Abstract
Background:
Combination therapy is commonly used for pulmonary arterial hypertension (PAH) treatment. We aimed to identify factors that may predict the need for future combination therapy.
Methods:
We conducted a retrospective chart review of consecutive patients with PAH in an aim to describe baseline clinical, echocardiogram, and hemodynamic characteristics of patients who eventually required combination therapy during the course of their disease and compared them to the ones who were maintained on monotherapy.
Results:
The monotherapy group was followed for an average of 31.8 ± 18.8 months and the combination therapy group was followed for an average of 28.7 ± 13.6 months. Among the 71 patients analyzed, a significantly higher number of patients who eventually required combination therapy belonged to World Health Organization functional class 3 (45% vs 37%) and 4 (23% vs 0) at baseline, compared with those on monotherapy (
Conclusion:
Patients with PAH who require combination therapy in the course of their disease have worse hemodynamics, PAH risk score, functional class, and end of 6MWT oxygen saturation at the time of presentation compared to patients maintained on monotherapy.
Introduction
Pulmonary arterial hypertension (PAH) is a devastating, life-threatening disease with death usually resulting from right heart failure. Over the past decade, a number of different effective therapies have become available. Until recently, available therapies belong to one of the 3 classes, that is, prostacyclin analogues (prostanoids), endothelin receptor antagonists (ERAs), and phosphodiesterase type 5 inhibitors, with recent addition of Riociguat. Riociguat was recently approved for PAH and chronic thromboembolic pulmonary hypertension. 1,2 Combination therapy utilizing drugs available from 2 or more of these classes has been in practice for quite some time. The rationale for using combination therapy is strong and a number of well-designed clinical trials have shown the addition of another therapeutic agent from a different class on background stable therapy improves 6-minute test (6MWT) distance, pulmonary hemodynamics, and World Health Organization (WHO) functional class. 3 –8 Results from a recently concluded trial of upfront combination therapy utilizing tadalafil and ambrisentan and comparing it to tadalafil or ambrisentan monotherapy alone (AMBITION) showed improvement in time to first clinical worsening primarily driven by reduction in hospitalization. In addition, significant improvements were also seen in 6MWT and N-terminal of the prohormone brain natriuretic peptide (NT-proBNP) but no change was seen in WHO functional class. Although the initial inclusion criteria for this trial was a standard definition of pulmonary hypertension (mean pulmonary artery pressure ≥ 25 and pulmonary vascular resistance (PVR) ≥ 240 dyne s/cm5), subsequently the PVR criteria was changed to ≥300 dyne s/cm5 with exclusion of patients who had 2 of the 3 comorbid conditions, namely, obesity, diabetes, and systemic hypertension. Although this trial provides some valuable insight into the role of combination therapy, a number of questions currently remain unanswered. Are there particular individuals who would be good candidates for upfront combination therapy or should it be applied across the board? What combination should be used? Is upfront combination better than add-on therapy? What is the optimum time to add-on therapy? Upfront combination therapy may have additional implications in terms of side effects, patient compliance, and the cost of therapy.
Keeping these questions in mind, we decided to look at our database in an effort to potentially answer one of the questions and identify factors that may predict the need for combination therapy in a specific patient.
Methods
After the institutional review board approval, we conducted a retrospective chart review of all patients who received outpatient treatment for group 1 PAH from July 2007 to July 2012. Pulmonary arterial hypertension was defined as right heart catheterization (RHC) documentation of resting mean pulmonary arterial pressure (mPAP) ≥25 mm Hg, PVR ≥240 dyne s/cm5, and pulmonary capillary wedge pressure (PCWP) or left ventricular end-diastolic pressure (LVEDP) of ≤15 mm Hg. Patients who presented to our center with an already established diagnosis and on treatment and patients with incomplete data or follow-up were excluded. Patients who were enrolled or completed a clinical trial were also excluded.
The patients were divided into 2 groups. Monotherapy group included patients who were initiated on a single PAH drug and maintained on that drug throughout the follow-up period and combination therapy group included patients who were initiated on a single drug but required addition of other classes of drug, either for clinical deterioration or inadequate response to therapy as determined by treating physician, during the follow-up period. The follow-up period in months was calculated up to the last documented visit in the clinic.
Data collected included age, gender, heart rate at first clinical visit, baseline 6MWT, Borg dyspnea index (BDI), and oxygen saturation (Sp
Mean and range or standard deviation was used to describe normally distributed continuous data. Median and interquartile range (25-75) were used to describe nonnormally distributed data and percentages were used to describe categorical data. A Fisher exact test and chi-square test were performed to analyze the relationship between categorical variables when appropriate. Logistic regression was used to identify possible predictors for need to combination therapy in future. Univariate analysis was conducted using Strata 13. The α level of significance was set to .05. The association was expressed in odds ratio and results also included 95% confidence interval. For the rest of the statistics, a
Results
We screened 130 patients and 59 were excluded either due to either incomplete data, follow–up, or were already on PAH therapy. Seventy-one patients met the inclusion criteria and were analyzed. Fifty-six (79%) patients were female and 15 (21%) patients were male. A total of 39 (55%) patients were maintained on monotherapy while 32 (54%) patients required combination therapy. The monotherapy group was followed for an average of 31.8 ± 18.8 months, and the combination therapy group was followed for an average of 28.7 ± 13.6 months. The average duration before a second agent was added on in the combination group was 6.3 ± 7.4 months.
There was no difference in age, sex, or WHO group between the 2 groups (Table 1). A significantly higher number of patients in the combination therapy group presented with WHO functional class 3 (45% vs 37%) and 4 (23% vs 3%) symptoms compared to the monotherapy group (Figure 1). There was also a difference in the mean REVEAL registry PAH risk score between the 2 groups with the mean score for the monotherapy group being 7.4 ± 1.7 while the mean score for combination therapy was 9.2 ± 1.7 (

Distribution of World Health Organization (WHO) functional class.

Difference between REVEAL PAH risk score calculated at first visit.
Baseline Demographics.

Abbreviations: APAH, associated pulmonary arterial hypertension; CTD, connective tissue disease; HIV, human immunodeficiency virus; PoPH, portopulmonary hypertension; CHD, congenital heart disease; SD, standard deviation.
End of 6MWT Sp
Baseline Clinical and Echocardiographic Variables.

Abbreviations: BPM, beats per minute; Sp
Pulmonary hemodynamic measurements during the baseline diagnostic RHC were also significantly different between the 2 groups with higher mPAP, lower PCWP or LVEDP, lower CO, and higher PVR noticed among the combination therapy group. There was no statistical difference in RAP measurements (Table 3).
Right Heart Catheterization-Derived Hemodynamic Data.
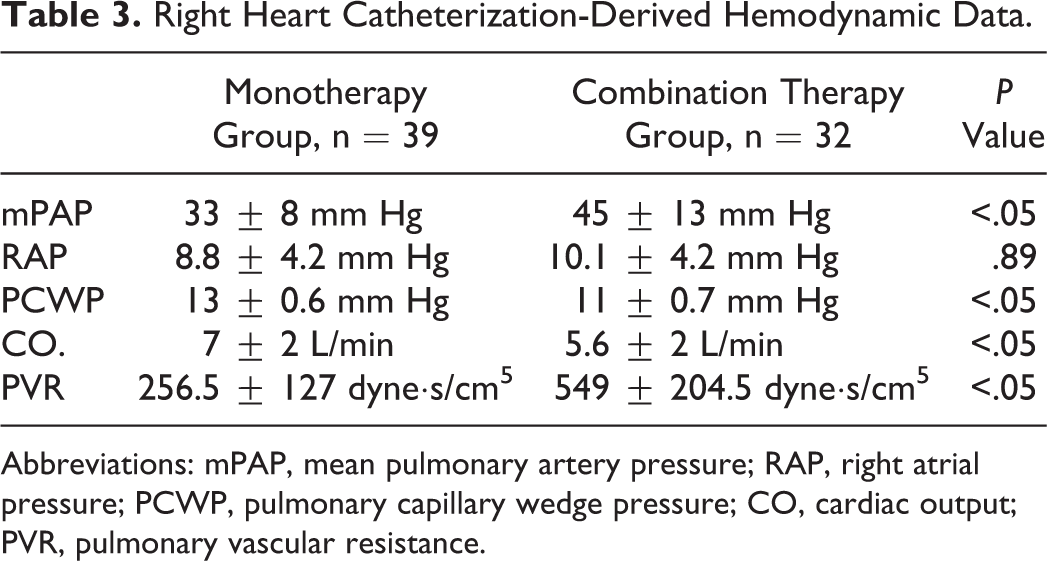
Abbreviations: mPAP, mean pulmonary artery pressure; RAP, right atrial pressure; PCWP, pulmonary capillary wedge pressure; CO, cardiac output; PVR, pulmonary vascular resistance.
Univariate analysis revealed that only PVR ≥ 300 dyne·s/cm5, mean PA presure of ≥ 40 mm Hg, echocardardiogram-derived estimated RVSP ≥ 60 mm Hg, PAH risk score ≥ 10, and end of 6 MWT saturation of ≤ 90% were predictors of future need of combination therapy (Table 5). Adjusted odd ratio analysis revealed that only PVR > 300 dynes/cm5 and echo derived estimated RVSP of > 60 mm Hg were statistically significant (Table 6).
Classes of Drug Used to Treat PAH.

Abbreviations: PAH, pulmonary arterial hypertension; SC, subcutaneous; IV, intravenous
Univariate Analysis of Predictors of Need for Combination Therapy.

Abbreviations: mPAP, mean pulmonary artery pressure; PVR, pulmonary vascular resistance; RVSP, right ventricular systolic pressure; 6MWT, 6-minute walk test; Sp
Adjusted Odds Ratio of Predictors of Need for Combination Therapy.

Abbreviations: mPAP, mean pulmonary artery pressure; PVR, pulmonary vascular resistance; RVSP, right ventricular systolic pressure; CI, confidence interval; OR, odds ratio; 6MWT, 6-minute walk test.
The most common class of drugs used in the monotherapy group was ERAs followed by phosphodiesterase type 5 inhibitors while in the combination therapy group, the most common class of the initial drug used was prostanoids followed by ERAs. We did not find any differences among the frequency of use of one class of drugs over the other in the combination group including when used as add-on therapy (Table 4).
Discussion
Combination therapy to target different pathways for treating PAH has been in practice since the development of multiple classes of therapy. Addition of a second therapeutic agent for combination therapy is suggested in a sequential manner when there is inadequate response to 1 PAH agent.
10
The second agent is usually chosen from a different class of PAH medications. Combination therapy appears to be a logical approach as multiple pathways can be targeted simultaneously and synergistic activity of different agents can potentially restrict the disease progression. Several placebo-controlled trials have looked at different combination of medications but primarily added onto background stable therapy and this has generated interesting data. BREATHE-2 (Combination of bosentan with epoprostenol in pulmonary arterial hypertension) was the first randomized, placebo-controlled, double-blind trial combining epoprostenol and bosentan compared to epoprostenol monotherapy.
5
The trial showed a nonstatistically significant trend toward reduction in PVR in the combination therapy group (−36% vs −22%,
Although retrospective, our study shows that there are significant hemodynamic and clinical differences between patients who require combination therapy in the course of their disease compared to those who can be maintained on monotherapy. These clinical and hemodynamic factors combined with REVEAL PAH risk score may provide us the ability to identify patients at risk of either failing or progressing on monotherapy and perhaps should be initiated on combination therapy up front or at least may help in designing future clinical trials. There are many patients who clinically do well on monotherapy and may not need a second agent added at all during the course of their disease and if these patients are not recognized and are included in the trials evaluating combination therapy, it may skew the information gained from the trials and unnecessarily expose patients to potential side effects of combination therapy. A number of randomized controlled trials have shown that the patients placed in placebo arm fail to catch up with the treatment arm, in terms of improvement in the 6 minute walk test.
18
A similar delay to wait and see if the first therapy is working in this population may likely have a significant impact on patient outcomes. In our study, prostanoid therapy was the most commonly used first therapy in the combination group, likely due to these patients being higher risk (functional class and hemodynamics) on presentation. Despite upfront prostanoid therapy, the need for another class of drug in this population was evident from this study. There are several limitations to this study. The retrospective nature limits the ability to derive solid conclusions. There are probably other factors related to initiation of combination therapy, besides failing or progressing on first therapy, we may have not identified. The groups were fairly similar and the connective tissue disease subpopulation mainly comprised of mixed connective tissue disease. There was only 1 patient with scleroderma in the combination group. Another factor to consider would be the follow-up duration, although the mean is not different across the groups, the individual follow-up periods may dictate when the combination is initiated. Functional class has been shown to correlate well with 6MWT distance but in our study there were more functional class 3 and some class 4 patients in the combination therapy group, but the 6MWT distance was not very different between the 2.
19
We are unable to explain this, but perhaps individual patient characteristics may have had an influence. Another factor to consider would be compliance with therapy playing a role in clinical failure of a specific drug, although hard to document and confirm, to the best of our knowledge all the patients included in the analysis were compliant with their medications. The univariate analysis done on this cohort showed that PVR >300 dyne s/cm5 was a significant risk factor. The same cutoff was used for inclusion criteria in the recently concluded AMBITION (A Study of First-Line Ambrisentan and Tadalafil Combination Therapy in Subjects With Pulmonary Arterial Hypertension) trial, the results of which were presented in the 2014 European Respiratory Society meeting and looked promising with reduction in clinical failure (decreased hospitalization) and clinical improvement (improvement in 6MWD and NT-proBNP). Further support for upfront combination therapy may also be reinforced by the recently presented abstract from the COMPASS 2 (Effects of the Combination of Bosentan and Sildenafil Versus Sildenafil Monotherapy on Pulmonary Arterial Hypertension) study, evaluating the effects of addition of bosentan to stable dose of sildenafil background therapy which did not meet the primary end point of reduction in morbidity and mortality, with an observed risk reduction of 17% versus placebo (
Conclusion
Patients with PVR ≥300 dyne s/cm5, echocardiogram-derived RVSP ≥60 mm Hg, mPAP ≥ 40 mm Hg during RHC, and end of 6MWT Sp
Footnotes
Author Contributions
Bajwa A contributed to conception and design, contributed to analysis and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Shujaat A contributed to conception and design, contributed to interpretation, critically revised the manuscript, and gave final approval. Cury JD contributed to conception, contributed to interpretation, critically revised the manuscript, and gave final approval. Jones LM contributed to design, contributed to interpretation, and drafted the manuscript. Qureshi T contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, and gave final approval. Seeram V contributed to conception, contributed to interpretation, and critically revised the manuscript. Al-saffar F contributed to design, contributed to analysis, and critically revised the manuscript.
Funding
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) received no financial support for the research, authorship, and/or publication of this article.
