Abstract
Human neuroinflammatory and neurodegenerative diseases, whose prevalence keeps rising, are still unsolved pathobiological/therapeutical problems. Among others, recent etiology hypotheses stressed as their main driver a chronic neuroinflammation, which is mediated by innate immunity-related protein oligomers: the inflammasomes. A panoply of exogenous and/or endogenous harmful agents activates inflammasomes’ assembly, signaling, and IL-1β/IL-18 production and neural cells’ pyroptotic death. The underlying concept is that inflammasomes’ chronic activation advances neurodegeneration while their short-lasting operation restores tissue homeostasis. Hence, from a therapeutic standpoint, it is crucial to understand inflammasomes’ regulatory mechanisms. About this, a deluge of recent studies focused on the NLRP3 inflammasome with suggestions that its pharmacologic block would hinder neurodegeneration. Yet hitherto no evidence proves this view. Moreover, known inflammasomes are numerous, and the mechanisms regulating their expression and function may vary with the involved animal species and strains, as well as organs and cells, and the harmful factors triggered as a result. Therefore, while presently leaving out some little-studied inflammasomes, this review focuses on the “other than NLRP3” inflammasomes that participate in neuroinflammation’s complex mechanisms: NLRP1, NLRP2, NLRC4, and AIM2. Although human-specific data about them are relatively scant, we stress that only a holistic view including several human brain inflammasomes and other potential pathogenetic drivers will lead to successful therapies for neuroinflammatory and neurodegenerative diseases.
Introduction
The innate immune system is the body’s first line of defense against cell or tissue acute damage inflicted by bacterial, viral, or fungal infections or stressors such as cell-released sterile products (e.g., DNA, ATP), dead cells, or metabolic imbalance products. Innate immunity detects these noxae via extra- and intracellular pattern recognition receptors (PRRs). PRRs sense conserved microbial or viral structures, termed pathogen-associated molecular patterns (PAMPs), host-derived compounds named damage-associated molecular patterns (DAMPs), or homeostasis-altering molecular patterns (HAMPs). These various stimuli induce a two-step process leading to the assembly and signaling activation of inflammasomes. In the priming first step (or signal 1), PAMPs, DAMPs, or HAMPs activate via nuclear factor κB (NF-κB) the synthesis of pro–IL-1β and pro–IL-18 (Martinon and others 2002; Kigerl and others 2014). Next, a polymorph signal 2 triggered by a range of factors—such as K+ efflux, high extracellular [K+]e, Ca2+ dyshomeostasis, mitochondrial and/or lysosomal dysfunction, reactive oxygen species (ROS) surplus formation, and oxidized mitochondrial DNA (Latz and others 2013; Silverman and others 2009)—induces the assembly of cytosolic high molecular weight protein oligomers, the inflammasomes, including the cysteine aspartate caspase 1. Couples of the latter undergo autocatalytic activation and next cleave pro–IL-1β and pro–IL-18 into their mature forms, with gasdermin D (GSDMD) also freeing its pore-forming N-terminal fragment (Fig. 1; de Vasconcelos and Lamkanfi 2020; Vanaja and others 2015). Inflammasomes’ signals trigger inflammatory responses aimed at restoring tissue homeostasis. However, when dysregulated for being too intense and long-lasting, such responses become detrimental (Chiarini and others 2020; Rathinam and others 2012) causing the following: metabolic disorders (e.g., type 2 diabetes [Gora and others 2021] and gout [Zou and others 2022]), heart disease (He and others 2012), autoimmune diseases (e.g., rheumatoid arthritis and systemic lupus erythematosus [SLE; Kong and others 2022; Shen and others 2018]), psoriasis (Carlström and others 2012), vitiligo (Levandowski and others 2013), hepatic fibrosis (Gaul and others 2020), nonalcoholic or alcoholic steatohepatitis (Knorr and others 2020), stroke (Fann, Lee, Manzanero, Chunduri, and others 2013; Fann, Lee, Manzanero, Tang, and others 2013), neurodegenerative diseases (e.g., Alzheimer [Halle and others 2008; Ising and others 2019], Parkinson [Wang and others 2022], multiple sclerosis [Malhotra and others 2020]), and allogeneic skin transplant rejection [Amores-Iniesta and others 2017], coronavirus-induced severe acute respiratory syndromes (SARS-CoV-2 included [de Rivero Vaccari and others 2020; Nieto-Torres and others 2015; Siu and others 2019]), and autoinflammatory syndromes due to gain-of-function mutations of genes encoding for inflammasome sensors (Booshehri and Hoffman 2019; Moghaddas and others 2018). Such PRRs form groups sharing structural motifs as the NOD-like nucleotide-binding domain and leucine-rich repeat–containing family of receptors (NLRs), the absent in melanoma 2–like receptors (ALRs), and the PYRIN receptors (de Zoete and others 2014; Guo and others 2015). The canonically signaling NLRs are the ones most intensely studied, being considered the most important. Human NLRs owning a PYRIN N-terminal homology domain (PYD) include 14 members: NLRP1, NLRP2, NLRP3, . . . NLR14. Physiologically, NLRs mostly assume an autoinhibited conformation (Hauenstein and others 2015), but brain NLRs are an exception to this. NLRs’ (i.e., NLRP1, NLRP2, NLRC1/2, NLRC4) autoinhibition ceases upon PAMP, DAMP, or HAMP detection: this starts the oligomeric inflammasomes’ assembly and signal transduction. N-terminal PYDs crucially bind and sequentially nucleate the oligomerization of the adaptor ASC protein (apoptosis-associated speck like protein endowed with a caspase recruitment domain [CARD]; de Alba 2019). Because the ASC gene also encodes a PYD, the ASC protein can form complexes with the NLRs PYD or CARD domains via CARD•CARD or PYD•PYD homotypic interactions. PYDs and CARDs are conserved protein interactive domains of about 80 to 90 amino acids forming six antiparallel α helices with a hydrophobic core and peripheral charges on their outer surfaces. Moreover, the CARD-endowed inflammasomal proteins activate NF-κB transcription-regulated genes encoding the inflammasomes’ structural proteins (Kesavardhana and Kanneganti 2017).
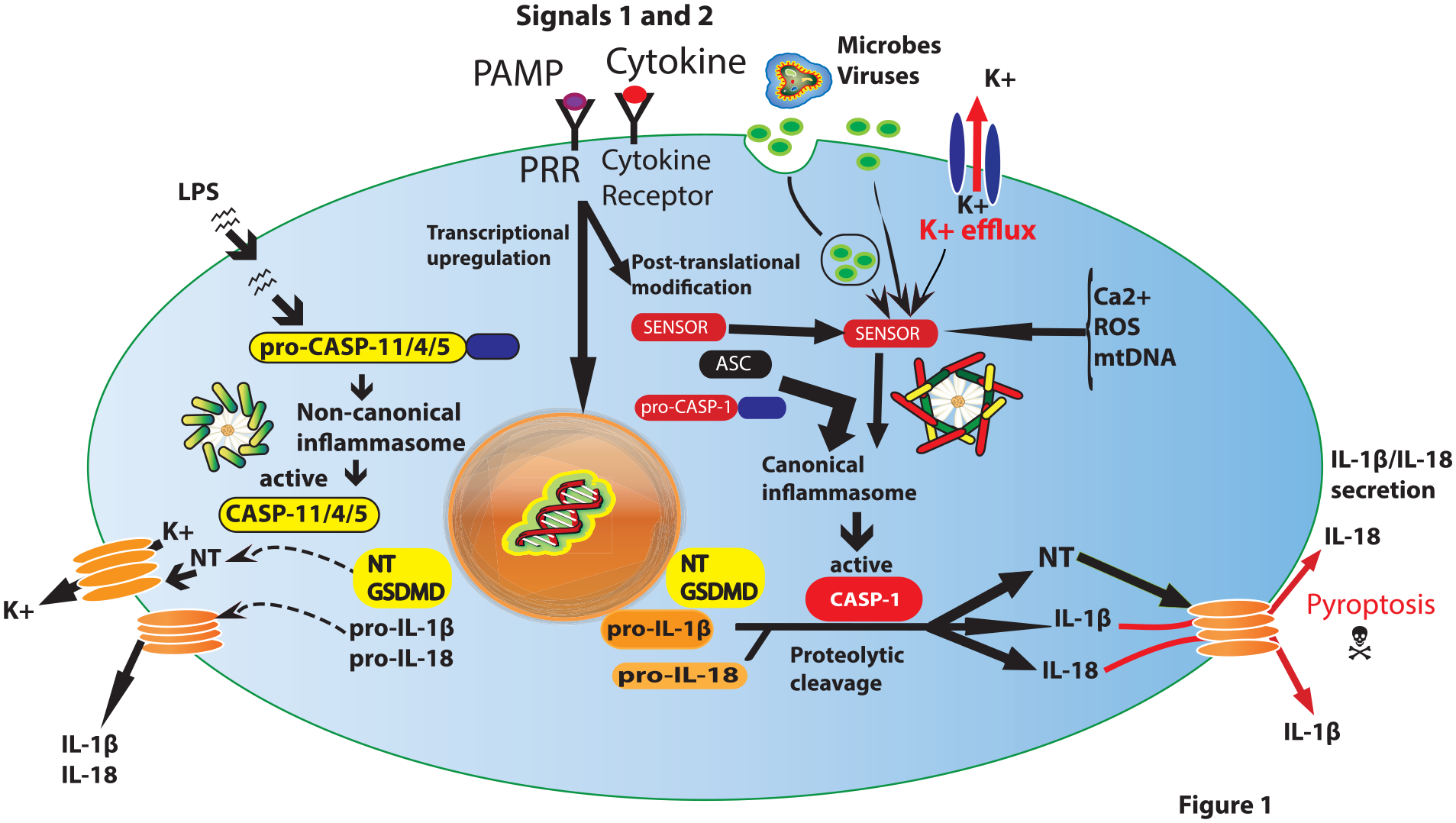
Inflammasome activation. Inflammasomes are multiprotein complexes generally encompassing a sensor (NLRs or ALRs) and an effector inflammatory caspase associated to an adaptor protein (ASC). Inflammasomes are defined as “canonical” when their assembly requires caspase 1 and as “noncanonical” when their assembly depends on human caspase 4 or 5 or murine ortholog caspase 11, directly bound to cytosolic LPS from Gram-negative bacteria. Physiologically, ALRs or NLRs are usually in autoinhibited conformations, except in the central nervous system, where inflammasomes are preassembled before any stimulation. A combination of two variable signals (e.g., PAMPs, cytokines, LPS, high K+ efflux) can induce the assembly of individual inflammasomes that provide a platform for caspase 1 autocatalysis and activation. Activated caspase 1 mediates proteolytic precursor cleavage and the secretion of mature IL-1β and IL-18 proinflammatory cytokines. Once triggered, caspase 1 also cleaves gasdermin D, whose N-terminal (NT) fragment forms pores in the membranes, which can result in pyroptosis. ASC = apoptosis-associated speck-like protein containing a CARD; CASP = caspase; GSDMD = gasdermin D; IL = interleukin; LPS = bacterial lipopolysaccharide; mtDNA = damaged mitochondrial DNA; PAMP = pathogen-associated molecular patterns; PRR = pattern recognition receptor.
Canonical inflammasomes’ CARD•CARD interactions muster via ASCs the precursors (or zymogens) of the protease caspase 1, causing their nucleated polymerization, and proximity-mediated autocatalytic self-cleavage, resulting in activated caspase 1 couples (Elliot and others 2009; Stehlik and others 2003). The latter cleave various proteins sharing the YVHD/FESD consensus sequence, including the precursors of interleukin 1β (IL-1β) and IL-18 and the human GSDMD and rodent GsdmD protein (Julien and Wells 2017; Shi and others 2015). The caspase 1–cleaved GSDMD/GsdmD N-terminal fragments oligomerize and form transmembrane pores through which mature IL-1β and IL-18 are released into the environment to trigger inflammation with K+ losses altering cells’ ion homeostasis. When persisting, such losses induce inflammatory cell death or pyroptosis, whose products intensify inflammation (Ding and others 2016). NLRP oligomerization, ASC recruitment, and caspase 1–nucleated polymerization/activation are no-return processes going on in a self-inducing prion-like fashion to drive inflammasomes’ canonical signals (Gambin and others 2018; Huber and others 2015).
Notably, other NLRs, such as NLRC3, NLRP6, NLRP12, and NLRX1, inhibit the NF-κB pathway to mitigate or suppress inflammation (Lupfer and Kanneganti 2013). Indeed, the latter inflammasomes are a crucial necessity to prevent chronic inflammatory disease. Other modulating mechanisms involve small PYD-only proteins (POPs) and/or CARD-only proteins (COPs), each of which owns a single PYD or CARD. The competitive binding of small-sized POPs and/or COPs can effectively hinder the PYD- and/or CARD-mediated interactions of multidomain proteins by blocking inflammasomes’ oligomeric assembly and signaling. Interestingly, POP1, POP2, and POP3, whose genes appeared about 25 million years ago, occur only in higher primates’ (chimpanzees and rhesus macaques) and humans’ genomes and bind inflammasomes’ PRRs or the ASC protein, hindering the assembly of PYD•ASC complexes (Atianand and Harton 2011). NLRP2P, a human unusually “resurrected” pseudogene (retrotransposon) closely related to the NLRP2 and POP2 genes, encodes POP4, which curtails the Toll-like receptor–stimulated production of the NF-κB–regulated cytokines tumor necrosis factor α (TNFα) and IL-6 but does not inhibit the NLRP3 inflammasome (Porter and others 2014). The three known human COPs—CARD16, CARD17, and CARD18—share various homology degrees with and bind caspase 1’s CARD, while only CARD16 binds ASC’s CARD (Devi and others 2020). Also, recent evidence showed that three main epigenetic mechanisms regulate inflammasomes’ activity via CpG islands DNA methylation, histones’ posttranslational changes, and noncoding RNAs’ expression (Poli and others 2020).
Conversely, the direct binding of the Gram-negative bacteria lipopolysaccharide (LPS) to the CARD domains of human caspase 4/5 or mouse caspase 11 results in the activation of noncanonical inflammasomes (Kayagaki and others 2011; Kayagaki and others 2013; Vigano and others 2015).
Concerning neuroinflammation, the various brain cell types—neurons, astrocytes, microglia, and pericytes—express a panoply of inflammasomes (de Rivero Vaccari and others 2014; Lech and others 2010; Minkiewicz and others 2013; Nyúl-Tóth and others 2017). A current view suggests that NLRP1 is most intensely expressed by neurons, NLRP2 by astrocytes, and NLRP3 by microglia. However, reality is more complex. Brain inflammasomes drive a neuroinflammatory response through the canonical and noncanonical release of IL-1β and IL-18 cytokines that causes pyroptosis (Kesavardhana and Kanneganti 2017). At odds with Saresella, La Rosa, and others (2016), a recent human postmortem investigation gained no proof that the NLRP3 inflammasome’s mRNA expression is changed in the temporal cortex of patients with Alzheimer’s disease (AD) as compared with sex- and age-matched controls (Tang and Harte 2021). While these interesting findings require extension and confirmation about the NLRP3 protein expression, they stress the need to focus the experts’ attention on the multiple roles of “other than NLRP3” brain inflammasomes in human neuroinflammatory and neurodegenerative diseases: the purpose of this review.
NLRP1 Inflammasome
NLRP1 (NALP1) was the first discovered inflammasome in mouse brain ischemic and rat spinal cord injuries (Abulafia and others 2009; de Rivero Vaccari and others 2008). NLRP1 components gather and signal in neurons and glial cells (astrocytes, oligodendrocytes, and microglia/macrophages). The activating transcription factor 4 (ATF4 or cAMP response element-binding protein 2 [CREB-2]) regulates Nlrp1 gene expression in rat spinal neurons and in THP1, K562, and JURKAT human cell lines (Lin and others 2016). ATF4 is the effector of the cells’ integrated stress response (ISR) driven by various stimuli (Box; Fig. 2). Injection of an adeno-associated heme oxygenase 1 (HO-1)–expressing virus following spinal cord injury downregulated the expression of ATF4 and NLRP1, thereby advancing in vivo rat neurons’ survival (Lin and others 2016).
Cells’ Integrated Stress Response.

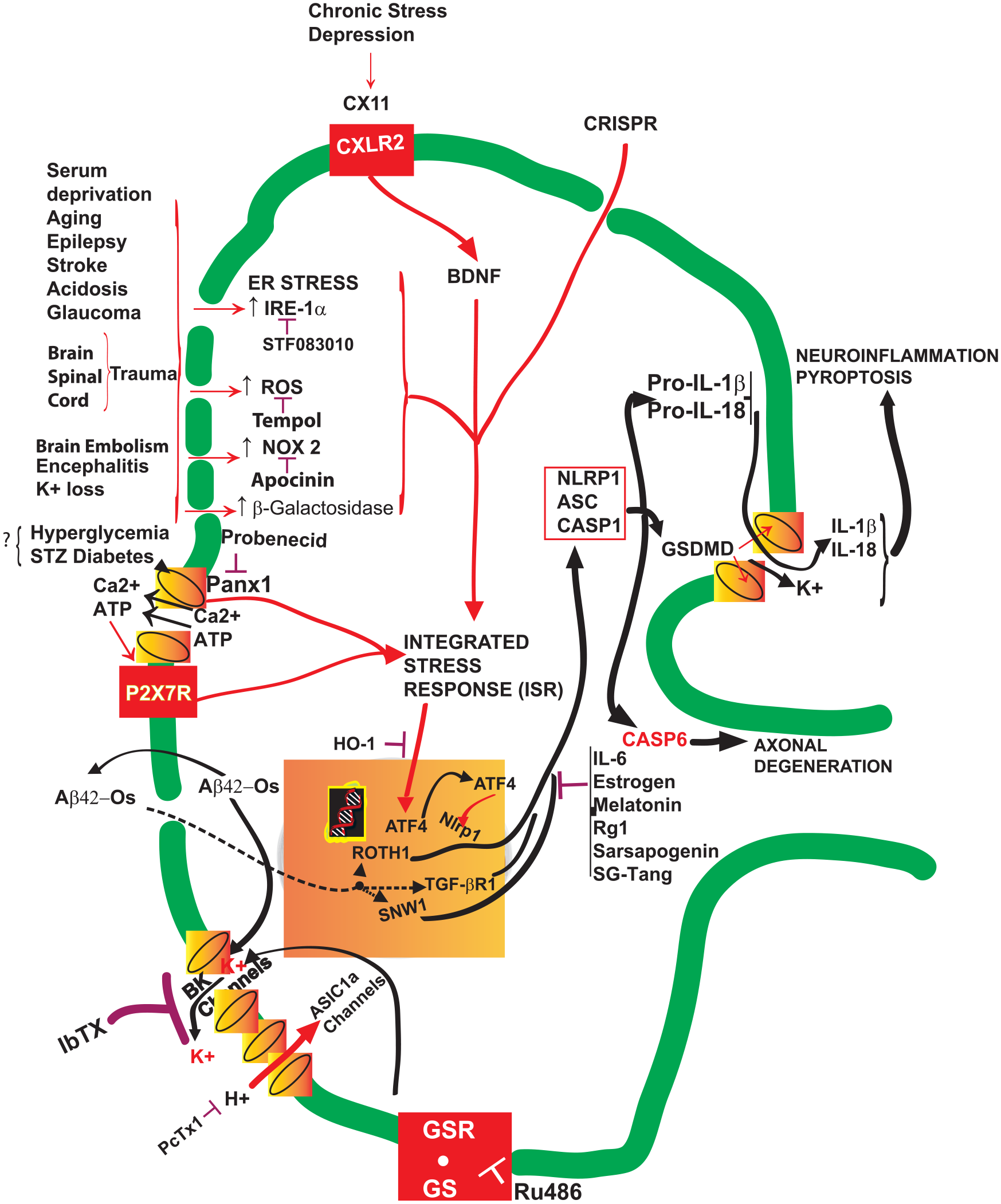
Factors influencing activation of the NLRP1 inflammasome. The graphic schematically depicts a neuron’s body and the initial portion of its axon. Plasma membrane, green. Nuclear envelope, yellow-orange. The several stressors activating the inflammasome are shown at the picture’s top and left sides. The assembled inflammasome is enclosed in a red frame. Aβ42-Os = amyloid β42 oligomers; ATF4 = activating transcription factor 4; BDNF = brain-derived neurotrophic factor; BK = big potassium channel; CASP = caspase; CX11 = C-X-C motif chemokine 11; CXCR2 = C-X-C motif chemokine receptor 2; CRISPR = clustered regularly interspaced short palindromic bacterial DNA repeats; GS = glucocorticoid (dexamethasone); GSDMD = gasdermin D; GSR = glucocorticoids receptor; HO-1 = adeno-associated heme oxygenase 1; IbTX = iberiotoxin; IRE-1α = inositol-requiring enzyme 1α; ISR = integrated stress response; NOX2 = NADPH oxidase 2; P2X7R = purinergic P2X receptor 7; Panx1 = pannexin 1; PcTX1 = psalmotoxin 1; ROS = reactive oxygen species; ROTH1 = atypical mitochondrial Ras homolog family member T1 gene; Ru486 = mifepristone; SG-Tang = Shaoyao Gancao Tang; STZ = streptozotocin; SWN1 = SWN domain-containing 1 gene; TGF-βR1 = transforming growth factor β receptor 1.
NLRP1’s N-terminal PYD enables a PYD•PYD–mediated NLR-nucleated oligomerization and the binding of the PYD- and CARD-equipped ASC adaptor proteins. Next, a CARD•CARD interaction allows pro–caspase 1 binding, followed by inflammasome’s signaling activation (Kesavardhana and Kanneganti 2017). A unique characteristic the NLRP1 inflammasome is a C-terminal CARD domain that binds a pro–caspase 1’s CARD with no ASC interposition (Schroder and Tschopp 2010). At NLRP1’s C-terminus there is a FIIND domain (function to find), whose autolytic cleavage at Ser1213 is needed for the inflammasome’s activation (Finger and others 2012).
Various Models of Brain and Spinal Cord Injuries
Various animal models of brain and spinal cord injury (trauma, thromboembolism, stroke, extracellular acidosis, hyperglycemia) have involved all the NLRP1 inflammasome components (Abulafia and others 2009; de Rivero Vaccari and others 2008; de Rivero Vaccari and others 2009; de Rivero Vaccari and others 2016; Freeman and Ting 2016; Wang and others 2015). Lines of evidence proved that cellular K+ losses were linked to NLRP1 signaling activation within in vitro and in vivo mouse hippocampal neurons (Zhang B, Zhang, Xu, and others 2017; Zhang B, Zhang, Wu, and others 2017). Chronic glucocorticoid (dexamethasone) treatment drove K+ ion loss through the BK (big potassium) voltage-gated large-conductance K+ channels. The consequently decreased intracellular K+ levels increased oxidative stress and NOX2 (NADPH oxidase 2), p22phox, p47phox, NF-κB, and phosphorylated NF-κB expression while activating NLRP1 inflammasome signaling and promoting neuroinflammation in the hippocampal neurons of mice. Inhibiting the glucocorticoid receptor with mifepristone (RU486) or NOX2 with apocynin or downregulating NLRP1 via siRNA or scavenging ROS with Tempol (Fig. 2) effectively suppressed the glucocorticoid-induced harmful effects on mouse hippocampal neurons while improving the behavioral and memory deficits in the treated mice (Hu W and others 2016; Sun and others 2019; Zhang B, Zhang, Xu, and others 2017; Zhang B, Zhang, Wu, and others 2017). Interestingly, extracellular acidosis activated neuronal H+-gated acid-sensing ion channels (ASICs), which increased BK channel currents and K+ ion efflux, activating NLRP1 signaling. Psalmotoxin 1 (PcTX1), a spider protein toxin from the Trinidad tarantula Psalmopoeus cambridgei that inhibits ASIC1a, and iberiotoxin (IbTX), a toxin from the eastern Indian red scorpion Hottentotta tamulus that inhibits BK channels (Fig. 2), effectively blocked the harmful effects of extracellular acidosis and safeguarded rat cortical neurons’ viability (Wang and others 2015). Injections of a NLRP1-neutralizing antibody into the postischemic brains of mice hindered NLRP1 inflammasome activation, decreased mature IL-1β and IL-18 levels, and kept neurons viable. These results attested that NLRP1 plays a pivotal role in postischemic neuroinflammation (Abulafia and others 2009; Fann, Lee, Manzanero, Tang, and others 2013; White and others 2017). Also, neuroinflammation driven by NLRP1 inflammasome activation underlay the chronic stress–elicited depressive-like behavior of mice related to the activated CXCl1/CXCR2/BDNF signaling pathway in the hippocampus. Nlrp1 knockdown suppressed CXCl1/CXCR2 upregulation and mitigated depressive-like behavior in mice (Song and others 2020). Yet, contusive (T6) spinal cord injuries in rats activated the synthesis and signaling of NLRP1 and NLRP3 inflammasomes; both such effects were mitigated by estrogen or melatonin (Majidpoor and others 2020).
Retinal glia activation and neuroinflammation triggered by acute ocular hypertension (glaucoma) involved NLRP1, NLRP3, and Aim2 inflammasomes in ganglionic neurons, astrocytes, and Müller glia coupled with the induction of GSDMD in rodents’ glia and ganglion neurons, followed by pyroptosis. These harmful effects were mitigated in the retinas of CASP 1-/-, CASP 4(11)del, and PANX 1-/- mice or wild type mice treated with probenecid (a pannexin 1 [panx-1] channel inhibitor; Pronin and others 2019). Panx-1 was also involved in NLRP1’s inflammasome activation in the cerebral cortex of streptozotocin (STZ)–induced diabetic rats, suggesting an added mechanism of neuronal damage in diabetes (Meng and others 2014).
In a neonatal rat model of hypoxic-ischemic encephalopathy NLRP1, active caspase 1, IL-1β, IL-18, and phosphorylated endogenous IRE-1α (inositol-requiring enzyme-1α)—a transmembrane sensor protein associated with endoplasmic reticulum stress—were upregulated, while miR-125-b-2-3p was downregulated. Administration of STF-083010, a powerful inhibitor of endoplasmic reticulum IRE-1α, neuroprotectively reversed all such effects, thus reducing neuronal pyroptosis. In turn, anti-miR-125 or NLRP1 activation by CRISPR (clustered regularly interspaced short palindromic bacterial DNA repeats) hindered STF-083010’s beneficial effects (Huang and others 2020).
Aging and AD
Studies conducted in progressively aging rodents (from 6 to 24 mo) showed that NLRP1 signaling activation concurred with increases in ROS levels, NOX2 and β-galactosidase activities, and neuronal pyroptosis in the hippocampi and cerebral cortices of mice aged 20 to 24 mo. These rodents also experienced learning, memory, and locomotion impairments. Such findings showed for the first time that NLRP1 inflammasome is involved in brain inflammaging (Sun and others 2021), an aging-related “physiologic” inflammation that may promote AD or Parkinson disease. They were confirmed by using primary in vitro cultures of hippocampal neurons (Xu T and others 2019).
Multiple reports attest that AD-related neuroinflammation involves the signaling of NLRP1 inflammasome. Remarkably, NLRP1 and NLRP3 mRNA expression was increased in the monocytes of mild and grave cases of human AD, being coupled with the overproduction of IL-1β and IL-18 (Saresella, La Rosa, and others 2016). The exposure to Aβ oligomers (Aβ-os), the primary AD drivers, induced cultured cortical neurons of Tg2576 mice to lose intracellular K+ ions via an efflux through the BK channels (Ray and others 2011). Similarly, the fall of [K+]i powerfully triggered the NLRP1 inflammasome canonical signaling and its upshots in rat neurons after spinal cord injury (de Rivero Vaccari and others 2008). Aβ peptides also activated the NLRP1 inflammasome canonical signaling within the pyramidal neurons and oligodendrocytes of AD model mice. In vitro–stressed (i.e., serum-deprived) human neurons assembled the NLRP1 but not the NLRP3 inflammasome: the former activated first caspase 1 and next caspase 6. Active caspase 6 immunocolocalized with NLRP1 in the same serum-deprived neurons in which the Aβ42/Aβ40 ratio increased driving pyroptosis. This mechanism linked caspase 6–induced axonal degeneration to the caspase 1–produced mature IL-1β/IL-18–evoking neuroinflammation (Fig. 2; Kaushal and others 2015). Likewise, stress due to serum deprivation did not affect NLRP3 but activated the NLRP1 inflammasome and its downstream caspase 1/caspase 6 pathway in cultured human neurons, thereby increasing the Aβ42/Aβ40 ratio values and driving neurons’ pyroptotic death and axons’ fragmentation. Strikingly, in postmortem human AD brain tissues, the number of NLRP1 inflammasome–expressing neurons was 25- to 30-fold higher than in non-AD brains (Kaushal and others 2015; Yap and others 2019). Moreover, NLRP1 levels were higher in the brains of B6C3-Tg (APPSwe/PS1dE9) AD model mice than in their wild type counterparts. In fact, the treatment of cultured cortical neurons with Aβ peptides raised their rate of NLRP1-mediated caspase 1–dependent pyroptotic death. Neurons’ pyroptosis and cognitive impairment remarkably decreased after the knocking down of Nlrp1 or pro–caspase 1 in the B6C3-Tg mice (Tan and others 2014).
A gene trap mutagenesis screen approach revealed the contribution of three genes—ROTH1 (atypical mitochondrial Ras homolog family member T1), TGF-βR1, and SNW1 (SNW domain containing 1)—as relevant contributors to pyroptotic neuronal death mediated by Aβ-induced NLRP1 signaling (Yap and others 2021). Yet, the NLRP1 expression levels did not change in an AD-like model induced via STZ injections in male Wistar rats (Saadi and others 2020). Such discrepant results about NLRP1 activation are in keeping with other lines of evidence that the regulation of specific inflammasomes involved in the neuroinflammation coupled with CNS injury and repair varies among animal species and according to specific experimental stimuli (Healy and others 2020).
Epilepsy and Rasmussen Encephalitis
NLRP1 and NLRP3 were activated in the neurons and glial cells residing in the sclerotic hippocampi (surgical samples) of patients affected by mesial temporal lobe epilepsy, thus revealing a pathogenetic role of such inflammasomes in this illness (de Brito Toscano and others 2021; Tan and others 2015).
Finally, Rasmussen encephalitis, whose causative factor is unknown, involves the activation of NLRP1 and NLRP3 inflammasomes and their downstream neuroinflammation-promoting pathways, mechanistically contributing to the illness in children (Ramaswamy and others 2013).
Altogether, these data stress NLRP1’s relevance in neuroinflammatory and neurodegenerative conditions suggesting it as a potentially relevant therapeutic target.
NLRP2 Inflammasome
The NLRP2 gene belongs to the NLR family of innate immune sensors for sterile stress signals (DAMPs) and PAMPs. The NLRP2 (NALP2, PAN1, PYPAF2, or CLR19.9) sensor protein that it encodes has C-terminal LRRs (leucine-rich repeats), a central NACHT (nucleotide-binding and oligomerization domain), and an N-terminal PYD (PYRIN domain). Several observations revealed that species differences affect the expression levels of NLRP2 as well as other inflammasomes (Peng and others 2012). In fact, NLRP2 protein levels were constitutively low in the brains of wild-type C57/BL6J male mice (Sun and others 2016). Conversely, humans express four NLRP2 isoforms (for details, see Kinoshita and others 2005). In astrocytes and neurons, the assembled NLRP2 inflammasomes include the NLRP2 receptor/sensor protein and the PYD-bound ASC protein, whose CARD domain binds pro–caspase 1 couplets, which next undergo self-activation (Fig. 3). The NLRP2 inflammasome complex is capable of interacting with and being activated by the P2X7 purinergic receptor, which is linked to the Panx-1 channel (Minkiewicz and others 2013).
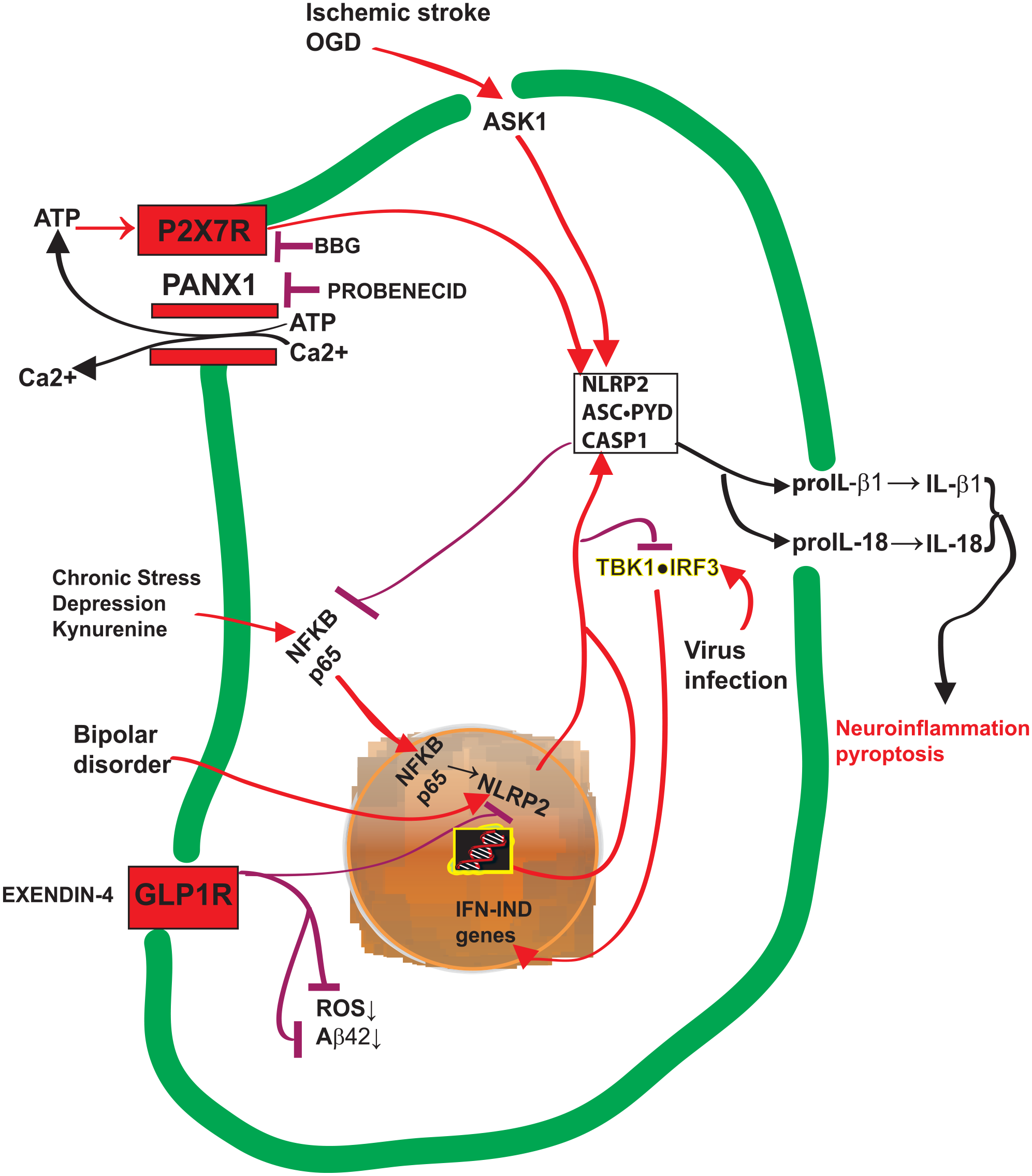
Factors regulating NLRP2 inflammasome’s activation and its outcomes. The graphic schematically depicts an astrocyte or a small-sized dorsal root ganglion neuron’s body. Plasma membrane, green. Nuclear envelope, yellow-orange. The various inflammasome-activating stressors are listed at the picture’s top and left. Aβ42 = amyloid β42; ASC = apoptosis-associated speck-like protein containing a CARD; ASK1 = apoptosis signal-regulating kinase; BBG = brilliant blue G; Casp = caspase; GLP-1R = glucagon-like peptide 1 receptor; IFN-IND = IFN-inducible genes; IRF3 = interferon regulatory factor 3; NFKB p65 = p65 subunit of nuclear factor κB; OGD = oxygen-glucose deprivation; Panx1 = pannexin 1; P2X7R = purinergic P2X receptor 7; PYD = PYRIN N-terminal homology domain; ROS = reactive oxygen species; TBK1 = TANK-binding kinase 1.
Available evidence shows that the functional control of the NLRP2 inflammasome depends on the cellular model investigated. The results of NLRP2 knockout in the human THP-1 monocytic cell line and NLRP2 hyperexpression in HEK 293T cells led Bruey and others (2004) to posit that NLRP2 inhibits NF-κB pathway activation at the IKKα complex level, as indirectly proven by a reduced expression of ICAM-1, an endogenous NF-κB target gene. Conversely, Fontalba and others (2007) posited that in a reciprocal regulatory loop, NF-κB’s p65 subunit transcription factor induced NLRP2 gene expression while NLRP2 hindered NF-κB’s pathway activation (Fig. 3).
Moreover, overexpressed NLRP2 protein formed complexes with TBK1 that hindered the phosphorylation of interferon regulatory factor 3 (IRF3) and consequently blocked the expression of IFN-inducible genes involved in the innate immunity antiviral response (Fig. 3). The NLRP2’s tight control of the IFN signaling cascade in response to viral infection would crucially check tissue harm and avert autoimmunity (Yang and others 2018). It is not understood whether this mechanism operates in human brain cells too.
Just like NLRP5 and NLRP7, the NLRP2 sensor partakes in activities unrelated to innate immunity, such as embryo development, tissue homeostasis, apoptosis, and graft-versus-host disease (Kufer and Sansonetti 2011). Particularly, NLRP2 regulates the 1) trophoblastic differentiation of human granulosa-induced pluripotent stem cells, 2) CD34+ hemopoietic progenitor differentiation into granulocytes, and 3) the transformation of mesenchymal stem cells and monocytes/macrophages into adipocytes (Chuang and others 2015).
Rodent NLRP2
Sun and others (2016) showed that NLRP2 expression, IL-1β and IL-18 production, and pyroptosis significantly increased in the cerebral cortex and corpus striatum of an ischemic stroke mouse model and in cultured oxygen-glucose–deprived (OGD) mouse astrocytes. Remarkably, the apoptosis signal-regulating kinase 1 (ASK1) was identified as an early activator and immune regulator inducing significant cell death after ischemic injury. ASK1 silencing remarkably reduced the NLRP2, IL-1β, and IL-18 levels in stroke model mice and in cultured mouse astrocytes. Therefore, the authors suggested that ASK1’s activity plays a crucial role in astrocytes’ NLRP2 inflammasome signaling and a proapoptotic role in mouse brain stroke (Cheon and others 2018). Nevertheless, ASK1’s role has hitherto remained unexplored in human brain stroke and other neuropathologic conditions.
The small-sized dorsal root ganglion neurons of the peripheral nervous system express the NLRP2 inflammasome, which may be involved in the development of pain hypersensitivity as observed in male C57BL/6 mice (Matsuoka and others 2019).
Interestingly, intraperitoneal injection of the tryptophan metabolite kynurenine to mice with chronic mild stress–induced depression or kynurenine administration to cultured mouse hippocampal astrocytes heightened by activating the NF-κB pathway NLRP2’s mRNA transcription and NLRP2 inflammasome’s signaling. Conversely, astrocytes’ NLRP2 knockdown abolished the kynurenine-induced depressive-like behavior of the mice. These results led the authors to suggest that NLRP2 plays a pivotal role in the kynurenine-elicited depression in mice (Zhang Q and others 2020). Whether kynurenine similarly affects NLRP2 expression in human astrocytes promoting depression remains to be verified.
Human NLRP2
It is not as yet understood whether and how NLRP2 affects human brain cell pyroptosis. Although the MAPK pathway activated NLRP2 signaling in human umbilical vein endothelial cells (Zhang and others 2019), it remains unproven that this mechanism operates in human brain cells too.
Interestingly, treating cultured human cortical primary astrocytes with ATP, a cell-released exogenous DAMP, activated the assembly of NLRP2 inflammasomal complexes and the release of mature IL-1β. Remarkably, both effects were suppressed by a siRNA-mediated NLRP2 silencing. The parallel finding that the untreated (control) human astrocytes also released low amounts of mature IL-1β showed for the first time that even under basal conditions, preassembled signaling NLRP2 inflammasomes were present in such cells. The authors posited that human astrocytes’ NLRP2 inflammasome importantly partakes in neuroinflammatory responses and hence is a potential therapeutic target to mitigate them (Minkiewicz and others 2013). However, it is noteworthy that quite little is known about the roles of NLRP2 inflammasomes in the various stages of human AD. The same consideration applies to AD model animals. This may depend on the fact that astrocytes’ roles in AD were quite often downplayed or neglected.
An NLRP2 gene dysregulation at an early stage of human fetal brain development permitted the distinguishing of bipolar disorder cases from healthy subjects (Vizlin-Hodzic and others 2017). Quantitative proteomic studies consistently showed that NLRP2 was the most significantly upregulated protein in neural stem cells and mature neural cells isolated from samples of patients with bipolar disorder (Truvé and others 2020).
Though still fragmentary, the important evidence about the functions of NLRP2 inflammasomes in human astrocytes and other brain cell types should entice neuroscientists to gain clarification on the pathophysiologic roles of this inflammasome.
Absent in Melanoma 2 (Aim2/AIM2) Inflammasomes
Viral and/or bacterial infections, cellular senescence, chronic inflammatory or autoimmune diseases (e.g., nephropathies, diabetes, atherosclerosis, AD, Parkinson, SLE), and tumorigenesis activate the cGaS (cyclic GMP-AMP synthase)/STING (stimulator of interferon genes) type I interferon (i.e., IFN-α/-β) stimulatory sequence-independent DNA (ISD) pathway (Duan and others 2011; Ishikawa and others 2009; Sharma and others 2019). Once activated, the ISD pathway induces the expression of the Aim2/AIM2–like receptor (ALR; murine/human) family genes. The latter encode for the evolutionarily conserved PYHIN (IFI200/HIN-200) p200 proteins, which are crucial innate immunity players acting protectively or harmfully (Owens and others 2014; Fig. 4). The PYHIN p200 proteins own a PYD domain and a C-terminal single or double DNA-binding HIN-200 domain (hematopoietic IFN-inducible nuclear 200-amino acid; Brunette and others 2012; Choubey 2012; Cridland and others 2012). Two successive oligonucleotide/oligosaccharide folds of the HIN-200 domain bind long double-strand DNA (dsDNA) fragments deriving from damaged nuclear and/or mitochondrial DNA or from infecting bacterial or viral pathogens (Veeranki and Choubey 2012). The HIN-200 domain also binds intranuclear DNA damaged sites (Hu B and others 2016; Jiang and others 2021).
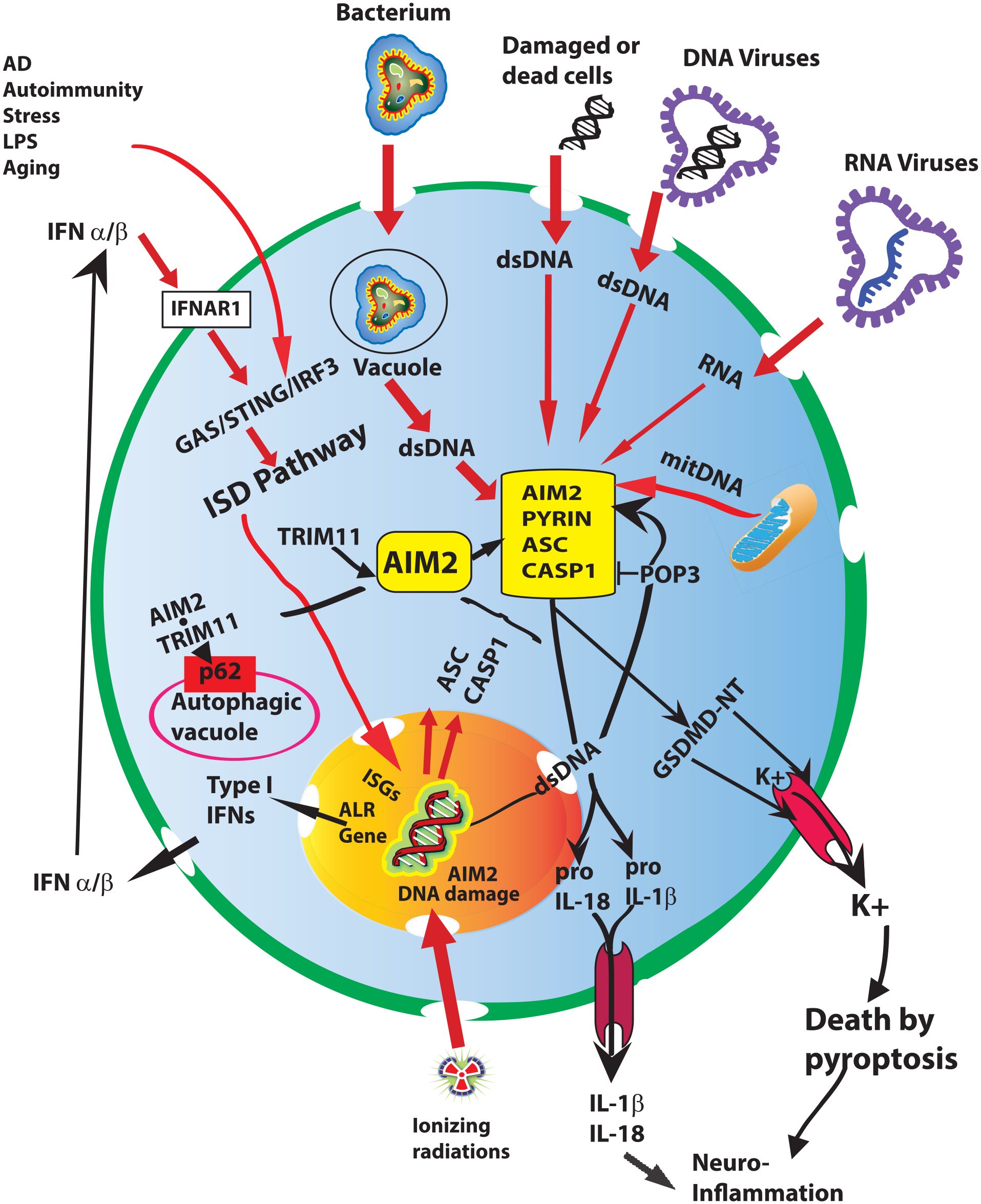
AIM2 inflammasome’s activation and its consequences. Schematic depiction of a generic neural cell. Plasma membrane, blue and nucleus, yellow-orange. The AIM2-activating stressors are listed at the picture’s top and left sides. Aβ42 = amyloid-β42; ASC = apoptosis-associated speck-like protein containing a CARD; Casp = caspase; dsDNA = double-strand DNA fragment; GAS = cyclic GMP-AMP synthase; IFNAR1 = interferon α and β receptor subunit 1; IFNs = interferons; IRF3 = IFN regulatory factor 3; ISD = IFN-stimulatory DNA pathway; ISGs = IFN-stimulated genes; PLM = promyelocytic leukemia protein; POP3 = PYRIN domain–only protein 3; pro = precursors of; PYD = PYRIN N-terminal homology domain; STING = stimulator of IFN genes; TRIM11 = tripartite motif containing 11 protein.
Rodents’ ALRs
Different from humans, there exists substantial diversity in the number of ALR genes among the various inbred mouse strains, which may justify their unlike susceptibility to modeled autoimmune diseases such as SLE (Rozzo and others 2001) and Aicardi-Goutières syndrome (Nakaya and others 2017). Conversely, Gray and others (2016) used C57BL/6 mice deprived of all 13 ALRs to show that such genes were unnecessary for the type I IFN responses to DNA virus and lentivirus infections and to transfected dsDNA ligands. Also, ALRs did not partake in the autoimmune pathology of the Trex1 (three prime repair exonuclease 1) knockout mouse model of Aicardi-Goutières syndrome.
The most investigated mouse ALRs are Aim2, Ifi202, Ifi204, and Ifi205 (Connolly and Bowie 2014; Cox and others 2015).
Aim2
Basal levels of Aim2, the most prominent PYHIN protein, are expressed by healthy neurons (Wu and others 2016), microglia, and astrocytes of mouse cerebral cortex and hippocampi (Cox and others 2015). Aim2 mRNA and protein expression significantly increased after an exposure to cytosolic dsDNA or IFN-β or acute Staphylococcus aureus infection (Cox and others 2015; Hanamsagar and others 2014). In addition, independent of its innate immunity roles, Aim2 promoted neuronal axon lengthening and dendrite shortening and variously changed animal behavior (Wu and others 2016). Moreover, once treated with synthetic dsDNA fragments, cultured mouse cortical or hippocampal neurons intensely expressed Aim2 mRNA and Aim2 conditionally secreted IL-1β. The Aim2 inflammasome consistently participated in neuronal pyroptosis (Adamczak and others 2014).
Heneka and others (2014) were the first to posit that DNA fragments released from damaged neural cells could worsen the course of neurodegenerative diseases. However, the studies on Aim2’s roles in AD model mice produced controversial results. AD model mice overexpressed type 1 IFNs and, via the heterodimeric IFN-α/β receptor, owned an activated Jak/Stat signaling pathway, which promoted the expression of IFN-stimulated genes (Mesquita and others 2015; Schoggins and Rice 2011; Taylor and others 2014). Aim2 and the Ifi204 gene–encoded p204 protein (Choubey and Lengyel 1992) were consistently type 1 IFN dependently overexpressed in a ME7 scrapie-infected C57BL/6 chronic neurodegeneration mouse model (Cox and others 2015). Conversely, Aim2’s deletion neither changed the increased IL-1β expression nor improved the declining cognitive functions (i.e., spatial memory), although it upregulated IL-6 and IL-18 expression via unknown mechanisms in 5XFAD model mice. Wu and others (2017) reported that Aβ peptide accumulation was lower but proinflammatory cytokines levels were higher in the cerebral cortex and hippocampi of Aim2-/- 5XFAD model mice than in their Aim2-intact 5XFAD counterparts. They concluded that brain amyloidosis and cytokine levels did not tightly correlate with Aim2 protein expression or with cognitive losses in 5XFAD model mice. Their results also implied that distinct inflammasomes could play unlikely roles in diverse physiologic and pathologic circumstances and/or disease model animals.
Cao and others (2021) reported a novel Aim2 inflammasome regulatory mechanism involving optineurin (OPTN), a 577–amino acid protein encoded by the Optn gene. Among its various functions, OPTN acts as a primary macroautophagy receptor involved in the selective elimination of irreparably damaged mitochondria (mitophagy). Thus, OPTN prevents the release of proapoptotic factors, mitochondrial dsDNA fragments, and ROS into the cytosol—the latter two compounds being involved in the activation of AIM2/Aim2 and NLRP3 inflammasome signaling (Yu and others 2014). Studies using cultured murine astrocytes and BV-2 microglial cells and APP/PS1 AD model mice revealed a diminished OPTN expression, causing a mitophagy deficit that led to the accumulation of defective mitochondria and activated the Aim2 inflammasome. These effects were fully reversed by the ectopic overexpression of OPTN, which hindered the serine/threonine RIPK-1 (receptor-interacting protein kinase 1)–mediated NF-κB inflammatory signaling (Cao and others 2021).
A recent study suggested a link between autophagy and Aim2 function. The charged multivesicular body protein 2A (CHMP2A) is a subunit of the ESCRT-III machinery (endosomal sorting complex required for the transport complex III) partaking in multivesicular body formation, endosomal cargo proteins sorting into multivesicular bodies, cytokinesis, and virus budding (Schmidt and Teis 2012). Following a transient cardiac arrest and circulation restoration, Aim2 inflammasome activation with a heightened release of IL-1β and IL-18 concurred with a drop in CHMP2A-dependent autophagy in rat brain neurons. In contrast, ectopic CHMP2A overexpression mitigated Aim2 activation and neuroinflammation, lessened ischemic harm, and hindered neuronal pyroptosis in the rat cerebral cortex (Shao and others 2021).
Recent evidence also outlined inflammasome involvement in neuropsychiatric disorders. A chronic stress restraint protocol heightened the Aim2, NLRP1, and NLRP3 proteins, as well as caspase 1 activity and IL-1β protein levels, in the frontal cortex of male Wistar rats. The antipsychotic drug paliperidone, a monoamine receptor antagonist, normalized the inflammasomes’ protein levels in these chronically stressed rats. The authors pointed out that drugs targeting inflammasomes’ signaling pathways are urgently needed to treat stress-related neuropsychiatric diseases (MacDowell and others 2021).
Finally, it is worth recalling here that the function of the TGF-β–activated kinase 1 (TAK1), a serine/threonine kinase of the MAPK (MAPKKK) family, is crucial for the activation of NF-κB and MAPK pathways by signals from Toll-like receptors, tumor necrosis factor receptor (TNFR), and IL-1 receptor I (IL-1RI; for details, see Xu and Lei 2021). In the microglia/macrophages of wild-type mice, the deletion of the (Mi/MΦ) TAK1 gene significantly lessened the harm otherwise evoked by an acute ischemic stroke. Usually, stroke entices the activation of the TAK1, Aim2, NLRP3, and NLRC4 inflammasomes and their associated downstream proinflammatory cascades. Erythropoietin administration hindered stroke-elicited activation of the TAK1, Aim2, NLRP3, and NLRC4 inflammasome signaling, resulting in neuroprotection (Heinisch and others 2021). Likewise, a study using living rats and in vitro primary rat cortical astrocytes and microglia showed that the hormonal steroids 17β-estradiol and progesterone selectively hindered Aim2 and NLRC4 inflammasome signaling activation otherwise triggered by an ODG exposure, thereby safeguarding neuron viability (Habib and others 2020).
Human ALRs
Humans own five ALR genes, of which AIM2, IFI16 (interferon inducible protein 16), and POP3 (PYRIN domain–only protein 3) and their respective proteins are reviewed here in regard to neuroinflammation and/or neurodegeneration (Fig. 4).
AIM2
Once formed, the cytosolic dsDNA•AIM2 complexes trigger the AIM2 inflammasome assembly (Wang and others 2020) and its downstream pathway, leading to pyroptosis. Physiologically, during neurodevelopment, this AIM2-mediated mechanism likely kills via pyroptosis any neuron bearing a heavily damaged DNA (Lammert and others 2020).
Interestingly, besides dsDNA fragments, AIM2 senses RNA arboviruses, such as West Nile virus, Chikungunya virus (Ekchariyawat and others 2015), and enterovirus 71 (EV71; Yogarajah and others 2017). EV71 is an envelope-devoid positive single-strand RNA virus causing hand, foot, and mouth disease and, as a complication, lethal encephalomyelitis. In SK-N-SH human neuroblastoma cells, EV71 infection upregulated AIM2 gene and protein expression and the AIM2 client genes CARD16 (caspase recruitment domain family member 16), pro–caspase 1, and pro–IL-1β and caused neuroblastoma cell pyroptosis. The authors hypothesized that in the event of EV71-induced encephalomyelitis, this upregulated AIM2 inflammasome-mediated signaling might act protectively by decreasing the proportion of virus-infected neurons and hence the intensity of viral replication in the inflamed brain areas. Their view should be strengthened by further investigations.
The AIM2 inflammasome’s regulatory mechanisms act at two levels. The tripartite motif–containing 11 (TRIM11) protein gathers with the AIM2 protein; the resulting AIM2•TRIM11 complex assembles with the p62 protein, a receptor of autophagic cargoes, which causes the selective autophagic degradation of AIM2, thus preventing the latter’s accumulation (Fig. 4; Liu and others 2016).
Moreover, it is well known that an acute exposure to ionizing radiations elicits the death of proliferating cells in the gut and bone marrow. ALR-knockout mice resisted such irradiation injuries, revealing that the AIM2 inflammasome assembles inside DNA-damaged nuclei and binds DNA harm sites, thereby inhibiting the DNA unpacking, which is crucially needed for DNA repair to occur. Consequently, ALRs advance genome instability, micronucleus formation, and cell death in ways disconnected from their innate immunity roles (Jiang and others 2021). Whether this mechanism operates within irradiated human brain cell nuclei remains to be ascertained.
Lark and LaRocca (2021) used postmortem human temporal cortex tissues to characterize the cell and tissue-specific effects of aging and AD on genes involved in exosome biogenesis. The results showed that patients with AD had reduced expression of the CHMP2A gene, which is involved in Tau protein trafficking, but increased expression of the SDCBP gene (syndecan-binding protein) encoding for syntenin 1, the core exosomes’ protein. In keeping with the results of Shao and others (2021), who studied a rat model of cerebral ischemia/reperfusion injury, Lark and LaRocca (2021) findings prove that an altered expression of genes involved in multivesicular bodies and exosome biogenesis is related to activated AIM2 inflammasome signaling in human patients with AD.
Moreover, activated AIM2 and NLRP3 inflammasomes sustain the neuroinflammation in autistic spectrum disorders, in which increases in proinflammatory IL-1β and IL-18 concur with altered levels of anti-inflammatory IL-37 and IL-33 cytokines (Saresella, Piancone, and others 2016).
IFI16
Like AIM2, the human ALR IFI16 protein sensor resides in the nucleus and cytosol (Wu and others 2017). The human IFI16-α isoform is mainly nuclear. The IFI16-β isoform, however, has two HIN domains but lacks the PYRIN domain, and like the murine p202, it colocalizes and interacts with AIM2 in the cytosol. By competing for the dsDNA binding site, IFI16-β hinders AIM2’s function (Wang and others 2018). Once bound to dsDNA, the IFI16 protein triggers the expression of IFN-α via the cGaS/STING/ISD pathway (Sun and others 2013). Chronic activation of the cGaS/STING-dependent ISD pathway was implicated as the cause of severe IFN-associated autoimmune human diseases, such as Aicardi-Goutières syndrome (Gall and others 2012) and SLE (Rozzo and others 2001). Moreover, the exposure to type 1 (IFN-α or IFN-β) or type 2 (IFN-γ) IFNs heightened the expression levels of IFI16, AIM2, ASC, and pro–caspase 1 proteins and increased the formation of cytoplasmic IFI16•AIM2 protein complexes. However, the ectopic IFI16-β isoform overexpression blocked pro–caspase 1 activation by the AIM2•ASC inflammasome in the cytosol of HEK-293T cells, revealing that the IFI16-β isoform could mediate the anti-inflammatory activity of type 1 IFNs (Veeranki and others 2011).
POP3
Finally, type 1 IFN-inducible POP3, one of the aforementioned PYD-only proteins, binds ASC, hindering its interaction with AIM2 or IFI16 and thereby inhibiting the buildup of AIM2 and IFI16 inflammasomes and their downstream effects in monocytes and macrophages. Conversely, POP3’s silencing advanced ALR inflammasomes’ assembly and proinflammatory cytokines’ maturation triggered by DNA fragments or DNA viruses (Khare and others 2014).
Clearly, further studies are needed to fully unravel the intricate roles of AIM2 and IFI16 inflammasomes in human brain diseases.
NLRC4 Inflammasome
The evolutionarily conserved NLRC4 inflammasome (IPAF or ICE protease-activating factor; Alfonso-Loeches and others 2014) is an innate immune system component initially thought to sense pathogenic bacteria (Franchi and others 2009). The NLRC4 inflammasome lacks the PYRIN domain, and in its stead it binds the NAIP sensor (neuronal apoptosis inhibitory protein). Unlike rodents, humans have a single NAIP gene. NAIP sensors bind PAMPs (e.g., flagellin, Cprl needle protein) from intracellular Gram-negative flagellate bacteria, such as Chromobacterium violaceum, Legionella pneumophila, and Salmonella genus, to activate the NAIP1•NLRC4 inflammasome (Fig. 5; Bauer and Rauch 2020). Infestation by the metastrongyloid nematode Angiostrongylus cantonensis also activates NLRC4 and NLRP1 inflammasomes and causes meningoencephalitis and pyroptosis in BALB/c mice (Lam and others 2020). Phosphorylation at Ser533 by protein kinase Cδ (PKCδ; Qu and others 2012) and ubiquitination (Kumar and others 2010) are crucial for NLRC4 inflammasome activation. The latter causes a caspase 1–mediated reduction in bacterial replication inside murine microglia cells (Jamilloux and others 2013). Notably, mouse astrocytes too express the NLRC4/IPAF inflammasome (Alfonso-Loeches and others 2014).
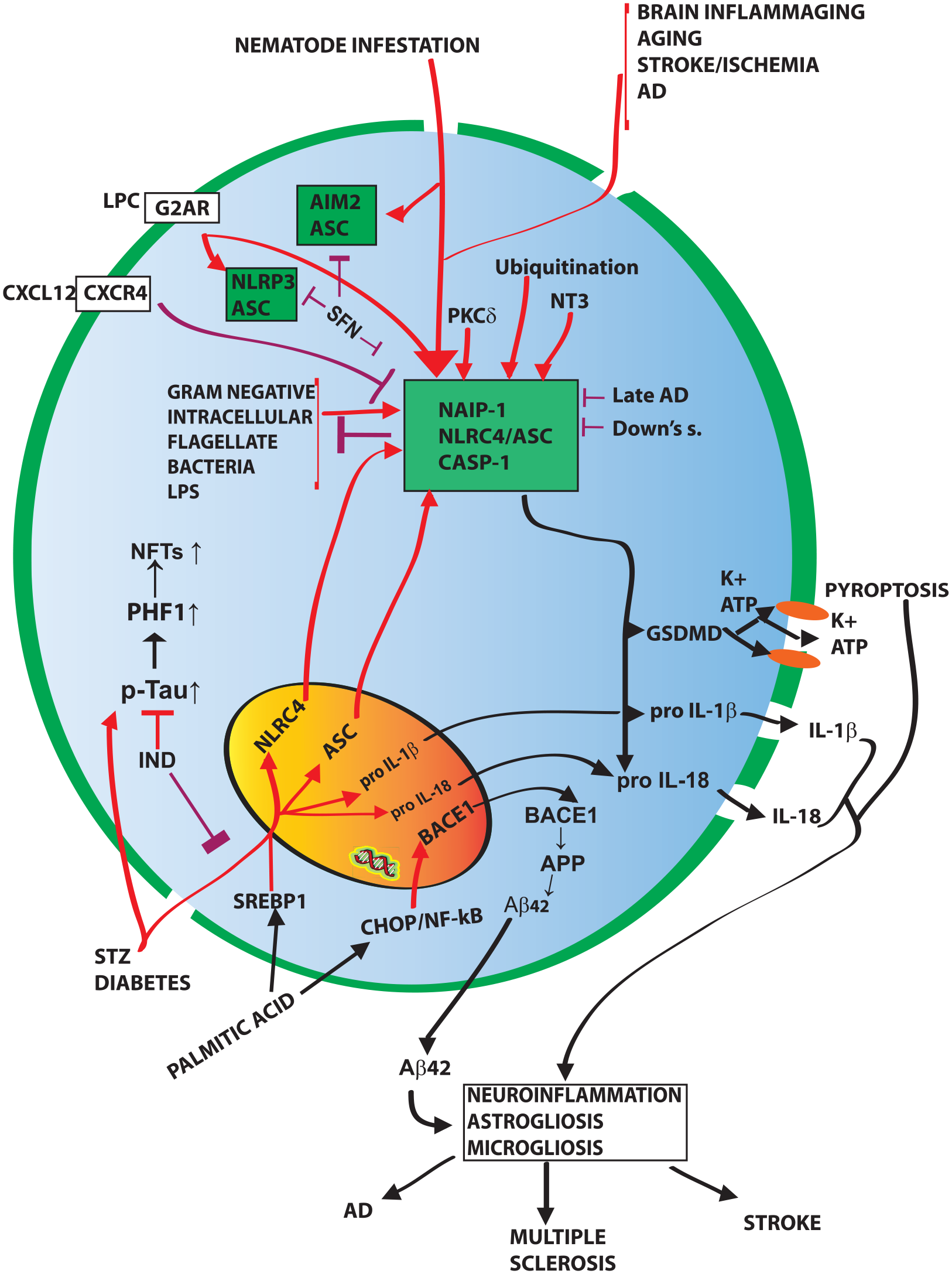
NLRC4 inflammasome’s activation: mechanisms and effects. The art depicts a generic neural cell (e.g., astrocyte or microglia). Plasma membrane, green. Nuclear envelope, black. The inflammasome-activating stressors are shown at the picture’s top and bottom left. Assembled NLRC4, AIM2, and NLRP3 inflammasomes are within black frames with a green background. Aβ42 = amyloid-β42; AD = Alzheimer disease; APP = amyloid precursor protein; ASC = apoptosis-associated speck-like protein containing a CARD; BACE1 = β-site amyloid precursor protein-cleaving enzyme 1 or β secretase; CASP = caspase; CHOP = C/EBP homologous protein; CXCL12 = stromal-derived factor 1α (SDF-1α); CXCR4 = chemokine receptor 4; G2AR = G protein–coupled receptor 132; GSDMD = gasdermin D; IND = indomethacin; LPC = lysophosphatidyl-choline; LPS = bacterial lipopolysaccharide; NAIP-1 = NLR family apoptosis inhibitory protein 1; NFTs = neurofilament tangles of p-Tau; PHF-1 = paired helical filament 1 (Tau) protein; PKCδ; protein kinase Cδ; pro = precursor of; p-Tau = phosphorylated Tau protein; s. = syndrome; SFN = sulforaphane; SREBP1 = sterol response element-binding protein 1; STZ = streptozotocin.
Mejas and others (2018) showed that the protein levels of the NLRC4 inflammasome, ASC, caspase 1, and IL-18 were higher in the cerebral cortical lysates from aged mice than in those from young mice, indicating that NLRC4 partakes in brain inflammaging and neurons’ pyroptosis. Moreover, NLRC4 functioned as a DAMP sensor involved with AIM2 in the neuroinflammation driven by an acute ischemic brain injury in a mouse model of focal ischemia induced by middle cerebral artery occlusion, while the NLRP3 inflammasome was not involved (Denes and others 2015). Although NLRC4 can directly interact with caspase 1, the NLRC4•ASC complex advanced the inflammasome’s assembly (Latz and others 2013) producing IL-1β and IL-18 and inducing pyroptosis (Mariathasan and others 2004; Qu and others 2012). In mouse BV2 microglia cells, activation of the CXCL12/SDF-1α•CXCR4 signaling complex (stromal-derived factor 1α•chemokine receptor 4) hindered the LPS-induced activation of NLRC4•ASC and NLRP3•ASC and its consequences (Roosen and others 2021).
Using as AD-like models with intracerebroventricularly STZ-injected male Wistar rats, Saadi and others (2020) assessed the expression of genes implicated in inflammasomal complexes: NLRP1, NLRP3, NLRC4, AIM2, ASC or PYCARD, IL-1β, IL-18, and CASP1. Significant NLRC4, ASC, and IL-1β gene expression increases occurred in the STZ-treated rats versus untreated controls. Additionally, the numbers of cells expressing caspase 1, IL-1β, and p-Tau proteins remarkably increased in the STZ-treated rats’ hippocampi (Fig. 5). The authors concluded that NLRC4 may be another target for AD therapy. Consistent with this view, Freeman and others (2017) showed that lysophosphatidyl-choline (LPC), an agent promoting neurodegeneration and demyelination, activated the NLRC4 and NLRP3 inflammasomes through the G2A receptor (GPR132 [G protein–coupled receptor 132]) in murine astrocytes and microglia in vitro and in vivo. Notably, NLRC4 and NLRP3 might therefore induce inflammatory astrogliosis and microgliosis in diseases such as AD, multiple sclerosis, and stroke (Fig. 5).
Interestingly, there are some discordant reports on the roles that the NAIP-1 sensor protein plays in human neurodegeneration. Lesnè and others (2005) reported that neurotrophin 3 (NT-3) upregulated NAIP-1 sensor protein expression, which, through the activity of Akt and PI3K and by restraining activation of caspases 3, 8, and 9, hindered the Aβ-driven apoptosis of primary mouse embryo cortical neurons.
Consistent with such findings, Christie and others (2007) showed that in the postmortem samples of hippocampal and entorhinal cortices of fully symptomatic AD cases, NAIP-1 protein levels were lower than in the brains of cases of minor cognitive impairment AD and controls. Concurrently, the levels of the paired helical filament 1 (PHF-1) protein, a marker of toxic hyperphosphorylated Tau protein neurofilament tangles, were higher in the same AD cases (Fig. 5). The authors suggested that NAIP-1’s fall increased the neurons’ risk to develop neurofilament tangles first and later to die by apoptosis, thereby advancing patients’ cognitive decline and Braak stages.
Notably, Down syndrome (DS) entails an early-onset AD-like neurodegeneration with an intensified neuronal apoptosis causing typical mental deficits. Also consistent with the aforementioned reports, Seidl and others (1999) showed that when compared with healthy controls, NAIP-1–like Western immunoreactivity was lower in the DS parietal and occipital cerebral lobe cortex and in the AD frontal and occipital lobes cortex. Therefore, decreases in neuroprotective NAIPs advance (and/or result from) DS and AD chronic neurodegeneration.
Importantly, Liu and Chan (2014) showed that pathogenic bacteria are not the unique activators of the NLRC4 inflammasome. Palmitate—a saturated fatty acid that is the main refined palm oil component used in several dairy products—awoke the NLRC4 inflammasome signaling and overproduction of mature IL-1β in primary rat astrocytes. The authors also showed that the postmortem brain tissues of a subgroup of patients with sporadic AD overexpressed NLRC4 and ASC. Additionally, NLRC4 silencing suppressed the IL-1β over release from astrocytes, which in turn reduced the primary neurons’ Aβ42 production. A later study from Marwarha and others (2018) revealed that the endoplasmic reticulum stress-associated transcription factor CHOP (C/EBP homologous protein) played a critical role along a CHOP/NF-κB signaling pathway in the palmitate-elicited upregulation of BACE1/β-secretase gene expression, which increased Aβ42 synthesis from amyloid precursor protein (APP) in human neuroblastoma SH-SYSYAPPSwe cells. A subsequent study (Marwarha and others 2019) revealed that palmitate also activated the SREBP1 transcription factor (sterol response element-binding protein 1), which increased BACE1/β-secretase gene expression and raised the Aβ42 production in mouse neuro-2a (N2a) neuroblastoma cells and in the hippocampus of living mice. As well, a palmitate-rich diet increased α-synuclein, tyrosine hydroxylase, and dopamine metabolites in the brains of m-Thy1 transgenic Parkinson model mice (Schommer and others 2018). Altogether these findings proved that, besides harming the cardiovascular system, a surplus of dietary palmitate activates the brain astrocytes’ NLRC4 inflammasome, thus advancing the AD- and Parkinson-related neuroinflammation. Hence, drastically reducing palmitate in dairy products and pharmacologically targeting the NLRC4 inflammasome could benefit patients experiencing neurodegenerative diseases.
The aforementioned data stress the importance of extending the knowledge about NLRC4 inflammasome roles in neuroinflammation and neurodegeneration.
“Other Than NLRP3” Inflammasomes’ Inhibitors
As summarized in Table 1, various agents inhibit these inflammasomes’ activation.
Inhibitors of Inflammasomes’ Activation and Their Effects in Preclinical Studies.
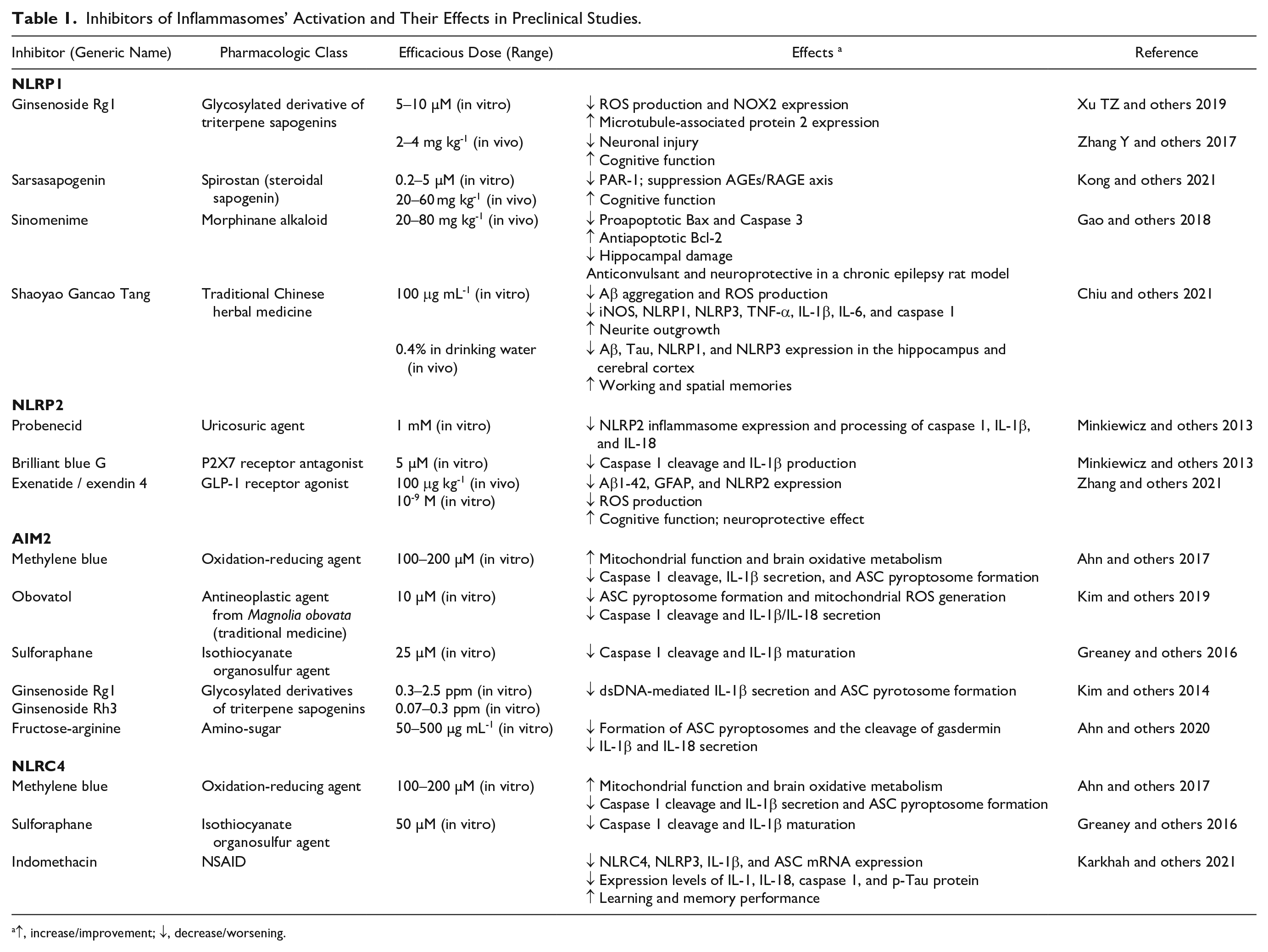
↑, increase/improvement; ↓, decrease/worsening.
NLRP1
To start with, ginseng saponin Rg1 of Panax ginseng hindered the activation of NLRP1 inflammasome signaling in cultured hippocampal neurons (Xu TZ and others 2019; Yi 2021) and in chronically dexamethasone-treated male mice (Zhang Y and others 2017). The natural steroid sarsasapogenin suppressed NLRP1 inflammasome signaling in the hippocampus and cerebral cortex by downregulating thrombin’s protease-activated receptor 1 (PAR-1), thus improving memory impairment in STZ-treated AD model rats (Kong and others 2021). Also, sinomenine (Cocculine), a Chinese and Japanese climbing plant Sinomenium acutum alkaloid used in traditional herbal medicine, was neuroprotective as it inhibited NLRP1 inflammasome; reduced IL-1β, IL-18, TNF-α, and IL-6 levels; and averted seizures in a chronic epilepsy rat model (Gao and others 2018). Moreover, in AD model cells and animals, the Chinese traditional medicine Shaoyao Gancao Tang, made of Glycyrrhiza uralensis and Paeonia lactiflora (at 1:1 ratio), suppressed NLRP1 and NLRP3 inflammasome signaling, mitigated neuroinflammation, promoted neurons’ survival and neurites’ outgrowth, and improved memory (Chiu and others 2021; Fig. 2).
NLRP2
The NLRP2 inflammasome teamed up with the P2X7 purinergic receptor and ATP-releasing Panx-1 channel in ATP-treated human astrocytes. In fact, the Panx-1 blocker probenecid or the P2X7 inhibitor Brilliant Blue G hindered NLRP2 inflammasome assembly and activation (Fig. 3; Minkiewicz and others 2013). Exenatide, a synthetic agonist of GLP-1R, mitigated piriform cortex neuroinflammation and improved cognitive and memory dysfunctions in 5xFAD model mice. Also, exendin-4 (the natural analog of exenatide) decreased the Aβ1-42–elicited oxidative stress and proinflammatory response in cultured mice astrocytes. Exenatide’s and exendin-4’s effects were coupled with NLRP2 inflammasome downregulation in the murine astrocytes (Zhang and others 2021). The last authors did not look for any concurrent changes of other inflammasomes.
AIM2/Aim2 and NLRC4
Methylene blue, an anti-inflammatory, antioxidant, and mitochondria- and brain-protective agent, inhibited NF-κB signaling and hence reduced canonical and noncanonical activation of the AIM2/Aim2, NLRP3, and NLRC4 inflammasomes in human and mouse macrophages (Ahn and others 2017). Also, obovatol, a biphenolic compound from Magnolia obovata (Japanese cucumber tree) used in traditional medicine to treat inflammatory diseases, inhibited AIM2 and NLRP3 inflammasomes by hindering their assembly with ASC (Fig. 4; Kim and others 2019). Sulforaphane (SFN), an isothiocyanate organosulfur agent found in cruciferous vegetables (broccoli sprouts) and sold as a dietary supplement, exerts anti-inflammatory, antioxidant, and apoptosis-preventive effects. According to Lee and others (2016), SFN blocked NLRP3 and NLRC4 activation but not that of the AIM2 inflammasome. Conversely, Greaney and others (2016) reported that SFN inhibited the AIM2 inflammasome, besides the NLRP3 and NLRC4 ones, by blocking the autoproteolytic activation of pro–caspase 1 (Fig. 5; Vomhof-Decrey and Picklo 2012). The reasons of these discrepant results about SFN effects on AIM2 activation remain unclear. Also, MCC950, a selective and powerful inhibitor of canonical and noncanonical NLRP3 activation, could not block the AIM2, NLRC4, and NLRP1 inflammasomes. Therefore, MCC950 is used to investigate NLRP3’s specific roles in human inflammatory and autoimmune diseases (Coll and others 2015). Even Rg1 and Rh3 ginsenosides, both steroidal saponins found in the Korean red ginseng extract, inhibited activation of the AIM2/Aim2 and NLRP3 inflammasomes in human and mouse macrophages in vivo and in vitro (Kim and others 2014). Additionally, by obstructing STAT3’s phosphorylation and subsequent binding to the Aim2 gene promoter, which would have activated Aim2’s transcription, Rg1 hindered LPS-evoked Aim2 inflammasome activation and microglia pyroptosis (Yao and others 2021). The amino-sugar fructose-arginine, a Korean red ginseng nonsaponin constituent, mitigated the Aim2 inflammasome’s activation but exerted no effects on NLRP2 or NLRC4 inflammasomes’ signaling in mouse macrophages transfected with exogenous dsDNA or infected with Listeria monocytogenes (Ahn and others 2020). Finally, using the STZ-induced AD-like rat model, Karkhah and others (2021) showed that indomethacin, a nonsteroidal anti-inflammatory drug, suppressed the expression of NLRC4, NLRP3, IL-1β, and ASC genes. Indomethacin reduced the hippocampal levels of caspase 1, IL-1β, and phosphorylated Tau protein while improving learning and memory in STZ-injected AD model rats. On the basis of such results, the authors suggested that NLRC4 and NLRP3 partake in AD’s neuroinflammation.
SARS-CoV-2 Brain Infection and Inflammasomes’ Activation
Human neuroinflammatory and neurodegenerative diseases are still an incomplete chapter, missing much information about old and novel pathologic patterns. An example of the latter is the recently identified infection of the human brain by the SARS-CoV-2. Indeed, SARS-CoV-2 was shown to infect human neural progenitor cells and cortical neurons grown in brain organoids via angiotensin-converting enzyme 2 (ACE2) and cluster of differentiation 147 (CD147) receptors (Chen and others 2021; Ribeiro and others 2021; Zhang BZ and others 2020). A potentially relevant neuropathologic mechanism of the SARS-CoV-2 infection may be the hyperactivation of the P2X7 receptors induced by excess extracellular levels of free ATP, which could activate NLRP2 and NLRP3 inflammasomes’ signaling in neural cells (Minkiewicz and others 2013; Ribeiro and others 2021). In the postmortem case series, SARS-CoV-2 virus nucleocapsid colocalized within neurons, astrocytes, oligodendrocytes, microglia, and blood-brain barrier endothelial cells, and the NLRP3 inflammasome was activated in CD68+ microglia (Cama and others 2021; Matschke and others 2020). Moreover, various SARS-CoV-2 virus proteins, such as viroporin E, ORF3a and ORF8a (open reading frames), and TNF receptor–associated factor 3 (TRAF3), did partake in the activation of NLRP3 but not of the AIM2 inflammasome, inducing macrophage pyroptosis (for details, see de Rivero Vaccari and others 2020; Shi and others 2019; Siu and others 2019). Importantly insoluble, ORF8b protein aggregates bound NLRP3/ASC specs; next, such protein complexes might cross-seed with Aβ-os and promote the spread of AD’s neuropathology (Venegas and others 2017). In addition, the Ca2+ surplus released through channels formed by the viroporin E protein in the endoplasmic reticulum/Golgi apparatus membranes could activate the NLRP3 inflammasome increasing IL-1β production in African green monkey kidney-derived Vero E6 cells (Nieto-Torres and others 2014). Yet, the IL-18 made via inflammasome activation acted protectively by hindering the replication of the SARS-CoV-2–like mouse hepatitis virus strain A-59 (MHV-A59; Zalinger and others 2017). From this, it is clear that our knowledge about inflammasomes’ roles in brain tissue viral infections due to SARS-CoV-2 and other viruses (e.g., influenza) is rather fragmentary, sometimes contradictory, and hitherto nearly exclusively concerns the NLRP3 inflammasome. Hence, further investigations on the roles of the “other than NLRP3” inflammasomes are in order in viral infections of the brain.
Conclusions and Future Perspectives
Human neuroinflammatory/neurodegenerative diseases still lack therapeutic regimens effectively preventing, healing, or even slowing their progression toward dementia and patients’ demise. Hence, they take a severe toll on the patient, family, society, economy, and health care (Wong 2020). Add to this the ongoing fearful epidemic-like increase in such diseases prevalence in aging people. Several factors contributed to this unsatisfactory situation—primarily, the often hardly recognizable etiologic factors involved, which differently associate in diverse patients and may be masked or confounded by collateral factors. This is compounded by the long-lasting course of neuroinflammation’s and neurodegeneration’s asymptomatic stage, during which the neuropathology silently consumes the brain’s functional reserve. Even acute-onset brain diseases such as stroke can underhandedly evolve into chronic neurodegenerative diseases if the patients survive long enough. Another factor was and is the persistently failed translation of the positive pharmacologic study results gained from animal models of neurodegenerative diseases to correspondingly ill human patients. The underlying reasons are multiple. Humans are not syngeneic, and their genomic variability and a host of acquired factors (epigenetic, environmental, dietetic, behavioral, infectious, toxic) likely require a precision medicine approach (i.e., patients’ stratification) to achieve significant therapeutic upshots. Moreover, human brains have evolved peculiar morphofunctional features (e.g., the neocortex) that other mammals partly or not at all share. This structural and functional diversity casts doubts on animal models as close-enough mirrors of human neurodegenerative diseases. However, undeniably most of the presently available data on such diseases have come and still come from experiments with animal models that, with human postmortem studies, have offered some enlightening glimmers on neural pathobiology. On such grounds, the high number of standing hypotheses on the pathogenesis of each human neurodegenerative disease must not surprise. We ourselves contributed one and still stand by it, based on the results of preclinical in vitro studies using untransformed human cerebral cortical astrocytes and neurons. We posit that the calcium-sensing receptor (CaSR) is a PRR and that the multipronged signaling from Aβ•CaSR complexes drives the AD proper intracerebral spreading of surpluses of Aβs, p-Taues, nitric oxide, VEGF-A, and neuroinflammation (Armato and others 2013; Chiarini and others 2020).
Because of the relevance of human brain pathobiology, in this work, we strove to report the human-related data available and to outline any differences between humans and rodents and among rodent strains. Current trends have been emphasizing the roles of neuroinflammatory processes driven by the activation/signaling of innate immunity-linked inflammasomes in chronic brain neurodegenerative diseases. As often happened in the past, the emphasis has fallen on neurons and microglia. As neuroinflammation is considered one leg of the pathogenetic tripod of AD (the other two being Aβs and p-Taues), it was an obvious suggestion to preclinically and then clinically test the effects of drugs targeting inflammasomes’ components on neuroinflammation and the AD course. The same suggestion has involved other acute and chronic brain diseases. Hence, a great deal of the attention has been and is still focused on the NLRP3 inflammasome as the crucial therapeutic target. Phase 1 clinical trials of NLRP3 inflammasome inhibitors have been completed, such as Inzomelid (from Inflazome), NT-0167 (from NodThera), IFM-2427 (from Novartis since 2019), and Dapansutrile (from Olatec Therapeutics). Their phase 2 clinical trials are ongoing, but hitherto no corresponding results have been published (Lünemann and others 2021). Conversely, recent evidence gained from a fresh human postmortem study did not support NLRP3 inflammasome involvement while stressing the astrocytes’ activation in the brains of patients with AD (Tang and Harte 2021). These last findings obviously require further confirmation. Notably, animal and human brains own a variety of cell types expressing quite a number of inflammasomes, whose regulatory mechanisms and reciprocal interactions remain largely undefined. In addition, the regulatory mechanisms of brain inflammasomes differ from those proper of other organs for the fact that the brain inflammasomes preexist as already preassembled complexes that do not require the priming step (signal 1), which ensures their faster response to PAMPs, DAMPs, or HAMPs (Abulafia and others 2009; de Rivero Vaccari and others 2008).
A topic thus far underinvestigated yet important concerns the reciprocal interactions occurring among diverse groups of inflammasomes—that is, whether and how the several inflammasomes influence one another’s expression and/or function. Notably, Heinisch and others (2021) showed that pharmacologically inhibiting the NLRP3 inflammasome increased the levels of Aim2 and NLRC4 proteins in murine BV-2 microglia cells. The authors suggested that reciprocal compensatory activities may occur between these inflammasomes. Conversely, Schölwer and others (2020) showed that a compensatory heightening of the Aim2 inflammasome’s expression with no change in the NLRC4 one occurred after oxygen-glucose deprivation in NLRP3-siRNA–depleted BV-2 murine microglial cells. The mechanisms underlying these discrepant NLRC4’s behavioral responses remained unclear. Bostanci and others (2009) showed that following infection by Porphyromonas gingivalis, a major oral bacterium causing periodontitis, NLRP3 inflammasome upregulation was counterbalanced by NLRP2’s downregulation in gum tissues from patients with gingivitis and cultured human Mono-Mac-6 monocytes. Besides, NLRP3 knockout did not suppress IL-1β overproduction in mice infected with Streptococcus pneumoniae, indicating its replacement by other (at the time undetermined) NLR-based or AIM2 inflammasomes but concurrently intensified lung edema, decreasing the infected mice’s chances of survival (Witzenrath and others 2011). The intervention of other inflammasomes has been suggested in NLRP3-suppressed but still IL-1β−producing mice with S.aureus pneumonia (Kebaier and others 2012).
As yet, it is unknown whether and how these interactions among inflammasomes take place in the animal and human brains in the course of any neuroinflammation and/or neurodegeneration. Indeed, in this review, we focused on “other than NLRP3” brain inflammasomes: NLRP1, NLRP2, NLRC4, and AIM2. We are aware that we left out still other inflammasomes, whose physiologic and pathologic roles have hitherto been little investigated. Therefore, notwithstanding the remarkable advances concerning the more intensely studied inflammasomes herein reviewed, including the NLRP3 one, significant knowledge gaps remain, particularly about the differences in expression, function, and complex regulatory mechanisms between animal and their human counterparts and about the mechanisms by which the primary neuropathologic drivers (e.g., Aβ-os) influence them. Such gaps need to be filled, and a host of inherent questions, including the pharmacologic approaches, must be solved about the multiple inflammasomes’ reciprocal interactions in the human neural cells and the human brain as a whole. An ultimate question also must be addressed—that is, whether it would be wiser to therapeutically target a single inflammasome or in its stead the primary drivers secondarily activating the brain cell inflammasomes or both together. The last approach is probably preferable. Therefore, we hope that a more comprehensive view of human brain inflammasomes’ workings and their upstream drivers will help understand the distinct pathogenesis of each human neuroinflammatory/neurodegenerative disease and hence pave the way to its effective therapeutic solution.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The FUR 2020 allotment from the MUR (Ministry of University and Research) given to A.C. and I.D.P. supported this research work. No funds were provided by private or commercial sources.
