Abstract
Prostaglandin I2 (PGI2) plays an important role in the clinical treatment of pulmonary arterial hypertension (PAH). However, the administration of PGI2 involves continuous intravenous infusion using an indwelling catheter, which limits the patient’s quality of life and increases the risk of infection. We therefore investigated whether human PGI2 synthase (hPGIS) gene transfer using an adeno-associated virus (AAV) vector is still effective in a mouse model of PAH and tested for differences in the therapeutic efficacy of PAH among AAV serotypes. The PAH was induced by subjecting mice to hypoxia (10% O2). Type 1 AAV expressing hPGIS (AAV1-hPGIS) or type 2 AAV expressing hPGIS (AAV2-hPGIS) was injected into the thigh muscle of mice. Both vectors expressing hPGIS produced strong hPGIS protein expression in the mouse thigh skeletal muscles after 8 weeks of hypoxia. The administration of AAV1-hPGIS or AAV2-hPGIS also significantly inhibited the hypoxia-induced increase in right ventricular systolic pressure, the ratio of right ventricular weight to body weight (RV/BW), and the ratio of RV weight to left ventricular plus septal weight (RV/LV + S), and significantly attenuated the hypoxia-induced increase in medial wall thickness of peripheral pulmonary arteries. Furthermore, there were no significant differences in the degree of amelioration in RV systolic pressure, RV/BW, RV/LV + S, and percentage of wall thickness of peripheral pulmonary arteries between AAV1-hPGIS and AAV2-hPGIS administrations. In conclusion, we revealed that type 1 and type 2 AAV are equally effective for the treatment of PAH in a hypoxia-induced mouse model. Gene-transfer therapy using AAV expressing hPGIS is, therefore, a potential therapeutic breakthrough for PAH.
Introduction
Pulmonary arterial hypertension (PAH) is a rare but life-threatening disease characterized by progressive pulmonary hypertension and right heart failure. 1 A study in 1991 of patients with idiopathic PAH 2 reported a median survival period of 2.8 years and 5-year survival rate of 34%. The pathogenesis of PAH includes pulmonary vasoconstriction, endothelial cell proliferation, smooth muscle cell proliferation, and in situ thrombosis. 3,4 Prostacyclin is a metabolite of arachidonic acid that has vasoprotective effects including vasodilation, antiplatelet aggregation, and inhibition of smooth muscle cell proliferation. 5 –8 Thus, continuous intravenous infusion of prostaglandin I2 (PGI2, prostacyclin) represented a therapeutic breakthrough for PAH. 9 –16 However, the duration of PGI2 activity is so short that patients with PAH require continuous infusion through an indwelling central venous catheter and this eventually limits the patients’ quality of life. 17 –20 Furthermore, there is a risk of catheter infection, which could exacerbate right heart failure.
Previously, we demonstrated that human PGI2 synthase (hPGIS) gene transfer using adeno-associated virus (AAV) vector is an effective treatment of PAH in mice. 21 Among the various virus vectors, AAV is known to be safe and has already been used in humans for the treatment of hemophilia, cystic fibrosis, and Parkinson disease. 22 –24 Gene therapy for hPGIS could, therefore, be a potent method for treating PAH. Gene therapy with hPGIS using AAV in clinical settings may not require the indwelling catheter, resulting in an improved quality of life for the patient and the avoidance of catheter infection.
There are several AAV serotypes, of which type 2 AAV (AAV2) is the most common one used in AAV-based research and in clinical settings for humans. 22 –24 We also demonstrated the efficacy of hPGIS gene transfer using AAV for the treatment of PAH, although we used type 1 AAV (AAV1) because it provides very high transduction rates in murine skeletal muscle cells and our study involved the intramuscular injection of AAV into the thigh muscle of mice. 21 Thus, it is important to compare treatment efficiency between AAV1 and AAV2 for the potential clinical application of AAV-based therapy.
Therefore, the purposes of this study were (1) to investigate whether PGIS gene transfer therapy using not only AAV1 but also AAV2 is effective for the treatment of mice with PAH and (2) to compare the treatment efficiency between AAV1 and AAV2.
Methods and Materials
AAV Vectors
The AAV vectors expressing hPGIS were constructed in good laboratory practice by Dynavec Corporation (Ibaragi, Japan).
Animal Experiments
Eight-week-old male C57/BL6 mice purchased from Japan CLEA (Tokyo, Japan) were used in this study. To generate PAH model, mice were kept in a tightly sealed chamber under hypoxia (10% O2) for 8 weeks, maintained using a hypoxic air generator (TEIJIN, Tokyo, Japan) and monitored with an O2 analyzer (JIKO-255, Tokyo, Japan). All experimental procedures and protocols were approved by the Animal Care and Use Committees of Keio University.
We randomly divided mice into 4 groups and assigned mice to 1 additional control group for comparison, mice injected with 0.9% saline (vehicle) under room air (control group, n = 10), PAH mice exposed to hypoxia and injected with AAV1 expressing enhanced green fluorescent protein ([EFGP] AAV1-EGFP group, n = 8), PAH mice exposed to hypoxia and injected with AAV2 expressing EFGP (AAV2-EGFP group, n = 8), PAH mice exposed to hypoxia and treated with AAV1 expressing hPGIS (AAV1-hPGIS group, n = 10), and PAH mice exposed to hypoxia and treated with AAV2 expressing hPGIS (AAV2-hPGIS group, n = 10) (Table 1).
Physiological Profiles of 5 Experimental Groupsa
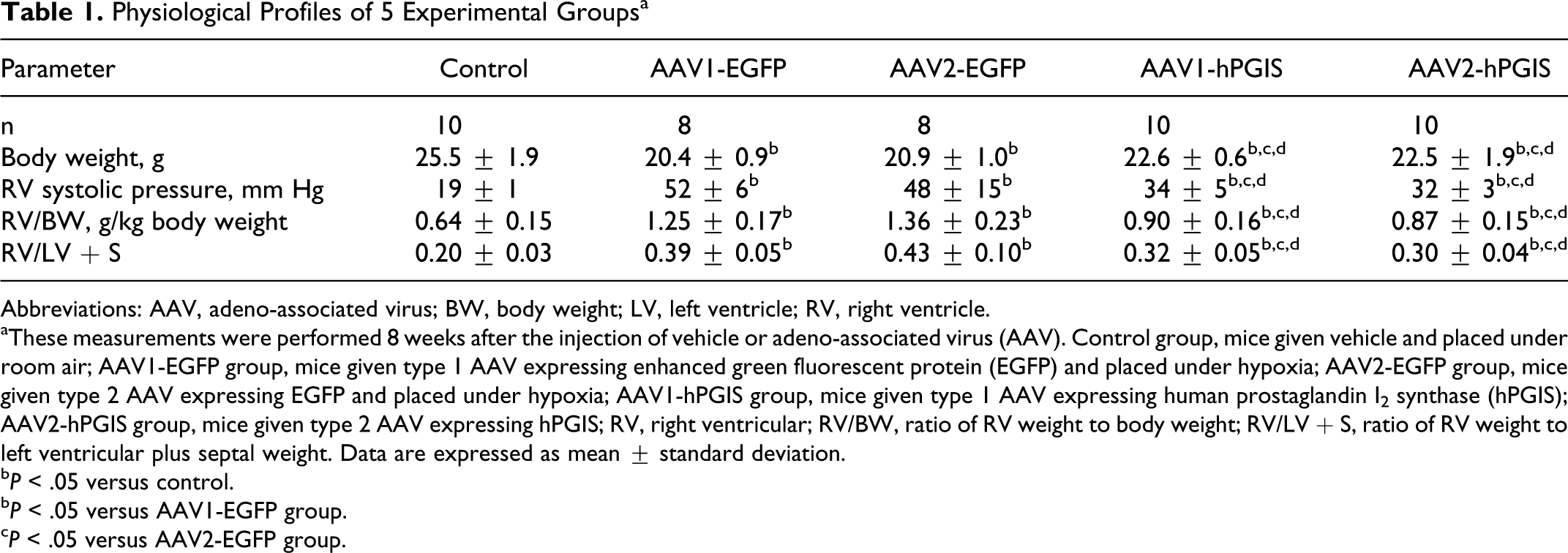
Abbreviations: AAV, adeno-associated virus; BW, body weight; LV, left ventricle; RV, right ventricle.
aThese measurements were performed 8 weeks after the injection of vehicle or adeno-associated virus (AAV). Control group, mice given vehicle and placed under room air; AAV1-EGFP group, mice given type 1 AAV expressing enhanced green fluorescent protein (EGFP) and placed under hypoxia; AAV2-EGFP group, mice given type 2 AAV expressing EGFP and placed under hypoxia; AAV1-hPGIS group, mice given type 1 AAV expressing human prostaglandin I2 synthase (hPGIS); AAV2-hPGIS group, mice given type 2 AAV expressing hPGIS; RV, right ventricular; RV/BW, ratio of RV weight to body weight; RV/LV + S, ratio of RV weight to left ventricular plus septal weight. Data are expressed as mean ± standard deviation.
b P < .05 versus control.
b P < .05 versus AAV1-EGFP group.
c P < .05 versus AAV2-EGFP group.
Experimental Protocols
After 24 hours of exposure to hypoxia, AAV1-EGFP, AAV2-EGFP, AAV1-hPGIS, or AAV2-hPGIS (1.0 × 10 10 vector genome/body) was administered into the right thigh muscle of mice by single injection. The vehicle (0.9% saline) was injected into the same site in the control group. Hemodynamics was measured at 8 weeks. To do this, light anesthesia with diethyl ether was administered. The mice were then intubated, mechanically ventilated with a Harvard respirator, and anesthetized with isoflurane. Left thoracotomy was performed and a 1.4-F microtip pressure transducer (SPR-671; Millar Instruments, Houston, Texas) was directly inserted into the right ventricle (RV), and the RV pressure was recorded using a polygraph system (Nihon Kohden, Tokyo, Japan). After hemodynamic measurements, the RV, left ventricle (LV), ventricular septum, lungs, and right thigh muscle were excised, weighed, and retained for further analysis. The ratio of RV weight to body weight (RV/BW) and the ratio of RV weight to LV plus septal weight (RV/LV + S) were calculated as indexes of ventricular hypertrophy.
Histological Analysis
Paraffin sections of 4-μm thickness were obtained from the middle region of the right lung and stained with elastic van Gieson for examination by light microscopy. The external diameter and medial wall thickness of the pulmonary arterioles were measured in 20 muscular arteries (ranging in size from 25 to 100 μm in external diameter). The medial wall thickness was expressed as follows: % wall thickness = ([medial thickness × 2]/external diameter) × 100. A lung section was obtained from individual mice in 5 groups (n = 5 each), as reported previously. 25 In addition, thigh muscle sections were stained with anti-human PGIS antibody, as described previously. 7 The percentage area of hPGIS staining in immunostaining images (original magnification × 200) was calculated using Image J software (n = 5 each).
Statistical Analysis
All data were expressed as mean ± standard deviation. Comparisons of parameters among the groups were made by 1-way analysis of variance, followed by Newman-Keul test. A value of P < .05 was considered statistically significant.
Results
The AAV-Mediated hPGIS Gene Expression In Vivo
There were no mice that died during the experiment. We performed immunofluorescent staining for hPGIS in histological sections of right thigh skeletal muscles from the AAV-injected mice. The mice administered with AAV1-hPGIS or AAV2-hPGIS showed strong hPGIS protein expression in their thigh skeletal muscles, while no signal was found in mice administered with AAV1-EGFP or AAV2-EGFP (Figure 1A). Furthermore, to compare the transduction rate of hPGIS gene, the percentage area of PGIS protein expression was calculated (Figure 1B). The PGIS protein expressions were significantly higher in mice administered with AAV1-hPGIS or AAV2-hPGIS than in mice administered with AAV1-EGFP or AAV2-EGFP. There was no significant difference in the PGIS expression between mice administered with AAV1-hPGIS and those with AAV2-hPGIS.

A, Immunohistochemical staining of human prostaglandin I2 synthase (hPGIS) in the right thigh muscle of mice injected with type 1 adeno-associated virus (AAV) expressing enhanced green fluorescent protein (EGFP) and placed under hypoxia (AAV1-EGFP group), mice injected with type 2 AAV expressing EGFP and placed under hypoxia (AAV2-EGFP group), mice given type 1 AAV expressing human prostaglandin I2 synthase (hPGIS) and placed under hypoxia (AAV1-hPGIS group), and mice given type 2 AAV expressing hPGIS and placed under hypoxia (AAV2-hPGIS group). Scale bars: 50 μm. B, The percentage area of hPGIS staining.
There was no evidence of tumor or angioma formation in muscle from any of the mice transferred with AAV1-EGFP, AAV2-EGFP, AAV1-hPGIS, or AAV2-hPGIS. Furthermore, there were no findings of inflammatory cell infiltrate and interstitial edema to suggest inflammation induced by AAV injection in any of the AAV-injected mice.
Effects of AAV-hPGIS on Pulmonary Hemodynamics and Right Ventricular Hypertrophy
Right ventricular systolic pressure was significantly increased after 8 weeks of sustained hypoxia in the AAV1-EGFP and AAV2-EGFP groups (Figure 2A). However, the increase was significantly attenuated in both the AAV1-hPGIS and AAV2-hPGIS groups. Similarly, the increase in RV/BW in the AAV1-EGFP and AAV2-EGFP groups was significantly attenuated in the AAV1-hPGIS and AAV2-hPGIS groups (Figure 2B), and the same was found for the hypoxia-induced increase in RV/LV + S (Figure 2C). There were no significant differences between the AAV1-hPGIS group and the AAV2-hPGIS group in RV systolic pressure, RV/BW, or RV/LV + S.

Effects of adeno-associated virus (AAV) expressing human prostaglandin I2 synthase (hPGIS) on right ventricular (RV) systolic pressure (A), the ratio of RV weight to body weight (RV/BW) (B), and the ratio of RV weight to left ventricular plus septal weight (RV/LV + S) (C) in mice injected with vehicle alone and placed under room air (control group), and in mice from the AAV1-enhanced green fluorescent protein (EGFP) group, AAV2-EGFP group, AAV1-hPGIS group, and AAV2-hPGIS group. Data are mean ± standard deviation (SD).
Effects of AAV-hPGIS on Vascular Remodeling
Representative photomicrographs show attenuated hypertrophy of the pulmonary vessel wall after sustained hypoxia in both the AAV1-hPGIS and AAV2-hPGIS groups compared with the AAV1-EGFP and AAV2-EGFP groups (Figure 3A). The demonstrated significant increase in percentage of wall thickness after sustained hypoxia in the AAV1-EGFP and AAV2-EGFP groups was also ameliorated in both the AAV1-hPGIS and AAV2-hPGIS groups (Figure 3B). The degree of amelioration in percentage of wall thickness was not significantly different between the AAV1-hPGIS group and the AAV2-hPGIS group.

A, Representative photomicrographs of peripheral pulmonary arteries 8 weeks after the injection of adeno-associated virus (AAV) expressing enhanced green fluorescent protein (EGFP) or AAV expressing human prostaglandin I2 synthase (hPGIS). The areas between arrowheads indicate wall thickness. Scale bars: 20 μm. B, Quantitative analysis of percentage of wall thickness in peripheral pulmonary arteries. Data are mean ± standard deviation (SD).
Discussion
In the present study, we demonstrated that (1) hPGIS is expressed in the thigh muscles of mice after a single injection of AAV-hPGIS; (2) a single injection of AAV1-hPGIS or AAV2-hPGIS ameliorated hypoxia-induced PAH; and (3) the efficacy of treatment for PAH was not significantly different between AAV1 and AAV2 administrations.
In recent years, AAV vectors have received wide attention because they are nonpathogenic and nontoxic in humans and show long-term gene expression in nondividing cells, confirming their strong efficacy, and safety for human use. In the present study, the strong gene expression of hPGIS in skeletal muscle 8 weeks after the injection of AAV-hPGIS (both AAV1-hPGIS and AAV2-hPGIS) demonstrated that a single injection is sufficient to achieve high efficiency of gene transfer in vivo. The present study also demonstrated that a single administration of AAV-hPGIS could markedly attenuate the development of hypoxia-induced PAH, as indicated by significant decreases in RV systolic pressure and RV weight. Furthermore, AAV-hPGIS also attenuated the increase in medial wall thickness of peripheral pulmonary arteries observed in PAH, thus administration of AAV-hPGIS offers an alternative approach to epoprostenol for treating severe PAH.
This study was also designed to directly compare the efficacy of AAV1-hPGIS and AAV2-hPGIS in a mouse model of PAH, considering that both AAV1 and AAV2 have documented benefits for potential therapeutic use in humans. 22 –24 Our results revealed long-lasting efficiency of gene expression in thigh muscles administered with either serotype and there was no significant difference in the PGIS protein expression between AAV1 and AAV2 vectors. Hemodynamics, RV weight, and medial wall thickness of peripheral pulmonary arteries were not significantly different between AAV1 and AAV2 vectors. These findings suggest that the efficacy of treatment for PAH would be similar between AAV1 and AAV2, although AAV2-hPGIS has the advantage that AAV2 has already been used safely in the human clinical setting. Thus, these findings suggest that AAV2 may be appropriate to use when the gene transfer therapy using AAV is applied for the treatment of PAH in the human clinical setting.
There are several study limitations in this study. The AAV-hPGIS was administered 24 hours after the exposure to hypoxia. Thus, our findings suggest that AAV-hPGIS significantly inhibited the development of PAH. However, the effects of AAV-hPGIS on the advanced PAH are still unclear. Furthermore, hemodynamics and histological analysis were performed at 8 weeks after the administration of AAV-hPGIS. In addition, there is a possibility that the antibody against AAV is formed during the long-term treatment period and diminishes the efficacy of AAV-hPGIS. Thus, the analysis of further long-term therapeutic effects of AAV-hPGIS including the detection of the antibody against AAV should be investigated in future studies. Moreover, more detailed analyses regarding the differences of AAV1-hPGIS and AAV2-hPGIS such as systemic blood pressure, expression of PGIS messenger RNA, PGI2 metabolites, and production of neutralizing antibody are desirable. For clinical use, further studies, such as acute and chronic toxicology analyses, central nervous system function testing, cardiovascular and respiratory safety pharmacology studies, sterility testing, and endotoxin tests are also necessary.
Conclusion
The present study revealed that type 1 and type 2 AAV are equally effective in the treatment of PAH, indicating that gene transfer therapy using AAV vectors expressing hPGIS represent a novel therapeutic strategy for PAH.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported in part by the Health and Labour Sciences Research Grant from the Ministry of Health, Labour, and Welfare of Japan, and by the Grant from Actelion Academia Prize 2010.
