Abstract
Background
Male breast cancer (MBC) represents a rare subtype of breast cancer, with limited prognostic factor studies available. The purpose of this research was to develop a unique nomogram for predicting MBC patient overall survival (OS) and breast cancer-specific survival (BCSS).
Methods
From 2010 to 2020, clinical characteristics of male breast cancer patients were obtained from the Surveillance, Epidemiology and End Results (SEER) database. Following univariate and multivariate analyses, nomograms for OS and BCSS were created. Kaplan-Meier plots were further generated to illustrate the relationship between independent risk variables and survival. The nomogram’s ability to discriminate was measured by employing the area under a time-dependent receiver operating characteristic curve (AUC) and calibration curves. Additionally, when the nomogram was used to direct clinical practice, we also used decision curve analysis (DCA) to evaluate the clinical usefulness and net clinical benefits.
Results
A total of 2143 patients were included in this research. Univariate and multivariate analysis showed that age, grade, surgery, chemotherapy status, brain metastasis status, subtype, marital status, race, and AJCC-T, AJCC-N, and AJCC-M stages were significantly correlated with OS. Lung metastasis, age, marital status, grade, surgery, and AJCC-T, AJCC-N, and AJCC-M stages were significantly correlated with BCSS. By comprising these variables, a predictive nomogram was constructed in the SEER cohort. Then, it could be validated well in the validation cohort by receiver operating characteristics (ROCs) curve and calibration plot. Furthermore, the nomogram demonstrated better decision curve analysis (DCA) results, indicating the ability to forecast survival probability with greater accuracy.
Conclusion
We created and validated a unique nomogram that can assist clinicians in identifying MBC patients at high risk and forecasting their OS/BCSS.
Introduction
Male breast cancer (MBC) is a rare malignancy subtype representing less than 1% of breast cancer (BC) cases and less than 1% of all male cancer. 1 The 5 year age-adjusted incidence of breast cancer in men is almost 1.2 per 100,000, according to recent data from the Surveillance, Epidemiology, and End Results (SEER) database, with an average age at diagnosis of 67 years compared to 62 years for females. 2 Overall survival (OS) estimates for MBC are substantially worse than those for female BC,3,4 highlighting the need for early screening and appropriate therapies for MBC. Despite the fact that MBC has some clinical and histological traits in common with female breast cancer, men were shown to have tumors of later onset with a lower grade, higher stage and more hormone receptor positivity (HR+) than women. Prognostic criteria and risk variables have been shown to be similar in men and women, both of whom demonstrated decreased breast cancer mortality and increased OS over time. 5
Currently, treatment options for MBC have been derived from strategies used in female breast cancer although, the clinicopathological characteristics of MBC are distinct from those of female breast cancer. 6 Given the increasing incidence of MBC in recent years, researchers have begun to pay attention to treatment options and prognostic indicators for this sub-group. 7 There are currently few detailed recommendations available for the treatment of MBC since available treatment options are mostly derived from female BC. 8 The National Comprehensive Cancer Network guidelines recommend that clinicians provide men with treatments similar to that afforded to postmenopausal women. However, many existing studies have shown that MBC is dissimilar to female disease, 9 thus men face unique challenges when it comes to cancer therapeutics. According to some research, OS may not be the most reliable indicator of MBC prognosis and survival rates; instead, breast cancer-specific survival (BCSS) may be better. 10 Some studies suggest that men and women have similar BCSS. 11 Using a sizable cohort from the SEER database, we aimed to examine the predictive variables of MBC patients in this study and create a unique risk classification method to predict their OS and BCSS. In addition, we validated the nomogram in a validation cohort and assessed its efficacy and clinical value using a battery of tests.
Materials and methods
Data Acquisition
SEER*Stat version 8.3.8 was utilized to obtain clinical data on MBC. Patients with MBC diagnosed between 2010 and 2020 were included in the database. This population-based database (https://www.seer.cancer.gov) gathers data on cancer patients from 18 registries, which together account for around 30% of the US population. We used sociological and clinicopathological data in this study, which was approved by the institutional review board of our university. Three keywords—male, breast cancer, and histological diagnosis—were used to identify target individuals. Figure 1 displays the patient selection flowchart. According to earlier research, BCSS is the period of time from diagnosis to MBC-related mortality.12,13 The flow chart for selection of study population.
Cohort Establishment and Variable Selection
By using the R software’s “createDataPartition” function, all eligible patients were recruited from the whole dataset and randomly split into training and validation cohorts at a ratio of 7:3. In this study, a predictive signature was created using the training cohort, and its predictive power and clinical value were confirmed using the validation cohort. Age at diagnosis, histologic type, tumor grade, laterality, surgery status, radiation status, chemotherapy status, bone metastatic state, lung metastatic state, liver metastatic state, brain metastatic state, race, marital status, subtype, ER, PR, HER-2, AJCC-T stage, AJCC-N stage, and AJCC-M stage were among the twenty variables that were extracted from the SEER database. Next, in order to choose the best variables for building predictive models, we used univariate and multivariate Cox regression analysis.
Statistical Analysis
The categorical variables were computed as counts and percentages, and the differences between the training and validation cohorts were compared using the Chi-square test. Independent predictors for individuals with MBC were found using univariate and multivariate Cox regression analyses. A nomogram was created using the best variables derived from multivariate analysis. The calibration plot was used for visualized comparison between prognosis predicted by nomogram and actual ones. Sensitivity and specificity were evaluated by receiver operating characteristics curve (ROC)-the area under the curve (AUC). Additionally, when the nomogram was used to direct clinical practice, we also used decision curve analysis (DCA) to evaluate the clinical usefulness and net clinical benefits. For all statistical studies, SPSS software version 23.0 for Windows andR software version 3.6.3 were used. P-values less than 0.05 were regarded as statistically significant.
Results
Clinical Characteristics of the Participants
Demographic and Clinical Characteristics of Patients With MBC(OS).

Demographic and Clinical Characteristics of Patients With MBC(BCSS).

Next, using a 7:3 randomization ratio, all patients were split into training cohorts (1500 patients) and validation cohorts (643 patients). A Chi-square test was used to determine whether there was a statistically significant difference between the 2 cohorts for BCSS, which was further divided into training cohorts (1106 patients) and validation cohorts (474 patients). For every variable considered, there were no differences between the training and validation cohorts.
Univariate and Multivariate Cox Regression Analysis
Univariate and Multivariate Cox Analyses on Variables for the Prediction of OS of Patients With Male Breast Cancer.
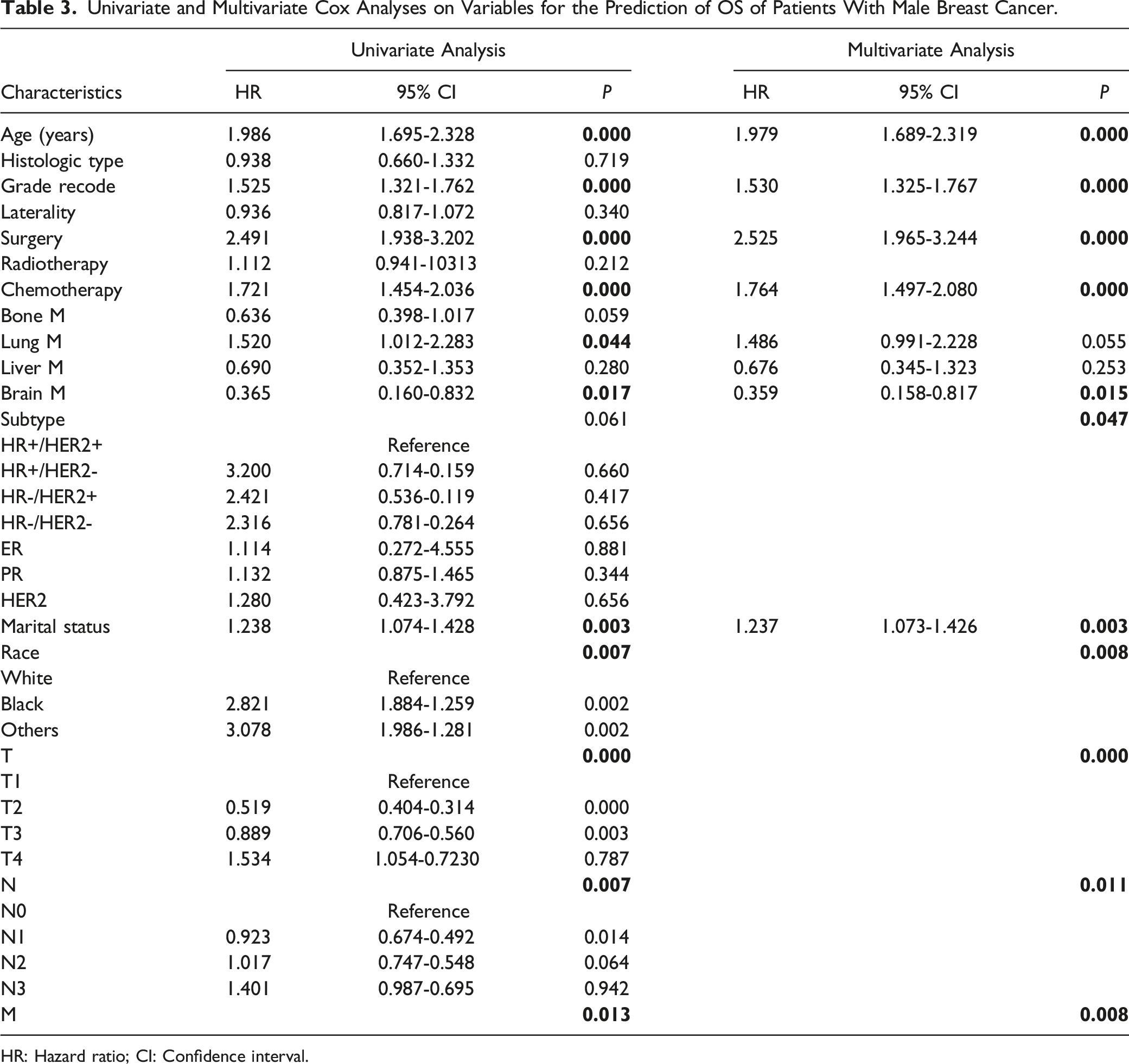
HR: Hazard ratio; CI: Confidence interval.
Univariate and Multivariate Cox Analyses on Variables for the Prediction of BCSS of Patients With Male Breast Cancer.

HR: Hazard ratio; CI: Confidence interval.
Nomogram Development and Validation
We constructed a nomogram to predict the survival probability of patients with MBC via R software, “rms ” and “regplot ” packages. An example of utilizing the nomogram to predict a patient’s chances of survival is shown in Figure 2. The independent predictive indicators found through multivariate analysis were used in this nomogram, to forecast each patient’s total points and predict chances of survival for 3, 5, and 10 years. In addition, we produced calibration and ROC curves to evaluate the nomogram’s calibration and discriminating skills in the 2 cohorts (Figure 3). It proved that the generated nomogram had a higher AUC in predicting the 3- (0.781 vs 0.748), 5- (0.746 vs 0.745), and 10- (0.755 vs 0.741) year survival probability of patients with MBC, regardless of whether they were in the training cohort or validation cohort. The results of 3-(0.836 vs 0.863), 5-(0.839 vs 0.843), and 10-(0.869 vs 0.802) year survival for BCSS are shown in Figure 3(C),(D). Furthermore, the calibration curves (Figure 4) showed strong consistency between the observed and expected OS/BCSS in the 2 cohorts. These aforementioned findings demonstrate that this nomogram has good predictive ability for survival in MBC patients. Establishment of overall survival (OS) and cancer-specific survival (BCSS) nomograms. A Construction of OS nomogram; B construction of BCSS nomogram. Receiver operating characteristics curve (ROC) comparison of OS nomogram. A3-,5-,10-year ROC of OS nomogram using training set; B 3-,5-,10-year ROC of OS nomogram using validation set; C 3-,5-,10-year ROC of BCSS nomogram using training set; D 3-,5-,10-year ROC of BCSS nomogram using validation set. Calibration plots of OS nomogram model. A 3-,5-,10-year calibration plot of OS using training set; B 3-,5-,10-year calibration plot of OS using validation set; C 3-,5-,10-year calibration plot of BCSS using training set; D 3-,5-,10-year calibration plot of BCSS using validation set.


Clinical Utility Evaluation of the Nomogram
The sensitivity and specificity of the prediction model form the basis of the ROC and calibration curve, which mean they are inadequate to distinguish between false positive and false negative situations. Therefore, DCA was extensively used to evaluate the net therapeutic benefits and clinical value of the nomogram. For a given range of threshold probabilities in the training cohort as well as the validation cohort, we found that the nomogram could contribute more net clinical benefits and predict better 3-, 5-, and 10- year OS/BCSS (Figure 5). By drawing a straight line, each independent risk factor corresponds to a specific score on the point axis of the line. The total score refects the sum of the scores for each factor and is plotted down to the 3-, 5- and 10-year survival axes, corresponding to the predicted probabilities of 3-, 5- and 10-year OS/BCSS. Decision curve analysis (DCA) of OS nomogram model. A 3-,5-,10-year DCA of OS using training set; B 3-,5-,10-year DCA of OS using validation set; C 3-,5-,10-year DCA of BCSS using training set; D 3-,5-,10-year DCA of BCSS using validation set.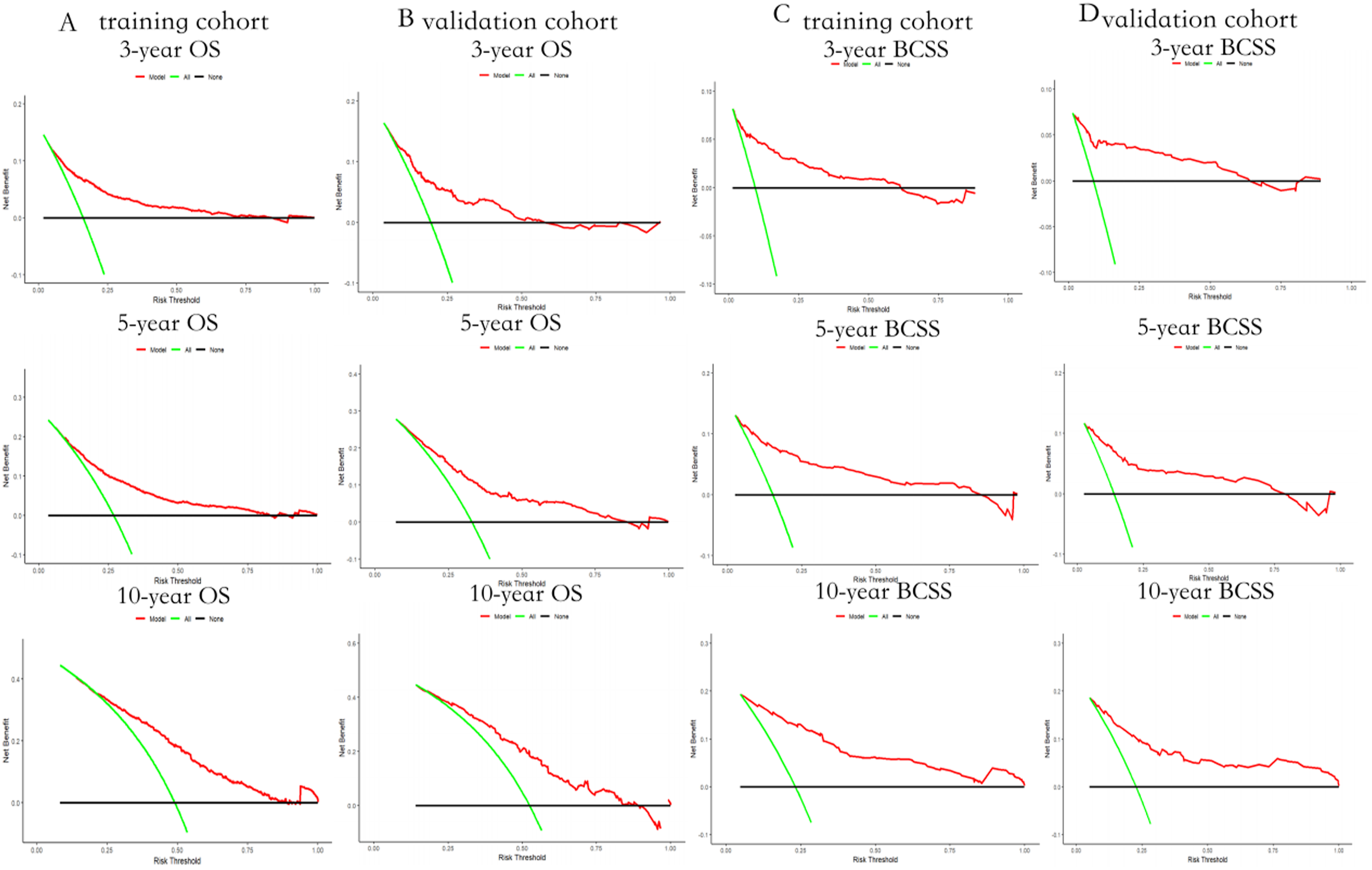
Risk Stratification Ability Assessment of the Nomogram
The risk stratification capacity of the created nomogram was assessed by dividing all patients into low- and high-risk groups based on the median of total points in the training cohort and the validation cohort. To illustrate the variations in survival between various risk groups, we also created Kaplan-Meier survival curves. The survival probability of patients in high-risk groups was found to be considerably lower than that of patients in low-risk groups for both OS and BCSS (Figure 6), indicating that the generated nomogram was likely to be accurate in identifying high-risk patients. Kaplan-Meier survival analysis for evaluating the risk stratification ability of the nomogram in patients with MBC. (A) OS Kaplan-Meier survival curve in the training cohort, (B) OS Kaplan-Meier survival curve in the validation cohort. (C) BCSS Kaplan-Meier survival curve in the training cohort, (D) BCSS Kaplan-Meier survival curve in the validation cohort. MBC, male breast cancer.
Discussion
MBC is an uncommon subtype of breast cancer, with little reported research regarding predictive factors and clinical prognosis. Using a significant MBC dataset from the SEER database, we investigated the clinical characteristics, prognosis, and prognostic variables of these patients. To estimate the likelihood of MBC survival, we created a nomogram based on many prognostic criteria and additionally carried out several validations to assess its clinical value and predictive power. We discovered that the created nomogram performs exceptionally well in predicting OS and BCSS in this patient cohort. Additionally, by calculating DCA, we found that MBC patients may benefit tremendously from clinical practices guided by the nomogram. Higher C- indices and convincing calibration plots for OS and BCSS predictions utilizing the SEER database were seen in the nomograms for this study.
Remarkably, in OS, 11 factors out of 20 were identified for the nomogram’s design (age, marital status, grade, AJCC-T stage, AJCC-N stage, AJCC-M stage, surgery, chemotherapy, brain metastasis status, breast subtype, and race). A similar feature was also seen in BCSS, where 8 factors out of 20 were identified for the nomogram’s development (age, grade, marital status,AJCC-T stage,AJCC-N stage,AJCC-M stage, surgery, and lung metastasis status). We found that in multivariate Cox regression analysis, unfavorable OS was connected with age 65 years or older, grade III-IV, advanced AJCC-T stage, N stage, M stage, brain metastasis, without surgery, without chemotherapy, and HR-/HER2-. Poor BCSS, on the other hand, was linked to extensive lung metastases, grade III –IV, advanced AJCC –T stage,N stage, M stage, and older age without surgery.
Previous studies intimated that variables such as age, a family history of breast cancer, elevated estrogen levels, orchitis/epididymitis, gynecomastia, Klinefelter syndrome, radiation exposure, obesity, external use of estradiol and testosterone, and several genetic risk factors (BRCA2, BRCA1, PALB2, CHEK2, BRIP1, ATM)9,14,15 may be correlated with OS in MBC. In a massive prospective cohort study, Brinton et al discovered that individuals who noted a first-degree relative with a history of breastcancer had an almost 2-fold increased risk of MBC. 16 MBCs are particularly vulnerable to hormonal imbalances that result in an overabundance of estrogen and a lack of testosterone. 17 In 1 report, 8 male patients with a diagnosis of breast cancer had mean total blood estradiol levels and mean free estradiol indices that were considerably higher (P < 0.01 and P < 0.01, respectively) than matched controls. 18 On the other hand, testosterone levels in men with breast cancer were not significantly decreased,and there was no consistent correlation established between estradiol levels and testosterone across studies. 19 Another substantial risk factor is a patient having Klinefelter syndrome, a chromosomal abnormality linked to an elevated estrogen-to- androgen ratio. 20
It is commonly recognized that the liver, brain, bone, and lungs are the organs most often affected when female breast cancer metastasizes. According to this study, bone was the most common site of metastasis, followed by liver and lung metastases. Brain metastases were found in only 0.3% of patients, findings consistent with the biological behavior of female BC. Moreover, multivariate analysis showed that lung metastases were linked to a worse BCSS, but brain metastasis was connected with an unfavorable OS in MBC. For these individuals, treating brain metastases may be beneficial for OS. According to previous findings, distant organ metastasis is uncommon in MBC, and thorough testing must be taken into account when establishing a diagnosis. Furthermore, since this patient subgroup has a reduced OS, they need precise and tailored care.
MBC is usually treated according to the same regimen as female breast cancer, which includes resection followed by chemotherapy, radiation, or endocrine treatment. 21 The great majority of studies demonstrate that these patients gain survival benefits from these therapies; nevertheless, these studies were comprised of individuals from various centers and altered background heterogeneity. Thus, there is ongoing debate regarding the prognostic significance of some therapies in MBC. Usually, sentinel node biopsy (SLNB), axillary lymph node dissection, or modified radical mastectomy are used to treat MBC. 22 The treatment option for T1 and T2 tumors is a simple mastectomy. Generally, males seldom get breast-conserving surgery (BCS) and the majority undergo mastectomies. 6 However, in some circumstances, mastectomies that preserve the skin, nipples, or breast tissue are performed. 9 Based on the TNM staging classification, only 18% of men with T1N0 tumors receive BCS, as per data from the SEER database. 23 BCS is a viable option despite its rarity and is linked to similar survival rates and improved esthetic results. 24 However, all males with clinically negative axillae should have SLNB. 25
Radiotherapy is usually offered to men with T3 or higher tumor stage, positive surgical margins, or 4 or more positive lymph nodes. According to this study, the prolongation of patient OS substantially correlated with both surgery and chemotherapy. Interestingly, no significant relationship was found between radiotherapy and OS/BCSS. Radiotherapy following mastectomy is a controversial topic in MBC. The role of adjuvant radiotherapy in MBC is unclear, but most women with breast cancer follow the recommended guidance. According to Soni et al, locoregional recurrence (LRR) rates without postmastectomy radiation treatment (PMRT) were 14% −25% in low-risk patients and 25% −50% in high-risk patients. PMRT was seen to increaseDFS andOS. The LRR ranged from 3.5% – 13% when using PMRT. 26 In patients with high-risk MBC (positive node, high stage, ≤2 mm or positive margin), Yu et al found reduced LRR rates with PMRT. 27 However, these studies did not show PMRT-induced benefits to OS. Nonetheless, adjuvant radiotherapy was reported to increase 5- and 10-year OS in both BCS and post-mastectomy patients in 1 study that analyzed the national cancer database. 28 According to Tirona et al, adjuvant RT should be performed on all males with BCS. 5 Although, there are no randomized studies to support adjuvant RT, a number of retrospective analyses have demonstrated better results regarding local recurrence after post-mastectomy radiation therapy, especially in cases of node-positivity. 22
There is a lack of prospective data on the use of adjuvant and neoadjuvant chemotherapy in MBC. According to a number of observational studies, men who underwent adjuvant chemotherapy appeared to have reduced recurrence rates and mortality risk. From 1974 to 1988, 31 men with stage II breast cancer and lymph node involvement participated in a single prospective study conducted by the National Cancer Institute, and they were followed for almost 20 years. All patients had a mastectomy followed by CMF (cyclophosphamide, methotrexate, and fluorouracil) for 12 cycles. This regimen led to higher 5-, 10-, and 20-year survival rates. 29 Similar results were seen in another smaller prospective study. 30 Patients who may benefit from adjuvant chemotherapy should be identified using characteristics to predict high-risk for recurrence, similar to those seen in women. These characteristics might include greater tumor size, higher grade, node positivity, and HR negativity. 31 Chemotherapy can be palliative or employed in cases of visceral crisis and rapidly progressive disease, unresponsive to endocrine treatment. Numerous retrospective studies have shown that chemotherapy is effective in treating metastatic MBC. One study examined fifty patients with metastatic MBC who had undergone at least 1 round of hormone treatment. Patients were given chemotherapy regimens that either included anthracycline or not. Considerable anti-tumor activity was seen in all groups with a 56% response rate overall. 32
Given that most MBCs express the ER, treating MBC often involves endocrine therapy, such as tamoxifen. Adjuvant treatment with tamoxifen is advised for a minimum of 5 years. Furthermore, the majority of metastatic MBCs are estrogen receptor-positive. 33 Aromatase inhibitors are not frequently utilized as first line therapy for MBC, in contrast to women. Aromatase inhibitors may partially reduce estrogen levels in gonad-intact males while increasing androgen levels. 34 Given that MBC is nearly universally AR-positive, this rise in androgens may be problematic. These factors make it advisable that an aromatase inhibitor be taken in conjunction with chemical or surgical castration if it is to be employed. 9
One of the most important survival predictors in MBC may be the tumor subtype. According to what is known about breast cancer, the worst prognoses have been associated with triple-negative subtypes. Generally speaking, tumor grade is acknowledged as a separate risk factor for BC prognosis. 35 Wang et al identified higher tumor grade as an independent predictor of reduced survival among female BC patients with bone metastasis, the same conclusion was reached in this study. 13 Numerous studies have documented a correlation between histologic subtype and survival in MBC. 36 Our univariate and multivariable analyses also revealed that triple-negative MBC patients had a considerably poorer prognosis and that MBC subtype was strongly related to OS; however, there was no statistically significant difference in the prognosis for patients with BCSS. Furthermore, race and marital status had a significant impact on prognosis. 37 Married patients in this research had considerably longer OS and BCSS. Married patients often have a better prognosis than single patients because they may receive greater financial and emotional support from their families, as well as more thorough therapy and follow-up. 38 Marriage may impact a patient’s prognosis through telomere length modification, and its influence on disease prognosis may extend beyond social and psychological factors. According to research by Whisman et al, elderly people whose marriages failed had shorter telomeres than others whose marriages remained stable. 39
As a prognostic statistical model, a nomogram can not only visually display the relevant indicators that affect the outcome in multifactor regression analyses but also predict the survival probability through a simple graphical representation, making the prediction simpler and more convenient. We selected the risk factors determining the prognosis of MBC patients through univariate and subsequent multivariate Cox regression analyses. The nomogram visualizesthe infuence of identifed risk factors and enables the survival prediction, with the multivariate regression analysis being the core of this model. The results of the internal verifcation show that when predicting the overall survival, the model has high degrees of discrimination and calibrated accuracy. Since the model can predict the risk of death well and is highly consistent with actual incidence data, it has a certain value for clinical applications.
As a retrospective study based on a single database, this study has several limitations. Firstly, the SEER database covers about 30% of the general USA population and results may not be applicable to the Chinese population. Secondly, some clinicopathological data such as smoking history, comorbidities, specific mixed pathological components, chemotherapy regimens, family history, menstrual history, fertility, genomic status, and concurrent diseases, were not included in the SEER database at the time of initial diagnosis. Furthermore, data on targeted treatment, chemotherapy, and endocrine therapy—all of which have a significant influence on the prognosis of patients with HER-2-positive breast cancer—were not published in the SEER database. Thirdly, we are aware that the application of endocrine and targeted therapy in BC significantly improves survival outcomes. Unfortunately, we were unable to assess the impact of targeted and endocrine treatment on MBC prognosis, due to a lack of treatment records. Could immunotherapy be beneficial for these patients? Perhaps further research is required to address this query. Lastly, this study’s prediction model has only been verified by internal data; more prospective studies or randomized controlled trials are required to substantiate its use.
Conclusion
MBC is an uncommon subtype of breast cancer. We examined possible prognostic predictors for patients with MBC; lung metastasis, age, marital status, grade, surgery, and AJCC-T, AJCC-N, and AJCC-M stages all correlated with BCSS. Meanwhile, we created a distinct nomogram, capable of precisely predicting the OS and BCSS of MBC patients. The constructed nomogram provided quantitative evidentiary support for survival prediction in different risk subgroups, which would help clinicians with reasonable risk management and selection of long-term survival strategies for patients. If our nomogram is used to direct clinical treatment, it may improve patient survival due to its strong predictive value. In addition, large-scale, prospective studies are necessary in the future to confirm our findings.
Footnotes
Acknowledgements
We thank Surveillance, Epidemiology and End Results (SEER) database supported by the Surveillance Research Program in National Cancer Institute’s Division of Cancer Control and Population Sciences. We thank the patients, their families, and the investigators who participated in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval Statement
All patient data in the NCI SEER is anonymized, and the study received a waiver for consent from the ethics committee due to its retrospective nature and guaranteed patient privacy.
