogrel for patients with a major bleed if the patient did not have NF alleles, although this finding was not statistically significant (44% vs 14%, P = .08).
Discussion
Our study is among the largest real-world cohorts of post-PCI patients on antiplatelet therapy with availability of genetic testing that focused on bleeding outcomes. Our methods allowed us to collect bleeding events more discretely and to categorize them based on the BARC definition. Ultimately, this led to a more granular depiction of bleeding events that would not have been captured with traditional bleed definitions such as Thrombolysis in Myocardial Infarction (TIMI) and Global Use of Strategies to Open Occluded Coronary Arteries (GUSTO). 13 We were also able to capture BARC 1 bleeds, which have not been previously reported in this type of study. Most previous studies relied upon TIMI and/or GUSTO criteria or BARC 2-5 definitions, thus missing more minor or “nuisance” bleeds that still may carry prognostic importance. 5,13 -16
In our study, we found that 33% of patients had any BARC bleeding within the first year of the index PCI. This rate exceeds that of other recent studies that evaluated pharmacogenomics guidance and was driven by BARC 1 and 2 bleeds. For comparison, the POPular Genetics study used PLATO major or minor bleeding as the primary safety outcome and reported rates of approximately 10% to 12%, whereas the BARC 2 to 5 rates were 10% to 13%, and BARC ≥3 rates were 2%. 14 In contrast, major bleeding (BARC ≥ 3) in our study was seen in 8% of patients. The TAILOR-PCI trial reported TIMI major or minor bleeding as the primary safety outcome, showing rates of approximately 2% to 3%, BARC 2, 3, or 5 rates of 2% to 3%, and BARC 3 or 5 rates of 2%. 15 Only the PHARMCLO trial used the BARC definition as the primary safety endpoint (BARC 3-5) and reported rates of approximately 4% to 7%. However, this study was prematurely terminated for technical reasons related to use of the study instrument. A retrospective single-center study found rates of clinically significant bleeding, defined by the GUSTO criteria, to be approximately 2% to 7% depending on P2Y12 inhibitor prescribed and whether patients were escalated or de-escalated. 16 Potential reasons for the higher bleed risk seen in our study may relate to having an older demographic with more co-morbidities, and a higher use of concomitant oral anticoagulation compared to prospective, randomized trials. However, the inclusion of BARC 1 bleeding events in our event definition is likely what contributed most to the higher prevalence of any bleeding. Furthermore, bleeding events in our study occurred at a median of 15 days post-PCI, which is earlier than other published studies and may also relate to the inclusion of all BARC bleeds. A recent study of post-discharge GUSTO bleeding after ACS (PCI or no PCI) showed that bleeding occurred at a median of 50 days after the index event. Furthermore, bleeding was associated with increased mortality and had a similar prognostic impact as post-discharge myocardial infarction. 12
Patients in our study who were prescribed ticagrelor or prasugrel had a significantly higher risk of any BARC bleeding (2-fold) and major bleeding (2.7-fold) compared to those on clopidogrel. This finding was independent of CYP2C19 metabolizer status, and consistent with previous randomized controlled trials. 7,8 Our findings are in contrast to those of a recent meta-analysis that showed no difference in bleeding between ALT and clopidogrel (regardless of genotype) for bleeding, however, a definition of TIMI major or minor bleeding was used. 19
Clopidogrel was the mostly commonly prescribed P2Y12 inhibitor in our study (72%). Real-world evidence shows that clopidogrel remains the most widely prescribed P2Y12 inhibitor after PCI by accounting for 44% to 72% of use. 20 Other estimates in the ACS setting show that clopidogrel is used in 51% to 70% of patients. 21 Evidence for lower use of prasugrel or ticagrelor has been reported in observational studies for reasons that relate to adverse effects (i.e., bleeding) and cost. 20,22 Despite clopidogrel being a mainstay of antiplatelet therapy, response is dependent on metabolic conversion by the polymorphic CYP2C19 enzyme to an active metabolite. In our study, 29% of patients had a NF allele (i.e., *2-*8).These alleles result in lower active metabolite concentrations, less inhibition of platelets and decreased efficacy compared to those without a NF allele. 4,23 Studies have demonstrated improved outcomes for patients with NF alleles who were prescribed ALT therapy versus clopidogrel. 5,6,14,24 Furthermore, a recent meta-analysis of PCI studies demonstrated that the effect of prasugrel or ticagrelor versus clopidogrel in reducing MACE is based primarily on the presence of CYP2C19 NF carrier status. 19 In our study, ticagrelor or prasugrel was prescribed in 33% and 26% of patients with and without a NF allele, respectively. Therefore, opportunities remain to further optimize antiplatelet use with genetic guidance.
While P2Y12 inhibitor switching is relatively common and often done for pragmatic reasons, such as bleeding, it is frequently accomplished in an empiric manner (without platelet function testing or genotyping). 25 -27 In our study, 36% of ticagrelor or prasugrel users were switched to clopidogrel within 1 year of follow-up. Those who had any bleeding were more likely to de-escalate versus those without (42% vs 32%). Conversely, escalation was far less common (7% discharged on clopidogrel were switched to ALT therapy). Use of CYP2C19 genotyping with clinical factors appears to provide utility as a de-escalation strategy to lower bleeding risk, particularly for those patients unable to remain on higher potency therapy (due to adverse effects or cost barriers). 2 Previous studies have showed that use of genotyping to guide de-escalation to clopidogrel in patients without a CYP2C19 NF allele was safe and effective. 14,16 A recent meta-analysis of guided (genetic or platelet function testing) versus standard antiplatelet therapy post-PCI showed that the de-escalation subgroup was associated with less overall bleeding, without any trade-off in efficacy, with guided therapy. 28 In our study, patients who received higher potency therapy despite not having a NF allele had an approximately two-fold higher risk of bleeding versus those on clopidogrel. This was the case in about 1 in 4 patients in our study, who would have potentially been appropriate for de-escalation based on their CYP2C19 genotype. Similar to other studies, we found no difference in MACE outcomes between prasugrel/ticagrelor and clopidogrel-treated patients in the non-carriers of the NF allele. This finding further strengthens the concept of genotype-guided de-escalation as a potential bleeding mitigation strategy. 5,6,14,19 Therefore, opportunities exist to further implement this strategy to mitigate bleeding risk in patients who do not carry a CYP2C19 NF allele and are on higher potency P2Y12 inhibitor treatment. The value of pharmacogenomic data in this setting is evident and should be integrated into clinical decision-making to improve patient outcomes, such as bleeding avoidance.
The main limitations of our study are related to the observational design. Although our pharmacogenomics service provided recommendations for P2Y12 inhibitor selection following the genotype result, treatment decisions were ultimately left to the prescriber. Our data were collected retrospectively and therefore relied on documentation within the electronic health record. However, we had access to data from a large integrated health system and manually reviewed and curated the index PCI visit and all subsequent visits within the 1-year study period. We also captured several clinical variables of interest known to affect both antiplatelet treatment selection and bleeding risk (e.g., age, anticoagulation, anemia, and history of GI bleed), and accounted for these in our regression analyses. However, other confounding factors could have been unaccounted for or were unable to be collected, including medication adherence. Platelet function testing was rarely performed as part of our clinical practice (<2% of patients with these data). We also did not collect labs such as white blood cell count, and C-reactive protein (which is not routinely collected in our practice). Pharmacogenomic testing covered variants of interest for CYP2C19, which is the most validated genetic determinant of clopidogrel response. Variants in other genes of interest have been associated with clopidogrel response (i.e., ABCB1, CYP2C9), however, these have not been consistently replicated and were therefore not tested in our study. We also did not have access to all visits which may have occurred outside of our health system. Regarding our primary outcome of bleeding, we determined the time to first major bleed for all patients, with minor bleeds captured up to the occurrence of a major bleed. As such, our analysis focused on time-to-first event rather than collecting total bleeding events. It is possible, therefore, that minor bleeds that occurred after the first major bleed were not captured. However, our method of data collection allowed us to better capture the incidence and severity of all bleeding outcomes, including those categorized as BARC 1 or “nuisance” bleeds.
Conclusion
In this cohort study, bleeding defined by BARC criteria was common and typically occurred soon after PCI. Bleeding risk was influenced by antiplatelet selection and was significantly higher in patients without a NF allele who received higher potency therapy versus clopidogrel. Therefore, CYP2C19 genotyping can be an important tool to mitigate bleeding risk by identifying post-PCI patients who would be appropriate for clopidogrel.
Footnotes
Author Contributions
Conception and design: JCC, JMS, AP, AJCS, and PEE. Data acquisition: LP. Data analysis/interpretation: JCC, JMS, AP, and PEE. All authors participated in the writing, reviewing, and editing of the manuscript and approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the American Society of Health System Pharmacists; American Heart Association [17MCPRP33400175]; National Institutes of Health [UL1TR001857]; and an anonymous nonprofit organization.
