Abstract
There are numerous barriers to white matter repair after central nervous system injury and the underlying mechanisms remain to be fully understood. In this study, we propose the hypothesis that inflammatory macrophages in damaged white matter attack oligodendrocyte precursor cells via toll-like receptor 4 signaling thus interfering with this endogenous progenitor recovery mechanism. Primary cell culture experiments demonstrate that peritoneal macrophages can attack and digest oligodendrocyte precursor cells via toll-like receptor 4 signaling, and this phagocytosis of oligodendrocyte precursor cells can be inhibited by using CD200-Fc to downregulate toll-like receptor 4. In an in vivo model of white matter ischemia induced by endothelin-1, treatment with CD200-Fc suppressed toll-like receptor 4 expression in peripherally circulating macrophages, thus restraining macrophage phagocytosis of oligodendrocyte precursor cells and leading to improved myelination. Taken together, these findings suggest that deleterious macrophage effects may occur after white matter ischemia, whereby macrophages attack oligodendrocyte precursor cells and interfere with endogenous recovery responses. Targeting this pathway with CD200 may offer a novel therapeutic approach to amplify endogenous oligodendrocyte precursor cell-mediated repair of white matter damage in mammalian brain.
Introduction
One of major findings in neuroscience in the past decade is that adult mammalian brain can be surprisingly plastic after stroke and brain injury. 1 In the context of functional recovery, white matter connectivity may be especially crucial, and many cells and processes contribute to endogenous mechanisms that promote axonal recovery. 2 But white matter recovery is often incomplete because repair processes are also impaired by the development of inhibitory responses in glial cells. Current knowledge is mostly focused on the potential role of inhibitory matrix substrates such as chondroitin sulfate proteoglycans and NOGO.3,4
Beyond axonal growth and reconnections, remyelination should also be an essential component of white matter recovery. Oligodendrocytes can be generated from V-SVZ cells in the adult brain, and newly generated oligodendrocyte precursor cells (OPCs) migrate toward the corpus callosum and the white matter tracts of striatum and fimbria fornix. 5 After white matter injury, recovery occurs when myelin sheaths are generated by oligodendrocytes that mature from OPCs that are capable of migrating into demyelinated areas to promote remyelination.6,7 But unlike the developing brain, OPCs are now moving into damaged tissue with a complex inflammatory milieu. How OPCs survive and respond in the inflammatory environment remains to be fully understood.
In this study, we used a combination of cell culture and the in vivo model to examine how OPCs respond to inflammatory macrophages in damaged white matter. Traditionally, “M2-like” macrophages are thought to perform tissue cleanup and debris removal duties that promote repair and recovery. In this study, our findings suggest that in addition to these beneficial actions, potentially deleterious “M1-like” macrophages can attack OPCs via toll-like receptor 4 (TLR4)-dependent pathways. By attacking OPCs, these macrophages prevent endogenous oligodendrocyte replacement and interfere with recovery. However, treatment with CD200-Fc may downregulate TLR4, prevent this macrophage attack on OPCs, and enhance white mater recovery.
Methods and materials
In vivo white matter injury models
All experiments were performed following an institutionally approved protocol in accordance with National Institutes of Health guidelines and with the United States Public Health Service’s Policy on Human Care and Use of Laboratory Animals and following Animals in Research: Reporting In vivo Experiments guidelines. 8 Male C57BL6 mice (11–12 weeks, Charles River Laboratories) were deeply anaesthetized with isoflurane (5–1.5%) in 30%/70% oxygen/nitrous oxide. Endothelin 1 (ET-1, Sigma, E7764) at a concentration of 10 µg/ml was injected through a 30-gauge needle over 5 min into the left corpus callosum as performed before (anterior: 0.5 mm from bregma, lateral: 1.0 mm from bregma, depth: 2.3 mm from the skull surface). 9 ET-1 is a potent vasoconstrictor and exhibits long-lasting actions for at least 48 h when injected into brain as a standard model for producing focally restricted ischemic injury. 10 Additionally, when injected into white matter area, ET-1 produces hallmarks of white matter injury observed in humans, such as axonal damage, demyelination, inflammation, and glial scar formation. 11 Myelin damage was detected by fluoromyelin staining (1:300, Life technologies, F34651). Since CD200-Fc treatment to target CD200R has been reported in an in vivo study, 12 , 13 we treated mouse CD200-Fc in our in vivo model. Mouse CD200 fusion protein (100 µg/kg, R&D systems, 3355-CD-050) or mouse control IgG (100 µg/kg, Santacruz, sc-2025) was injected intraperitoneally on days 1, 5, and 10 after ET1 injection.
Macrophages (Mϕ) isolation
Mϕs were isolated from rat peritoneal cavity. After collecting cells from the cavity, cells were seeded on noncoated six-well plates and grown in RPMI medium 1640 containing 10% FBS, 1% penicillin/streptomycin. Twenty four hours later, attached cells were used for further experiment.
OPCs/oligodendrocyte/microglia culture
OPCs/oligodendrocyte/microglia cultures were prepared as previously described.14,15 Briefly, cerebral cortices from one to two-day-old Sprague-Dawley rats were dissected, minced, and digested. Dissociated cells were plated in poly-
GST-pull down assay
GST-pull down assay experiment was performed between His-tagged NG2 recombinant protein (1 µg, R&D systems, 2585-PG-050) and GST-tagged TLR4 (0.1 µg, Abnova, H00007099-Q01) or GST control (0.1 µg, Santacruz, sc-4033). Total amount of 400 µl of each combination was incubated with 10 µl of a 50% slurry of glutathione-agarose beads for 4 h at 4℃ with end-over-end mixing. Agarose beads were collected after centrifugation at 1000g for 10 s and washed three times with 50% PRO-PREP lysis buffer (iNtRON Biotechnology, 17081) diluted by PBS. NG2 recombinant protein associated with the GST fusion protein was detected by SDS-PAGE-western blot method.
Macrophage-mediated bacteria phagocytosis assay
Rat peritoneal macrophages were stimulated by interleukin-1β (IL-1β, 100 ng/ml) for 24 h and the ability of nonstimulated or IL-1β-stimulated macrophages to incorporate bacterial particles was assessed by Vybrant Phagocytosis Assay Kit (Life technologies, V-6694). E. coli particles (K-12 strain) labeled by fluorescein was added to macrophages for 2 h and washed twice with PBS to remove free floating debris. Finally, the incorporation of bacterial particles in macrophage was analyzed by fluorescence microplate reader at Ex (488 nm)/Em (515 nm).
Macrophage-mediated OPC or myelin debris phagocytosis assay
OPCs were labeled by diI-ac LDL (Invitrogen, L3484) for 2 h at 37℃ incubator and washed three times with PBS. Isolated macrophages were directly co-cultured with OPCs in neurobasal media containing glutamine, 1% penicillin/streptomycin, 10 ng/ml PDGF, 10 ng/ml FGF, and 2% B27 supplement onto poly-
LDH cytotoxicity assay
OPCs or mature oligodendrocytes were co-incubated with IL-1β-stimulated rat peritoneal macrophages or IL-1β-stimulated rat brain microglia for 24 h. Since LDH release is commonly observed during phagocytosis,16,17 extracellular LDH was detected by LDH Cytotoxicity Detection kit (Roche Applied Science, 11644793001). Then the absorbance of the culture medium was measured with a microplate reader at a test wavelength of 570 nm and a reference wavelength of 630 nm. Macrophage or microglia was identified by CD68 antibody (1:100, Abcam, ab955) and OPCs were labeled by diI-ac LDL, PDGFRα antibody (1:200, R&D Systems, AF1062), or Olig2 antibody (1:200, Santa Cruz, sc-19969). To determine which cells, OPCs or macrophages, were killed and releasing LDH, propidium iodide (PI 1:300, Invitrogen, P3566) staining for 10 min prior to fixation (prefixation PI staining)18,19 was performed.
WST assay
Cell proliferation/viability was assessed by WST reduction assay (Dojindo), which detects dehydrogenase activity of viable cells. The cells were incubated with 10% WST solution for 1 h at 37℃. Then the absorbance of the culture medium was measured with a microplate reader at a test wavelength of 450 nm and a reference wavelength of 630 nm.
FACS analysis
For analyses of in vivo brain samples, tissues are minced and then digested at 37℃ for 30 min with an enzyme cocktail (Collagenase type IV; Sigma-Aldrich, C5138, DNase I; Sigma-Aldrich, D4263) as previously described. 20 Single cell suspensions were prepared by filtering through a 40 µm strainer. Cell suspensions are preblocked with 3% BSA and then incubated with the following primary antibodies against TLR4 (1:100, Abcam, ab45104), NG2 (1:100, Abcam, ab81104), PDGFRα (1:200, BD biosciences, 558774). F4/80 (1:100) was purchased from Biolegend (123102). Fab-specific secondary antibodies tagged by phycoerythirin (PE), fluorescein isothiocyanate, and allophycocyanin from Jackson laboratories are incubated for 30 min at room temperature. Labeled cell populations are measured by FACSCalibur (BD biosciences). FACS data are analyzed by Cellquest pro software (BD biosciences). FACS analysis is performed using a variety of controls including unstained samples, isotype antibodies, and single stained samples for determining appropriate gates, voltages, and compensations required in multivariate flow cytometry.
Western blot analysis
Western blot procedure was performed as described before. 20 After blocking, membranes were then incubated overnight at 4℃ with following primary antibodies: anti-NG2 (1:500, Santa Cruz, sc-166251), monoclonal anti-MBP (1:500, Abcam, ab62631), monoclonal antibeta actin (1:5000, Sigma, A2228) were obtained from Cell Signaling Technology after incubation with peroxidase-conjugated secondary antibodies (GE Healthcare, NA931 anti-mouse, or NA934 anti-rabbit, or NA935 anti-rat) and visualization by enhanced chemiluminescence (Thermo Scientific, 34078).
Immunocytochemistry or immunohistochemistry
Immunocytochemistry or immunohistochemistry was performed as described before. 20 After staining with primary antibody, fluorescent-tagged secondary antibody, nuclei were counterstained with or without 4,6-diamidino-2-phenylindole (DAPI), and coverslips were placed. Immunostaining was analyzed with a fluorescence microscope (Nikon ECLIPSE Ti-S) interfaced with a digital charge-coupled device camera and an image analysis system or using Carl Zeiss Laser Scanning Confocal Microscope Pascal 5 LSM and Pascal 5 LSM software Version 3.2.
Electron microscopy
Tissues were fixed in 2.0% glutaraldehyde in 0.1 M sodium cacodylate buffer, pH 7.4 (Electron Microscopy Sciences, Hatfield, PA) overnight at 4℃. They were rinsed in buffer, postfixed in 1.0% osmium tetroxide in cacodylate buffer for 1 h at room temperature, rinsed in buffer again and dehydrated through a graded series of ethanol to 100%. They were then infiltrated overnight with Eponate resin (Ted Pella, Redding, CA) in a 1:1 solution of Eponate:propylene oxide. The following day they were placed in fresh Eponate for several hours and then embedded in Epon overnight at 60℃. Thin sections were cut on a Leica EM UC7 ultramicrotome, collected onto formvar-coated grids, stained with uranyl acetate and lead citrate, and examined in a JEOL JEM 1011 transmission electron microscope at 80 kV. Images were collected using an AMT digital imaging system (Advanced Microscopy Techniques, Danvers, MA). Using the calibrated measurement tool within the AMT Image Capture Engine V601 software package, myelin measurements were drawn (inner axonal diameter from myelin edge to myelin edge, and total axonal diameter) and values (mm) embedded on images at magnification x60,000 at the time of data collection.
Statistical analysis
Results were expressed as mean ± SEM. When only two groups were compared, Student’s t-test was used. Multiple comparisons were evaluated by Tukey–Kramer’s test after one-way or two-way ANOVA. P < 0.05 was considered to be statistically significant. All experiments and analyses were conducted in a randomized and blinded manner in accordance with NIH guidelines on rigor and reproducibility.
Results
Acute injury in white matter upregulates endogenous OPCs populations as part of the compensatory response for repair and recovery.21,22 But how well do these newly generated cells survive the inflammatory environment that is known to exist in damaged brain tissue?
23
Endothelin-1 (ET-1) was stereotaxically injected into the corpus callosum of adult male C57Bl6 mice in order to induce focal ischemic damage and demyelination. As the lesion developed over the first few days after ET-1 injection (Figure 1(a)), macrophages were observed to accumulate within the damaged white matter zone (Figure 1(b)). Confocal microscopy suggested that macrophages often surrounded PDGFRα-positive OPCs (Figure 1(c)), hence raising the possibility that this inflammatory process may somehow interfere with the endogenous progenitor cell response. Cell counts demonstrated that OPC density began to decrease from day 3 to day 7 (Figure 1(d)), along with an increase in accumulated TLR4-expressing macrophages (Figure 1(e)). Correspondingly, GSTpi-positive mature oligodendrocytes showed a decreasing trend over 14 days after white matter injury (Figure 1(f)).
Reduction of OPCs and macrophage accumulation after focal white matter ischemia: (a) Stereotaxic injection of endothelin-1 (ET-1) into the corpus callosum induced focal demyelination in the white matter tracts. DAPI (blue) staining showed cell accumulation in the lesion area. Scale bar: 200 µm. (b) Immunohistochemistry showed an accumulation of F4/80-positive macrophages in demyelinated white matter on day 3 after ET1 injection. Scale bar: 100 µm. (c) PDGFRα-positive OPCs (green) were surrounded by F4/80-positive macrophages (red) on day 7 postinjury. Scale bar: 50 µm. (d) Flow cytometry analysis (FACS) showed that the number of OPCs (NG2/PDGFRα-double positive cells) was decreased on day 7. Quantitation of the FACS data confirmed the significant decrease of OPCs. *P < 0.05 versus sham (sham; n = 3, day 3; n = 4, day 7; n = 5, day 14; n = 4). (e) FACS analysis demonstrating an accumulation of TLR4-positive macrophages (F4/80-positive cells) on day 7 in the injured white matter. Quantitation of the FACS data confirmed the significant increase of total macrophages and TLR4-positive macrophages. *P < 0.05, **P < 0.01 versus sham (sham; n = 3, day 3; n = 4, day 7; n = 4, day 14; n = 4). (f) FACS analysis showed a gradual reduction in GSTpi-positive mature oligodendrocytes over the course of 14 days after ET-1 injection (sham; n = 3, day 3; n = 4, day 7; n = 4, day 14; n = 4). SSC: side scatter. All data are presented as mean ± SEM.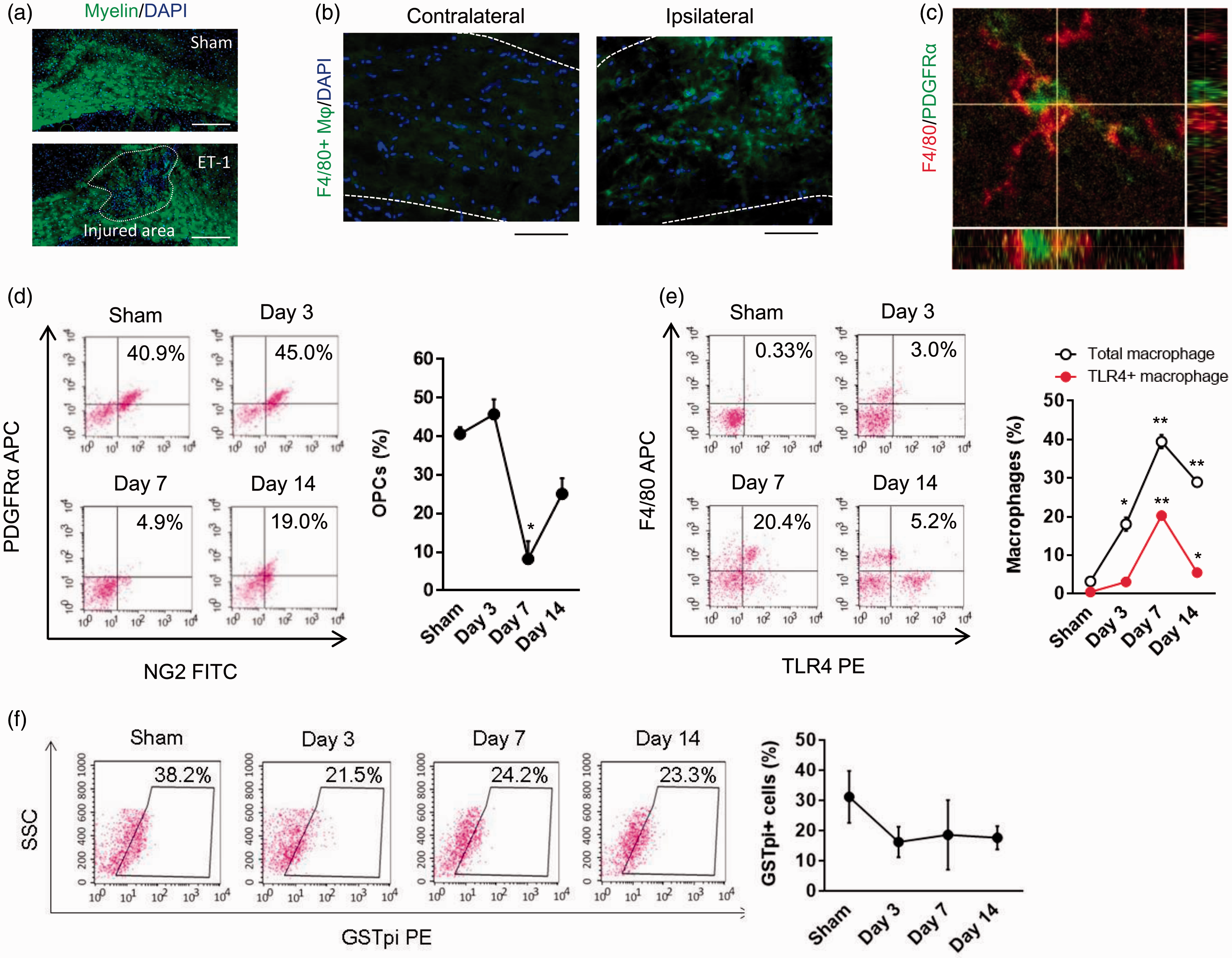
The temporal profile of decreasing OPCs along with accumulating macrophages as well as the spatial co-localization of these cells over time suggests that macrophages can attack OPCs. To assess the mechanisms that may underlie this phenomenon, we turned to cell culture models. First, we showed that within the range of concentrations used here, ET-1 did not appear to directly kill OPCs (Suppl. Figure 1), suggesting that the effects documented in our in vivo model may be due to white matter ischemia and not cytotoxicity of ET-1 per se. Next, we conducted in vitro phagocytosis assays where rat peritoneal macrophages were stimulated with interleukin-1beta (IL-1β) to increase phagocytic activity and TLR4 expression (Figure 2(a) and (b)). When macrophages were then added onto primary rat OPCs, they attached to, ingested, and digested the live OPCs (Figure 2(c) to (f)). Conditioned media collected from macrophages did not kill OPCs (Figure 2(g)), suggesting that direct cell-on-cell attacks may be required. In contrast, macrophages did not attack mature oligodendrocytes in our system (Figure 2(e)). Additionally, we investigated whether brain microglia attacked OPCs. In our cell culture system, IL-1β-stimulated brain microglia could attack OPCs, but the degree of phagocytotic attack was smaller compared to macrophages (Figure 2(h)).
OPC phagocytosis by macrophage in vitro: (a) Rat peritoneal macrophages were stimulated with interleukin-1beta (IL-1β 100 ng/ml) for 24 h. IL-1β significantly enhanced E. coli (K-12) phagocytic function in macrophages (control; n = 4, IL-1β; n = 3). (b) FACS analysis was performed using TLR4 antibody conjugated with PE. Membrane expressing TLR4 in macrophages was upregulated by IL-1β stimulation (100 ng/ml) for 24 h. 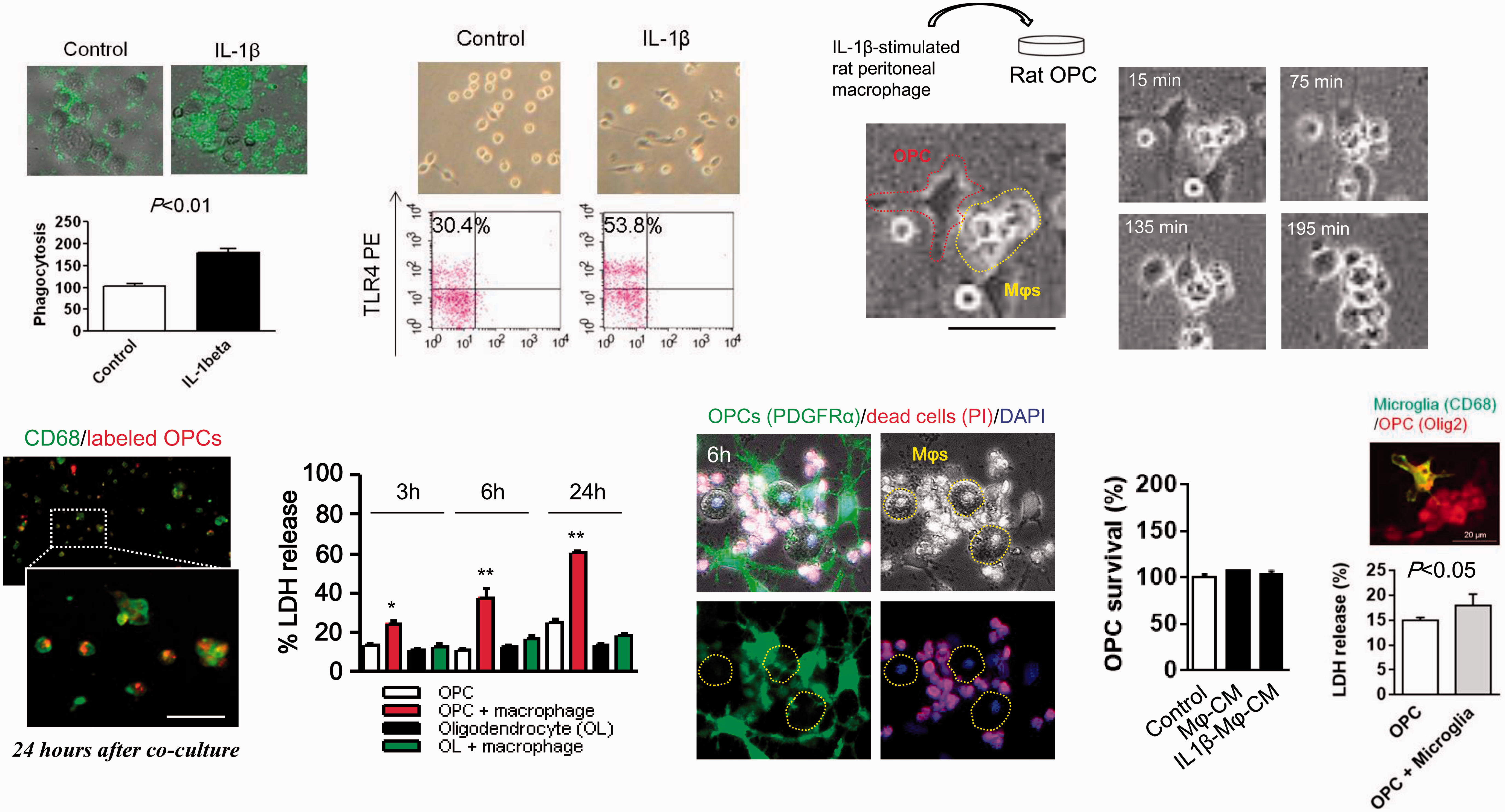
Because of this differential vulnerability between OPCs and oligodendrocytes, we next asked whether the ability of macrophages to target susceptible cells may be mediated by the expression of immature signals in OPCs such as NG2 (Figure 3(a)). TLRs comprise a family of proteins known to be essential for macrophage function,
24
so we used a GST-pull-down assay to screen this TLR family for NG2-binding ability. Our initial data suggest that TLR4 can bind to NG2 proteins (Figure 3(b)). Moreover, TLR4 from macrophages appeared to overlap with NG2 in OPCs when these cells were co-cultured (Figure 3(c)), and application of anti-TLR4 or anti-NG2 neutralizing antibodies reduced OPC phagocytosis (Figure 3(d) and (e)).
Macrophage TLR4 interacts with NG2 to promote OPC phagocytosis in vitro: (a) Western blot analysis confirmed that OPCs expressed NG2, and mature oligodendrocytes expressed MBP in our culture system. (b) In the cell-free system, His-tagged NG2 bound to GST-tagged TLR4 assessed by western blot after GST pull-down. (c) Immunocytochemistry showed that NG2 positive fraction was incorporated in macrophages 24 h after macrophage/OPC co-culture. Scale bar: 50 µm. (d) Blockade of TLR4 (10 µg/ml) or NG2 (5 µg/ml) with a neutralizing antibody attenuated macrophage attack on OPCs. (e) Treatment with neutralizing antibody significantly decreased LDH release at 90 min after the co-culture (n = 4). **P < 0.01. All data are presented as mean ± SEM.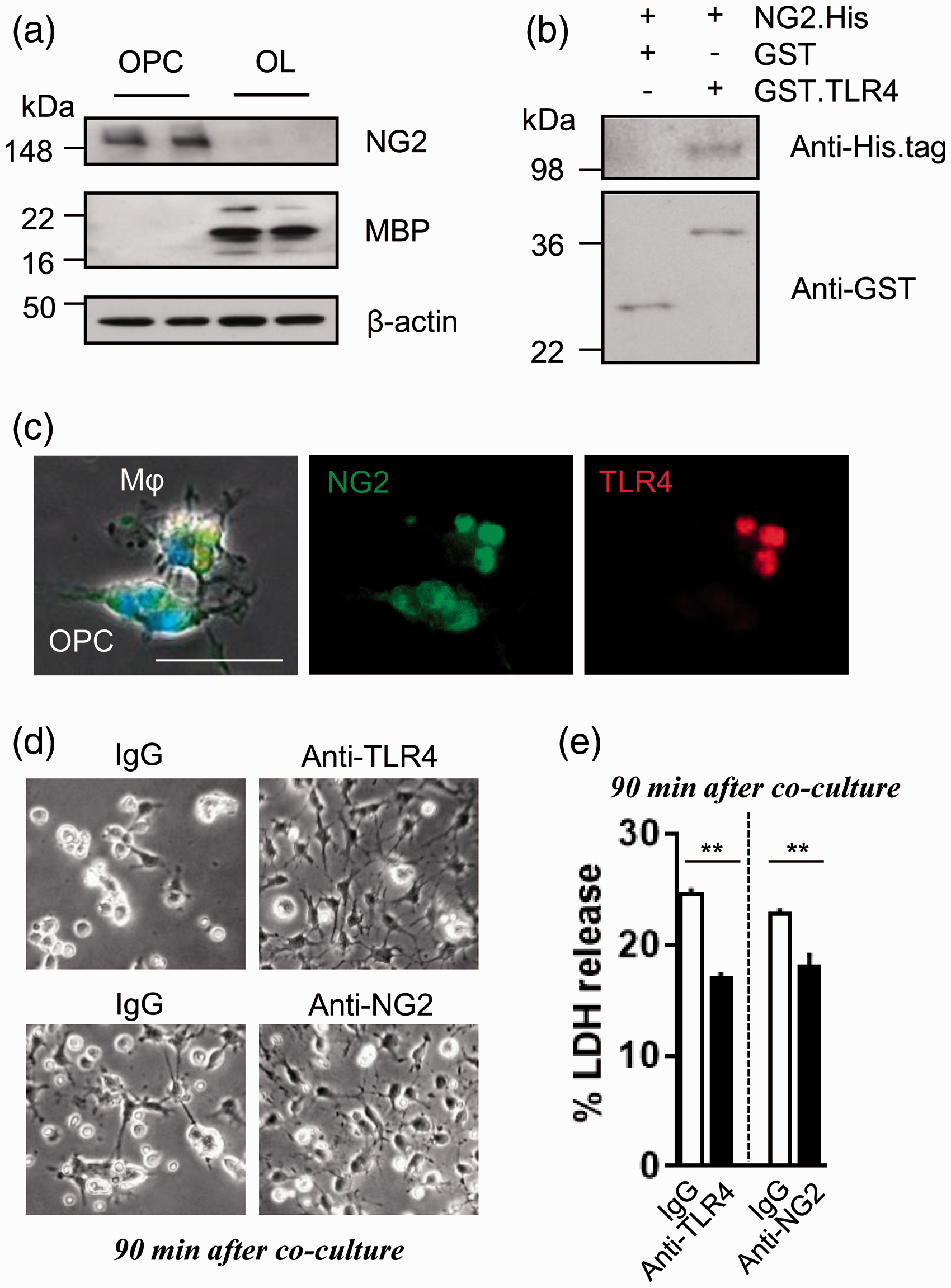
If macrophages can attack and phagocytose OPCs via TLR4 signaling, is it possible to prevent this deleterious process and promote white matter recovery by suppressing the macrophage response? To answer this question, we focused on the immunoglobulin superfamily CD200, which is known to interact with its receptor CD200R on immune cells to deactivate inflammation after stroke.
23
Cultured macrophages expressed CD200R (Figure 4(a)), and treatment with the fusion protein CD200-Fc reduced their surface levels of TLR4 (Figure 4(b)). Next, conditioned media from IL-1β-stimulated macrophages following IgG or CD200-Fc treatment were collected and transferred into OPCs. We confirmed that CD200-Fc-stimulated macrophage conditioned media did not affect OPC proliferation (Figure 4(c)). Interestingly, treatment with CD200-Fc significantly decreased the ability of macrophages to attack and ingest OPCs (Figure 4(d)) without affecting the ability of macrophages to incorporate myelin debris (Figure 4(e)), suggesting that CD200-Fc may protect OPC from macrophage attack through downregulating TLR4 expression and function.
CD200-Fc suppresses OPC phagocytosis by macrophage without affecting clearance of myelin debris in vitro: (a) Immunocytochemistry showed that rat peritoneal macrophages (CD68 positive cells) expressed CD200 receptor. (b) IL-1β (100 ng/ml)-stimulated macrophages were co-incubated with CD200-Fc (10 µg/ml) for 24 h. FACS analysis showed that CD200-Fc treatment significantly suppressed TLR4 expression on macrophage cell surface. (c) Macrophage conditioned media were collected from IL-1β (100 ng/ml)-stimulated macrophages treated by IgG or CD200-Fc (10 µg/ml). Conditioned media did not increase OPC proliferation. (d) FACS confirmed that macrophages ingested OPCs at 24 h after starting the co-culture. Co-incubation of CD200-Fc (10 µg/ml) significantly reduced the OPC phagocytosis by macrophages in vitro. (IgG; n = 7, CD200-Fc; n = 6). (e) Fluorescent myelin debris was prepared from mature oligodendrocytes stained with fluoromyelin. CD200-Fc (10 µg/ml) did not affect the incorporation of myelin fragment in IL-1β (100 ng/ml)-stimulated macrophages (IgG; n = 4, CD200-Fc; n = 4). All data are presented as mean ± SEM.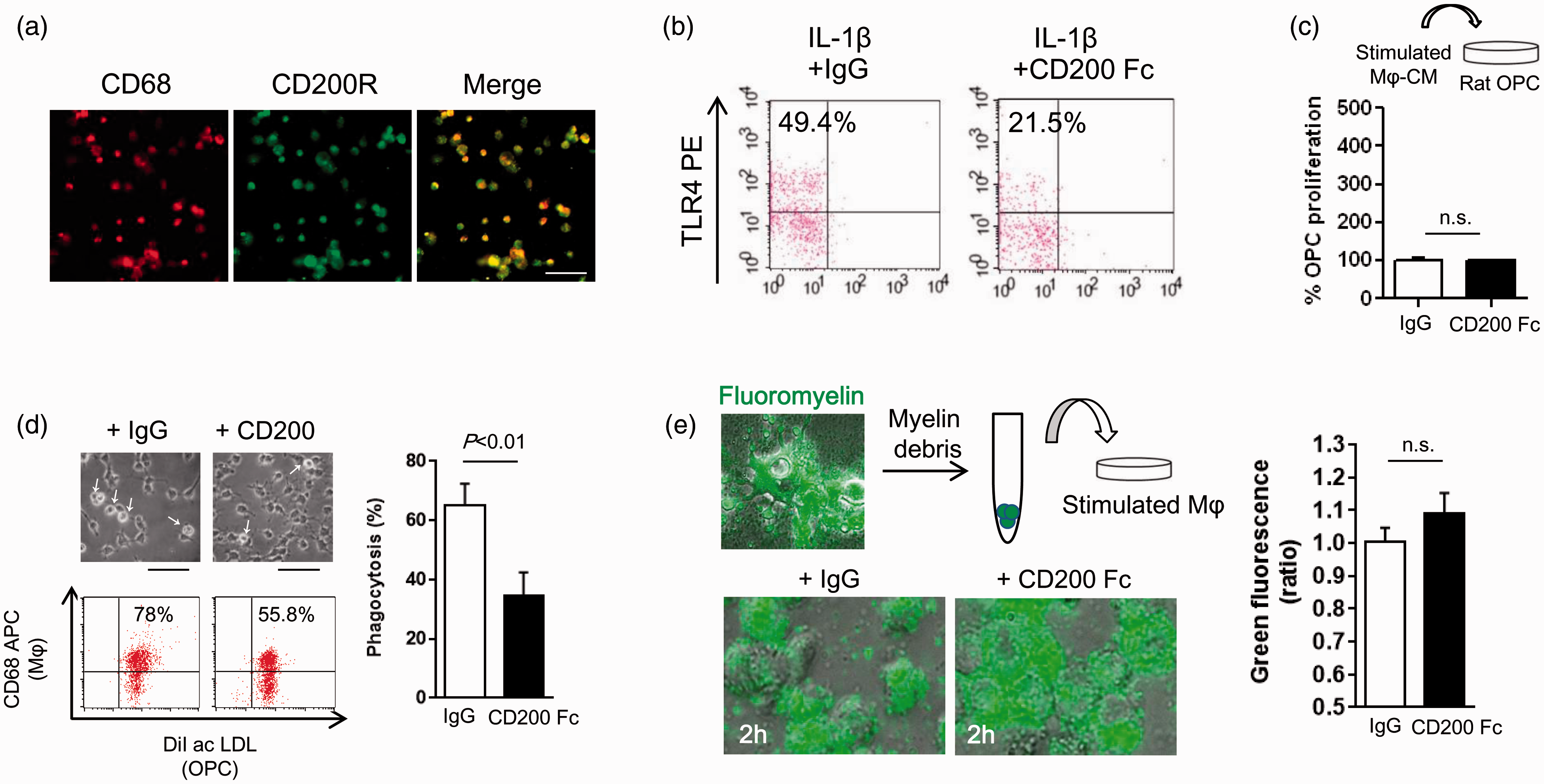
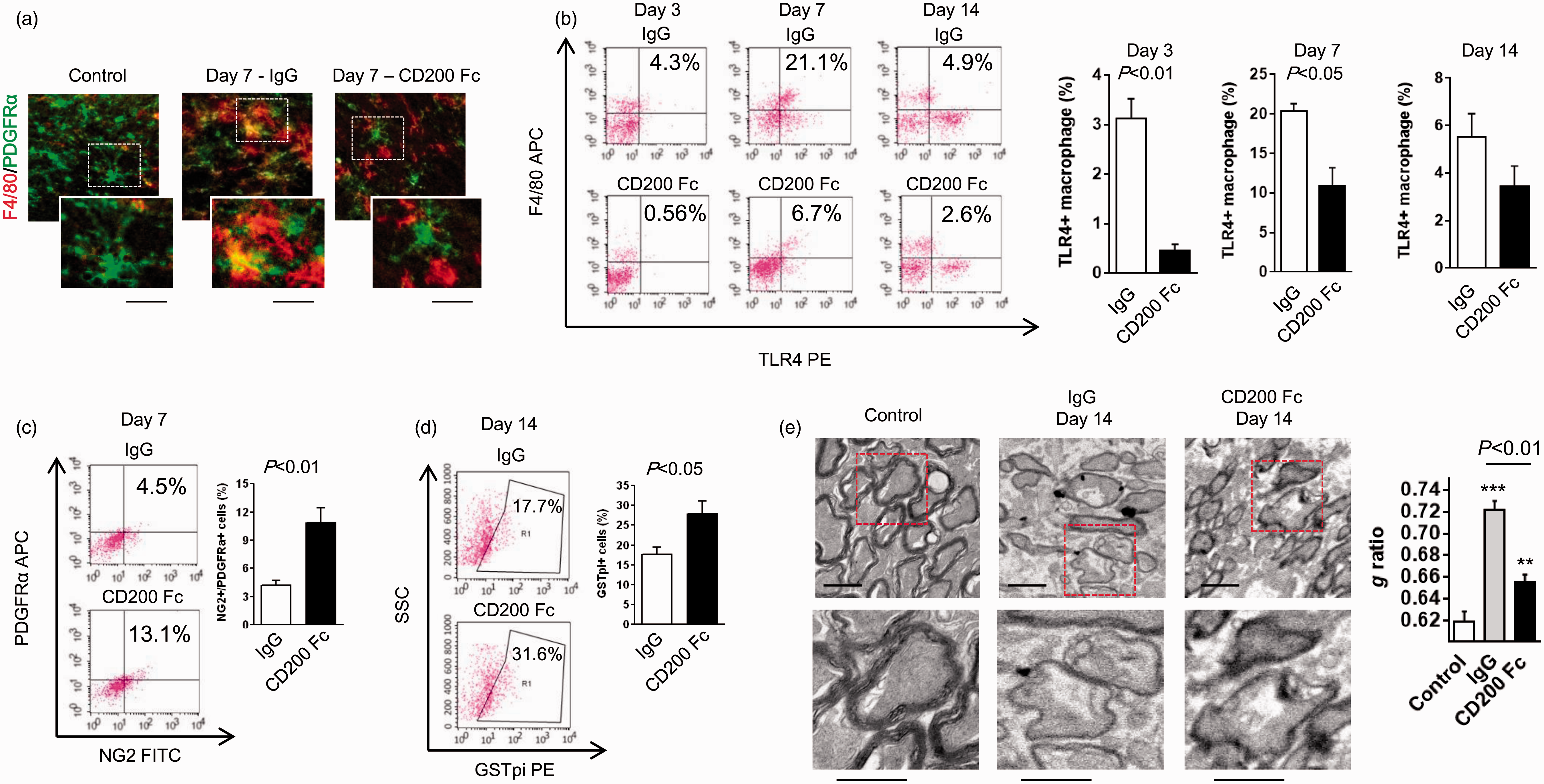
Based on these in vitro findings, we then asked whether CD200-Fc administration could similarly suppress macrophages and promote white matter recovery in vivo. Mice were stereotaxically injected with ET-1 to induce focal ischemic injury in the corpus callosum and then were treated with CD200-Fc fusion protein (100 µg/kg intraperitoneal) or control IgG at 1, 5, and 10 days later. To assess whether CD200-Fc suppressed TLR4 expression in blood monocyte/macrophages, FACS analysis was performed on blood samples collected at three days after injury (Figure 5(a)). This analysis showed that treatment with CD200-Fc successfully increased plasma CD200 protein level (Figure 5(b)). After three days following ET-1 injection, the number of circulating F4/80 positive monocyte/macrophages found in blood was significantly increased and accompanied with upregulation of TLR4 expression compared to sham-operated control group in FACS analysis (Figure 5(c)). Treatment with CD200-Fc suppressed TLR4 expression at day 3 after ET-1 injection (Figure 5(c)), suggesting that intraperitoneally injected CD200-Fc may be able to ameliorate circulating monocytes/macrophages before they target toward the injured corpus callosum. Confocal microscopy images showed that macrophages surrounding OPCs were decreased in corpus callosum of CD200-treated mice (Figure 6(a)). Mice that were treated with CD200-Fc showed a significant suppression in TLR4 positive macrophage accumulation within the lesioned white matter compared to controls (Figure 6(b)) at days 3 and 7 after ET-1 injection. Concomitantly, the number of OPCs and mature oligodendrocytes were also preserved in the CD200-Fc-treated brains (Figure 6(c) and (d)). Finally, using electron microscopy, we confirmed that CD200-Fc treatment significantly improved myelination at day 14 after ET-1-induced white matter stroke (Figure 6(e)), suggesting that protecting OPCs from macrophage attack may facilitate newly generated oligodendrocytes for white matter remodeling.
Intraperitoneal administration of CD200-Fc reduced TLR4 expression in blood monocyte/macrophages after white matter ischemia in vivo: (a) Western blot or FACS analysis was performed using blood samples from ET-1-induced white matter stroke in mice. CD200-Fc (100 µg/kg) or mouse IgG (100 µg/kg) was administered on day 1 after ET-1 injection, and then TLR4 expression in blood monocyte/macrophages was assessed on day 3. (b) Western blot analysis confirmed that intraperitoneal treatment with CD200-Fc increased plasma CD200 expression (IgG; n = 3, CD200-Fc; n = 3). (c) FACS analysis revealed that CD200-Fc did not strongly affect the count of total monocyte/macrophage in the blood, but significantly reduced TLR4 expression compared to IgG-treated group at day 3 after ET-1 injection. *P < 0.05, **P < 0.01 versus sham. (Total F4/80 or F4/80/TLR4; sham n = 3, IgG n = 4, CD200-Fc n = 4). All data are presented as mean ± SEM. CD200-Fc treatment enhances remyelination after ET-1-induced white matter ischemia in vivo: (a) Confocal microscope images indicated that treatment with CD200-Fc (100 µg/kg) at days 1 and 5 after ET-1-injection into mouse corpus callosum preserved PDGFRα-positive OPCs (green) at day 7. Scale bar: 20 µm, macrophage: F4/80 (red). (b) CD200-Fc treatment significantly suppressed the accumulation of TLR4-positive macrophages in the ET-1-damaged white matter area (day 3—IgG; n = 4, CD200 Fc; n = 4, day 7—IgG; n = 4, CD200 Fc; n = 3, day 14—IgG; n = 4, CD200 Fc; n = 4). (c) FACS analysis showed that the number of OPCs was significantly preserved after multiple treatments with CD200-Fc on day 7 after white matter injury (IgG; n = 3, CD200 Fc; n = 3). (d) The number of GSTpi-positive mature oligodendrocytes was significantly increased after multiple treatments with CD200-Fc on day 14 after ET-1 injection (IgG; n = 5, CD200 Fc; n = 3). (e) Electron micrograph of axons in the corpus callosum from day 14 after ET-1 treatment. Quantification of g ratio showed the significant improvement of myelination in CD200 Fc-treated mice (control; n = 100 axons, IgG; n = 108 axons, CD200-Fc; n = 101 axons). Scale: 1 µm. All data are presented as mean ± SEM.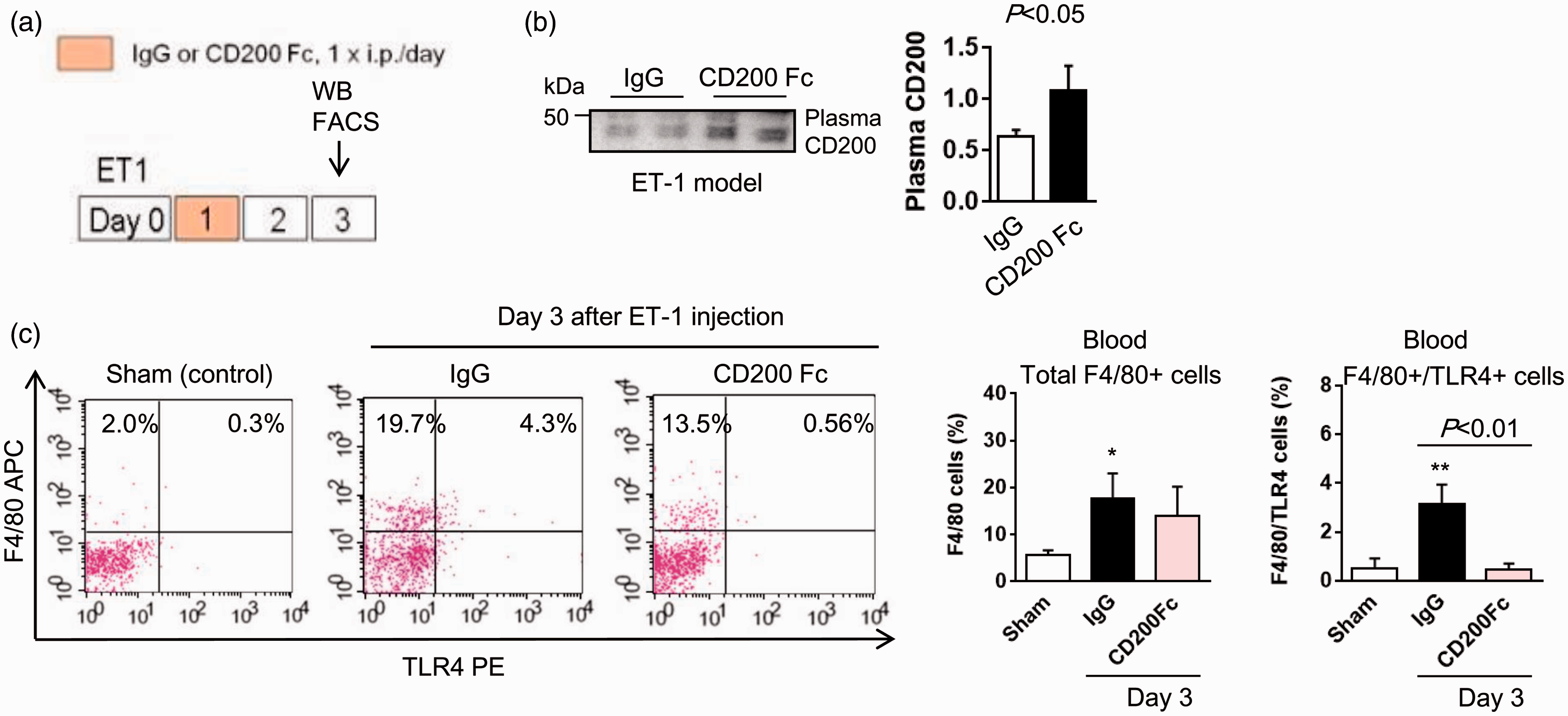
Discussion
After stroke and brain injury, endogenous attempts at self-repair are complex and poorly understood. Many cell types and intercellular signals are involved, and often, these pathways comprise a shifting balance between beneficial and deleterious responses. Furthermore, beyond cell–cell signaling within injured brain per se, dynamic crosstalk between brain and systemic responses such as circulating blood cells may also be important. 25 In particular, monocyte/macrophage cells may contribute to both brain damage and remodeling26,27 depending on the inflammatory circumstance. 23 The present study may be consistent with this overall conceptual framework. Our primary cell culture and in vivo data suggest that macrophages can attack OPCs via TLR4-NG2 signaling, and treatment with CD200-Fc administration boosts white matter recovery by suppressing the macrophage attack and phagocytosis of susceptible OPCs. Macrophages are traditionally thought to be somewhat beneficial by mediating tissue cleanup and debris clearance, thus facilitating repair and recovery. Our findings here suggest that in addition to positive “M2-like” phenomenon, activated macrophages may also possess detrimental “M1-like” actions in damaged white matter.
TLRs are traditionally considered as sensors for danger-associated molecules produced by bacteria and viruses to discriminate between “self” and “nonself” in innate immune system. More recently, emerging data demonstrate that TLRs signaling may have critical roles in both detrimental inflammatory responses and beneficial repair processes after CNS injury. For example, TLR4-deficient mice show smaller infarction along with less inflammatory substances such as iNOS and cyclooxygenase compared to wild type. 28 LPS did not affect cell survival of oligodendrocyte precursors, but microglial TLR4 activation by LPS caused oligodendrocyte precursor injury. 29 On the other hand, TLR4 and TLR2 mutations may cause impairments of myelination and long-term recovery of locomotor function after spinal cord injury, 30 suggesting that TLRs may give various impact upon the oligodendrocyte function and homeostasis after CNS injury. Our current findings may provide a novel mechanism that macrophage-expressing TLR4 can directly interact with OPC-derived NG2 and promote OPC phagocytosis in acute stage thus interfering endogenous oligodendrogenesis in late stage after white matter injury. This TLR4-NG2-mediated attack on OPCs may indeed comprise an underlying mechanism that potentially explains the damaging effects of macrophages in damaged white matter in our model systems.
The key role of neuroimmune regulatory proteins (NIRegs) has well been evidenced in the control of macrophage or microglia activation. For example, death signaling pathways based on CD95L and CD95 interaction transmit an apoptotic signal to target T cells. 31 CD47-SIRPα signaling behaves as “don’t eat me” signals to prevent unwarranted phagocytosis in macrophage or microglia. 32 Siglecs are known as a class of receptors that can bind to sialylated glycoproteins and glycolipids contain immunoreceptor tyrosine-based inhibitory motifs in their cytoplasmic tail which may negatively regulate the function of phagocytes, including microglia. 33 More recently, the concept of macrophage polarization toward different phenotypes after CNS injury has also been explored. In general, macrophage phenotype can be characterized as classically activated pro-inflammatory forms (M1-like) or alternatively activated forms (M2-like). Inflammatory microglia/macrophages promote secondary injury, 34 whereas M2-like macrophages may participate to debris cleanup and remodeling. 35 Recent report showed the important role of CCR2 in the phenotype polarization in monocyte. Blockade of CCR2 increased M1 phenotype and resulted in increased infarct volume along with exacerbated functional deficits. 36 Additionally, it has been reported that HDAC inhibition shifted microglia/macrophage to M2 phenotype and prevented white matter injury. 37 As a compartment of NIRegs, CD200 can bind to CD200 receptor which predominantly expresses on myeloid lineage cells and suppress the inflammatory responses. 38 In this study, we demonstrate another example which CD200-Fc administration boosts white matter recovery by suppressing macrophage attack and phagocytosis of susceptible OPCs through TLR4 downregulation. Additionally, CD200-Fc may not alter the ability of debris clearance in macrophages. Thus, these novel pathways may lead to new therapeutic opportunities for promoting white matter recovery after injury and disease.
Nevertheless, there are several issues that warrant further consideration. First, we only focus on macrophages as a major representative cell of the inflammatory response. But other cells may also participate in white matter injury. For example, many studies have implicated microglia in a wide spectrum of CNS disorders.39,40 In our OPC cultures, microglia can attack OPCs, but compared to macrophages, the degree of phagocytotic attack was smaller. The difference of cellular responses should be further investigated, especially in blood where besides monocytes, potentially beneficial cells such as endothelial progenitor cells may also be recruited.20,41 To study the effects of CD200 on all cell types is outside our scope for now. Whether and how CD200 affects other inflammatory and circulating cell types bearing CD200 receptors should be carefully examined in future studies. Second, our data suggest that TLR4 in macrophages would recognize NG2 in OPCs for the initiation of their phagocytosis. However, other brain cells such as pericytes may also express NG2. 42 Activated NG2 positive pericytes trigger the chemotactic migration of leukocytes and upregulation of TLRs, integrins, MMPs on neutrophils and monocytes. 43 The importance of NG2-positive pericytes in CNS disorders is well recognized now. 42 Whether and how our hypothesized CD200 mechanism affects pericytes should be further investigated. Third, rat peritoneal macrophages attacked OPCs but not mature oligodendrocytes in our in vitro model system, potentially due to the differential expression of NG2. But exactly how mature oligodendrocytes able to avoid macrophage attack remains to be fully elucidated. After CNS injury, degenerated myelin is phagocytosed by activated microglia or macrophages. However, healthy oligodendrocytes express CD47 that may serve as a “don’t eat me” signal to prevent phagocytosis by microglia or macrophages. 44 Besides our hypothesis which is that OPC-derived NG2 enhances the ability of macrophages to target OPCs, endogenous NIRegs expression in mature oligodendrocytes might be an important complementary mechanism that allow oligodendrocytes to avoid macrophage attack. Fourth, we should also consider the roles of CD200 in vascular remodeling after white matter damage. Neurovascular and gliovascular remodeling are important facets in the chronic phase of almost all CNS diseases.45,46 In the so-called oligovascular niche in white matter, trophic coupling between OPCs and vascular compartments regulates both angiogenesis and oligodendrogenesis. 14 Our current study demonstrates that CD200 may promote oligodendrogenesis recovery mechanisms. Future studies may need to examine whether this mechanism may also affect angiogenesis via oligovascular signaling. Finally, it must be acknowledged that there are no perfect models for white matter injury and disease. In this study, data from both ET-1 injections in vivo and primary OPC/macrophage cultures in vitro were consistent with hypothesis. But further studies in other model systems are warranted in order to assess the generalizability of our hypothesized phenomenon.
The proliferation and maturation of progenitor cells may provide an important endogenous protective and repair mechanism in CNS disease. However, this internally programmed protective system may be self-limited because of the continuing development of a potentially hostile inflammatory environment in damaged or diseased tissue. In gray matter, compensatory neurogenesis may be dampened by activated microglia.47,48 Here, we show that an analogous phenomenon may operate in damaged white matter. Accumulated macrophages appear to recognize and attack susceptible OPCs via TLR4-NG2 signaling. Treatment with CD200-Fc may downregulate TLR4, dampen the macrophage attack on OPCs, and promote white matter recovery. Further investigation into these mechanisms may lead to novel therapeutic opportunities for modulating the balance between positive versus negative effects of macrophage inflammation, and promote white matter repair in CNS diseases.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is
Acknowledgement
The authors thank Dr S. T. Carmichael for helpful discussions on white matter stroke modeling.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
KH contributed to manuscript preparation, experimental design/analysis, and conducted experiments. LDP, JHS, NM, TM, and YT, contributed to conducted experiments. SS and CW contributed to data analysis. DB, KVL, KWK, KA, and EHL contributed to manuscript preparation and experimental design.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
