Abstract
Introduction
Breast cancer, predominantly seen as a female disease, also affects men, though very infrequently. Male breast cancer (MBC) accounts for approximately 1% of all breast cancers and 1% of cancer diagnoses among men globally, representing a unique subset within oncology.1,2 The rarity of MBC has contributed to a significant knowledge gap in its pathophysiology, leading to a reliance on data extrapolated from female breast cancer research for treatment protocols.3,4 However, emerging evidence suggests that MBC has distinct clinical and molecular characteristics compared to its female counterpart, warranting a gender-specific approach to its management.5,6
The genetic and epigenetic landscape of MBC, including differences in hormone receptor expression and variations in gene mutations, influences its clinical behavior and response to therapy. 4 These differences highlight the necessity for dedicated studies to tailor treatment strategies for MBC patients. Moreover, the identification of high-risk male groups remains a challenge, often leading to delayed diagnoses and suboptimal outcomes, particularly in resource-limited settings. 7 The psychosocial burden of MBC is also notable, as affected men may experience emasculation and isolation due to the perception of breast cancer as a female disease.8-10
While female breast cancer has been extensively studied, resulting in improved detection methods and therapies, MBC has not received the same level of attention. This disparity is evident in clinical guidelines and trial designs, which have historically excluded or underrepresented men.7,11 Recent initiatives by the Food and Drug Administration (FDA) and other regulatory bodies have begun to address this issue by advocating for the inclusion of men in breast cancer clinical trials, an approach that promises to provide more gender-inclusive data.12,13
This retrospective analysis conducted at a community academic institute in central Illinois aims to examine MBC epidemiological trends, clinical presentation, treatment modalities, and patient outcomes over 21 years, as well as to compare these trends against the national female data. By analyzing this, we hope to contribute valuable insights into the natural history of MBC and its management, potentially influencing future guidelines and therapeutic strategies tailored to men with breast cancer.
Methodology
This retrospective study was conducted at a community Academic institute in Central Illinois. We sought to analyze the epidemiology, treatment patterns, and outcomes of male breast cancer (MBC) patients over two decades, from January 2000 to December 2021. This study aimed to provide a more comprehensive understanding of MBC in an academic setting, which may reflect a different patient demographic than those seen in national data.
We retrospectively identified and included all cases of men aged 18 years and older with histologically confirmed MBC who had received any treatment for their condition at our institution. We excluded patients lost to follow-up if their medical records did not contain sufficient information for a complete review, and transsexual individuals as potential biases may be introduced due to hormonal replacement therapies. This study was reported following the relevant Equator - STROBE guidelines. 14
Utilizing REDCap electronic data capture tools hosted at our institution, we systematically collected patient data. This included demographics, clinical presentation, imaging and biopsy results, histopathological features, treatment modalities (including surgery, chemotherapy, radiation therapy, and hormonal therapy), recurrence information, and survival outcomes.
The study protocol was reviewed and approved by the Southern Illinois University School of Medicine Institutional Review Board (IRB #22-259). Given the retrospective nature of the study, informed consent was waived. All patient data were de-identified to maintain confidentiality and comply with HIPAA regulations.
Statistical Analysis
Descriptive statistics were utilized to summarize patient characteristics, clinical features, and outcomes. We reported categorical variables as counts and percentages, and continuous variables using means, medians, ranges, and standard deviations as appropriate. Categorical variables included age at diagnostic biopsy (under or over 65 years), race (white, black, other), first sign of disease (breast mass, pain, nipple inversion, asymptomatic), mammogram result (normal, abnormal), ultrasound (normal, abnormal), breast biopsy side (left, right), hormone receptor status of ER, PR, and HER-2 neu (negative, positive), histology of tumor (invasive ductal, invasive mucinous, invasive lobular), grade (I, II, III), TNM (X,0,1,2,3,4), Oncotype Dx test result (<16, 16-25, >25), surgery (lumpectomy, mastectomy), axillary exploration (sentinel lymph node biopsy, axillary lymph node dissection), clinical stage (I, II, III, IV), neoadjuvant and adjuvant treatment (yes, no). Additionally, continuous variables included age at diagnostic biopsy, BMI, and time from first sign of disease to diagnostic imaging in months.
Survival analysis was conducted to evaluate differences in recurrence-free survival (RFS) and overall survival (OS) across categorical groups. Categorical groups of interest included age group, race, clinical stage, grade, and hormone receptor status. Overall survival time was calculated from date of diagnostic biopsy until date of death, censoring those alive at their last known follow up date. Recurrence free survival time was calculated as date of diagnostic biopsy until date of recurrence, censoring those without recurrence at the last known clinical assessment date. Only patients with available documented information and follow up were included per analysis. Kaplan-Meier survival curves were generated to estimate overall survival probability at 5 years. The log-rank test was used to compare survival distributions between groups and assess statistical significance. Censoring was applied for patients who were lost to follow-up or did not experience the event (recurrence/death) during the study period. Survival estimates and standard errors were reported at each time point. All tests were two-sided, and a P-value of less than 0.05 was considered statistically significant. Statistical analyses were performed using SAS software, version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
TNM Staging. Percentages in Parentheses

Histologically, invasive ductal carcinoma was the predominant type among 75 patients (n = 6 missing), found in 90.7% of patients. Tumor pathology grades among 68 patients (n = 13 missing) were distributed as follows: Grade 1 in 7.4%, Grade 2 in 47.1%, and Grade 3 in 45.6%. MammaPrint/BluePrint molecular subtyping was done on one patient, identifying a Luminal B tumor. Oncotype Dx testing performed in 17 patients showed low scores in 35.3%, intermediate in 41.2%, and high in 23.5%. Foundation One genetic testing was performed in the tumor tissue of seven patients, identifying mismatch repair deficiency in 28.6% and ATM mutation in 14.3%. Germline BRCA mutations were investigated in seven patients, detecting BRCA2 mutations in six cases (85.71%). Other constitutional mutational tests were not commonly available for many of our patients at the time of diagnosis.
Clinical Characteristics of Male Breast Cancer Patients.

Of 76 patients, 50.0% ultimately received radiation therapy while the rest either declined therapy (4.0%) or were not offered radiation therapy due to early-stage disease (46.0%). The remaining five patients did not have sufficient information to be included in this analysis. Of those treated, 33 patients (86.8%) received a standard-dose regimen of 5040 gray delivered in 28 fractions while the rest were missing this information.
De-novo stage IV metastatic disease was present in 10.4% of the 77 patients with available clinical TNM staging, while metastatic sites included bone (50.0%), lungs (25.0%), and liver (12.5%); other patients did not list exact location of metastatic disease. Treatment for metastatic disease included tamoxifen, anastrozole with goserelin, everolimus with exemestane, or fulvestrant, followed by various chemotherapy regimens. Palbociclib was administered to 7.0% of patients, while zoledronic acid was used in 4.0%.
There were 77 patients with available clinical follow-up data for assessment of recurrence. A total of 16 patients experienced recurrent disease. The median time to recurrence was 3.2 years (range 81 days to 9.4 years), of which early recurrences (within 5 years) occurred in 81.3% of cases, whereas 18.8% experienced late recurrences (5+ years). Recurrence patterns showed that 31.3% of these patients had local recurrences, while 68.8% had distant recurrences. Median follow-up time for those not experiencing recurrence was 4.1 years (range 17 days to 22.5 years). The 5-year recurrence-free survival (RFS) rate for all 77 patients was 76.2% (standard error [SE]: 5.9%) (Figure 1). Recurrence Free Survival in Total Study Population at Risk for Recurrence. Kaplan-Meier Curve Representing Recurrence Free Survival of Subjects at risk. Number of Patients at Risk at the Bottom of the figure.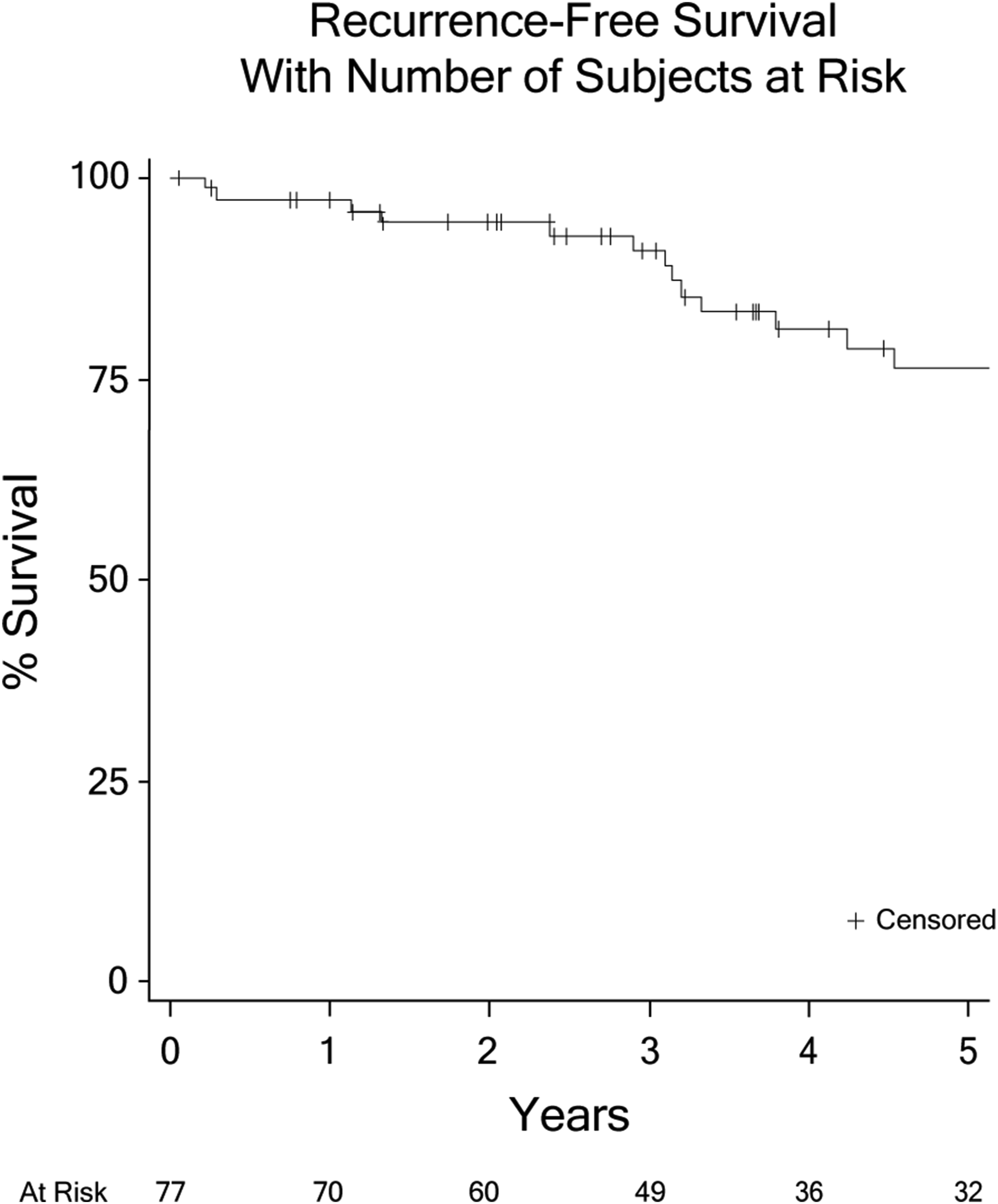
Recurrence Free Survival and Overall Survival in Patients with Respect to Their Clinical Characteristics] Groups Compared Using Log-Rank Test.
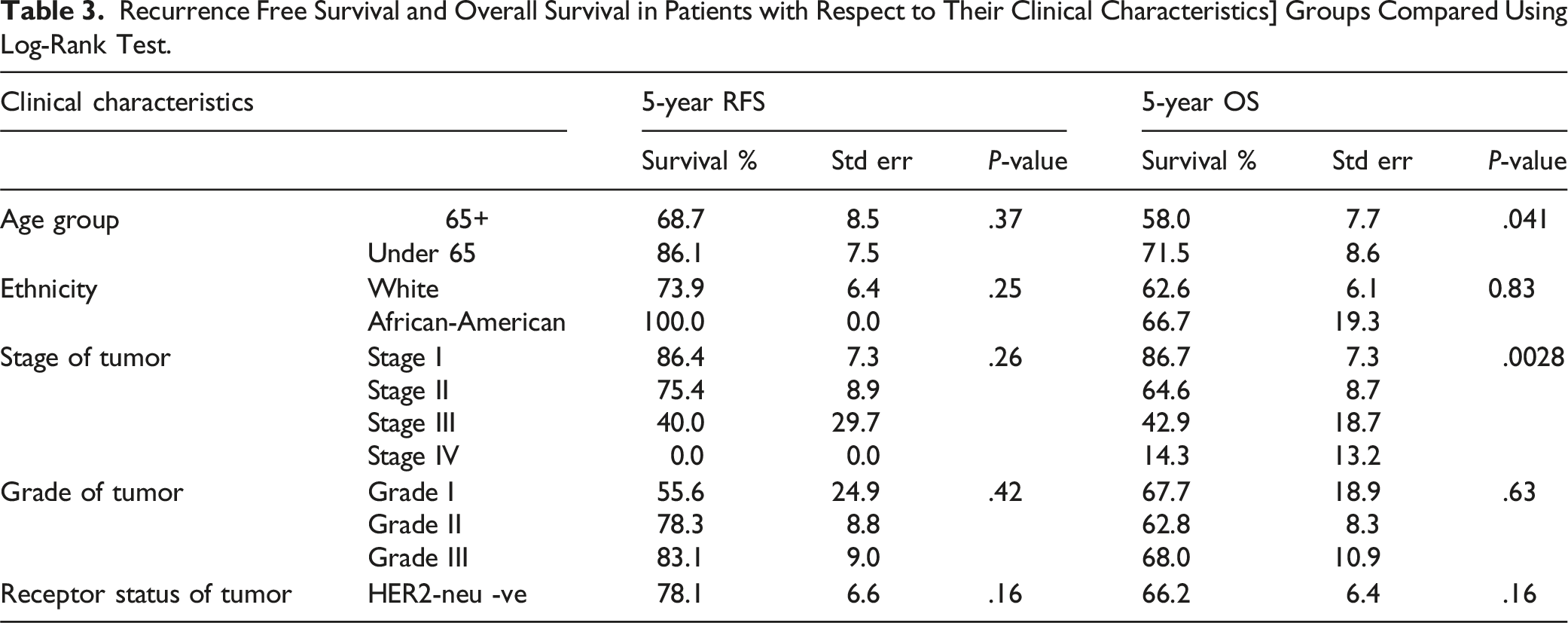
At the study’s conclusion, 45 patients were deceased. The median time from initial biopsy to death was 3.7 years (range 22 days to 18.0 years). Median follow-up time for those not deceased was 4.9 years (range 17 days to 22.6 years). The estimated 5-year overall survival (OS) rate was 63.4% (SE: 5.8%) (Figure 2). When evaluating OS based on demographic and clinical factors, patients under 65 years old had a 5-year OS of 71.5% (SE: 8.6%), while those older than 65 was 58.0% (SE: 7.7%). Moreover, the 5-year OS among the African-American group was 66.7% while in the White population was 62.6%. Survival rates at five years across stage of disease were 86.7% for Stage I, 64.6% for Stage II, 42.9% for Stage III, and 14.3% for Stage IV. By tumor grade, the 5-year OS was 67.7% for grade I, 62.8% for grade II, and 68.0% for grade III. In terms of hormone receptor status, patients who were HER-2 neu negative had a 5-year OS of 66.2%, in contrast to 49.1% in HER-2 neu positive patients (Table 3). Statistical analysis revealed that age significantly influenced survival outcomes, with a marked difference OS between patients below 65 and those above 65 (P = .041) (Figure 3). Additionally, a significant association in OS was identified across the different tumor stages (P = .0028) (Figure 4). Overall Survival in Total Study Population at Risk for Death. Kaplan-Meier Curve Representing Overall Survival of Subjects at risk. Number of Patients at Risk at the Bottom of the figure. Overall Survival in Patients Below 65 years of Age vs Patients Older than 65. Kaplan-Meier Curves Representing Overall Survival of Subjects at Risk by Age Group. Solid Line, Patients Over 65 years old. Dashed Line, Patients Under 65 years old. Groups Compared Using Log-Rank test. Number of Patients at Risk at the Bottom of the figure. Overall Survival in Patient Population Suffering From Stage I-Stage IV Disease. Kaplan-Meier Curves Representing Overall Survival of Subjects at Risk by Clinical Stage. Thin Dashed Line, Stage I. Thin Solid Line, Stage II. Thick Dashed Line, Stage III. Thick Solid Line, Stage IV. Number of Patients at Risk at the Bottom of the figure. Groups Compared Using Log-Rank Test.


Moreover, in order to analyze the patients’ RFS and OS based on their breast cancer molecular subtype, we grouped all patients with positive hormonal receptor status and considered them as Luminal (n = 71; due to the limited information available, we could not differentiate between Luminal A and B); as well as patients with HER-2 positive as HER-2 enriched (n = 9); one patient had triple negative breast cancer and was not included in the statistical analysis. With this approach, we did not find any statistical differences between groups (RFS: P = .5; OS: P = .2).
Discussion
Male breast cancer (MBC), although relatively rare, presents unique challenges in understanding and managing the disease due to the scarcity of prospective clinical data. Accounting for approximately 1% of malignancies in men, and less than 1% of all breast cancer diagnoses, male breast cancer’s rarity underscores the importance of studies like ours to inform clinical practice. 1 It is estimated that 2790 new male breast cancer cases would be diagnosed in the United States, resulting in approximately 530 deaths while the female population is expected to have 310 720 new cases and 42 250 deaths. 1 Additionally, according to national data, the median age at diagnosis in males is 68 years and 62 years in females, which is consistent with the data of our population (67 years).2,15
Several risk factors have been associated with male breast cancer, including high estrogen exposure, androgen deprivation states, and genetic predispositions.6,16,17 For example, the inherited BRCA2 germline mutation is present in 3.7%–40% of MBC and in 20% of high risk females with breast cancer4,18; In addition, recurrent sporadic mutations in PIK3CA, HER2C2, MAP3K1 is present in 12% of luminal type A cancers in males and 40%–50% in females. 19 Moreover, PIK3CA and GATA3 were the most frequent mutations present in luminal type B breast cancer in males, roughly up to 20% while in females TP53 and PIK3CA were the most frequent mutations, being present in 32% of the luminal type B breast cancer cases.4,20 BRCA2 germline mutations were prevalent in 6 out of 7 cases (85.71%) of our study population. Moreover, according to the literature, Luminal A phenotype is the most frequent phenotype in up to 75% of male breast cancers and 68% of female breast cancers; while basal phenotype is very rare.4,21 Our study identified the presence of luminal type B identified in one patient through BluePrint testing. Additionally, ATM mutations or mismatch repair deficiencies were observed in three patients via Foundation One testing. However, this data may be biased in our population as genetic analysis was only performed in patients with specific risk factors that required further testing.
The histologic profile in our patient population mirrors the literature, with invasive ductal carcinoma being the most common subtype observed in up to 90% of MBC patients as compared to 70% in female breast cancer patients. 16 The hormone receptor positivity in our cohort was high, with 91% and 90% of patients expressing estrogen and progesterone receptors, respectively, which is slightly higher than the 80% reported in women. 22 Notably, a higher-than-expected 12% of our patient population had HER-2 neu-positive tumors as compared to the 5% national average of HER2 prevalence in MBC, but similar to the 14% seen in women.3,7,11,13 This underlines the heterogeneity of HER-2neu positive breast cancer and the potential value of molecular profiling to inform prognosis. 23
Diagnosis delays are a critical issue in male breast cancer due to the lack of screening protocols such as women have, often leading to advanced-stage disease (delays of >6 months) and a poorer prognosis (delays of >2 months).7,24 The patients in our population that presented with a breast mass (70.3%) had an average timeframe of 2.4 months between the discovery of the mass and obtaining diagnostic imaging. In contrast, patients without a breast mass (16.2%) delayed seeking medical attention an average of 1.1 years, which is in the lower reported range in the literature for males (1-10 years), 10 but greater than the reported in the female population (12 days). 25 This data emphasizes the lack of awareness of breast diseases in men, which can translate into diagnosis delays and poorer prognosis. 24 In our study, the high sensitivity of mammography (96%) and ultrasound (98%) for detecting abnormalities emphasizes the importance of these diagnostic tools. 7 Moreover, the stage distribution in our study is reflective of national trends, with 82% presenting with early-stage disease. 8 However, the proportion of stage IV disease in male breast cancer remains higher compared to female breast cancer (9% in male vs 6% in female), highlighting the need for awareness efforts. 15
Oncotype DX testing in our study performed only on 17 patients revealed a mean score of 14.6, which, compared to the literature, is slightly higher than the score of 13.4 found in female breast cancer, though lower than the 18.8 found in the other studies on MBC. 26
Treatment paradigms for male breast cancer largely derive from evidence in female breast cancer due to the lack of male-specific randomized clinical studies.6,17 Surgical management in our population followed conventional practices, with mastectomy being the predominant approach.2,5,11,12 Radiation therapy was appropriately considered for patients with positive lymph nodes or larger tumors, and the majority of those offered radiation accepted it. 16
Our study was consistent with treatment guidelines that corroborate the efficacy of tamoxifen as the cornerstone of endocrine therapy for hormone receptor-positive male breast cancer.6,11,27 Of 41 patients who received tamoxifen and had available information, 43.9% completed their five year regimen while 56.1% continued and adhered to treatment until the progression of disease, intolerable side-effects, or death. According to national data, the compliance rate of tamoxifen is only 60.2% in men with breast cancer after five years and 50% among women. 28 Despite potential adverse effects leading to discontinuation, our data suggest that maintaining tamoxifen therapy is crucial for improving disease-free and overall survival.29-31 The underuse of adjuvant tamoxifen and the challenges of delayed diagnosis underscore the need for heightened awareness and adherence to treatment protocols.
While aromatase inhibitors are generally reserved for the metastatic setting due to their potential to activate androgen receptor-positive tumors,32,33 their combination with GnRH agonists/antagonists or the use of fulvestrant may offer alternative options for tamoxifen-intolerant patients, although the quality of life and sexual function may still be impacted by these agents.34,35
Advancements in therapeutic approaches are mirrored by the declining mortality rates in male breast cancer in the USA and Western Europe. However, disparities in outcomes persist, likely due to variations in access to care and diagnostic delays. 36 The use of chemotherapy remains a choice for high disease-burden patients, and our patient population with metastatic disease management aligns with these strategies, emphasizing the importance of individualized care. Additionally, emerging therapies, such as PARP inhibitors and AR-targeted treatments, show promise, especially considering the genetic landscape of male breast cancer and the frequency of BRCA mutations.37-39
Our study’s 5-year 63.4% OS, which is lower compared to the national cancer database between 2004 and 2016 (OS 72.8% in males and 83.4% in females), 40 may be attributed to the relatively high number of patients who stopped adjuvant hormonal therapy due to side-effects and the higher percentages of HER-2 positive patients. Moreover, the lack of differences between the recurrence-free survival by clinical stages may be attributed to the high number of censored patients with early disease stages. This highlights the need for diligent application of evidence-based practices and patient adherence. 41 Patient adherence, in general, has led to improved medical outcomes. DiMatteo et al conducted a meta-analysis that led to their conclusion; they noted a 26% improvement in medical outcomes with patient adherence. 42 Numerous oncologic studies have shown improved survival with patient adherence to regimens and physician adherence to treatment guidelines in a variety of cancers.43-45
The small number of patients, as well as the nature of retrospective studies, may limit the scope of our study; however, due to the rarity of this disease, it is important to collectively identify treatment trends and survival. Moreover, a meta-analysis of these studies, and eventually multi-center prospective trials, may be required to truly set this diagnosis apart from female guidelines.
Conclusion
Our study showed similar demographics, histology, and treatment guidelines when compared with the national literature. Patient compliance with therapeutic interventions plays an important role in overall survival and recurrence free survival. Notably, patients with HER-2 neu-positive tumors who consistently engaged in anti-HER2 neu therapy, as well as those with hormone receptor-positive and HER-2 neu-negative tumors who maintained hormone-blocking therapy, were noted to have improved survival. This finding gains particular importance in light of the higher Oncotype Dx scores typically seen in male breast cancer patients, which are often associated with a poorer prognosis. Furthermore, the well-established correlation between advanced cancer stages and diminished 5-year survival rates underscores the critical role of early detection and treatment compliance in this study. Our results highlight the necessity for targeted strategies to enhance patient compliance, as well as the potential for personalized treatment plans to mitigate the risks associated with higher Oncotype Dx scores and advanced cancer stages. Future research should aim to elucidate the underlying reasons for treatment non-adherence in male breast cancer patients and to develop interventions that can improve both adherence and outcomes in this unique population. Additionally, further focus should be aimed to study recurrence-free survival by disease stage as there is limited information in the literature.
In conclusion, our study underscores the necessity for ongoing research into the unique aspects of male breast cancer to improve diagnostic, therapeutic, and management strategies. The integration of genetic counseling and testing, along with the development of targeted therapies, holds the potential to improve care for this patient population. Our findings advocate for a concerted effort to enhance disease awareness, promote treatment adherence, and ultimately improve outcomes for male breast cancer patients. It is through dedicated and patient-centric research that we can hope to reduce mortality rates and improve the quality of life for those affected by male breast cancer.
ORCID iDs
Ricardo Cossyleon https://orcid.org/0000-0002-6918-9230
Krishna Rao https://orcid.org/0000-0002-1788-264X
Statements and Declarations
Ethical Approval
This study received ethical approval from the Southern Illinois University School of Medicine Institutional Review Board on Dec 05, 2022 (IRB approval #22-259). This is an IRB-approved retrospective study, all patient information was de-identified, and patient consent was not required. Patient data will not be shared with third parties.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
