Abstract
Background and objectives
Multiple sclerosis (MS) is a chronic autoimmune disease causing neuroinflammation and neurodegeneration. The apolipoprotein E4 (APOE4) allele, a major genetic risk factor for late-onset Alzheimer's Disease, accelerates cognitive decline and neuroinflammatory processes, including blood–brain–barrier disruption. This study explores the impact of APOE4 on later-life health in MS patients using biomarkers from the UK Biobank.
Methods
MS patients were grouped by APOE4 carrier status: MS-E4 and MS-nonE4. Age- and sex-matched non-MS controls (control-E4, control-nonE4) were included for comparison. Analyses assessed retinal nerve fiber layer thickness (RNFL) from optical coherence tomography (OCT), blood biomarkers, cognitive performance, and brain magnetic resonance imaging (MRI) metrics.
Results
MS-E4 patients exhibited worse outcomes, including thinner RNFL, higher blood glial fibrillary acidic protein (GFAP) and neurofilament light chain levels, slower cognitive reaction times, and more white matter hyperintensities. GFAP had significant interactions between MS and APOE4 status, correlating with neurodegenerative markers.
Discussion
APOE4 exacerbates neurodegeneration and neuroinflammation in MS, evident in retinal OCT, cognitive testing, and MRI findings. Similar effects were observed in healthy APOE4 carriers. These results highlight the utility of multi-domain biomarkers for MS diagnosis and long-term management, emphasizing less invasive tools for monitoring disease progression.
Introduction
Multiple sclerosis (MS) is a common neuroimmune disease affecting nearly 1 million people in the US, predominantly among young adults in their 20s and 30s.1,2 Clinically, MS is a heterogeneous condition characterized by diverse subtypes, including relapsing-remitting, primary progressive, secondary progressive, and progressive-relapsing variants, each exhibiting distinct clinical trajectories. 3 While disease-modifying therapies have been developed to reduce morbidity and mortality, particularly in relapsing-remitting MS, a definitive cure remains elusive.4,5
The etiology of MS is largely unelucidated, involving a combination of genetic predispositions and environmental factors such as Epstein-Barr virus infection, smoking, and obesity. 6 Understanding the genetic architecture of MS is essential for identifying prognostic markers and tailoring therapeutic approaches. Although no single gene explains its pathogenesis or inheritance, several genetic loci have been implicated in the genetic basis of MS. 7 Among these, the apolipoprotein E (APOE) gene, specifically the E4 variant (APOE4), is a prominent candidate due to its established role in neurodegenerative diseases and its association with cognitive decline in MS patients. 8 However, the exact mechanism by which APOE4 influences MS severity is unclear and requires further investigation.
In addition to standard clinical and radiographic criteria for diagnosing MS, novel biomarkers were identified to better monitor disease activity and severity. These include advanced minimally invasive, inexpensive, and readily available tools such as retinal optical coherence tomography (OCT) and blood biomarkers like glial fibrillary acidic protein (GFAP), neurofilament light chain (NfL), and Chitinase 3-like-1 (CHI3L1).9–11 This study investigates the impact of the APOE4 allele on long-term health outcomes in MS patients, compared to healthy controls, focusing on retinal tomography, neuroimaging, blood biomarkers, and cognitive tests, using data from the UK Biobank (UKB).
Methods
Study cohort description
The UKB is a multisite longitudinal cohort study from the United Kingdom recruited through the National Health Service, with the primary aim to facilitate comprehensive exploration into the genetic and environmental factors influencing prevalent diseases in later stages of life. Ethical approval for the UKB was granted by the research ethics committee (REC reference for UKB 11/NW/0382), with participants offering written informed consent. This analysis included participants with available demographic data, APOE genotype, blood proteomics, and imaging records. Patients with prior histories of cerebral-vascular accidents (stroke) or brain cancer were excluded from the cohort. MS was defined according to the lCD-10 code G35. For the secondary analysis, age- and sex-matched subjects without known MS diagnosis were selected as control subjects to further investigate effects on common biomarkers based on APOE4 carrier status. Demographic information such as age, sex, date of MS diagnosis, and date of initial encounter were obtained. Educational status was also examined and stratified according to the International Standard Classification of Education (ISCED) category 12 (Table 1).
General representation of how educational attainment is categorized in the UK biobank dataset.

Biomarker data
Retinal OCT data were downloaded with a focus on the retinal nerve fiber layer thickness (RNFL) thickness in micrometers. The RNFL measures were combined from each eye and accounted for as two measures for each patient. Reaction time in seconds was assessed by 12 rounds of the game snap, which has been found to be a reliable test of processing speed as compared with other available tests. 13 Magnetic resonance imaging (MRI) scans in the UKB were performed 5–10 years after the initial encounter and were analyzed in the study subjects with available imaging outcomes. Specific blood markers were obtained through Olink for the initial encounter: GFAP, CHI3L1, and NfL. The normalized relative values provided by the UKB were then transformed into quantitative protein concentrations using a natural log transformation.
Statistical analysis
The primary analysis consists of group-level comparison of MS patients with or without E4 allele (MS-E4 vs. MS-nonE4). In the secondary analysis, two additional groups of subjects without MS (age- and sex-matched controls), split based on their APOE4 status, were included. The groups were compared with regard to their reaction time at both the initial visit (instance 0) and at time of MRI for those with available data (instance 2), RNFL thickness, plasma markers, and MRI findings. Significance level was established at 95% confidence interval (p < 0.05). Group-level comparisons were performed in Jamovi Version 2.5 (The Jamovi project 2024 Sydney, Australia) using a linear regression model. Analysis of blood markers, RNFL thickness and MRI was adjusted for covariates including sex and number of years with MS. Body mass index (categorized into WHO classes: normal weight, overweight, and obese [Class I–III]) was also included as a covariate in the linear models to account for its influence on GFAP, CHI3L1, and NfL levels. Education was additionally adjusted when comparing reaction times. For retinal OCT data, RNFL thickness was initially analyzed using both left and right eyes as separate observations, given the frequent unilateral involvement in MS. To account for the repeated-measures nature of this data, a sensitivity analysis was performed by averaging RNFL thickness across both eyes for each subject. Results remained statistically significant and consistent across both approaches, supporting the robustness of our findings. Chronological age was used in secondary analysis instead of number of years with MS. We also included the interaction term MS × APOE4 carrier status on selected biomarkers. Partial correlation adjusted for age and sex was used to examine relationships between biomarkers and reported as Spearman's Rho values. Multiple comparisons for primary endpoints (blood markers, RNFL, and reaction time instance 0) were addressed using Benjamini–Hochberg false discovery rate (FDR) correction. Exploratory analyses (MRI and reaction time instance 2) were reported with unadjusted p-values due to limited sample sizes. Plots were generated using GraphPad Prism version 10.0.0.
Data availability
Anonymized data not published within this article will be made available by request from any qualified investigator. Data is available from the UKB after submitting an application (ukbiobank.ac.uk/register-apply/).
Results
Participant characteristics
Out of a total of 502,367 patients in the UKB, 21,311 were excluded for having a prior history of stroke or brain cancer. Of those remaining, 1988 patients were diagnosed with MS prior to the initial encounter, of which 188 had available APOE genotyping and subsequently constituted the subject pool for the study. 140 patients were identified as not having the APOE4 allele while 48 were identified as having the allele (Figure 1). For the secondary analysis, 48 APOE4 and 140 non-APOE4 control subjects were matched to the primary study population as described above.

Flowchart for screening eligible participants for the study.
Primary analysis—apolipoprotein E4 effect in multiple sclerosis patients
Blood markers
To investigate the impact of APOE4 on MS-related biomarkers, we compared MS-E4 carriers to MS-nonE4 subjects across three key blood biomarkers (Table 2 and Figures 2A–C). Compared to MS-nonE4 patients, MS-E4 patients had higher GFAP (p < 0.001), higher NfL (p < 0.001), and lower CHI3L1 (p < 0.001). Interaction between MS status and APOE4 carrier status was also analyzed with regard to blood markers with both MS patients and healthy controls. Only GFAP showed significant positive interaction (regression coefficient β = 0.38, p = 0.02). A significant correlation was found between GFAP and both MS status and APOE4 status (GFAP-MS status correlation: rho = 0.17, p = 0.01; GFAP-APOE4 status correlation: rho = 0.20, p = 0.003). These results highlight the influence of the APOE4 allele on the extent of neurodegeneration in MS.
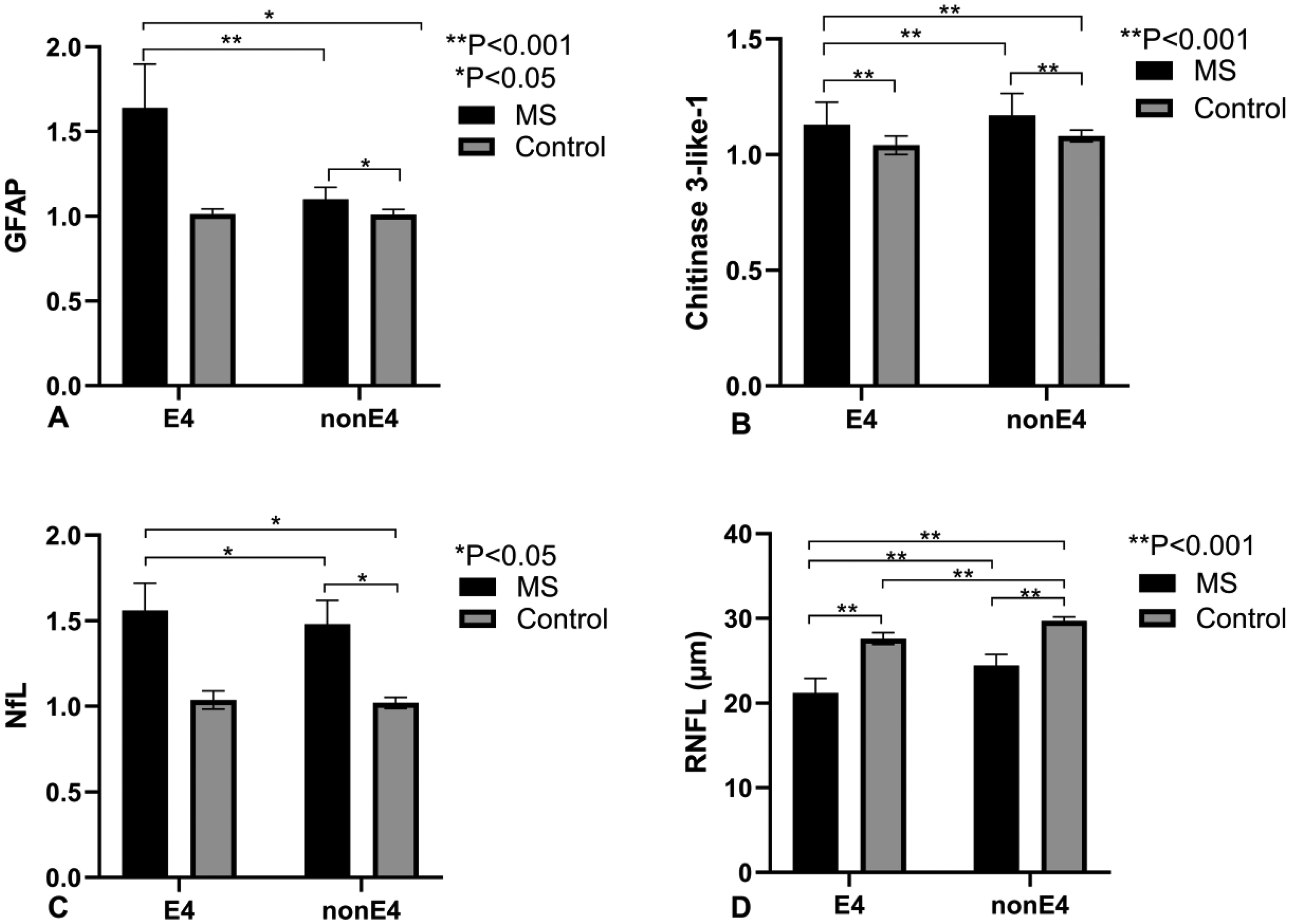
Comparison of biomarker levels and retinal nerve fiber layer (RNFL) thickness between multiple sclerosis (MS) patients and controls, stratified by APOE ε4 status (E4 vs. nonE4). A) Glial fibrillary acidic protein (GFAP) levels in MS (black bars) and controls (gray bars) show significantly elevated GFAP in MS patients for both E4 (**p < 0.001) and nonE4 (*p < 0.05) groups. Differences are significant between E4 and nonE4 groups within both MS and control populations. B) Chitinase 3-like-1 levels are higher in MS across both E4 and nonE4 groups (**p < 0.001), with significant pairwise differences (**p < 0.001) between E4 and nonE4 in both MS and controls. C) Neurofilament light (NfL) levels are elevated in MS compared to controls in both E4 and nonE4 groups (*p < 0.05), with noted differences between MS and controls in each allele group. D) RNFL thickness (µm) is reduced in MS compared to controls for both E4 and nonE4 groups (**p < 0.001), with significant intra-group differences (**p < 0.001) between E4 and nonE4. Error bars indicate standard error of the mean (SEM), with significance levels indicated for each comparison. This data highlights APOE ε4's influence on biomarker levels and RNFL thickness in MS. APOE4: apolipoprotein E4.
Summary of statistical analyses and results.
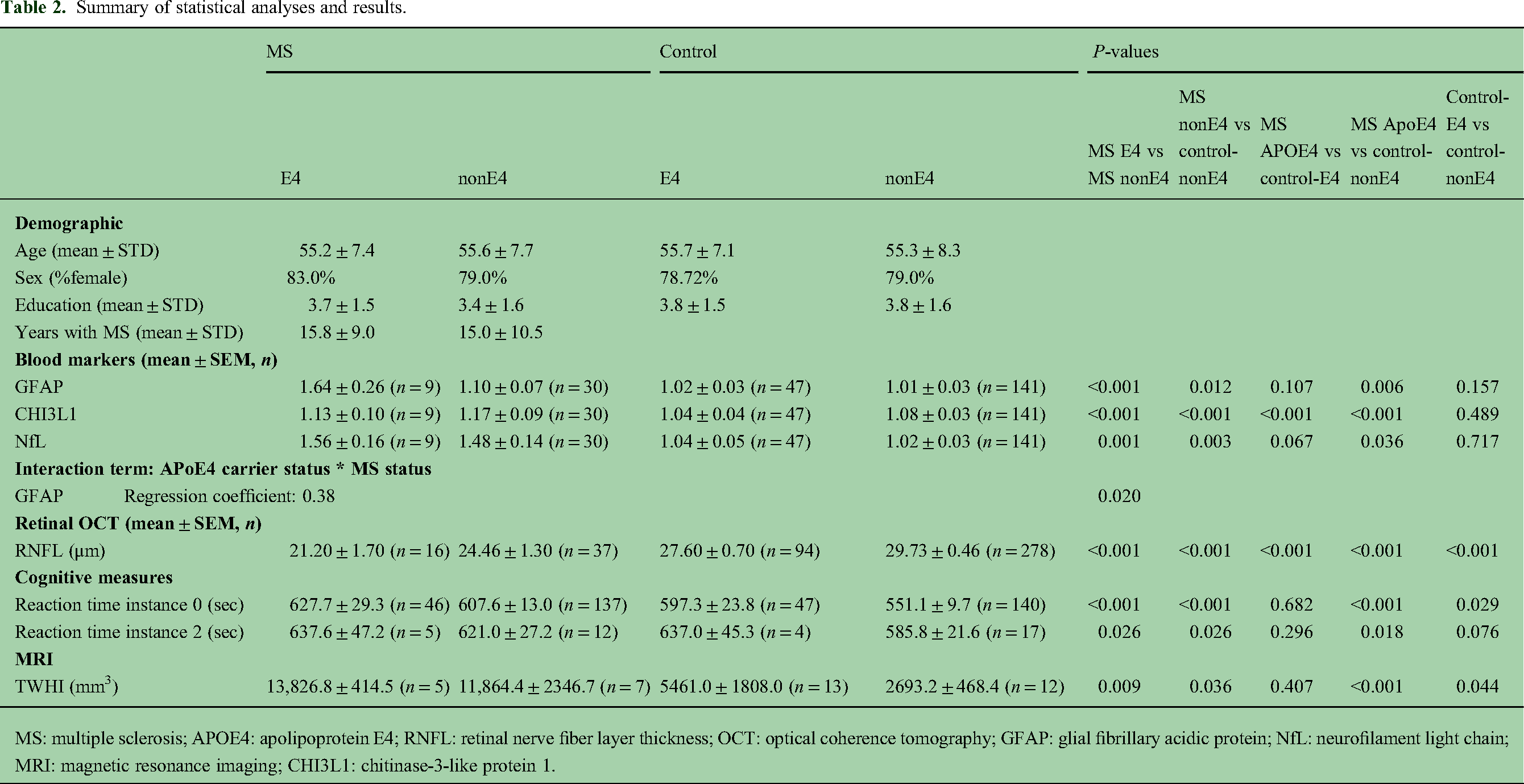
MS: multiple sclerosis; APOE4: apolipoprotein E4; RNFL: retinal nerve fiber layer thickness; OCT: optical coherence tomography; GFAP: glial fibrillary acidic protein; NfL: neurofilament light chain; MRI: magnetic resonance imaging; CHI3L1: chitinase-3-like protein 1.
Retinal optical coherence tomography
We analyzed retinal OCT data to assess the impact of APOE4 on retinal nerve fiber layer (RNFL) thickness in MS patients (Table 2 and Figure 2D). Eight MS-E4 subjects had OCT data of both the left and right eye, while for MS-nonE4, 18 patients had OCT data of both eyes with one additional patient having only right eye data. The RNFL was significantly thicker in patients without the APOE4 allele irrespective of the laterality of the eye. The average thickness for the MS-E4 group was 21.2 μm compared to 24.5 μm in the MS-nonE4 group (p < 0.001). A sensitivity analysis using the average RNFL thickness across both eyes per subject confirmed the original findings, with all between-group comparisons remaining statistically significant (p < 0.001), thereby validating the repeated-measures approach. These findings suggest that the presence of the APOE4 allele may be associated with a greater demyelination and axonal loss as suggested by the thinner RNFL in MS-E4 patients.
Cognitive status
We assessed attention and processing speed by comparing reaction times in MS patients with and without the APOE4 allele (Table 2 and Figures 3A and B). At baseline (instance 0), MS-E4 patients had longer reaction times than MS-nonE4 patients (p < 0.001), which was maintained at follow-up (instance 2) (p < 0.001). These findings suggest that the presence of the APOE4 may be associated with greater cognitive impairment among MS patients.
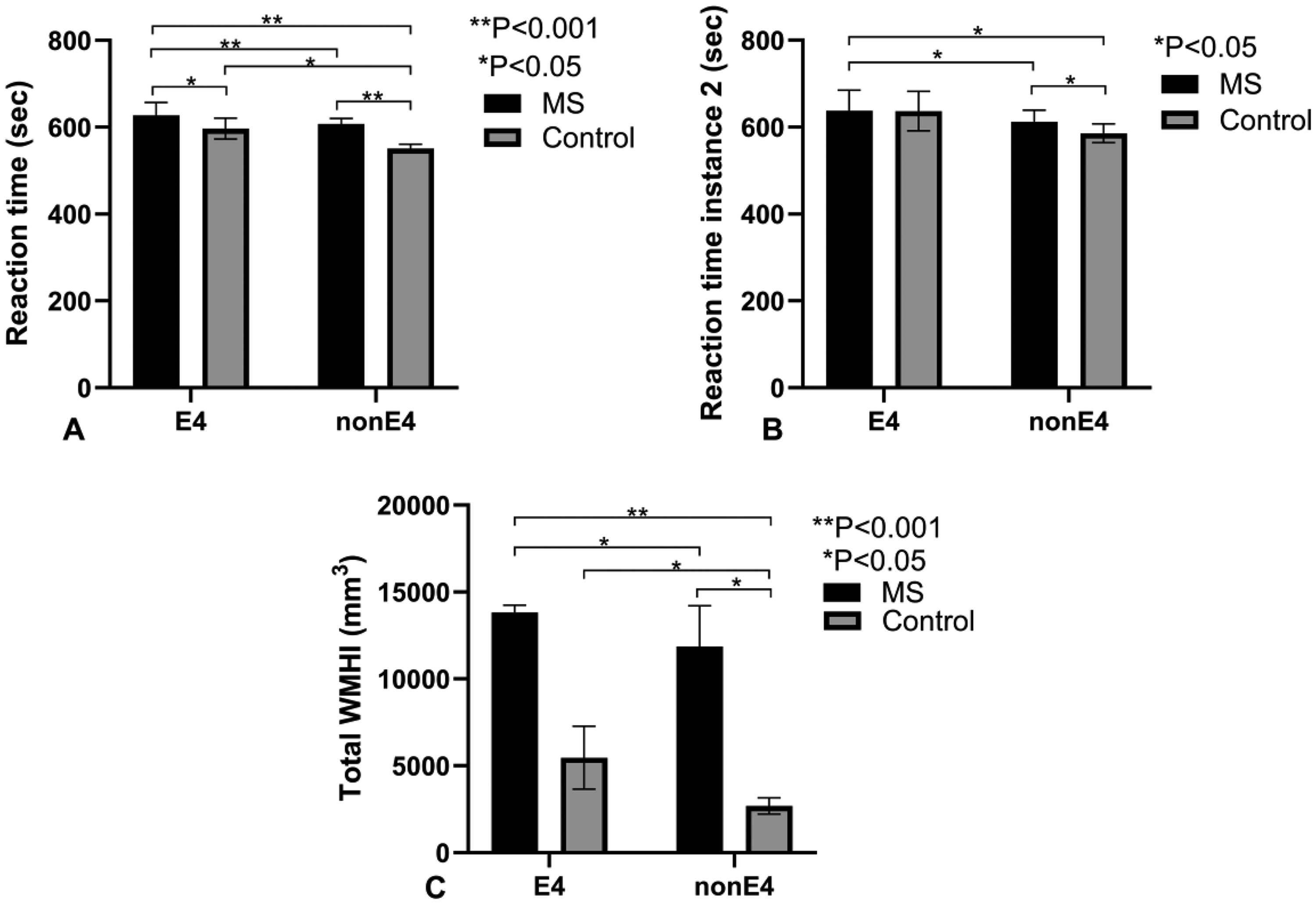
This figure consists of three panels comparing reaction times and total white matter hyperintensity index (WMHI) volumes between multiple sclerosis (MS) patients and control subjects, stratified by APOE ε4 allele status (E4 vs. nonE4). In all panels, the error bars represent the standard error of the mean (SEM), and statistical significance levels are clearly marked for comparisons between groups. A) Reaction time (seconds) in the first instance is presented for E4 and nonE4 groups in both MS patients and control subjects. The MS patients (black bars) exhibit significantly longer reaction times compared to control subjects (gray bars) in both E4 and nonE4 groups. Statistical significance is indicated by *p < 0.05 and **p < 0.001, with detailed pairwise comparisons showing significant differences between E4 and nonE4 groups within both MS and control categories. B) Reaction time (seconds) in the second instance is shown for E4 and nonE4 groups in MS patients and control subjects. The pattern is similar to the first instance, with MS patients showing longer reaction times than controls across both E4 and nonE4 groups. Significant differences (*p < 0.05) are noted in specific comparisons between MS and control subjects. C) Total WMHI volume (mm³) is depicted for E4 and nonE4 groups among MS patients and control subjects. MS patients display significantly higher WMHI volumes compared to controls within both E4 and nonE4 groups, with highly significant differences (**p < 0.001) and several pairwise comparisons indicating *p < 0.05. The data suggest a pronounced increase in WMHI volume among MS patients, particularly those with the E4 allele. APOE4: apolipoprotein E4.
Magnetic resonance imaging findings
MRI was used to assess the impact of the APOE4 allele on white matter disease burden in MS patients. MRI studies were available for 12 subjects in the study, 5 MS-E4 and 7 MS-nonE4. Notably, the MRIs are obtained 10 years after the initial encounter. Results are summarized in Table 2 and Figure 3C. MS-E4 patients were noted to have more white matter hyperintensity index (WMHI) (p = 0.009). These findings indicate that MS-E4 patients exhibit a greater burden of WMHI compared to MS-nonE4 patients.
Primary endpoints remained significant after FDR correction (see Supplementary Table S1), while exploratory outcomes are reported with unadjusted p-values.
Secondary analysis—apolipoprotein E4 in non-multiple sclerosis subjects
Multiple sclerosis-nonE4 vs control-nonE4
MS-nonE4 patients were compared to control-nonE4 subjects with regard to blood markers, OCT, reaction time, and MRI data. GFAP, CHI3L1, and NfL were all elevated in the MS-nonE4 group (GFAP: p = 0.017; CHI3L1: p < 0.001; NfL: p = 0.005). RNFL was found to be thinner for the MS-nonE4 group than control-nonE4 (p < 0.001). Reaction time was longer in the MS-nonE4 group (p < 0.001). MRI data also showed higher volume of WMHI in the MS-E4 group (p = 0.036). These results demonstrate that all tested markers, including blood biomarkers, RNFL thickness, reaction time, and MRI findings, showed significant differences between MS-nonE4 patients and control-nonE4 subjects, underscoring the pronounced impact of MS in the nonE4 groups (Table 2 and Figures 2 and 3).
Multiple sclerosis-E4 vs control-E4
MS-E4 patients were compared to control-E4 subjects on blood markers, OCT, reaction time and MRI data. Unlike in the nonE4 comparison, only CHI3L1 was higher in the MS group compared to control (p < 0.001). RNFL was thicker in the control-E4 group (p < 0.001). Reaction times or MRI outcomes were not different between the groups (Table 2 and Figures 2 and 3). These findings indicate that, in contrast to the nonE4 groups, most markers tested did not show significant differences between MS-E4 patients and control-E4 subjects, with only CHI3L1 levels and RNFL reaching statistical significance, highlighting a less pronounced distinction in neurodegenerative and neuroinflammatory profiles within the E4 carrier groups.
Multiple sclerosis-E4 vs control-nonE4
MS-E4 patients were compared to control-nonE4 subjects with regard to blood markers, OCT, reaction time, and MRI data (Table 2 and Figures 2 and 3). GFAP, CHI3L1, and NfL were all significantly elevated in the MS-E4 group (GFAP: p < 0.001; CHI3L1: p < 0.001; NfL: p < 0.001). RNFL was found to be significantly thinner in the MS-E4 group (p < 0.001). Reaction time was longer in the MS-E4 group (p < 0.001). MRI data also showed a significantly greater volume of WMHI in the MS-E4 group (p < 0.001). These results highlight the significant differences between MS-E4 patients and control-nonE4 subjects across multiple measures, including elevated blood markers, longer reaction times, thinner RNFL, and increased WMHI.
Control-E4 vs control-nonE4
Lastly, the two control groups (with and without the APOE4 allele) were compared with regard to blood markers, OCT, reaction time, and MRI data. No differences were found in the blood markers GFAP, CHI3L1, or NfL. The RNFL was thinner in the Control-E4 group (Figure 4). Reaction time was significantly longer in the control-E4 group (p = 0.005). MRI findings showed significantly greater volume of WMHI in the control-E4 group as well (p = 0.044). These findings suggest that while blood markers were comparable between the control groups, the presence of the APOE4 allele in controls was associated with thinner RNFL, longer reaction times, and a greater volume of WMHI.

Retinal nerve fiber layer images of two healthy control subjects. Both images are of the left eye. The image on the left is taken from a person without the APOE4 allele while the picture on the right is taken with a person with the APOE4 allele. APOE4: apolipoprotein E4.
Discussion
This study investigated the role of APOE4, a well-established genetic risk factor for late-onset Alzheimer's Disease, in influencing the progression and severity of MS. The findings suggest that APOE4 may exacerbate chronic neuroinflammation and accelerate cognitive decline in MS. Specifically, we found that 1) key biomarkers, such as GFAP and NfL, were elevated in MS-E4 patients, suggesting greater neuronal injury and inflammatory response; 2) CHI3L1, a novel inflammatory protein, emerged as a promising blood-based biomarker; 3) retinal OCT showed thinner RNFL in APOE4 carriers with MS; and 4) APOE4 carriers had longer reaction time regardless of MS diagnosis. Collectively, these results underscore a more severe disease phenotype in MS-E4 patients compared to their non-APOE4 counterparts and demonstrate that APOE4 may contribute to a measurable neuronal injury even in healthy carriers without neurological diagnoses.
GFAP and NfL, key markers of neuronal injury and astrocytic activation, were significantly elevated in MS patients carrying ApoE4 compared to their nonE4 counterparts, suggesting more pronounced neurodegeneration and autoimmune activity. GFAP, an astrocyte cytoskeletal protein, has been shown to be higher in the serum of patients with primary progressive MS as opposed to relapsing-remitting MS and correlating with white matter lesion load on MRI. 14 The significant interaction between MS and APOE4 carrier status with respect to GFAP levels is particularly noteworthy. NfL, a cytoskeletal protein released from injured neurons, was previously shown to be elevated in MS patients compared to healthy control in both cerebrospinal fluid (CSF) and plasma, with levels correlating with the Expanded Disability Status Scale (EDSS). 15 In addition, both GFAP and NfL have been shown to be elevated in the serum of patients with mild cognitive impairments and have been suggested as potential early prognostic biomarkers to predict Alzheimer's disease development. 16 While MRI remains the gold standard for diagnosing MS, the increased WMHI observed in MS-E4 patients at follow-up further supports the notion of a more aggressive disease course in APOE4 carriers. These biomarkers, combined with MRI observations, provide strong evidence for the contribution of APOE4 to neurodegenerative processes in MS and support their continual use and examination as non-invasive and inexpensive biomarker for MS. It is worth noting that the absence of significant differences between the MS-E4 and control-E4 groups on some biomarkers may reflect limited power due to small sample sizes. Nonetheless, the consistent pattern of elevated GFAP, NfL, and WMHI in MS-E4 compared to control-E4 is directionally aligned with our hypothesis. Moreover, the significant differences between MS-nonE4 and control-nonE4 support the notion that APOE4 may independently predispose to neurodegenerative changes. Prior studies have demonstrated that APOE4 is associated with increased vulnerability to axonal injury and impaired neuronal repair mechanisms—even in cognitively normal individuals—suggesting a baseline predisposition to central nervous system (CNS) damage regardless of underlying disease status.17–19 Additionally, the observed structural and cognitive differences between control-E4 and control-nonE4—despite minimal serum biomarker differences—imply that APOE4-related CNS changes may precede detectable alterations in peripheral blood. This could reflect a temporal order of disease progression between tissue-level pathology and biomarker release into the serum, or limitations in peripheral sensitivity for early neurodegenerative changes.
CHI3L1, another inflammatory marker, is a glycoside protein secreted from glial cells, astrocytes in particular, and plays a crucial role in the inflammatory response seen in MS as well as other neurodegenerative diseases. 20 Previous studies showed that CSF levels of CHI3L1 are higher in MS patients than healthy controls and correlate with the EDSS. 20 CSF CHI3L1 levels are also elevated during remission periods than during acute attacks in MS patients. 11 Our data are consistent with these findings, showing increased serum CHI3L1 levels in MS patients compared to controls. However, the lower levels observed in the MS-E4 group compared to MS-nonE4 are unexpected and may reflect APOE4-associated alterations in astrocyte function or neuroinflammatory signaling. While CHI3L1 levels are generally elevated in the blood of MS patients, their association with disease progression and severity has been inconsistent across plasma-based studies. 21 Furthermore, Donder et al. reported higher serum CHI3L1 levels in patients with clinically isolated syndrome compared to those with established MS, suggesting that serum CHI3L1 may be more dynamic in early inflammatory disease states. 22 Lower serum CHI3L1 in E4 carriers may reflect diminished astrocytic secretion or different inflammatory kinetics.11,21 Future studies using more direct indicators of astrocyte activation, such as translocator protein PET imaging, may help clarify the relationship between serum CHI3L1 and CNS gliosis in E4 carriers.
The detection of these protein biomarkers in the plasma poses questions regarding the mechanism of relocation of said proteins from CSF to blood. While NfL is present in the peripheral neurons as well as central, it has been shown that along with GFAP, a purely CNS protein, it can be used to predict MRI abnormalities in traumatic brain injury. 23 As such, these proteins have been suggested as potential indicators of blood–brain–barrier (BBB) permeability. 24 Similarly, CHI3L1, has been implicated as a central player in the disruption of the BBB though both an APOE4 pathway and neuroinflammatory pathway. Since BBB disruption due to endothelial cell dysregulation and cytokine-mediated inflammation has been implicated in both MS and APOE4 studies,25,26 it is reasonable to postulate that our results may underscore a more substantial BBB integrity loss in MS-E4 patients.
Retinal OCT findings, RNFL thickness in particular, have been shown to correlate clinically with visual deficits, disability, and MRI findings. 27 The retina, being the most easily visualized window into the cerebrum, is a very common autoimmune target in MS with approximately 20% of patients presenting with optic neuritis as the first symptom and almost 90% of MS patients showing evidence of retinal damage on post-mortem examination. 28 As noted in prior literature, 29 our results support the findings of significantly thinner RNFL in MS subjects compared to healthy controls in all groups. In the MS populations, we find compelling evidence of an APOE4 carrier effect on retinal nerve myelination with significantly thinner RNFL in the MS-E4 group compared with the MS-nonE4 group. Interestingly, healthy controls with the APOE4 allele had significantly thinner RNFL thicknesses than nonE4 controls, which can be explained by the inherent predisposition of APOE4 carriers for neurodegenerative processes and supports the ongoing utilization of retinal OCTs in neurodegenerative and neuroinflammatory diseases. 30
Cognitive decline is common in MS, with previous research demonstrating slower reaction time and processing speed in MS patients compared to controls.31–35 Additionally, reaction time is known to be one of the most affected cognitive domains in MS.36,37 Our study confirms this overall finding but highlights a more pronounced impact of the APOE4 allele on reaction time than MS itself. Control subjects without APOE4 demonstrated significantly faster reaction times compared to those with the allele, both with and without MS, suggesting a potential cognitive disadvantage linked to APOE4. This pattern persisted upon retesting 10 years later, although the difference between healthy controls with and without APOE4 lost significance. In MS patients, those carrying APOE4 consistently had longer reaction times at both time points. Given numerous studies linking processing speed to memory and attention,32,33,38 these results suggest that reaction time, while not directly correlated with other biological markers, may reflect deficits in these cognitive domains, particularly in individuals with the APOE4 allele. However, it is important to acknowledge that reaction time may also be influenced by motor function. Future studies using the UKB or other datasets could incorporate objective motor assessments, such as grip strength, to better isolate the cognitive component of reaction time and clarify APOE4's role in cognitive decline.
This study has several limitations. First, it does not account for the treatments administered to MS patients, including the use of disease-modifying therapies known to alter biomarker levels and MRI progression, which could impact results. Moreover, the study did not distinguish between different forms of MS which would influence response to therapy and have been shown to influence biomarker levels as well as clinical profile. 21 This is a key limitation of UKB data that should be addressed in further analysis using different datasets, particularly as new therapies emerge. Second, the number of patients who underwent brain MRIs was limited, and no spinal MRIs were available. Future studies could incorporate longitudinal imaging, including both spinal and brain MRIs, to better monitor disease progression. Third, the study's demographic was predominantly female (∼78%) and white, reflecting the UKB study population. A more diverse cohort would strengthen the generalizability of the findings. Fourth, while reaction time is a well-recognized proxy for information processing speed in MS, it may be influenced by motor confounders and does not fully capture other cognitive domains such as memory, executive function, or language. Additionally, data were largely drawn from a single time point, with MRIs conducted around 10 years after the initial assessment. Longitudinal data was sparse, and limited power could have weakened certain associations. Future studies should incorporate serial evaluations of emerging disease severity markers. Finally, the study did not differentiate between homozygous and heterozygous APOE4 carriers due to sample size limitations. Larger studies are needed to explore the impact of the number APOE4 allele copies on disease outcomes.
In conclusion, this study demonstrated the greater disease severity and worse health outcomes in MS patients who are APOE4 carriers and highlights APOE4's impact on neurodegeneration at pre-clinical stages of disease. This study also supports the integration of biomarkers and retinal OCT in MS diagnostics and long-term management.
Supplemental Material
sj-docx-1-mso-10.1177_20552173251387833 - Supplemental material for Apolipoprotein E4 and its later-life health effects on the multiple sclerosis population
Supplemental material, sj-docx-1-mso-10.1177_20552173251387833 for Apolipoprotein E4 and its later-life health effects on the multiple sclerosis population by Dean Zeldich, Chia Hsin Cheng, Yi Guan, Sriman Narain and Bang-Bon Koo in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Disclosures
The authors report no conflicts of interest or disclosures relevant to this work.
Declaration of generative AI and AI-assisted technologies in the writing process
During the preparation of this work the authors used OpenAI in order to proofread and improve the readability of the article. After using this tool/service, the authors reviewed and edited the content as needed and take full responsibility for the content of the published article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by CDMRP research awards W81XWH-22-1-0488 and HT9425-24-1-0934. This research has been conducted using the UK Biobank Resource under application number 102369.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
