Abstract
The mammalian brain comprises two structurally and functionally distinct compartments: the gray matter (GM) and the white matter (WM). In humans, the WM constitutes approximately half of the brain volume, yet it remains significantly less investigated than the GM. The major cellular elements of the WM are neuronal axons and glial cells. However, the WM also contains cell bodies of the interstitial neurons, estimated to number 10 to 28 million in the adult bat brain, 67 million in Lar gibbon brain, and 450 to 670 million in the adult human brain, representing as much as 1.3%, 2.25%, and 3.5% of all neurons in the cerebral cortex, respectively. Many studies investigated the interstitial WM neurons (IWMNs) using immunohistochemistry, and some information is available regarding their electrophysiological properties. However, the functional role of IWMNs in physiologic and pathologic conditions largely remains unknown. This review aims to provide a concise update regarding the distribution and properties of interstitial WM neurons, highlight possible functions of these cells as debated in the literature, and speculate about other possible functions of the IWMNs and their interactions with glial cells. We hope that our review will inspire new research on IWMNs, which represent an intriguing cell population in the brain.
Keywords
Introduction
Brains of mammals consist of two structurally and functionally distinct parts: the gray matter (GM) and the white matter (WM). Both the GM and the WM contain neuronal and glial elements alongside the blood capillaries. A major difference between these two areas is that the density of neuronal cell bodies and synapses is significantly higher in the GM, while the density of myelinated neuronal axons is higher in the WM. However, various studies using immunohistochemistry for neuronal markers have revealed a population of neurons residing within the WM areas of the mammalian brain, including humans (Garcia-Marin and others 2010; Judas and others 2010b; Kostovic and Rakic 1980; Meyer and others 1992; Smiley and others 1998), monkeys (Mortazavi and others 2016; Swiegers and others 2019), cats (Valverde and Facal-Valverde 1988), rats (Clancy and others 2001; Reep 2000; Valverde and others 1989), mice (Hoerder-Suabedissen and Molnar 2013; von Engelhardt and others 2011), and bats (Bhagwandin and others 2020). Those neurons were first described by Theodor Meynert as early as 1867, as neurons in the subcortical WM, and were later introduced as “interstitial cells” by Ramón y Cajal, who described them in the cerebellum and in the trigeminus of a cat (Cajal 1896). Notably, WM neurons were also described in the spinal cord, the spinal nerves, and the cranial nerves (Das and Kreutzberg 1969; Judas and others 2010a; Sosa and Andrew 1947). Throughout the literature, various terms have been employed to describe them, such as WM (interstitial) cells/neurons, neurons in the WM, interstitial neurons of the WM, and interstitial or infracortical WM neurons (Akbarian and others 1996; Duchatel and others 2019; Kostovic and others 2011; Mortazavi and others 2016; Sedmak and Judas 2021; Suarez-Sola and others 2009; Swiegers and others 2019; Yang and others 2023). Acknowledging the range of names attributed to these cells, in this review, we have chosen to use the name interstitial WM neurons (IWMN) and emphasize the need for a unified nomenclature.
Despite their early discovery and the advent of advanced molecular and cellular techniques, little is still known regarding the molecular properties and the functional role of IWMNs. In this review, we provide a concise update regarding distribution and properties of IWMNs in normal and diseased brains and highlight possible functions of these cells.
The Traditional Differentiation of White Matter and Gray Matter and the First Hints of Neurons within White Matter.
The brain of vertebrates contains two kinds of tissue: gray matter (GM) and white matter (WM). Segregation into GM and WM occurs during development and represents a ubiquitous feature of the vertebrate brain. In humans, around gestational week 7, radially migrating neurons from the ventricular (VZ) and subventricular zones (SVZ) trigger the development of the cortical plate (CP), which initially consists of a thin superficial marginal zone (MZ) and an underlying subplate (SP) (Budday and others 2015). The marginal zone contains tangentially migrated cells, which hinder the radial migration of developing cortical neurons, causing them to accumulate below, that is, between the marginal zone and the subplate (Fig. 1) (Duque and others 2016). As development proceeds, the marginal zone neurons and the underlying cortical neurons form the GM (Fig. 1) (Molnar and others 1998; Rakic and Zecevic 2003). The area between the subplate and the proliferative ventricular and subventricular zones is called the intermediate zone (IZ) (Fig. 1). It contains axons of long-range projection neurons, few migrating cells, and former preplate cells. As development proceeds, the intermediate zone becomes the WM (Budday and others 2015; Bystron and others 2008). For reasons that are not fully understood, some neurons from the fetal subplate zone do not migrate to the developing GM and end up in the adult WM (Duque and others 2016). Together with the neurons from the fetal intermediate zone (fetal WM), they form the neuronal population of the adult WM. This population is called the interstitial white matter neurons (IWMNs).
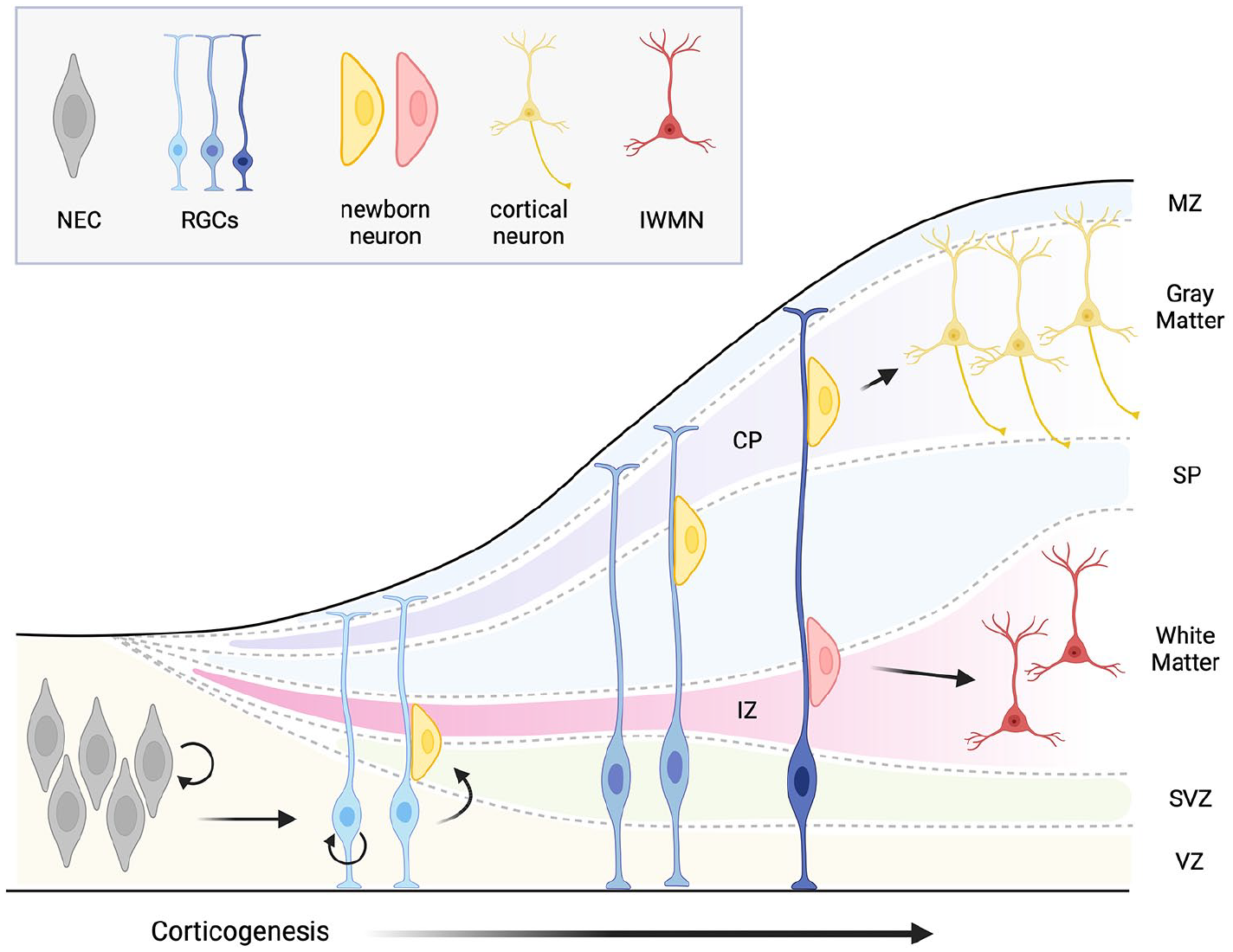
Simplified view of cortical development showing segregation of the white and gray matter, and appearance of the interstitial white matter neurons. CP, cortical plate; IWMN, interstitial white matter neuron; IZ, intermediate zone; MZ, marginal zone; NEC, neuroepithelial cell; RGG, radial glial cell; SP, subplate; SVZ, subventricular zone; VZ, ventricular zone. Created with BioRender.com.
Origin, distribution, and subpopulations of IWMNs in the brain
In rodents, monkeys, and humans, the IWMNs arise during embryonic development and remain into adulthood and normal aging. While their relative density decreases during the first year of life in monkeys and humans, it remains relatively stable throughout adulthood and normal aging (Kostovic and Rakic 1980; Mortazavi and others 2016). There are approximately 10 to 28 million IWMNs in the brain of adult bats, 67 million in lar gibbon, and 450 to 670 million in the adult human brain, representing 1.3%, 2.25%, and 3.5% of the total neuron population in the cerebral cortex, respectively (Bhagwandin and others 2020; Sedmak and Judas 2019, 2021; Swiegers and others 2019).
Based on their location and developmental origin, IWMNs in the brain may be arbitrarily divided into two populations: the superficial (gyral) and the deep IWMNs (Fig. 2). The superficial population encompasses the IWMNs located within the sulcal and gyral areas, directly below the sixth neocortical layer, and represents the descendants of the subplate cells (Judas and others 2010a, 2010b). The deep population consists of IWMNs clustered in the corpus callosum, anterior commissure, internal and external capsule, and deeper parts of the WM. This population encompasses neurons that reside within the deep WM already in the fetal brain and continue to reside there throughout life (Judas and others 2010a, 2010b).
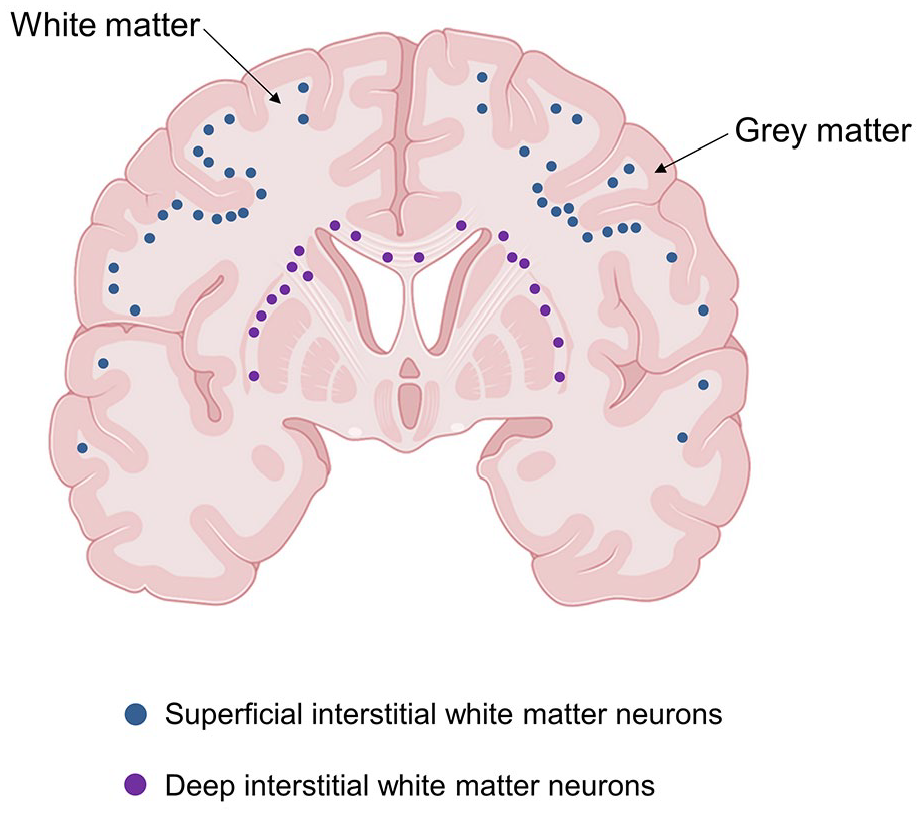
Schematic drawing showing two populations of interstitial white matter neurons (IWMNs) in human brain classified based on their location and developmental origin. The superficial (gyral) population (blue dots) encompasses the IWMNs located within the sulcal and gyral areas directly below the cortical gray matter. Those IWMNs are considered the descendants of the subplate cells. The deep population (violet dots) encompasses the IWMNs located in the corpus callosum, anterior commissure, and internal and external capsule. Those IMWNs are thought to reside within the deep WM already in the fetal brain and continue to reside there throughout life. Created with BioRender.com.
Many studies report that, although the IWMNs are distributed throughout the WM, their density is higher directly beneath the cortical GM and decreases gradually toward the center of the gyrus and the sulci in humans and monkeys (Kostovic and Rakic 1980; Sedmak and Judas 2019; Suarez-Sola and others 2009) (Table 1). The density of the IWMNs may also vary depending on the cortical area. For instance, their incidence was found to be higher within the temporal lobe versus the occipital and the frontal lobe, as well as below the primary motor cortex versus the visual cortex (Meyer and others 1992; Rojiani and others 1996). The following densities of the superficial IWMNs were reported in human brain: 2660 ± 153.2 neurons/mm3 in the frontal cortex, 2515 ± 144.2 neurons/mm3 in the cingulate cortex, 1774 ± 121.6 neurons/mm3 in the visual cortex, and 1726 ± 98.89 neurons/mm3 in the temporal cortex (Garcia-Marin and others 2010). However, there was no difference in the density of the IWMN within the deep WM of the examined cortical areas (Garcia-Marin and others 2010). Another interesting observation was made in a very recent study that aimed to investigate whether there is a correlation between the numbers of cortical neurons and IWMNs (Ahmed and others 2024). The authors counted NeuN-immunolabeled neurons in GM and WM within seven coronal sections (50 µm thick, ~1 cm apart), from anterior to posterior of the right hemisphere of the three macaques, and found that for each IWMN, there were ~5 cortical neurons in layer 5 and ~3 cortical neurons in layer 6a (Ahmed and others 2024). The results were consistent independently of the cortical area or location (deep or superficial) of the IWMNs (Ahmed and others 2024). The authors suggested that there may be a link between the alterations in the density of IWMNs and GM cortical neurons of layers 5 and 6a during diseases (e.g., schizophrenia or epilepsy) (Ahmed and others 2024).
Neurochemical Markers Used to Label the IWMNs.
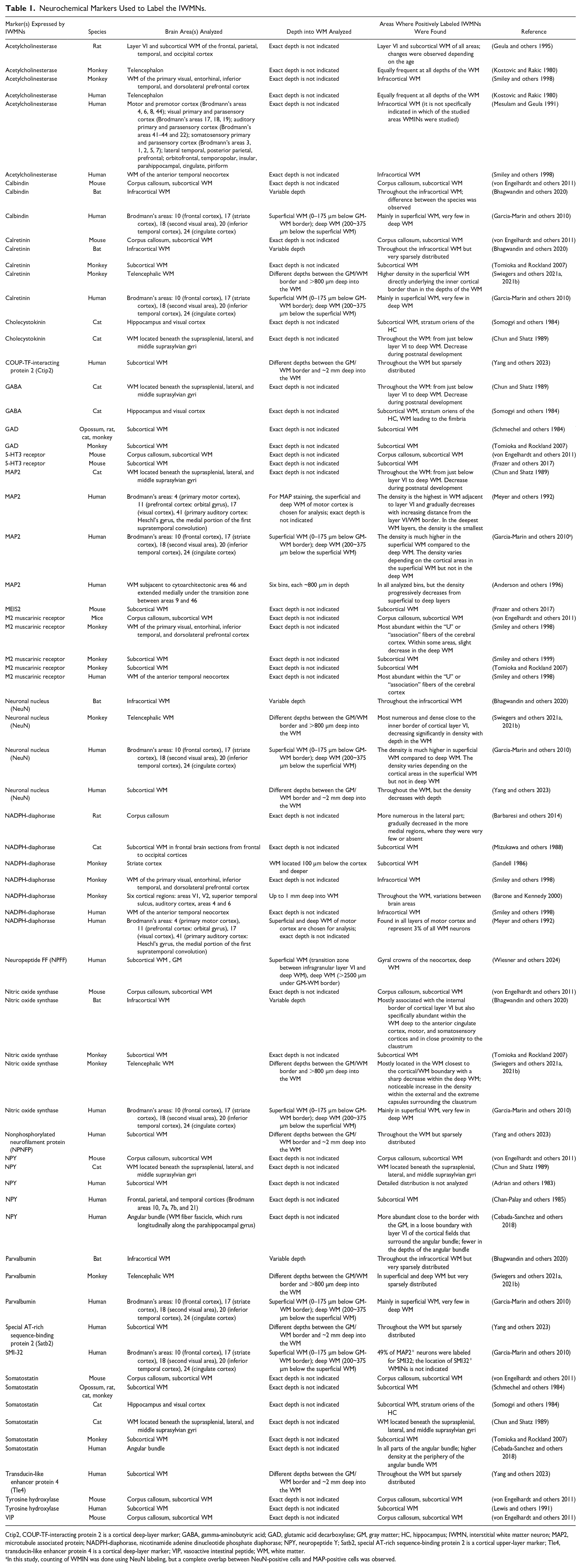
Ctip2, COUP-TF-interacting protein 2 is a cortical deep-layer marker; GABA, gamma-aminobutyric acid; GAD, glutamic acid decarboxylase; GM, gray matter; HC, hippocampus; IWMN, interstitial white matter neuron; MAP2, microtubule associated protein; NADPH-diaphorase, nicotinamide adenine dinucleotide phosphate diaphorase; NPY, neuropeptide Y; Satb2, special AT-rich sequence-binding protein 2 is a cortical upper-layer marker; Tle4, transducin-like enhancer protein 4 is a cortical deep-layer marker; VIP, vasoactive intestinal peptide; WM, white matter.
In this study, counting of WMIN was done using NeuN labeling, but a complete overlap between NeuN-positive cells and MAP-positive cells was observed.
Recently, some alternative hypotheses regarding the origin of IWMNs have been proposed, suggesting that IWMNs may derive from the ventral telencephalon and migrate toward the cortex or that they originate from the subependymal zone located along the wall of the lateral ventricle (Duchatel and others 2019). Interestingly, the rostromedial telencephalic wall (RMTW) of embryonic mouse brain was described as an alternative site for neurogenesis of subplate projection neurons as well as GABAergic interneurons (Pedraza and others 2014). While most of the RMTW-derived cells are destined to become GABAergic interneurons in the cortical layers or become part of the subplate layer during further development, it could be speculated that some of these cells become IWMNs. Further experimental studies are needed to provide more information regarding possible additional origins of the IWMNs.
Morphology of IWMNs
IWMNs in monkeys and humans are morphologically heterogeneous (Judas and others 2010b; Kostovic and Rakic 1980; Meyer and others 1992). Their cell bodies can assume round, elongated, or pyramidal shape, and size may vary significantly (i.e., between 60 and 250 µm2), with an average size of 115 to 130 µm2 (Borra and others 2020; Mortazavi and others 2016). IWMNs may elaborate few or many dendrites and may orient their dendrites parallel to the WM fibers (bipolar) or radiate them in multiple directions (multipolar). Smaller bipolar and fusiform neurons with fewer dendrites tend to be located deeper in the WM, while larger polymorphic cells with several radial dendrites are more common closer to the cortical layers (Garcia-Marin and others 2010; Hoerder-Suabedissen and Molnar 2015).
Morphological complexity of IWMNs may exhibit variation in adult versus developing brains. Age-related alterations have been observed in the area, perimeter, and circularity of IWMNs’ cell soma, as well as in length, numbers, and ramification of their dendrites (Judas and others 2010b). Age-dependent changes in size, shape, and complexity of IWMNs may follow different trends depending on the brain region. For instance, in monkeys, the cell soma area of superficial IWMNs decreases by approximately 20% in frontal and parietal cortical areas but increases by approximately 30% in temporal areas between 6 and 28 years of age (Mortazavi and others 2016). However, no alterations of cell soma area with age were detected for IWMNs located in the deep WM (Mortazavi and others 2016).
Neurochemical markers expressed by IWMNs
Immunohistochemical and morphological studies in different species (mice, rats, bats, cats, monkeys, and humans) show that IWMNs express various neurochemical markers, which are summarized in Table 1. Those markers include 1) acetylcholinesterase, a cholinergic enzyme that hydrolyzes neurotransmitter acetylcholine into acetic acid and choline (Geula and others 1995; Kostovic and Rakic 1980; Mesulam and Geula 1991; Smiley and others 1998); 2) Ca2+-binding proteins: calbindin, calretinin, and parvalbumin (Bhagwandin and others 2020; Garcia-Marin and others 2010; Swiegers and others 2021a, 2021b; Tomioka and Rockland 2007; von Engelhardt and others 2011); 3) peptide hormone cholecystokinin (Chun and Shatz 1989; Somogyi and others 1984); 4) neurotransmitter gamma-aminobutyric acid (GABA) (Chun and Shatz 1989; Somogyi and others 1984); 5) glutamic acid decarboxylase (GAD), an enzyme that produces GABA by catalyzing the decarboxylation of glutamate (Schmechel and others 1984; Tomioka and Rockland 2007); 6) microtubule associated protein (MAP2) that stabilizes growth of microtubules (Anderson and others 1996; Chun and Shatz 1989; Garcia-Marin and others 2010; Meyer and others 1992); 7) M2 muscarinic receptor that is coupled to Gi protein and causes inhibition of adenylyl cyclase upon activation but may also be involved in modulation of G protein-coupled inwardly rectifying potassium channels (Smiley and others 1998; Smiley and others 1999; Tomioka and Rockland 2007; von Engelhardt and others 2011); 8) nitric oxide synthase (NOS), a key enzyme in the nitric oxide synthesis (Bhagwandin and others 2020; Garcia-Marin and others 2010; Swiegers and others 2021a, 2021b; Tomioka and Rockland 2007; von Engelhardt and others 2011), and/or nicotinamide adenine dinucleotide phosphate (NADPH)–diaphorase, which is thought to be an immunochemical marker of NOS activity (Barbaresi and others 2014; Barone and Kennedy 2000; Meyer and others 1992; Mizukawa and others 1988; Sandell 1986; Smiley and others 1998); 9) the peptides: somatostatin (SOM) (Cebada-Sanchez and others 2018; Chun and Shatz 1989; Schmechel and others 1984; Somogyi and others 1984; Tomioka and Rockland 2007; von Engelhardt and others 2011), neuropeptide Y (NPY) (Adrian and others 1983; Cebada-Sanchez and others 2018; Chan-Palay and others 1985; Chun and Shatz 1989; von Engelhardt and others 2011), and vasoactive intestinal peptide (VIP) (von Engelhardt and others 2011); and 10) tyrosine hydroxylase, an enzyme that catalyzes the conversion of the amino acid L-tyrosine to L-3,4-dihydroxyphenylalanine (L-DOPA) (Lewis and others 1991; von Engelhardt and others 2011). Other markers identified in IWMNs include COUP-TF–interacting protein 2 (Ctip2), nonphosphorylated neurofilament protein (NPNFP), special AT-rich sequence-binding protein 2 (Satb2), and transducin-like enhancer protein 4 (Tle4) (Yang and others 2023); 5-HT3 receptor (Frazer and others 2017; von Engelhardt and others 2011); NeuN (Bhagwandin and others 2020; Garcia-Marin and others 2010; Swiegers and others 2021a, 2021b; Yang and others 2023); and SMI-32 (Garcia-Marin and others 2010).
A population of interneurons expressing 5HT3A receptors have been identified in the WM of mice (Table 1). The 5HT3A receptors are coupled to G proteins and mediate neuronal depolarization and excitation. A subpopulation of IWMNs expressing the 5HT3A receptors coexpresses the transcription factor MEIS2 (Table 1), which could be considered a subclass-specific marker of these cells (Frazer and others 2017).
A most recent study discovered that neuropeptide FF (NPFF) labels a population of cells in the superficial WM (and sparsely in the GM) of the neo- and allocortex in humans (Wiesner and others 2024). Microscopic investigation of these cells in the WM revealed them to be pyramidal-like with multipolar cell soma; they were predominantly located in the gyral crowns, stipulating them to be a subset of IWMN.
The expression of specific molecular markers is often associated with either an excitatory or an inhibitory neuronal phenotype, raising the question of whether IWMNs are excitatory (glutamatergic) or inhibitory (GABAergic). Currently, the answer to this question is not fully clear. Based on some studies, IWMNs may be heterogeneous and encompass glutamatergic and GABAergic subpopulations of cells (Connor and others 2011; Kostovic and others 2011; Sedmak and Judas 2021). The GABAergic identity of IWMNs appears plausible because they label positively for several markers of GABAergic interneurons, including GABA, calbindin, calretinin, and parvalbumin (Table 1). However, immunochemical evidence supporting the glutamatergic phenotype of IWMNs seems to be less clear. Studies claiming the glutamatergic identity of IWMNs based their conclusions on positive immunolabeling of IWMNs for MAP2 and SMI32. Although those two molecules are usually considered markers of glutamatergic neurons, they have also been detected in GABAergic neurons (Kelly and others 2019; Lin and others 2015). Immunolabeling for vesicular glutamate transporter 1 (vGLUT1), which is a marker of glutamatergic neurons, was detected around the cell soma or opposite to the dendrites of IWMNs (Garcia-Marin and others 2010; Vigneault and others 2015), indicating that IWMNs may receive glutamatergic synaptic input rather than verifying the glutamatergic identity of IWMNs themselves. Yet, a finding of vGLUT1 “faintly stained cell bodies reminiscent of interstitial cells” in the cerebellar WM (Vigneault and others 2015) may indeed indicate that IWMNs can be glutamatergic. Further, indications of a potential glutamatergic phenotype of IWMNs have emerged from research on temporal lobe epilepsy (see below).
It is also worth considering that IWMNs may be capable of co-releasing GABA and glutamate, in analogy to other neurons that co-transmit information upon co-release of multiple neurotransmitters in the brain (Wallace and Sabatini 2023). Glutamate and GABA, for example, may be co-released by the neurons in the ventral tegmental area, supramammillary nucleus, and entopeduncular nucleus (Kim and others 2022; Root and others 2014; Wallace and Sabatini 2023; Wallace and others 2017). Furthermore, research using a combination of mouse breeding strategies and in situ hybridization found that 30 brain regions have neurons that express the genes required for synaptic vesicle packaging of both glutamate (vGluT2/3;
Cortical Layer 6b Neurons and IWMNs.
In 1922, Lorente de Nó described a layer of “polymorphous” cells in the lower part of neocortical layer 6 in mice and introduced the terminology “layer 6b” for the sublamina containing these cells (Lorente de Nó 1922) (Fig. 3). Layer 6b (L6b) neurons are thought to be the remnants of the subplate that develops during early periods of corticogenesis and partially persists into adulthood (Chen and others 2012; Marx and others 2017; Tolner and others 2012). L6b neurons (in the sensory cortex) express several marker genes that are also expressed in the subplate, including the connective tissue growth factor (Ctgf), the orphan nuclear receptor 1 (Nurr1), neurexophilin 3 (Nxph3), heparin sulfatase (Sulf2), complexin 3 (Cplx3), and monooxygenase Dbh-like 1 (MoxD1). Recently, a detailed review specifically dedicated to the L6b neurons has been published that includes information on their origin, molecular markers, synaptic connectivity, and neuromodulation (Feldmeyer 2023).
We refer to the L6b neurons here to draw the attention of the readers to an ongoing discussion in the literature regarding whether the L6b neurons in rodents may be homologous to the interstitial white matter neurons (IWMNs) in monkeys and humans and to inspire new thinking and new experiments. For example, Ohtaka-Maruyama (2020) refers to L6b neurons as white matter neurons, while the study by Bruguier and others (2020) states, “Whether layer 6b in rodents is homologous to primate subplate or interstitial white matter cells is still debated.”
This discussion and the uncertainties are mainly based on the following facts: 1) both L6b neurons and IWMNs originate from the subplate and are present in the postnatal/adult brain as the remnants of the subplate, even after the subplate as a defined developmental structure disappears; 2) the border between the gray and the white matter may not be always clearly defined, making it potentially challenging to clearly distinguish between the superficial IWMNs and the L6b neurons based on their location, in each specific case; and 3) some evidence points to similarities between the L6b neurons and the IWMNs with regard to their axonal projections. For example, in mice, a subset of cortical L6b neurons selectively innervates higher-order thalamic nuclei (Hoerder-Suabedissen and others 2018; Zolnik and others 2024; Zolnik and others 2020), and a link between the subplate/cortical L6b neurons and psychiatric disorders has been proposed, suggesting that altered connectivity involving these neurons contributes to the pathophysiological mechanism of autism or schizophrenia (Eastwood and Harrison 2005; Hoerder-Suabedissen and Molnar 2013; Nagode and others 2017). In turn, IWMNs in adult rhesus monkeys were also found to project axons to the thalamus, and many studies proposed that IWMNs play a role in schizophrenia and autism (see text for details).
Currently, the question of possible homology/similarity between the L6b neurons and the IWMNs remains unresolved. Further studies performing a comprehensive comparison of the IWMNs versus the L6b neurons and their axonal projections neurons in rodents and primates would be very useful.
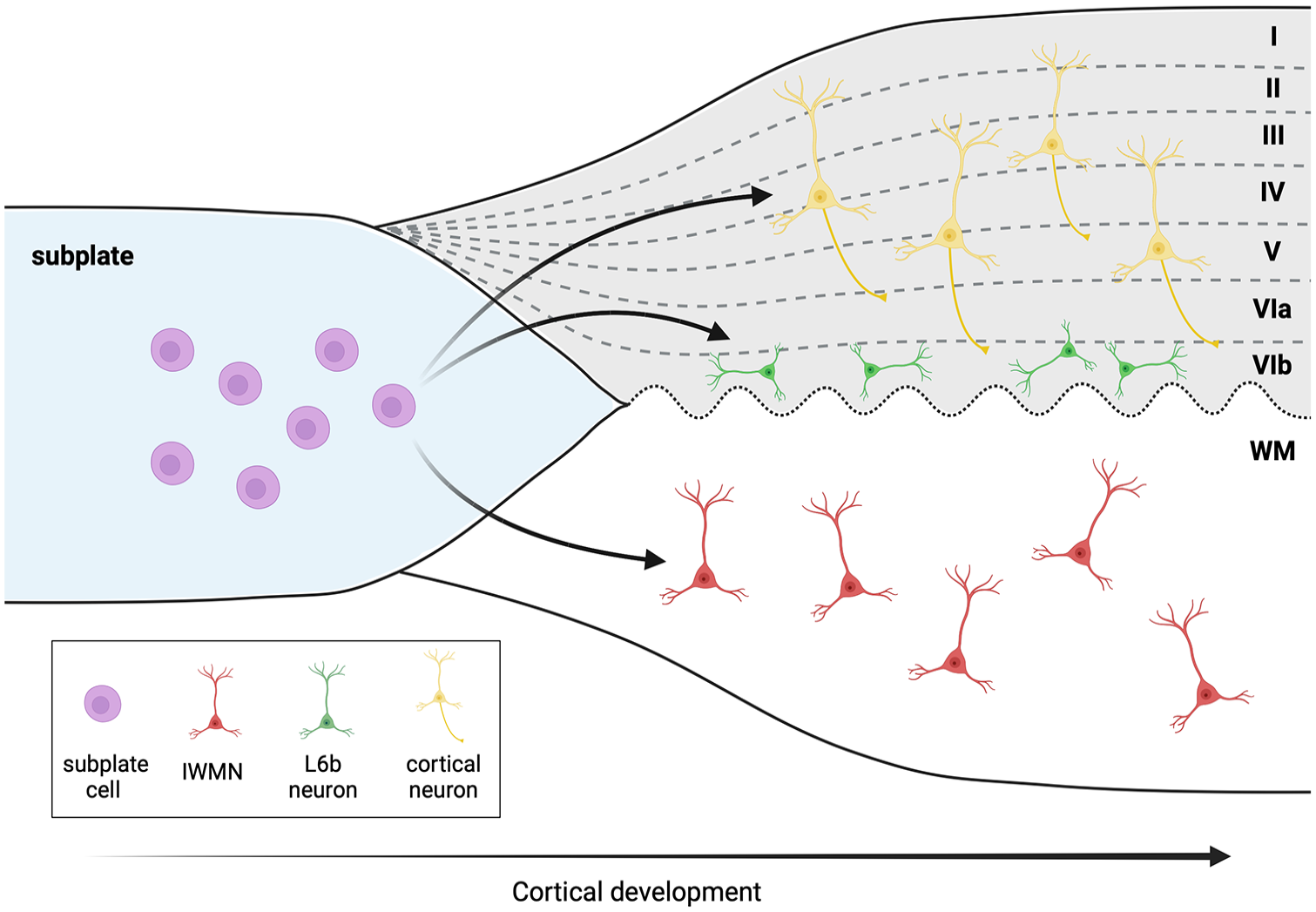
Layer 6b neurons and the interstitial white matter neurons (IWMNs). During cortical development, the IWMNs in the white matter (WM) and the layer 6b neurons (L6b) in the cortex persist as a remnant of the subplate cells. It is currently debated in the literature whether both cell types share the same common progenitor cells and whether the L6b neurons in rodents may be homologous to the IWMNs in monkeys and humans. Some studies refer to layer L6b neurons as cells located between the gray matter (GM) and the WM. The waved dotted line indicates that the border between the WM and the GM is not always clearly distinguishable. Created with BioRender.com.
Physiological role of IWMNs in the brain
Current knowledge regarding physiology and functions of IWMNs in the adult brain is very limited. Major reasons for this are 1) difficulty to target IWMNs in adult brains for invasive and especially noninvasive functional assays (e.g., patch-clamp or multielectrode array recordings in living animals, electroencephalography in humans) because of their relative scarcity and/or deep location within the brain and 2) lack of knowledge regarding specific molecules exclusively expressed in IWMNs (and not in other neurons) and could be used to precisely target and manipulate IWMNs.
Below we highlight three possible functions of IWMNs (Fig. 4) that have been debated in the literature: 1) regulation of excitation, inhibition, and neuronal wiring; 2) regulation of microcirculation; and 3) regulation of myelination. For detailed information, consider the more comprehensive reviews (Kubo 2020; Luhmann and others 2018; Suarez-Sola and others 2009).
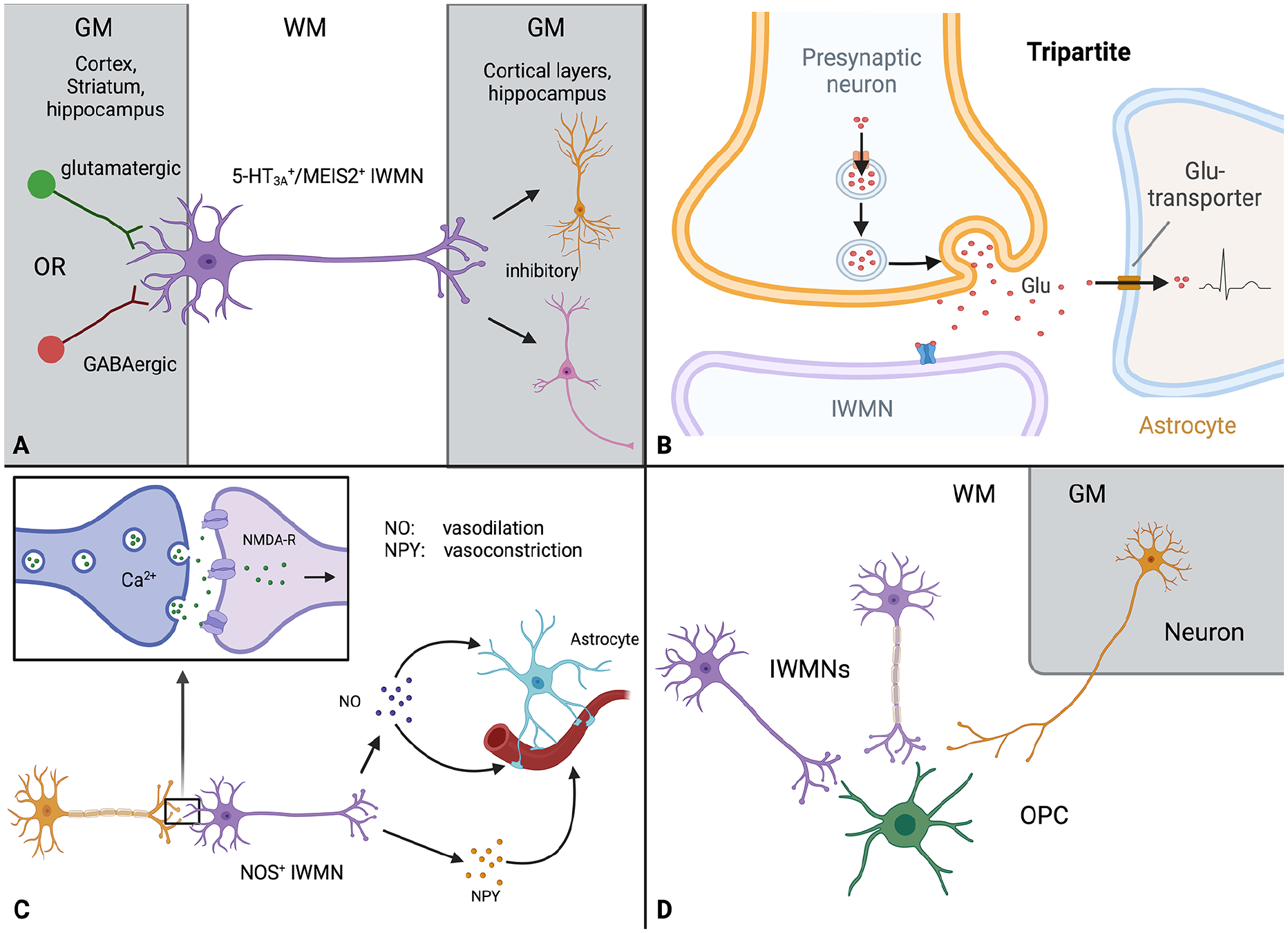
Roles and functions of interstitial white matter neurons (IWMNs) in the brain. (A) Suggested synaptic wiring of IWMNs. Studies have indicated that IWMNs receive glutamatergic and GABAergic synaptic input from the cortex, striatum, and hippocampus. A subtype of 5-HT3A and MEIS2-expressing IWMNs was shown to have dendrites in the white matter as well as to connect with pyramidal cells and interneurons in the gray matter (cortical layers and hippocampus) and are inhibitory. (B) Synapses on IWMNs may be tripartite, involving a presynaptic neuron, a postsynaptic IWMN, and an astrocyte. These tripartite synapses may mediate fast signaling within neuronal and neuron-glia circuits connecting gray and white matter. (C) Possible role of IWMNs in the regulation of microcirculation. Some IWMNs show positive labeling for NOS and NPY. In the corpus callosum of adult monkeys, IWMNs are structurally associated with blood vessels and may be involved in the regulation of the microcirculation, either directly through NO and/or NPY release or via the astrocytes. (D) IWMNs and myelination. Similar to what occurs in cortical neurons, the axons of some IWMNs may be myelinated while the axons of others may not. IWMNs may form synapses with white matter oligodendrocyte progenitor cells (OPCs), similar to long-range projection neurons from other brain areas. We could not find information regarding myelination of IWMNs and IWMN-OPC synapses. Created with BioRender.com.
IWMNs in regulation of excitation, inhibition, and neuronal wiring
Synaptic wiring of IWMNs and their integration into the neuronal circuits
Studies using intracellular tracers demonstrated that dendrites and/or axons of IWMNs may be located in the adjacent WM, cortex, striatum, and hippocampus of juvenile or adult mice, rats, ferrets, cats, and monkeys (Borra and others 2020; Clancy and others 2001; Higo and others 2007; Luth and others 1995; Meyer and others 1992; Rockland 1985; Tomioka and others 2005; von Engelhardt and others 2011).
Electrophysiological studies of IWMNs in mice focused on a subpopulation of IWMNs located in the corpus callosum and expressing the ionotropic serotonin receptor (5-HT3A) (Frazer and others 2017; von Engelhardt and others 2011). Some of these 5-HT3A–expressing IWMNs (~30%) were positive for calretinin or VIP and very rarely for calbindin, NPY, or SOM but not for parvalbumin. In contrast to the 5-HT3A neurons in the GM, the 5-HT3A–expressing IWMNs express transcription factor MEIS2 (Frazer and others 2017). The 5-HT3A–expressing IWMNs have their dendrites in the WM, striatum, and hippocampus and project their axons to the pyramidal cells and interneurons located in the cortex (Frazer and others 2017; von Engelhardt and others 2011). With regard to their function, the 5-HT3A–expressing IWMNs are inhibitory: they provide feedforward inhibition of principal neurons and interneurons in the cortex, and they receive glutamatergic and GABAergic inputs from cortical neurons (Fig. 4A) (Frazer and others 2017; von Engelhardt and others 2011). The 5-HT3A receptors in IWMNs seem to be located extra- or presynaptically and may regulate neurotransmitter release (von Engelhardt and others 2011).
Electrophysiologic recordings of IWMNs in juvenile rats showed that IWMNs exhibited short-duration action potentials followed by distinct fast after-hyperpolarizations (Clancy and others 2001). In response to sustained (300–800 ms) depolarizing pulses, IWMNs showed little or no spike-frequency adaptation, and their firing frequency (53 Hz) was higher than that in pyramidal neurons (31 Hz) (Clancy and others 2001; Torres-Reveron and Friedlander 2007). Biocytin labeling of electrophysiologically characterized IWMNs demonstrated that they project axons to the adjacent WM, to all cortical layers, and to the hippocampus and that axons of IWMNs have synaptic bouton-like swellings (Clancy and others 2001). Dendrites of IWMNs were found in the WM, cortical layers V and VI, and the hippocampus (Clancy and others 2001; Torres-Reveron and Friedlander 2007). IWMNs receive GABAergic synaptic input, as well as glutamatergic synaptic input mediated by AMPA/kainate receptors; the contribution of NMDA receptors to the synaptic input remains less clear (Torres-Reveron and Friedlander 2007).
No electrophysiologic recordings from IWMNs have been performed in slices from the human brain. However, immunolabeling for vGLUT and vesicular GABA transporter (vGAT) demonstrated that IWMNs in the superficial and deep human WM are innervated by glutamatergic and GABAergic axon terminals (Garcia-Marin and others 2010). Electron microscopy further revealed that both glutamatergic and GABAergic axon terminals are located around the soma and the dendrites of the IWMNs. Only symmetric (presumably GABAergic) synapses were identified perisomatically, while both symmetric (presumably GABAergic) and asymmetric (presumably glutamatergic) synapses were found on the dendrites of the WM cells (Garcia-Marin and others 2010).
Taken together, electrophysiologic and morphologic data show that IWMNs in the rodent and human brain are integrated into the neuronal circuits. Furthermore, some evidence suggests that neuronal circuits involving IWMNs are plastic and dynamic. For instance, in rodent brain slices, synaptic inputs to IWMNs undergo short- and long-term plasticity, indicating that synaptic strength at IWMN synapses can be modulated by neuronal activity. Specifically, upon paired-pulse stimulation of axons contacting the IWMNs, the synaptic inputs to IWMNs show short-term facilitation, while in response to 1 Hz conditioning, they show long-term depression (Torres-Reveron and Friedlander 2007). In the hippocampus and cerebellum, long-term depression is thought to be an important mechanism behind learning and memory. Although the functional significance of synaptic plasticity at synapses onto IWMNs remains unknown, a recent study using human brain samples proposed that somatostatin- and NPY-positive IWMNs may contribute to the basic wiring of the hippocampus through which autobiographical and spatial memories are stored in the infant brain (Cebada-Sanchez and others 2018).
Another interesting but unresolved question is whether integration of IWMNs into the neuronal circuits may be modulated by external stimuli. It has been proposed that during integration of subplate cells into the cortical circuitry, the rearrangement of their neurites is modulated by the sensory activity. Specifically, the neonatal ablation of a whisker row prevented the neurites of the subplate cells to change from an intra- to extra-barrel pattern in the region corresponding to whisker deprivation (Molnar and others 2020; Pinon and others 2009). The dynamics of neuronal microcircuits within the barrel cortex are well known from many studies, with the important findings that plasticity differs within the local populations of cortical neurons, and alterations of activity result from sensory deprivation and learning (Margolis and others 2014). But more and more evidence is accumulating that external behavioral stimuli modulate tissue properties not only of the GM but also of the WM and that learning correlates with structure and/or function of the WM. For instance, most recent studies found that WM structure predicts language performance and learning success (Sanchez and others 2023), and WM fiber tract of the cortical visual system is enhanced in visual artists (Hong and others 2023). A detailed discussion on this topic is outside of the scope of this review, but broader literature on the topic and many interesting reviewers are available. It would be certainly exciting to discover the role of IWMNs during WM plasticity and to find out how various behavioral stimuli modulate their structural and functional properties as well as their connectivity.
Interaction of IWMNs with astrocytes: are synapses onto IWMNs tripartite?
Many synapses in the brain are tripartite because they include not only pre- and postsynaptic neuronal elements but also the smallest processes of astrocytes (Araque and others 1999). Astrocytes perform multiple functions at synapses, including removal of neurotransmitters and release of molecules, which may modulate synaptic function (Rose and others 2017). Astrocytes spatially segregate synapses (to a different extent in various brain regions), ensuring their structural and functional independence from neighboring synapses and limiting neurotransmitter spillover (Tzingounis and Wadiche 2007), and thus shape synaptic transmission and influence information processing in the brain.
Astrocytes reside also in the WM, but their structural and functional relationships with IWMNs remain unknown. WM astrocytes do not show a sponge-like appearance that is typical for GM astrocytes and is determined by numerous tiny processes, many of which are located at synapses (Fattorini and others 2017; Kohler and others 2021; Matyash and Kettenmann 2010; Oberheim and others 2009; Raff and others 1983; Reichenbach 2005). However, experiments using dye-filling of individual WM astrocytes demonstrate that they possess some very thin processes (Fig. 5). WM astrocytes express many similar (or analogous) proteins and perform many similar functions as GM astrocytes (Bugiani and others 2022; Kohler and others 2021). For instance, WM astrocytes express functional glutamate transporters: they show glutamate transporter currents in response to photolysis of MNI-d-aspartate, which are inhibited in the presence of the glutamate transporter antagonist TBOA (Regan and others 2007), and they also show glutamate-induced sodium transients sensitive to antagonists of glutamate transporters (Moshrefi-Ravasdjani and others 2017). Hence, it is plausible that WM astrocytes extend processes to synapses formed on IWMNs and are involved in the modulation of synaptic function in the WM Fig. 4B.
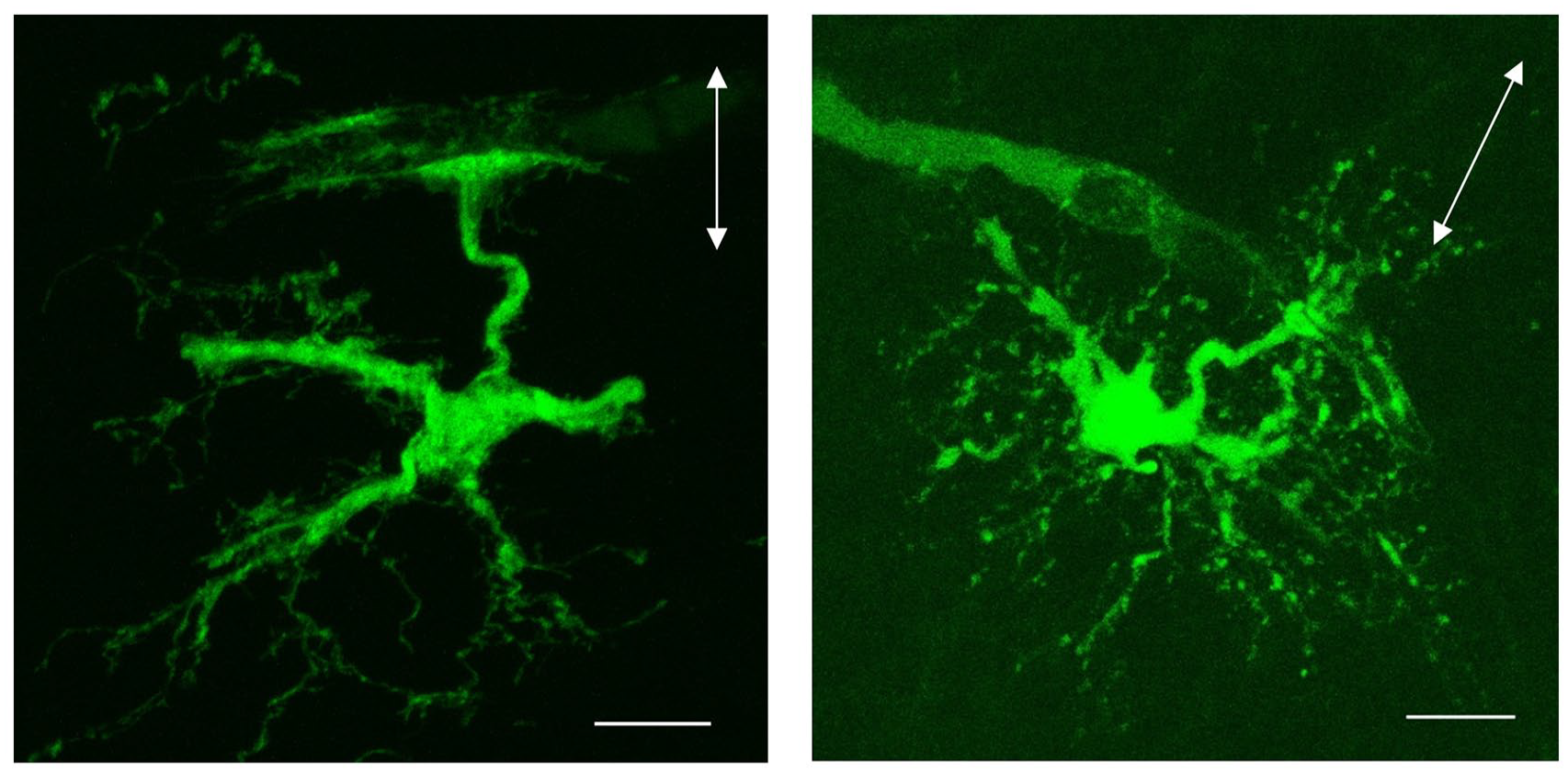
Examples showing a morphologic appearance of fibrous astrocytes in the white matter. The images represent maximum intensity projections of astrocytes recorded in the corpus callosum, in slices prepared from P7 and P13 mouse brains. The cells were filled with Lucifer Yellow during patch-clamp recordings. Arrows indicate the orientation of the fibers in the corpus callosum; arrowheads indicate astrocytic end-feet on blood vessels. Scale bars 10 µm.
IWMNs in regulation of microcirculation
Some IWMNs are positive for NOS and NADPH-diaphorase (Barbaresi and others 2014; Clancy and others 2001; Rockland and Nayyar 2012). NOS is an enzyme involved in the synthesis of nitric oxide, which regulates blood flow through vasodilatation. Activation of NOS is induced by the influx of calcium (e.g., via NMDA receptors) and requires NADPH as a co-substrate. In the corpus callosum of adult monkeys, NOS/NADPH-diaphorase–expressing IWMNs are structurally associated with blood vessels (Rockland and Nayyar 2012), suggesting that they may release nitric oxide and regulate blood flow directly or via activation of astrocytes (Fig. 4C), as has been proposed for GM neurons (Attwell and others 2010; Tomioka and Rockland 2007). Interestingly, some NOS/NADPH-expressing IWMNs are positive for neuropeptide Y, a potent vasoconstrictor (Suarez-Sola and others 2009). Probably, IWMNs release nitric oxide and NPY from different compartments (cell soma vs. axons), potentially contributing to the coupling of microvessels to neuronal activity (Suarez-Sola and others 2009) (Fig. 4C).
Interaction of IWMNs with oligodendrocyte lineage cells and possible role of IWMNs in regulation of myelination
Neurons and oligodendrocyte lineage cells are involved in multiple modes of interaction with each other, in GM and WM of the brain. Oligodendrocytes myelinate axons of many neurons, ensuring fast propagation of action potentials and providing tropic support for axons (Thornton and Hughes 2020). Neurons can modulate the proliferation and differentiation of developing myelinating cells, as well as the myelination process itself, through various mechanisms, thereby contributing to formation and functionality of neuronal circuits (Thornton and Hughes 2020). The density of oligodendroglial cells in the WM is high, and hence it is intriguing what the nature of interactions between oligodendrocytes and IWMNs is and whether axons of IWMNs are myelinated.
Are IWMNs involved in synaptic signaling with oligodendrocyte progenitor cells?
In the brain and spinal cord, fast synaptic signaling exists between neurons and oligodendrocyte progenitor cells (OPCs) (Kula and others 2019; Thornton and Hughes 2020). Initially, synapses between neurons and oligodendrocyte lineage cells have been described in the human cerebral cortex using electron microscopy and were proposed to be cholinergic (Tusques and others 1976). Later, electrophysiologic studies demonstrated that neuron-OPC synapses are glutamatergic and/or GABAergic depending on the brain region. In the WM, axon-OPC synapses have been described in the corpus callosum, the optic nerve, and the cerebellar WM of rodents, as well as in the human fimbria (Gallo and others 2008; Karadottir and others 2008; Kukley and others 2007; Ziskin and others 2007; Zonouzi and others 2015). The origin of neurons providing synaptic signaling to the WM OPCs can be inferred based on the general anatomic evidence regarding brain connectivity: for optic nerve OPCs, retinal ganglion neurons; for callosal OPCs, pyramidal neurons located in cortical layers II to III and V, as well as in the thalamus; for the cerebellar WM OPCs, the cerebellum itself, as well as various other brain and spinal cord regions; and for OPCs in the human fimbria, the hippocampal neurons. Optogenetic and trans-synaptic tracing studies verified that neurons establishing synapses with callosal OPCs are located in the cerebral cortex and the thalamus (Gibson and others 2014; Mount and others 2019; Ortiz and others 2019). It is intriguing whether IWMNs also form synapses with OPCs (Fig. 4D) and whether axon-OPC synaptic input in the WM represents a mixture of two inputs: one from the axons of long-range projection neurons located in the GM and another one—from the axons of local IWMNs. To test for synaptic signaling between IWMNs and WM OPCs, it will be necessary to perform paired recordings from the visually identified and connected two cell types, as has been done previously to study neuron-OPC synapses in the cortex (Orduz and others 2015). Alternatively, a photosensitive ion channel channelrhodopsin-2 (ChR2) may be expressed in IWMNs, and patch-clamp recordings of OPCs may be combined with optogenetic stimulation of IWMNs. Such studies would certainly advance our understanding of axon-glia circuitry in the brain.
Do IWMNs interact with mature myelinating oligodendrocytes and influence myelination?
Two major questions are of interest here: 1) whether axons of IWMNs are myelinated and how this myelination is regulated and 2) whether IWMNs can influence the function of oligodendrocytes, myelinating their own or neighboring axons. We could not find studies addressing these questions, and older studies performing electron microscopy (Kostovic and Rakic 1980; Valverde and Facal-Valverde 1988) have not specifically investigated the myelination of IWMN axons, but some findings allow putting forward a few hypotheses.
Axons of some GM neurons projecting through the WM are myelinated while axons of other neurons are not. The same may hold true for the axons of IWMNs (Fig. 4D). The rules determining this selective myelination of some axons versus the others are not fully understood. Interestingly, for a long time, the importance of myelination has been acknowledged mainly for neurons with long axons (e.g., glutamatergic long-range projection neurons), while myelination of local neurons (e.g., GABAergic interneurons) has come to light only recently. Within the population of GABAergic interneurons, axons of parvalbumin-positive interneurons are more frequently myelinated than axons of VIP- or SOM-positive cells (Benamer and others 2020; Call and Bergles 2021; Micheva and others 2016). If this holds true also for IWMNs, it is possible that axons of many GABAergic IWMNs stay unmyelinated because fewer IWMNs label positively for parvalbumin than for other markers of GABAergic neurons (von Engelhardt and others 2011).
Can IMWNs influence myelination of other axons? SOM-positive IWMNs may release SOM and thus impact development and/or function of oligodendrocyte lineage cells. Rodent oligodendrocytes express receptors for SOM, which are involved in the regulation of K+-channels in these cells in vitro (Karschin 1995; Marques and others 2016). In the human brain, an inverse relationship between the density of SOM receptors and the myelination process in the fiber tracts has been described (Carpentier and others 1999), although it is not known whether the two processes are causally related. In the mouse brain WM, OPCs receive synaptic input from SOM-positive interneurons located in the cortex (Mount and others 2019) and may also receive input from SOM-positive IWMNs. This input may be involved in the regulation of OPC development and function.
NPY is another molecule that IWMNs express and may release to regulate myelination (Cebada-Sanchez and others 2018). Reduced numbers of oligodendrocytes and myelin basic protein (MBP) were detected in mice treated with NPY-receptor 1–specific antagonist, while injection of recombinant NPY resulted in a higher number of myelinated axons and increased MBP expression (Hashimoto and others 2011). In NPY knockout mice, proliferation of cells in the subcallosal zone, which generates oligodendrocytes, was decreased (Laskowski and others 2007). These findings, although not directly focusing on IWMNs, make it tempting to suggest that NPY released by IWMNs may influence the development and function of oligodendrocyte lineage cells in the WM.
IWMNs during aging
In humans, WM volume decreases, starting from the age of ~60 years, and aging WM becomes more susceptible to age-related conditions (e.g., stroke, traumatic brain injury, and neurodegeneration) (Liu and others 2016; Liu and others 2017; Pakkenberg and Gundersen 1997; Seidler and others 2010). Alterations in IWMN may contribute to these conditions. Studies in monkeys showed that both deep and superficial IWMNs undergo morphometric changes with age: cell soma area and perimeter were smaller and had a more circular shape in older versus young monkeys (Mortazavi and others 2016). Furthermore, IWMNs of the deep WM moderately (although not statistically significantly) decreased in density with age (Mortazavi and others 2016). Given putative involvement of IWMNs in cortical circuitry and vascular regulation, morphologic age-related alterations in IWMNs may have an impact on their function in monkeys and also in humans.
IWMNs during diseases
Neuropathologic alterations in IWMNs occur during diseases. The majority of the studies available on this topic to date focus on schizophrenia and autism spectrum disorders (ASDs), but there are also reports regarding IWMNs in multiple sclerosis, epilepsy, multiple system atrophy, and Alzheimer’ disease. Discovery of these alterations is very important because they emphasize that WM damage contributes to the pathophysiologic mechanism of many diseases, which for a long time have been considered GM diseases.
IWMNs in schizophrenia and autism spectrum disorders
Altered development of cortical trajectory and connectivity caused by genetic and/or environmental factors is considered a key mechanism in the pathophysiology of schizophrenia and ASDs (Calderoni and others 2016; Chisholm and others 2015; Crippa and others 2016; Hutsler and Casanova 2016; Hutsler and Zhang 2010; Stachowiak and others 2013). Changes in the WM microstructure occur in both schizophrenia and ASDs (Koshiyama and others 2020).
Many studies investigating the postmortem brain WM of patients with schizophrenia found an increase in IWMN density (Akbarian and others 1993a, 1993b; Akbarian and others 1996; Anderson and others 1996; Connor and others 2009; Duchatel and others 2019; Eastwood and Harrison 2003, 2005; Ikeda and others 2004; Joshi and others 2012; Kirkpatrick and others 1999; Kirkpatrick and others 2003; Kubo 2020; Rioux and others 2003; Yang and others 2011) (Table 2), while other studies found no change or reduction in the count or density of IMWNs (Beasley and others 2002; Beasley and others 2009; Bertram and others 2007; Duchatel and others 2019; Hoistad and others 2013; Kubo 2020; McFadden and others 2016; Molnar and others 2003; Tsai and others 2020) (Table 2). Diverse types of alterations in the deep versus superficial WM were also described (Table 2). The IWMNs were usually investigated at a depth of up to 4.8 mm beneath the GM and were quantified using the following markers: NADPH, NADPHd, MAP2, SMI, NPY, NeuN, NRG-1α, cresyl-violet, SOM, GAD65/67, and Nissl (Table 2).
Alterations in IWMNs in Schizophrenia.
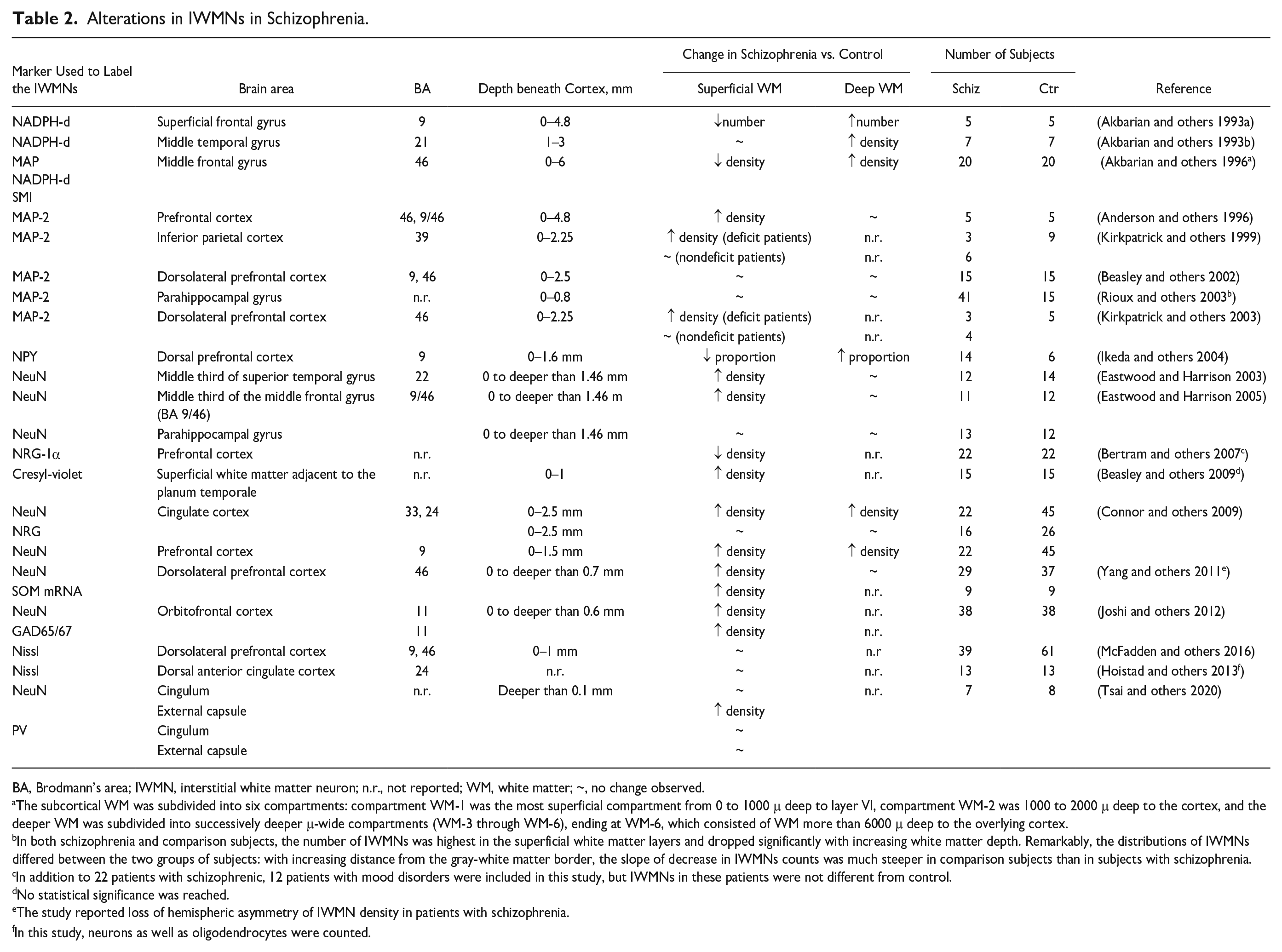
BA, Brodmann’s area; IWMN, interstitial white matter neuron; n.r., not reported; WM, white matter; ~, no change observed.
The subcortical WM was subdivided into six compartments: compartment WM-1 was the most superficial compartment from 0 to 1000 μ deep to layer VI, compartment WM-2 was 1000 to 2000 μ deep to the cortex, and the deeper WM was subdivided into successively deeper μ-wide compartments (WM-3 through WM-6), ending at WM-6, which consisted of WM more than 6000 μ deep to the overlying cortex.
In both schizophrenia and comparison subjects, the number of IWMNs was highest in the superficial white matter layers and dropped significantly with increasing white matter depth. Remarkably, the distributions of IWMNs differed between the two groups of subjects: with increasing distance from the gray-white matter border, the slope of decrease in IWMNs counts was much steeper in comparison subjects than in subjects with schizophrenia.
In addition to 22 patients with schizophrenic, 12 patients with mood disorders were included in this study, but IWMNs in these patients were not different from control.
No statistical significance was reached.
The study reported loss of hemispheric asymmetry of IWMN density in patients with schizophrenia.
In this study, neurons as well as oligodendrocytes were counted.
Studies investigating postmortem brain tissue of patients with autism or ASDs reported an increased number of superficial and deep IWMNs (Avino and Hutsler 2010; Bailey and others 1998; Casanova and others 2006; Duchatel and others 2019; Kubo 2020; Oblak and others 2011; Simms and others 2009) (Table 3). Brains of patients with ASD also showed irregularities of the GM-WM boundaries probably reflecting altered neuronal migration and/or higher numbers of neurons remaining in the WM (Avino and Hutsler 2010).
Alterations in IWMNs in ASD.
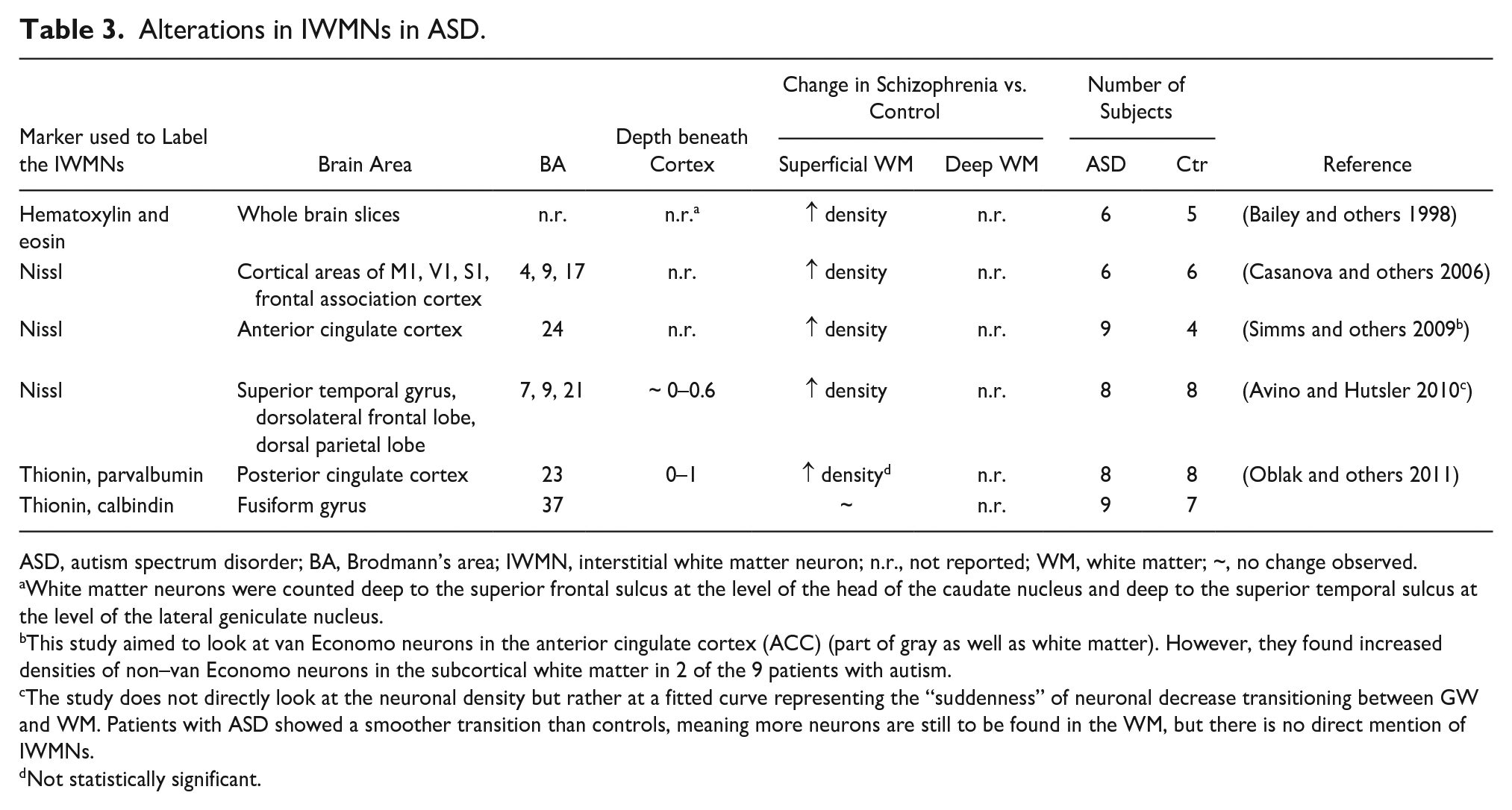
ASD, autism spectrum disorder; BA, Brodmann’s area; IWMN, interstitial white matter neuron; n.r., not reported; WM, white matter; ~, no change observed.
White matter neurons were counted deep to the superior frontal sulcus at the level of the head of the caudate nucleus and deep to the superior temporal sulcus at the level of the lateral geniculate nucleus.
This study aimed to look at van Economo neurons in the anterior cingulate cortex (ACC) (part of gray as well as white matter). However, they found increased densities of non–van Economo neurons in the subcortical white matter in 2 of the 9 patients with autism.
The study does not directly look at the neuronal density but rather at a fitted curve representing the “suddenness” of neuronal decrease transitioning between GW and WM. Patients with ASD showed a smoother transition than controls, meaning more neurons are still to be found in the WM, but there is no direct mention of IWMNs.
Not statistically significant.
Several hypotheses may explain the mechanisms triggering alterations in the IWMNs during schizophrenia and ASDs (Kubo 2020; Serati and others 2019). The major among them are 1) failure of migration of excitatory and inhibitory neocortical neurons; 2) deregulation of apoptosis of subplate neurons, leading to their increased survival and, subsequently, to a higher count of IWMNs; and 3) elevated adult neurogenesis and dysregulation of neuron generation by the outer radial glial cells (Kubo 2020; Serati and others 2019). Changes in neuronal migration and/or apoptotic death in the brains of patients with schizophrenia or ASDs may be triggered by genetic and/or environmental factors. Many genes are involved in deregulation of these processes in patients with psychiatric disorders (Bourgeron 2016; Escudero and Johnstone 2014; Harkin and others 2017), and reelin (
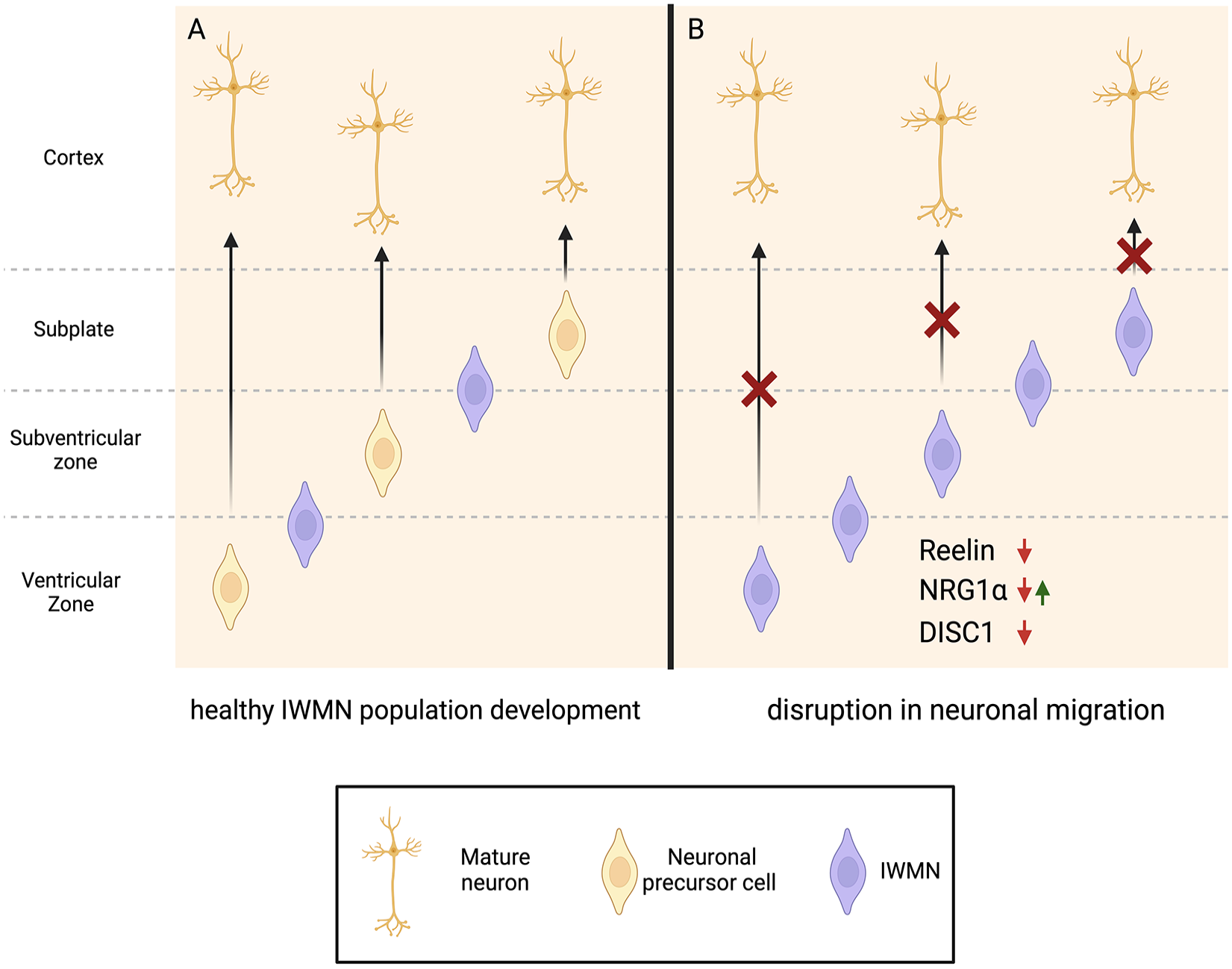
Hypothetical scheme suggesting the influence of genetic factors on interstitial white matter neuron (IWMNs) in schizophrenia. (A) In healthy individuals, only some neuronal precursor cells fail to migrate to the cortex, eventually maturing to IWMNs. (B) Down-regulation of Reelin and DISC1, as well as changes (either up- or down-regulation) in NRG1, could potentially lead to disruption of migration and development of neuronal precursor cells, leading to an increased density of IWMNs in the (sub)ventricular zone and subplate (which becomes the white matter in later development) that are associated with schizophrenia. Created with BioRender.com.
Reelin is a large extracellular matrix glycoprotein that controls cell-cell interactions and regulates neuronal migration in the developing brain.
Another gene of interest in schizophrenia and ASD is NRG-1, the epidermal growth factor family member that interacts with the ErbB receptor (Shi and Bergson 2020; Stefansson and others 2004) (Fig. 6). In mice, inhibition of NRG1-ErbB4 signaling leads to behavioral alterations consistent with neuropsychiatric disorders, as well as to changes in oligodendrocyte number, reduced myelin thickness, slower axonal conduction velocity, and morphologic changes in the corpus callosum (Roy and others 2007). Alterations in the levels of specific NRG-1 isoforms result in schizophrenia-like behavior (Dominguez and others 2018). In patients with schizophrenia, either an increase or a decrease in the level and/or signaling of NRG-1/Erb4B was reported, while NRG-1 single-nucleotide polymorphisms risk allele was associated with lower WM volume in schizophrenia (Cannon and others 2012). The density of NRG-1–expressing IWMNs was reduced in the prefrontal cortex of patients with schizophrenia (Bertram and others 2007). Considering that NRG-1 is a motility factor in the epidermis, the altered NRG-1 expression or function may affect migration and correct placement of IWMNs (Bertram and others 2007). Increased serum levels of NRG-1 were found in patients with ASD (Esnafoglu 2018), suggesting that it may be important in ASD pathogenesis.
The
The 22q11.2 microdeletion syndrome (velocardiofacial, DiGeorge syndrome) is worth mentioning here as another genetic alteration linked to ASD and psychosis (Jalbrzikowski 2021). A case study in the postmortem brain tissue of an infant with this syndrome detected an increased number of IWMNs, suggesting that genetic variants may have an impact on the increased IWMN densities (Wu and others 2014).
Among the nongenetic and/or environmental factors causing an increase in IWMNs in schizophrenia or ASD are fetal or neonatal hypoxia/ischemia, inflammation during pregnancy, maternal immune activation, and premature birth. Analysis of postmortem brain tissue from preterm infants showed WM injury and an increased number of neurons in the WM presumably caused by altered migration of cells through the WM to the cortex (Kubo and others 2017). In animals, immune activation during pregnancy leads to increased density of IWMNs (Duchatel and others 2016). Hence, inflammation triggered by infections during pregnancy in humans was proposed to preclude the successful migration of developing neurons from the subplate to the cortex and cause them to remain in the WM (Duchatel and others 2019; Kubo 2020).
An alternative, or additional, cause underlying the dysregulated numbers of IWMNs in schizophrenia may be the increased generation of neurons from a special type of nonepithelial radial glia (Connor and others 2011).
Why and how do altered numbers of IWMNs may contribute to the pathophysiologic mechanism of schizophrenia and ASDs? The IWMNs may influence functional connectivity between the limbic pathways and the prefrontal cortex because they are distributed between the axons of long-range projection neurons running through the WM (Kostovic and others 2011). The GABAergic IWMNs, if increased in number during schizophrenia, may contribute to the augmented inhibition of pyramidal neurons in the prefrontal cortex to which they are connected (Kostovic and others 2011). Notably, several studies in adult rhesus monkeys indicated that IWMNs project axons to the thalamus, based on their findings of sparse IWMNs labeling in the cingulate, prefrontal cortex, and supplementary motor area upon retrograde tracer injections into medial dorsal or ventral anterior/ventral lateral thalamic nucleus (Giguere and Goldman-Rakic 1988). Abnormalities in thalamocortical connectivity have been found in many studies as a robust feature of schizophrenia (Abram and others 2023). Altered numbers of IWMNs in schizophrenia may contribute to those abnormalities with an altered number or strength of their connections within the thalamocortical circuitry.
IWMNs in multiple system atrophy
Multiple system atrophy (MSA) is a rare progressive neurodegenerative disease with unknown etiology that usually occurs in adults. Major clinical features of this disease are cerebellar ataxia, Parkinsonism, corticospinal dysfunction, and autonomic failure. A significant loss of IWMNs has been found in patients with MSA, compared to neurologically healthy control subjects, using stereological cell counting in postmortem brain WM (Nykjaer and others 2017). In vivo structural magnetic resonance imaging (MRI) and diffusion tensor imaging study detected widespread alterations of WM microstructure in patients with MSA; moreover, oligodendrocytes in these patients accumulate α-synuclein, forming glial cytoplasmic inclusions (Del Campo and others 2021). Damage to myelinating cells may lead to demyelination and axonal degeneration, and it may trigger astrogliosis and microgliosis. The cascade of pathologic changes in glia may contribute to damage and loss of IMWNs (Fig. 7). In contrast, it is possible that alterations in glial cells and loss of IWMNs in patients with MSA are not intercausal processes but occur in parallel to each other and perhaps aggravate each other.
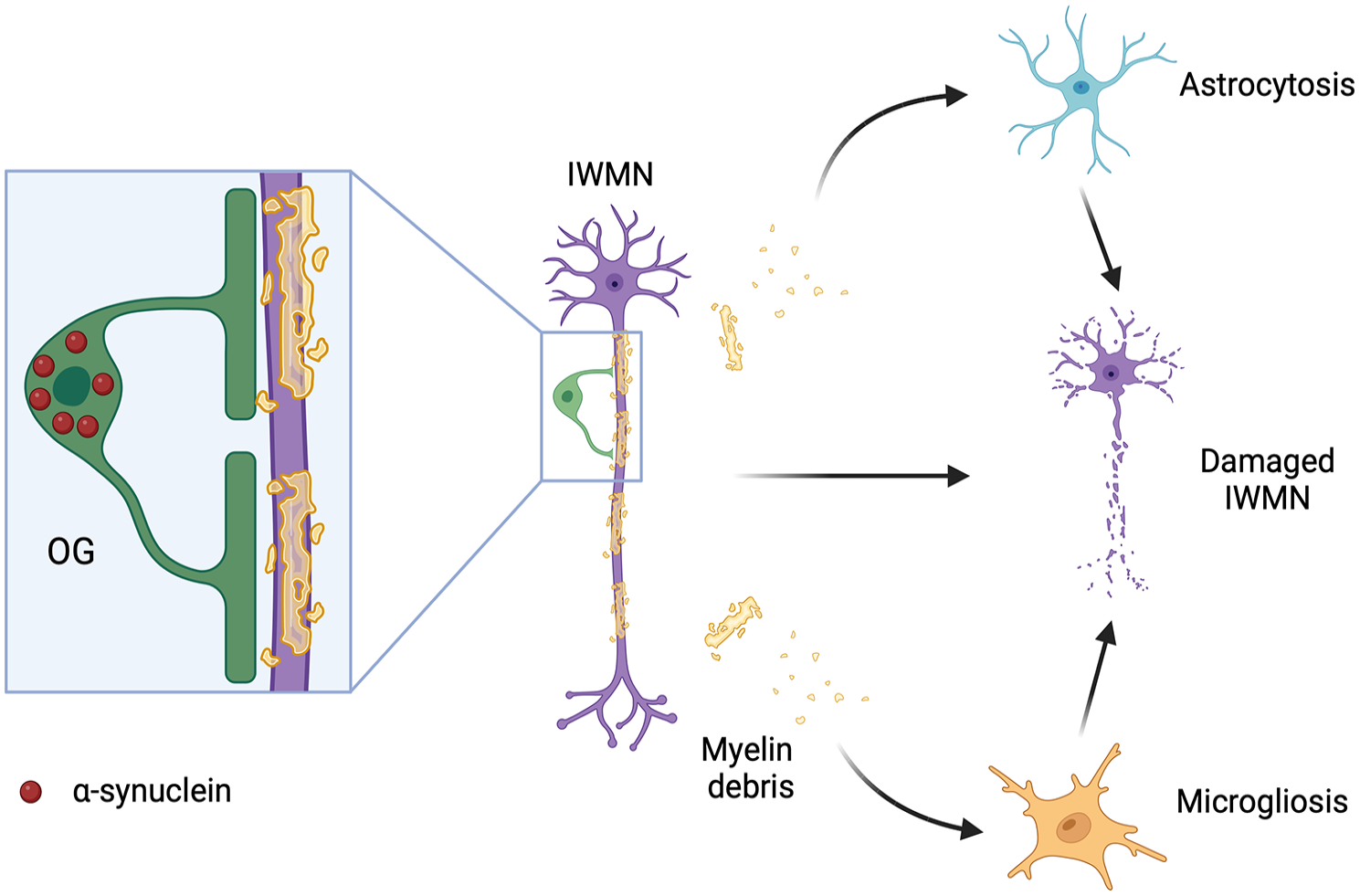
Hypothetical mechanism of interstitial white matter neuron (IMWN) damage in multiple systems atrophy. Accumulation of α-synuclein in oligodendrocytes in the white matter leads to the formation of glial cytoplasmic inclusions and damage to the myelinating cells. This, in turn, leads to demyelination and axonal degeneration, resulting in damage and/or loss to IWMNs. Alternative pathologic pathways may include astrogliosis and microgliosis. Created with BioRender.com.
IWMNs in Alzheimer’s disease
Alzheimer’s disease (AD) is a neurodegenerative disorder characterized by extracellular deposition of amyloid-β plaques and intraneuronal neurofibrillary tau-containing tangles, both of which contribute to neuroinflammation, neuronal loss, and neurological dysfunction (Griffiths and Grant 2023; Mangalmurti and Lukens 2022). Alterations in the WM are part of the pathophysiologic mechanism of AD. Some studies even propose that AD originates in the WM, myelin breakdown and axonal damage in the WM precede alterations in the GM, and demyelination in the WM occurs before the presence of amyloid-β plaques and neurofibril tangles (Alves and others 2015; Reisberg and others 1999; Sachdev and others 2013). In support of this theory, neuroimaging studies found abnormalities in the WM without structural changes of the GM in patients with preclinical AD (Alves and others 2015; Balthazar and others 2009; Guo and others 2010; Nasrabady and others 2018). A distinctive subset of SOM+ neurons with AD-related abnormal tau was detected in human WM, suggesting that a subpopulation of IWMNs may be specifically affected or involved in the AD process (van de Nes and others 2002). Moreover, a recent study investigating a potential subset of NPFF-positive IWMNs showed a reduction of these cells in the superficial and deep WM of the frontal gyrus of patients with AD compared to controls (Wiesner and others 2024). Damage to the IWMNs may be a part of the pathophysiologic mechanism of AD.
IWMNs in epilepsy
Epilepsy encompasses a group of neurological disorders characterized by recurrent seizures due to abnormal electrical activity of the brain. Immunohistochemical studies demonstrated an increased density of IWMNs in patients with temporal lobe epilepsy (TLE) (Emery and others 1997; Richter and others 2016; Thom and others 2001). Furthermore, in patients with TLE, density of a presynaptic marker synaptophysin correlated with the density of IWMNs and was significantly higher than in control samples (Soki and others 2022). Density of IWMNs and synaptophysin also correlated with verbal memory scores in patients with TLE (Soki and others 2022). A significant increase in expression of NPNFP, as well as in the density of GABAergic and glutamatergic synapses on IWMNs, was found in patients with pharmaco-resistant epilepsy with focal cortical dysplasia and in patients with TLE (Yang and others 2023). The NPNFP-positive IWMNs exhibited an enlarged cell soma. The authors suggested that the imbalance between synaptic excitation and inhibition of dysmorphic IWMNs could contribute to altered synaptic networks in epilepsy (Yang and others 2023) (Fig. 8). Notably, a subpopulation of IWMNs positively labeled for the brain-specific T-box transcription factor (Tbr1) was found in brain tissue samples from patients with TLE, as well as from control subjects (Richter and others 2016). Tbr1 is highly expressed in early-born glutamatergic neurons of the subplate and plays a key role in the formation of cortico-thalamic connections (Hevner and others 2002). Therefore, expression of Tbr1 in IWMNs suggests that those cells (Richter and others 2016) are glutamatergic and may contribute to altered synaptic circuits (e.g., impaired connections between the temporal cortex and the thalamus) in patients with TLE.
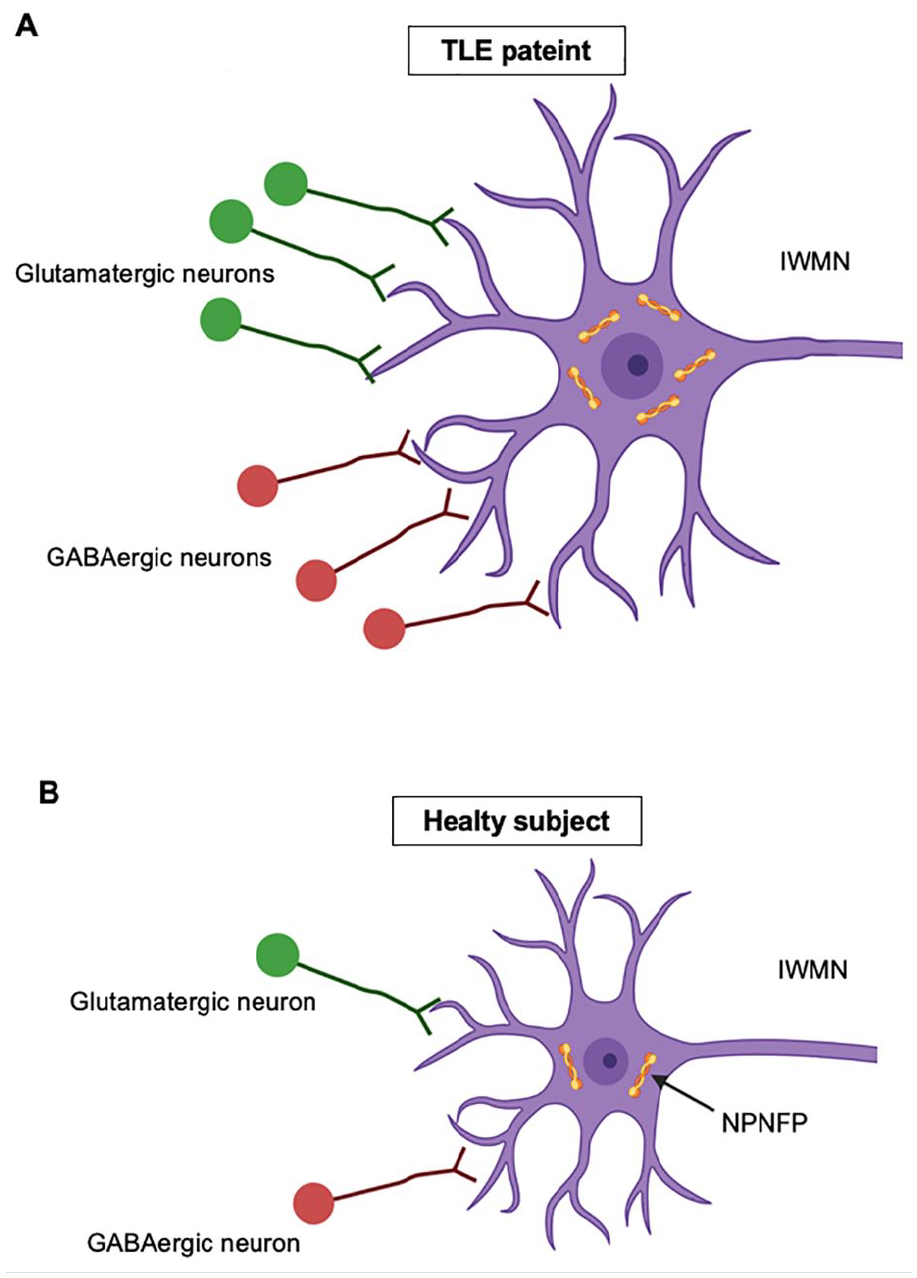
Cellular changes of interstitial white matter neurons (IWMNs) in temporal lobe epilepsy (TLE). (A) Schematic drawing of an IWMN in patients with TLE. (B) Schematic drawing of an IWMN in control subjects. Note that IWMNs in patients with TLE show enlarged soma and increased expression of non-phosphorylated neurofilament protein (NPNFP) compared to control subjects. In addition, higher densities of glutamatergic and GABAergic synapses have been observed in IWMNs of patients with TLE. The imbalance of excitation and inhibition on IWMNs may contribute to altered synaptic networks in epilepsy. Created with BioRender.com.
Currently, it is not clear whether alterations in IMWNs and in neuronal networks involving the IWMNs are beneficial or detrimental during epilepsy, and whether those alterations represent an active part of the pathophysiologic mechanism of seizures or a secondary symptom—an epiphenomenon. Some studies claimed that an increased density of IWMNs was associated with a worse outcome of patients with TLE following surgery (Kasper and others 1999). Other studies reported that a higher density of IWMNs was associated with a more favorable postsurgical outcome of patients with TLE (Hardiman and others 1988; Soki and others 2022; Thom and others 2001). Resolving these problems remains a task of future research.
IWMNs in multiple sclerosis
Multiple sclerosis (MS) is a demyelinating disease affecting muscle control, causing pain, fatigue, and cognitive problems. The cause may include genetic, environmental, and infectious factors (Dobson and Giovannoni 2019; Lassmann 2018).
In human patients with MS, both a decrease and an increase of MAP2+ IWMNs have been described. In many acute and chronic WM MS lesions, IWMNs were not detected; if they were present, their MAP2+ dendrites appeared fragmented and perikarya shrunken (Chang and others 2008). However, in a subset (~25%) of chronic WM MS lesions, NeuN+/MAP2+ neurons and immature NeuN+/MAP2– neurons were found (Chang and others 2008). In the lesions, densities of MAP2+ and NeuN+ neurons were increased by 72% and 130%, respectively (Chang and others 2008). The perikarya and the dendrites of IWMNs were closely opposed by synaptophysin-positive synaptic boutons, and the punctate synaptophysin staining in chronic MS lesions was increased compared to normal-appearing WM (Chang and others 2008). The identified axodendritic and axosomatic synapses on IWMNs were symmetric, indicating that IWMNs receive inhibitory innervation (Chang and others 2008). The authors proposed that while inflammatory demyelination destroys IWMNs in acute and many chronic WM MS lesions, in some cases, neurogenesis may be triggered (Chang and others 2008). The reasons for this remain unclear.
An important issue that has not been discussed in the literature so far is the interactions between IWMNs and oligodendrocyte lineage cells during MS. These interactions may exist in multiple forms, e.g., myelination of IWMN axons by oligodendrocytes, synaptic signaling between IWMNs and OPCs. etc, and may be altered during MS and contribute to pathophysiologic mechanisms of the disease.
Conclusion and future perspectives
IWMNs remain the least studied nerve cells in the central nervous system. While there are many studies describing distribution and morphology of IWMNs, our knowledge regarding functions of IWMNs and their interaction with other neuronal and glial cells remain very sparse. Evidence is now accumulating that IWMNs are affected during various diseases, including neurodegenerative and neuropsychiatric diseases, but it remains unclear whether damage to IWMNs is among the triggers of these diseases or only accompanies other pathologic changes.
We think that in the future, it will be especially important to develop new animal models and novel technical approaches allowing for specific targeting of IWMNs in the brain of animals and humans in order to discover their physiologic and pathophysiologic functions and modes of integration into the neuronal and neuron-glia circuits. For example, it will be important to shed more light on the molecular markup of IWMNs and to find out whether they express specific markers that distinguish them from other cortical neurons. This may be achieved, for example, by extracting individual IWMNs from the WM using magnetic-activated or fluorescence-activated cell sorting and applying advanced sequencing technologies such as single-cell RNA sequencing to establish the transcriptomic profiles of IWMNs. If candidate genes specific for IWMNs are found, the corresponding protein expression may be tested and new markers for IWMNs may be developed. Developing the approaches that specifically label IWMNs (but not other neurons) in animal models in vivo will be especially valuable because this would allow targeting the IWMNs for imaging (e.g., calcium imaging) and patch-clamp recordings in slices and in vivo. Imaging and electrophysiologic approaches currently remain the best ways to study function and connectivity of cells in the nervous system. Trans-synaptic viral labeling approaches (e.g., rabies viruses) may be also useful for studying connectivity of IWMNs. These techniques have been used in many studies investigating connections within and between various brain areas, but the focus of those studies is usually on the GM tissue while the WM tissue is usually left out. Studying the WM in experiments using trans-synaptic tracing may help understand how IWMNs are integrated into the neuronal circuitry.
Footnotes
Acknowledgements
We thank Boris Zalc (Paris, France) for pointing to and discussing the initial discovery by French neuroscientists of the neuron-glia synapses in the human cerebral cortex.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Our research is supported by IKERBASQUE Basque Foundation for Science, the Basque Government PIBA Project (PIBA 2020_1_0030), the MCIN project PID2019-110195RB-I00, and the MCIN/AEI /10.13039/501100011033/FEDER Una manera de hacer Europa (project PID2022-140726NB-I00).
