Abstract
Apolipoprotein A-IMilano (ApoA-IM), a naturally occurring Arg173 to Cys mutant of ApoA-I, has been shown to reduce atherosclerosis in animal models and in a small phase 2 human trial. We have shown superior atheroprotective effects of ApoA-IM gene compared with wild-type ApoA-I gene using transplantation of retrovirally transduced bone marrow in ApoA-I/ApoE null mice. In this study, we compared the antiatherogenic efficacy of ApoA-IM gene transfer using Recombinant adeno-associated virus (rAAV) 2 or rAAV8 as vectors in ApoA-I/ApoE null mice. Mice received a single intravenous injection of 1.2 × 1012 vector genomes of AAV2 or AAV8 vectors expressing ApoA-IM or control empty vectors (12 mice/group). Circulating levels of ApoA-IM were higher in recipients of AAV8 compared with AAV2 at 4, 12, and 20 weeks postinjection. Qualitative polymerase chain reaction analysis of RNA collected from different tissues showed that the AAV8-mediated gene transfer resulted in a more efficient transgene expression in the heart, brain, liver, lung, spleen, and kidney of the recipient mice compared with AAV2. Intravenous AAV8-ApoA-IM injection reduced atherosclerosis in the whole aorta (P < .01), aortic sinuses (P < .05), and brachiocephalic arteries (P < .05) compared with the vector control, whereas there was no statistically significant reduction in atherosclerosis in mice receiving intravenous AAV2-ApoA-IM. The ApoA-IM gene was expressed in the aortic tissue of mice receiving AAV8 ApoA-IM but not in those receiving AAV2 ApoA-IM. Immunostaining showed that compared with the vector control, there was reduced macrophage content in the brachiocephalic (P < .05) and aortic sinus plaques (P < .05) of AAV8 ApoA-IM recipients but not in the recipients of AAV2 ApoA-IM. Thus, intravenous injection of AAV8 is more effective than intravenous injection of AAV2 in the expression of ApoA-IM gene. These data provide support for the potential feasibility of this approach for atheroprotection in humans.
Introduction
Apolipoprotein A-I (ApoA-I) is the major structural protein component of high-density lipoprotein (HDL), and apolipoprotein A-IMilano (ApoA-IM) is a rare naturally occurring Arg173 to Cys173 mutant of ApoA-I carried by a small number of inhabitants of Limone sul Garda. 1,2 This mutation appears to confer greater resistance to atherosclerosis in the carriers, despite being associated with markedly lower HDL levels. 2 –4 We have previously shown superior atheroprotective effects of ApoA-IM gene compared with the wild-type ApoA-I gene in ApoA-I/ApoE null mice. 5 The precise reasons for these superior atheroprotective effects are unclear but may include greater cholesterol efflux-promoting capacity and/or antioxidant/anti-inflammatory actions. 6,7 Some laboratories, using different models, different vectors or route of administration, and short duration of treatment, have shown comparable atheroprotective effects of ApoA-IM and wild-type ApoA-I. 8,9
A series of experimental studies from our laboratory and later from other laboratories have established rapid atheroprotective effects of intravenous infusion of recombinant ApoA-IM in animal models, and these results were also supported by a small phase 2 human trial. 10,11 –16 This finding suggests that therapy based on ApoA-IM could have potential as an effective antiatherogenic strategy. 17
Adeno-associated virus (AAV) is a parvovirus that infects humans and some other primate species. Adeno-associated virus has been shown to cause only a mild immune response and is not known to cause any disease in humans. It can infect both dividing and nondividing cells and may incorporate its genome into that of the host cell. These features make AAV a very attractive candidate as a vector for gene transfer and for the creation of an isogenic human disease model. Indeed clinical trials using AAV for the treatment of cystic fibrosis, hemophilia, and muscular dystrophy showed evidence of gene transfer and expression of human clotting factor IX in patients with hemophilia B. 18 We have previously reported that AAV2 can be used to express ApoA-IM. 19 However, the gene expression level was low, and although we used different promoter constructs to enhance the level of gene expression, the expression level of ApoA-IM remained low, suggesting that AAV2 vector has a limited ability to express high level of ApoA-IM. This prompted us to investigate other AAV serotypes. We now report that AAV8 vector is more efficient than AAV2 vector for intravenous ApoA-IM gene therapy of atherosclerosis and have shown that its expression is highest in the heart compared with the other organs.
Materials and Methods
Production of Recombinant AAVs
The AAV2 and AAV8 vectors encoding ApoA-IM were constructed based on vectors previously constructed and utilized in our laboratory for the purpose of ApoA-IM expression. 19,20 Briefly, the AAV-packaging plasmid XX2 (AAV rep2 and cap2) or p5E18-VD287 (AAV rep2 and cap8) was mixed with adenovirus helper plasmid XX6-80 and plasmid-encoding ApoA-IM to be used for transfection of NautCell packaging cell line. The expression of human ApoA-IM gene is driven by a cytomegalovirus (CMV) immediate–early promoter/enhancer that includes the enhanced green fluorescent protein marker gene. The empty plasmid for each serotype was used as a vector control. The recombinant AAV (rAAV) vectors were transfected into NautCells using Effectene Transfection Reagent (Qiagen, Qiagen Inc., Valencia). The transfected cells were harvested, the cell extracts were prepared, and the rAAVs were purified by discontinuous iodixanol density gradient centrifugation. The viral preparations were concentrated and desalted by centrifugation through the 100,000 MWCO Amicon ultra-15 centrifugal filter devices (EMD Millipore Corp., Billerica).
The viral genome titers were determined by dot blot hybridization using RNA Detector Northern Blotting Kit (KPL Gaithersburg) according to the manufacturer’s instructions. The rAAV2/2 titer was 6.2 × 1012 genome copies/mL and the rAAV2/8 titer was 5.6 × 1012 genome copies/mL. Viral transducing units (Tu) were determined by transducing the 293 cells in the presence of rAAV vectors followed by fluorescence-activated cell sorting analysis.
Animal Procedures and Study Design
Six- to eight-week-old ApoA-I/ApoE double-knockout (KO) mice (weighing 15-21 g) of either gender were used for this study. The animals were randomly divided into 6 groups and injected (tail vein) with viral preparations at a dose of 1.2 × 1012 genome copies in 320 µL phosphate-buffered saline. Animals were placed on a high-fat diet containing 42% fat and 0.15% cholesterol 4 weeks after injection for the total period of 20 weeks, at which time the mice were euthanized. Blood samples were periodically collected from the retro-orbital plexus at least 4 times, once before and at the indicated times after injection. The use of experimental animals was in accordance with the guidelines of the Cedars-Sinai Medical Center Institutional Animal Care and Use Committee.
Biodistribution of ApoA-IM
The transfection efficiency of different viruses was compared by detecting plasma level of ApoA-IM in the control and animals injected with the viruses using enzyme-linked immunosorbent assay (ELISA) using antihuman ApoAI monoclonal antibody (EMD Millipore Corp., Billerica) essentially as we described. 19 For the expression of ApoA-IM gene, total RNA (n = 3-4 mice/group) was extracted from different tissues of mice with TRIzol (Life Technologies, Grand Island, NY) and then analyzed by PCR for the expression of ApoA-IM. We have used either qualitative polymerase chain reaction (qPCR), reverse transcription PCR (RT-PCR), or nested PCR depending on the tissue to be examined. The qPCR and RT-PCR were performed with the following primer sequences (223 bp): sense, 5′-aag gac ctg gcc act gtg ta-3′; antisense, 5′-tct cct cct gcc act tct tc-3′. The nested PCR was performed with “outer” primers (301 bp): forward, 5′-aaggacctggccactgtgta-3′; reverse, 5′-tctcctcctgccacttcttc-3′ and “inner” primers (190 bp): forward, 5′-tgg atg tgc tca aag aca gc-3′; reverse, 5′-agg ccc tct gtc tcc ttt tc-3′.
Assessment of Atherosclerosis
Atherosclerosis was assessed in the whole aorta, brachiocephalic artery, and aortic sinuses. The extent of the atherosclerotic lesions in the total aorta was quantified after Oil red O staining, as described previously. 19 Briefly, the vascular tree was dissected intact and then the luminal surface was exposed. After tissue fixation, the adventitial tissue was removed and aorta was stained with Oil red O for lipids. The aortas were placed on a glass slide and images were captured using a Spot digital camera (Scientific Instrument Company, Sunnyvale, California). The extent of the atherosclerotic lesions was quantified using Image-Pro Plus (version 4; Media Cybernetics, Silver Spring, Maryland). Lesions were reported as percentage of the total aortic area consisting of thoracic aorta and abdominal aorta ending at the iliac bifurcation.
For brachiocephalic artery sections, the aortic arch was fixed in 4% paraformaldehyde overnight, then embedded in optimal cutting temperature (OCT), and 8-µm serial sections were used for staining and lesion assessment. For aortic sinus lesion assessment, 8-µm serial sections of OCT-embedded aortic sinus were stained with Oil red O and hematoxylin. The quantitative analysis of lesion area for innominate artery and aortic sinus was measured by Image-Pro Plus using manual tracing of the lesion, which was identified by a combination of lipid staining and histological morphology (mean of 8 sections/mouse, 5 mice/group).
To determine the macrophage content, serial sections from aortic arch and sinus were stained with anti-MOMA-2 antibody (AbD Serotec, Raleigh) and counterstained with hematoxylin; the positive areas in innominate artery and aortic sinus lesion were analyzed by Image-Pro Plus.
Data Analysis
Data are from representative experiments and are expressed as mean ± standard deviation. Statistical test for significance was done using an unpaired t-test or 1-way analysis of variance followed by a Tukey test for pairwise comparisons.
Results
Circulating Levels of Transgene
Plasma samples were collected from each group of mice at various times postinjection, and the level of ApoA-IM was measured by ELISA (Figure 1). The plasma levels of ApoA-IM were significantly higher in AAV8 recipients compared with AAV2 recipients at 4 weeks (P < .001), 12 weeks (P < .001), and 20 weeks (P < .01) postinjection.

AAV8-ApoAI-M transgene is expressed at a significantly higher level than AAV2 ApoA-IM in mouse blood plasma. The ApoA-I−/−/ApoE−/− double-knockout (KO) mice were injected with the viral vectors encoding ApoA-IM. The blood plasma samples were collected at the indicated times postinjection and analyzed by enzyme-linked immunosorbent assay (ELISA) for the presence of ApoA-IM protein. Each bar represents mean ± standard deviation from 12 mice/group. AAV indicates adeno-associated virus; ApoA-IM, apolipoprotein A-IMilano.
Organ Distribution of Transgene Expression
At the time of euthanasia, various organs were collected, snap frozen, and total RNA was extracted and analyzed using qPCR with primers specific to ApoA-IM (Figure 2). The level of AAV8- and AAV2-mediated transgene expression was as follows: brain: 11.85 ± 2.4 versus 0.95 ± 0.2, P = .05; heart: 102.3 ± 24.20 versus 0.9 ± 0.5, P = .05; liver: 32.14 ± 14.56 versus 1.37 ± 0.22, P = .2; lung: 16.49 ± 10.75 versus 1.86 ± 1.8, P = .25; spleen: 5.41 ± 1.59 versus 3.39 ± 1.69, P = .4; and kidney: 1.96 ± 0.8 versus 0.81 ± 0.18. As expected, no ApoA-IM was noted in the vector control groups.

Tissue distribution of ApoA-IM in the double-knockout (KO) mice injected with different viral vectors. The mouse groups were injected with AAV8 or AAV2-encoding ApoA-IM or control vectors for 24 weeks when the mice were euthanized. The indicated organs were collected; total RNAs were analyzed by qPCR. The qPCR data are normalized against glyceraldehyde 3-phosphate dehydrogenase. Data represent mean ± standard deviation from 10 to 12 mice/group. AAV indicates adeno-associated virus; ApoA-IM, apolipoprotein A-IMilano.
Aortic Transgene Expression
Total RNA was extracted from the aortas of mice injected with AAV8 control vector or AAV8-ApoAIM vector. The expression of apoA-IM was determined by RT-PCR. Although no ApoA-IM gene expression was detected in the 4 aortas of the mice injected with the control vector, a 223-bp fragment identifying ApoA-IM was detected in the aortas of 3 of the 4 mice (Figure 3A), showing that the ApoA-IM gene is expressed in the aorta.

ApoA-IM gene expression in monocytes and aorta. A, The double-knockout (KO) mouse groups were injected with either an AAV8 vector control (5 mice/group) or an AAV8 vector expressing ApoA-IM (6 mice/group). The peripheral blood was collected from the mouse groups; total RNA was extracted and analyzed using RT-PCR with primers amplifying a 304-bp fragment within the ApoA-IM gene. This was followed by nested PCR where the 304-bp amplified products were further amplified using nested primers that are located within 304 bp, generating a nested 190-bp product. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as control in all experiments. B, Expression of ApoA-IM gene in the aortas of AAV8-treated mice. Total RNA was extracted from the aortas dissected from the groups of mice (4 mice/group) injected with vector control (VC) or vector expressing ApoA-IM (A-IM) that had been on high-fat diet for 20 weeks. The expression of ApoA-IM was detected using RT-PCR with primers specific to the gene. GAPDH was used as a control. AAV indicates adeno-associated virus; ApoA-IM, apolipoprotein A-IMilano.
Transgene in Circulating Mononuclear Cells
To determine whether viral vectors can be detected in the peripheral blood mononuclear cells (PBMCs), cells were collected from 6 mice receiving either AAV8 control or AAV8-ApoA-IM vectors that were fed a high-fat diet for 12 weeks. The expression of ApoA-IM was determined by nested RT-PCR. We used nested PCR to analyze expression of ApoA-IM because the amount of total RNA isolated from PMNC from each mouse is small. First, we used outer primers that amplified a 304-bp PCR product corresponding to the ApoA-IM gene. We did not detect amplification of the 304 bp band in the isolated PMNC (Figure 3B). However, when we used nested primers that are located within the target sequence of outer primers, we noted amplification of a 190-bp band corresponding to ApoA-IM in the PBMC from 4 mice (Figure 3B). No transgene expression was detected in the groups of mice injected with AAV8 vector control; glyceraldehyde 3-phosphate dehydrogenase was used as an internal control.
Effect on Atherosclerosis
The aortas were dissected and stained with Oil red O and the extent of aortic atherosclerosis was measured as percentage of aorta with plaque. Quantitative analysis showed that there was no statistically significant difference in aortic atherosclerosis between the AAV2 vector control group and the AAV2-Apo-AIM group (P > .05; Figure 4A and C). In contrast, the mean aortic lesion area was reduced by 42% in AAV8 ApoA-IM when compared with the AAV8 vector control group (7.7% ± 0.6% vs 13.5% ± 1.1%; P < .01; Figure 4B and C). The extent of aortic atherosclerosis in AAV8 ApoA-IM recipients was significantly less compared with AAV2 ApoAIM recipients (P < .05; Figure 4C). The AAV8 ApoA-IM recipients also had a significant reduction in the size of aortic sinus (P < .05) and brachiocephalic artery plaques (P = .05) compared with AAV8 vector controls, whereas AAV2 ApoA-IM recipients had no significant reduction in aortic sinus or brachiocephalic artery plaque size compared with AAV2 vector control (Figure 5A and B).

Evaluation of atherosclerosis in groups of mice injected with AAV2 or AAV8 vectors. A, Representative micrographs of the aorta from double KO mice injected with either AAV2 vector control or AAV2 expressing ApoA-IM. Aortas were isolated from the mouse groups that were injected with the different viral vectors, fed high-fat diet for 20 weeks, and then euthanized. The aortas were dissected and stained with Oil red O to measure their lipid content. B, Representative Oil red O staining of the aorta from mouse groups injected with either AAV8 vector control or AAV8 expressing ApoA-IM. C, Quantitative analysis of atherosclerosis in the aortas of mouse groups injected with different viral vectors. The double KO mice were injected with the indicated viral vectors and were fed high-fat diet 4 weeks postinjection. After 20 weeks on high-fat diet, the mice were euthanized, and their aortas were isolated and stained with Oil red O. The mean lesion areas were determined as described in Method section. AAV indicates adeno-associated virus; ApoA-IM, apolipoprotein A-IMilano; KO, knockout.

Quantitative analysis of atherosclerosis in brachiocephalic artery and aortic sinus lesions from mouse groups injected with different viral vectors. The brachiocephalic artery (panel A) and aortic sinuses (panel B) were collected from the mice injected with different viruses and fed a high-fat diet as described in the legend of Figure 4. The lesions were quantified as described earlier.
Plaque Lipid Content and Macrophage Content
The lipid content in aortic sinus and brachiocephalic artery plaques was significantly reduced (P < .05) by AAV8 ApoA-IM compared with AAV8 vector control (Figure 6A and B). However, AAV2 ApoA-I M recipients did not show any significant reduction in aortic sinus or brachiocephalic plaque lipid content.
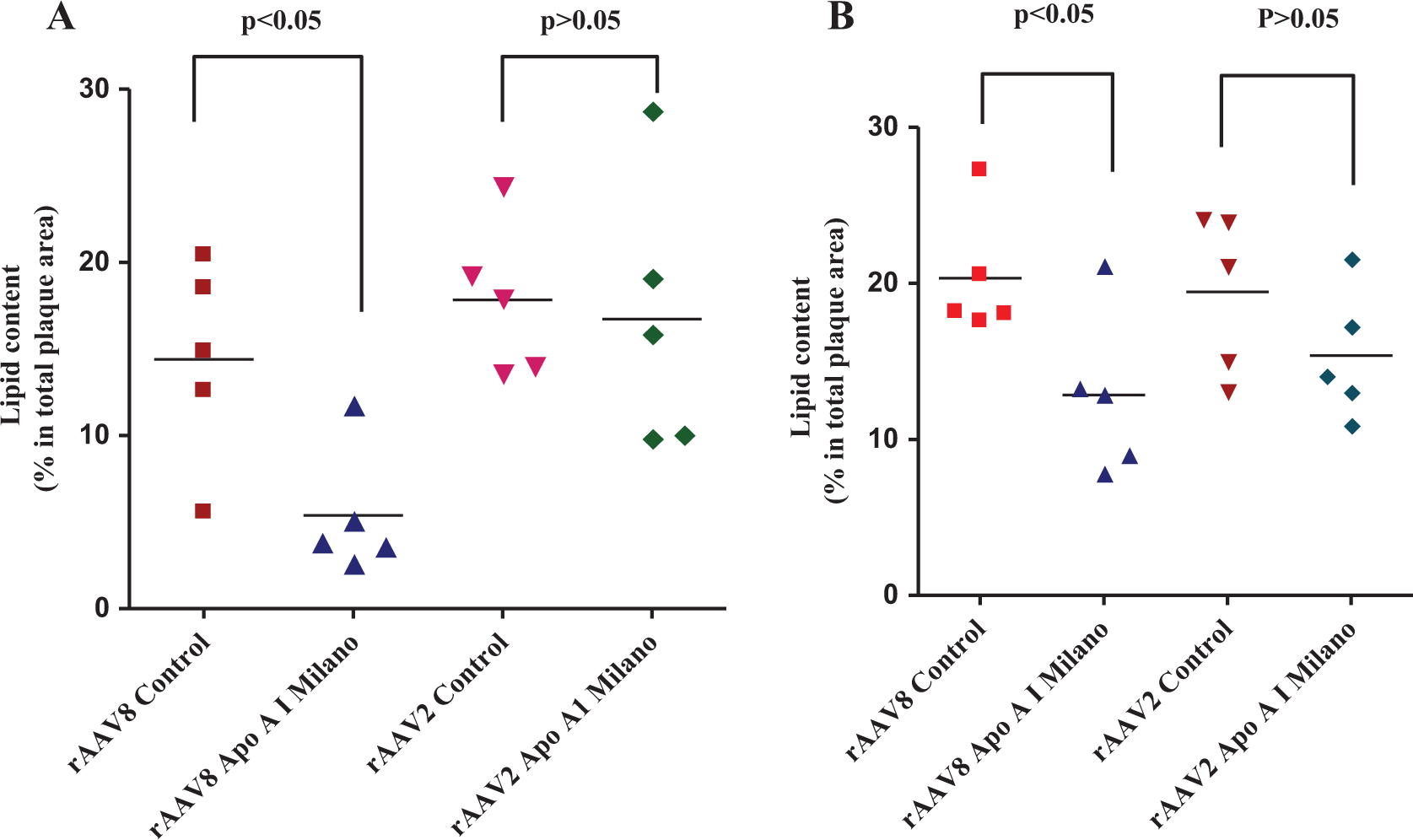
The lipid content of brachiocephalic artery and aortic sinus lesions from mouse groups injected with different viral vectors. The lipid content of lesions in the brachiocephalic artery (panel A) and aortic sinuses (panel B) was determined by Oil red O staining and quantified as described earlier. The lipid content was found to be significantly lower in the mouse group injected with the AAV8 vector compared with the AAV2 group. AAV indicates adeno-associated virus.
The macrophage immunoreactivity in aortic sinus and brachiocephalic artery plaques was significantly (P < .05) reduced by AAV8 ApoA-IM compared with AAV8 vector control (Figure 7A and B). However, AAV2 ApoA-I M recipients did not show any significant reduction in aortic sinus or brachiocephalic plaque lipid content compared with AAV2 vector controls (Figure 7A and B).

The macrophage content of brachiocephalic artery and aortic sinus lesions from mouse groups injected with different viral vectors. The macrophage content in the brachiocephalic artery (panel A) and aortic sinuses (panel B) was measured by staining the respective sections with antibody specific to macrophages (MOMA-2). The positive immunoreactivity was quantified as described earlier.
Plasma Cholesterol
To determine whether expression of ApoA-IM by the AAV vectors affect plasma cholesterol level, we collected plasma from mice before injection with the virus and after injection when the mice were placed on high-fat diet for 8 and 20 weeks. Plasma cholesterol was measured from each group of mice (n = 12/group). The average plasma cholesterol in the mouse groups before high-fat diet was 152 ± 12 mg/dL. After feeding high-fat diet for 8 weeks, the average plasma cholesterol level increased to 1155 ± 120 mg/dL in groups of mouse injected with AAV8 control virus or AAV8-ApoA-IM (Figure 8). The cholesterol level in these groups of mice was reduced to 824 ± 52 mg/dL after 20 weeks of high-fat diet feeding. The differences between these groups of mice were not statistically significant. Similarly, mice injected with AAV2 vectors that were fed high-fat diet for 8 weeks had average plasma cholesterol level of 1068 ± 16 mg/dL, and the cholesterol levels reduced to 792 ± 5.5 mg/dL when the high-fat diet continued for 20 weeks, a statistically insignificant difference. These results suggest that the effect of AAV8-ApoA-IM on atherosclerosis is independent of plasma cholesterol level.

The plasma cholesterol level between groups of mice injected with adeno-associated virus (AAV) viruses. The plasma were collected from mouse groups (n = 10-12/group) before injection and 8 or 20 weeks on high-fat diet after injection. The cholesterol levels were measured as described in Method section. The data show statistically insignificant difference in the cholesterol level among the mouse groups.
Discussion
Apolipoprotein A-IM gene is an attractive gene for amelioration, stabilization, and regression of atherosclerosis because of its pleiotropic biological actions. These include promotion of reverse cholesterol transport, anti-inflammatory and immune-modulating effects, and antioxidant actions as well as its proven efficacy in animal models. 17 Gene transfer is a potentially attractive approach to harness the atheroprotective effects of ApoA-I and ApoA-IM avoiding the need for large-scale manufacturing of recombinant protein for intravenous injection.
In this study, we demonstrate that compared with a control vector, intravenous injection of AAV8-encoding ApoA-IM gene produces significant reductions in atherosclerosis in the whole aorta, aortic sinuses, and brachiocephalic arteries in a hyperlipidemic mouse model without significant changes in circulating cholesterol levels. We further show that AAV8-encoding ApoA-IM gene also reduces plaque lipid content and plaque macrophage immunoreactivity consistent with a more stable plaque phenotype. This study also demonstrates that intravenous injection of AAV2-encoding ApoA-IM produced much smaller reductions in atherosclerosis, which were not statistically different from vector controls and were significantly less than those achieved with AAV8. We selected AAV vectors because they have several advantages as outlined earlier and are currently being evaluated in several gene therapy trials in humans. In this study, we used ApoA-I−/−/ApoE−/− double KO recipient mice in order to eliminate the interplay between endogenous ApoA-I activity and exogenous apoA-IM activity; therefore, the biological activity of ApoA-IM that we observed would be exclusively related to the activity of the delivered gene. In addition, using the double KO mice allowed us to investigate tissue distribution of the virally produced ApoA-IM without any interference from endogenous ApoA-I. Our previous studies using an AAV2 vector for the expression of ApoA-IM in vitro required the development of specific CMV promoter construct to achieve a high level of gene expression 20 ; we used this promoter construct again in these experiments.
The precise reasons for the greater effectiveness of intravenous AAV8 compared with AAV2 for gene transfer, as observed in this study, currently remain unclear but may be related in part to higher systemic levels of transgene and or greater tissue tropisms of AAV8 for the heart and vasculature. Thus, the plasma levels of ApoA-IM in AAV8-ApoA-IM recipient mice increased 4 weeks after injection (∼80 ng/mL); remained high for 12 weeks; and declined 20 weeks postinjection. In contrast, the plasma level of ApoA-IM in the AAV2 vector recipient group was comparatively low (∼20 ng/mL) 4 weeks after the injection and decreased further when measured at 12- and 20-week time points. Furthermore, biodistribution studies also demonstrated higher levels of transgene expression in the heart and aorta of AAV8 recipients compared with AAV2 recipients. Of particular interest is our observation that circulating mononuclear cells expressed the transgene in AAV8 recipients but not in AAV2 recipients, suggesting the possibility that transgene expression in mononuclear cells may contribute to the greater atheroprotective effects of AAV8 since a number of prior studies have shown that macrophage-specific expression of ApoA-I or ApoA-I M is highly effective in reducing atherosclerosis in murine models, despite very low systemic transgene levels. 5,21 These data collectively suggest that intravenous AAV8 vector may be a better gene delivery system than AAV2 vector in the murine model, which we have employed. Unlike 2 previous studies of AAV8-mediated ApoA-I or ApoA-IM gene transfer, which used an intraportal injection, this is the first study of intravenous delivery of AAV8-encoding ApoA-IM gene. 9,22
In summary, we demonstrate that ApoA-IM gene transfer using intravenous AAV8 is more effective than AAV2 in reducing atherosclerosis and favorably modifying plaque phenotype in a murine hypercholesterolemic model. Further investigation of this strategy is warranted as a novel atheroprotective intervention.
Footnotes
Acknowledgments
The authors gratefully acknowledge the support of the Heart Foundation, the Eisner Foundation, the Spielberg Foundation, Corday Foundation, and the Skirball Foundation.
Authors’ Note
FT, LW, AA, and MY carried out the experiments described in the article. BGS and PKS were involved in the development of hypotheses, designed experiments, and wrote the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The evolution of this approach was initially supported by program project grant from NHLBI Grant 1POHI60898.
