Abstract
Purpose
To evaluate the clinical course and long-term outcomes of patients with ipsilateral breast tumor recurrence (IBTR) after breast-conserving surgery (BCS) and identify independent prognostic factors for further recurrence.
Methods
In this retrospective study, we reviewed the records of 327 patients who experienced IBTR after undergoing BCS for breast cancer at Asan Medical Center during 1990–2013. Overall survival (OS) after IBTR and cumulative incidence rates of recurrences after IBTR were calculated. The association of clinicopathological factors with survival and the development of further recurrence after IBTR was determined in multivariate analysis.
Results
At a median follow-up of 127.7 months, 97 patients experienced recurrence after IBTR. The 5-year and 10-year cumulative incidence rates of recurrence after IBTR were 32% and 41%, respectively. The 5-year and 10-year OS rates after IBTR were 86.6% and 70.3%, respectively. In multivariate analysis, hormone receptor negativity was associated with decreases in OS after IBTR (hazard ratio [HR] 2.83, 95% confidence interval [CI] 1.18–6.78). Patients with longer disease-free interval (DFI) had decreased risks of second recurrence (HR .99, 95% CI .99–1.00), and second locoregional recurrence (LRR) (HR .98, 95% CI .97–.99). Lymphovascular invasion (LVI) of IBTR was associated with increased recurrence rates (second recurrence-free survival, HR 3.58, 95% CI 2.16–5.94; second LRR free survival, HR 5.21, 95% CI 2.77–9.78; second distant metastasis-free survival, 2.11, 95% CI 1.04–4.30) and lower survival rates (OS after IBTR, HR 4.64, 95% CI 2.23–9.67).
Conclusions
Despite subsequent recurrences during long-term follow-up, the survival rates after IBTR remained high. Patients with hormone receptor-negative tumors, shorter DFI, and tumors that present LVI of IBTR had higher risks for recurrence and poor survival rates after IBTR. The study findings may help in understanding the course and prognosis of IBTR patients and identifying high-risk IBTR to establish management strategies.
Introduction
Breast cancer is the most commonly diagnosed cancer in women worldwide. 1 Breast-conserving surgery (BCS) has been established as the standard surgical treatment for patients with early-stage breast cancer. Randomized clinical trials conducted in the 1980s showed that the long-term survival outcomes of BCS and radiotherapy (RT) are similar to that of mastectomy.2,3 With the advent of screening, there has been an increase in the number of patients eligible for BCS because most newly diagnosed patients have relatively small tumors. 4 The rate of BCS has therefore increased during the past years, and ∼60% of contemporary patients with early-stage breast cancer receive BCS.5,6 Moreover, there has been a decrease in breast cancer-related death and local recurrence rate after BCS. 7
Despite the advances in early-stage breast cancer surgery and treatment, approximately 20% of patients experience recurrence 8 and the widespread prevalence of BCS inevitably harbors the risk of ipsilateral breast tumor recurrence (IBTR). During a 10-year follow-up, IBTR was noted to occur in approximately 5%–10% of patients undergoing BCS and receiving RT.2,3,9 Despite the increase in the survival rate of patients with recurrent breast cancer over time, 10 several studies have shown that the occurrence of IBTR is associated with an increase in the risk of systemic metastasis and a decrease in the survival rate of patients.9,11-13 Risk factors for secondary local relapse have also been identified,14-16 but the details of the clinical course of patients after IBTR remain unclear.
Patients who experienced IBTR have different tumor characteristics and treatment histories, which significantly affect their clinical courses. Therefore, the optimal management of IBTR remains controversial. In the absence of standardized treatment guidelines, clinicians should strive to predict the prognosis of patients with IBTR based on retrospective clinical data and to estimate the risk for further recurrence. Fear of recurrence significantly affects the quality of life of long-term breast cancer survivors.17,18 Therefore, there is an increasing need to determine the outcomes after IBTR and identify the patient populations that are at high risk for a second event. Therefore, it would be clinically useful to know the long-term progress of patients who experience IBTR.
In the current study, we analyzed the data of 327 patients with IBTR who underwent BCS for breast cancer at our institution between 1990 and 2013. We aimed to delineate the patterns of disease relapse and long-term outcomes after IBTR and identify the clinicopathological factors that could affect subsequent recurrence and survival after IBTR.
Materials and Methods
Patients
Between January 1990 and December 2013, a total of 10 622 patients with stage 0-III breast cancer underwent BCS at Asan Medical Center. Of these patients, 449 (4.2%) developed IBTR. Patients who had IBTR with synchronous other types of recurrences (locoregional recurrence [LRR] and distant metastasis [DM]) (n = 106) were excluded. We also excluded patients treated with neoadjuvant chemotherapy (n = 16). Therefore, 327 patients were included in the final analysis. Patients who had contralateral breast cancer (n = 8) were excluded from survival analysis.
We retrospectively reviewed clinicopathological data including age, tumor size, lymph node status, histologic grade, nuclear grade, presence of lymphovascular invasion (LVI), and immunohistochemical (IHC) status of estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2). IBTR was defined as a recurrent carcinoma in situ or invasive carcinoma that occurred after BCS in the ipsilateral breast. No distinction was made between true recurrence and new primary cancer, which are the two types of IBTR. ER or PR positivity was defined as nuclear staining ≥1% or an Allred score of 3–8 based on the results of IHC staining. Hormone receptor positivity was defined by either ER or PR positivity, while hormone receptor negativity was defined by both ER and PR negativity. For HER2, positivity was defined as 3+ on IHC staining or HER2 gene amplification by fluorescence in situ hybridization (FISH). Because FISH was not used at our institution in the early study period, HER2 grade 2+ (equivocal) without FISH results was defined as an unknown HER2 status. The surgical margin was regarded as positive when an invasive or noninvasive tumor was present at the resection margin. Pathological staging was determined according to the American Joint Committee on Cancer Staging Manual, 7th edition. Each patient received different modes of treatment, including combined treatment of surgery and adjuvant therapy such as chemotherapy, RT, and endocrine therapy. This study was reviewed and approved by the Institutional Review Board of Asan Medical Center (2017-1341). Informed consent was waived because the study was based on retrospective clinical data.
Endpoints
The study endpoints were second recurrence-free survival (RFS), second IBTR-free survival (IBTRFS), second locoregional recurrence-free survival (LRRFS), second distant metastasis-free survival (DMFS), and overall survival (OS) after recurrence. LRR was defined as recurrence in the chest wall, skin, or regional lymph node areas, including the axillary, supraclavicular, or internal mammary areas. Any other site of recurrence was considered as DM. Second RFS was defined as the date of the first IBTR to any relapse. Second IBTRFS was calculated only for patients who underwent repeat lumpectomy (n = 148) and was defined as the date of the first IBTR to the second IBTR. Second LRRFS was defined as the date of the first IBTR to LRR. Second DMFS was defined as the date of the first IBTR to DM. OS after IBTR was defined as the period from IBTR to death from any cause. Disease-free interval (DFI) was defined as the time from the initial surgery to the first IBTR. Only the patients with second IBTR and LRR as the second recurrence were considered to be at risk for subsequent LRR or DM. Because DM is considered non-curable and no additional IBTR or LRR has consequences, subsequent recurrences after DM were not considered. For patients with synchronous recurrence, more extensive recurrence was considered as the type of recurrence.
Statistical Analyses
Continuous variables are presented as mean ± standard deviation. Survival curves were calculated using the Kaplan–Meier method and the significance of survival differences was verified using the log-rank test. Multivariate Cox regression analysis was performed to estimate the hazard ratios (HRs) and to identify independent prognostic factors. Multivariate analysis adjusted for age at initial operation, initial tumor size, nodal status, hormone receptor status, HER2 status, tumor grade, LVI, age at IBTR, DFI, invasive tumor size of IBTR, adjuvant therapy after IBTR. All reported P values were two-sided, and P values <.05 were considered statistically significant. All statistical analyses were performed using IBM SPSS Statistics for Windows, version 25.0 (Armonk, NY: IBM Corp).
Results
Initial Characteristics of the Study Patients
Initial characteristics of patients who developed ipsilateral breast tumor recurrence after breast-conserving surgery (n = 327).

Data are shown as number (%) unless specified otherwise.
Abbreviations: IBTR, ipsilateral breast tumor recurrence; SD, standard deviation; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; CMF, cyclophosphamide, methotrexate, and fluorouracil; AC, anthracycline and cyclophosphamide.
Patient Characteristics Related to Ipsilateral Breast Tumor Recurrence
Ipsilateral breast tumor recurrence characteristics of patients who developed IBTR after breast-conserving surgery (n = 327).

Data are shown as number (%) unless specified otherwise.
Abbreviations: IBTR, ipsilateral breast tumor recurrence; SD, standard deviation; DFI, disease-free interval; HER2, human epidermal growth factor receptor 2; CMF, cyclophosphamide, methotrexate, and fluorouracil; AC, anthracycline and cyclophosphamide; TC, taxane and cyclophosphamide.
Clinical Course of Ipsilateral Breast Tumor Recurrence
The median duration of follow-up from the time of initial diagnosis was 127.7 months (range, 6.0–320.9), and the median duration of follow-up from the time of diagnosis of IBTR was 55.9 months (range, .1–233.3). The 5-year and 10-year rates of OS after IBTR were 86.6% and 70.3%, respectively. The 5-year and 10-year rates of second RFS were 67.3% and 59.6%, respectively (Supplementary Figure 1).
Of the 319 patients included in the survival analysis, 222 did not show recurrence since the IBTR. In contrast, 97 (30.4%) patients experienced recurrence events after IBTR; 30 patients had second IBTR, 41 had LRR, and 26 had DM. Of the 30 patients who had a second IBTR, 20 had no events since the second IBTR, 7 had developed LRR, and 3 had developed DM. Of the 41 patients who had LRRs as a second event, 26 had no further relapse and 15 had developed DM (Figure 1). Clinical course of ipsilateral breast tumor recurrence. NED, no evidence of disease; LRR, locoregional recurrence.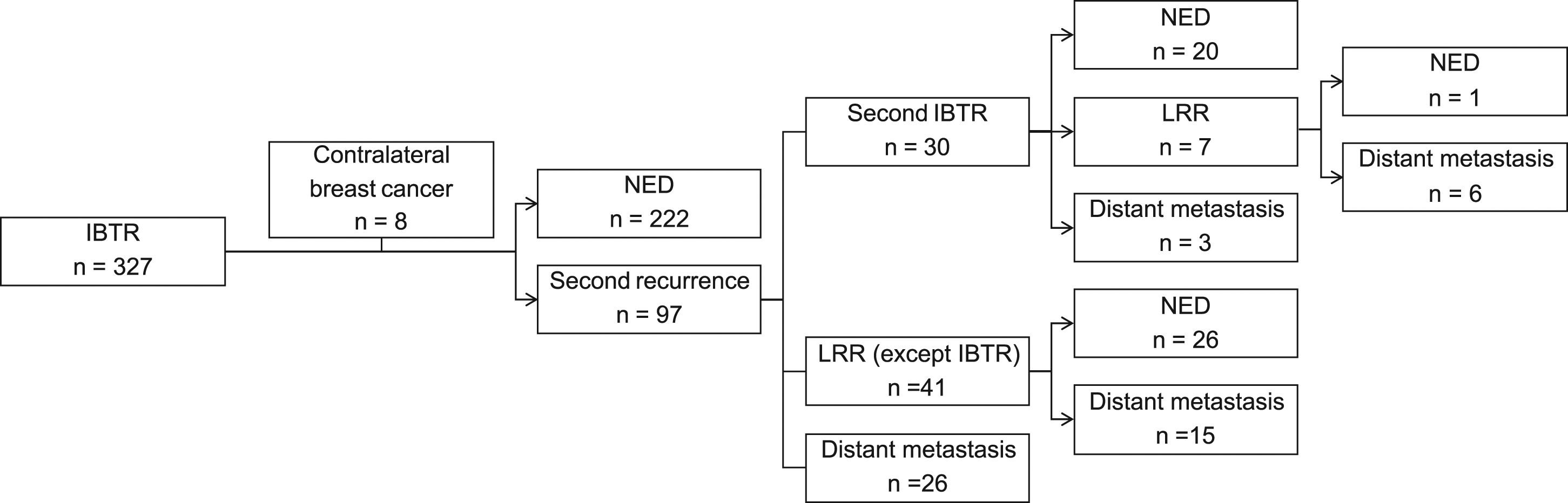
Cumulative incidence rates of recurrence, locoregional recurrence, and distant metastasis after ipsilateral breast tumor recurrence.

Abbreviation: IBTR, ipsilateral breast tumor recurrence.
Prognostic Factors in Predicting Second Recurrence After Ipsilateral Breast Tumor Recurrence
Multivariate Cox regression analysis for second RFS, second LRRFS, second DMFS, and OS after IBTR.

Abbreviations: RFS, recurrence-free survival; LRRFS, locoregional recurrence-free survival; DMFS, distant metastasis-free survival; OS, overall survival; IBTR, ipsilateral breast tumor recurrence; HR, hazard ratio; CI, confidence interval; HER2, human epidermal growth factor receptor 2; LVI, lymphovascular invasion; DFI, disease-free interval.
aSignificant value.

Kaplan–Meier survival analysis according to lymphovascular invasion of ipsilateral breast tumor recurrence for (A) second recurrence-free survival, (B) second locoregional recurrence-free survival, (C) second distant metastasis-free survival, and (D) overall survival after ipsilateral breast tumor recurrence.

Kaplan–Meier survival analysis according to initial hormone receptor status for (A) second recurrence-free survival, (B) second locoregional recurrence-free survival, (C) second distant metastasis-free survival, and (D) overall survival (OS) after ipsilateral breast tumor recurrence.

Kaplan–Meier survival analysis according to the disease-free interval for (A) second recurrence-free survival, (B) second locoregional recurrence-free survival, (C) second distant metastasis-free survival, and (D) overall survival after ipsilateral breast tumor recurrence.
Multivariate Cox regression analysis for second IBTRFS.

Abbreviations: IBTRFS, ipsilateral breast tumor recurrence-free survival; HR, hazard ratio; CI, confidence interval; HER2, human epidermal growth factor receptor 2; LVI, lymphovascular invasion; IBTR, ipsilateral breast tumor recurrence; DFI, disease-free interval.
aSignificant value.
Discussion
Survival rates after breast cancer diagnosis have improved dramatically over the past 20 years owing to advances in early diagnostic tools and treatments.19-21 Because the survival durations of breast cancer patients have been extended and the local control and survival rate are relatively high even after IBTR, it is important to understand the post-IBTR course and the need for longer follow-up have emerged.
In this study, we analyzed 327 patients with IBTR, of whom 97 experienced subsequent recurrence events after IBTR at a median follow-up of 127.7 months. The cumulative incidence rate of 10 years of secondary recurrence after IBTR was 41%. Of the 97 patients who had a second recurrence, 25 had a third recurrence event. Patients with IBTR had a median follow-up duration of 55.9 months after IBTR, and their 5-year and 10-year OS rates after IBTR were 86.6% and 70.3%, respectively. These data show favorable survival outcomes of IBTR with long-term follow-up. In multivariate analysis, hormone receptor status showed significant differences in survival. DFI was associated with the risks of second recurrence and second LRR. Moreover, LVI of IBTR was an independent prognostic factor for recurrence and survival.
Several studies have identified factors that can predict the long-term prognosis of patients who develop IBTR after BCS. Shen et al 22 analyzed 120 patients with IBTR in whom clinicopathological factors were assessed by univariate and multivariate analyses for their association with disease-specific survival (DSS). The DSS was significantly associated with time to IBTR ≤48 months on univariate analysis. LVI positivity in the recurrent tumor was the single independent predictor of DSS on multivariate analysis (relative risk 4.6, 95% CI 1.5–14.1), which is consistent with the findings of our study. Our study identified that the LVI of IBTR is a strong predictor of all types of relapse and poor prognosis. Panet-Raymond et al 23 reported that LVI-positive status in the recurrent tumors was a significant independent predictor of diminished OS (HR 2.46, P < .001). However, Ishitobi et al 24 analyzed 65 patients who underwent repeat lumpectomy without RT after IBTR and reported that the LVI of IBTR has no significant association with the risk of second IBTR; however, this study had 65 fewer cases and the patient group selection was different. LVI has been frequently identified as a prognostic factor for initial breast cancer. In our study, unlike the LVI of IBTR, initial LVI was not a significant factor for relapse and survival after IBTR. This result is not conclusive because there were many missing values of the initial LVI that were not reported in the early period of the current study. Further research related to this finding is needed.
Komoike et al 25 analyzed 172 patients who developed IBTR and 51 patients who developed subsequent DM after IBTR. They reported that the initial nodal status and interval to IBTR were independent risk factors for DM. Several studies have reported that DFI was correlated with subsequent distant disease and OS in patients with local recurrence.23,26-28 Our results showed that shorter DFI was associated with higher risks of further recurrence after IBTR. Additionally, in multivariate analysis, DFI was not associated with OS after IBTR, but was associated OS (HR .97, 95% CI .96–.98) (data not shown). It is notable that DFI itself as a continuous variable was an independent risk factor for recurrence after IBTR regardless of the DFI cutoff value. Our findings suggest that the risk of developing subsequent recurrence may be predicted by DFI at the time of diagnosis of IBTR.
Hormone receptor status is a well-known prognostic factor for the survival of breast cancer patients. In our study, survival after IBTR were better in patients with hormone receptor-positive tumors. These results are consistent with those of previous studies on survival after recurrence.29-31 In our multivariate analysis, hormone receptor status did not significantly affect further recurrence after IBTR. In patients with hormone receptor-positive tumors, recurrence is known to steadily occur for up to 15 years after completing a 5-year endocrine therapy. 32 In recent guidelines, extended endocrine therapies beyond 5 years are recommended considering the individual recurrence risk in patients with hormone receptor-positive breast cancer. 33 Among the 178 patients who received endocrine therapy in our study cohort, 72 patients discontinued initial endocrine therapy before 5 years due to recurrence, and 33 patients discontinued initial endocrine therapy for reasons other than recurrence (eg, self-discontinuation, side effect). No patients received extended endocrine therapy beyond 5 years during the study period. Considering the effects of long-term endocrine therapy on hormone receptor-positive tumors and the potential for late recurrence, patients with hormone receptor-positive tumors may require a longer follow-up duration to identify the factors associated with further recurrence after IBTR.
Given the heterogeneous nature of IBTR, several treatment options are available. The CALOR trial studied the role of chemotherapy in patients with isolated local recurrent breast cancer and reported improvements in the 5-year disease-free survival rate compared to that with no chemotherapy (69% vs 57%). 34 Similarly, in our study, chemotherapy after IBTR was associated with better second IBTRFS (HR .24, 95% CI .06–.98). In addition, endocrine therapy after IBTR was also associated with better second IBTRFS (HR .27, 95% CI .10–.72) and second LRRFS (HR .39, 95% CI .17–.89). These results are similar to those of a randomized trial that compared tamoxifen to controls for the treatment of isolated LRR after mastectomy. 35 In that study, the 5-year disease-free survival rate was 61% in the tamoxifen arm and 40% in the control arm. This difference was primarily due to the reduction in the occurrence of further local relapses (P = .01). However, no significant differences in OS were reported.
Re-irradiation is still controversial as there are limited data on its therapeutic potential and toxicity. In our study, 24 (7.4%) patients received re-irradiation after IBTR. The decision for re-irradiation was made by a multidisciplinary team for the following reasons: long DFI, positive resection margin of the IBTR, and not completing the previous RT. Although several studies on recurrent breast cancer re-irradiation have reported promising results,36-38 more data are needed to determine the therapeutic benefits of re-irradiation for improving local control and survival in IBTR patients.
Our results also show that factors such as young age, large initial tumors, and initial lymph node metastasis were prognostic factors for recurrence after IBTR. These factors are widely known predictors for IBTR and other LRRs.9,13 However, there is limited information regarding the effects of recurrence after IBTR. In our study, young age (≤35 years) was correlated with a short second LRRFS (HR 4.44, 95% CI 1.43–13.82). Larger initial tumor size (>2 cm) was correlated with a short second RFS (HR 2.31, 95% CI 1.47–3.65) and second LRRFS (HR 2.17, 95% CI 1.15–4.09). Positive lymph nodes were correlated with a short second IBTRFS (HR 2.89, 95% CI 1.11–7.56) and second LRRFS (HR 2.05, 95% CI 1.05–3.99). There were no significant differences in DM or survival with respect to any of the above factors. Previous studies have reported different results for further relapse after recurrence for these factors.25,26,28,39 In the absence of definitive predictors, risk stratification for further recurrence and survival after IBTR should consider several of the previously mentioned factors.
Of the total patients, 46.2% underwent repeat lumpectomy, which is not the current standard surgery for IBTR. We analyzed the cumulative incidence of second IBTR and second IBTRFS in patients who underwent repeat lumpectomy. The clinicopathological features of second IBTRs are presented in Supplementary Table 1, and the 5 year and 10-year cumulative incidence rates of second IBTRs were 12% and 17%, respectively (Supplementary Table 2). We have previously reported that long-term survival outcomes of repeat lumpectomy and salvage mastectomy were similar after propensity score matching. Our institution performed repeated lumpectomy mainly in patients with initially low T stage, hormone response, HER2-negative tumors at initial diagnosis, and small-sized recurrent tumors. 40
Because of the low frequency of isolated IBTR, most previous studies were small-sized. Our study followed up a considerable number of IBTR patients for a long period. Previous studies have mainly focused on the relationship between IBTR and second IBTR or IBTR and DM. In our study, we have subdivided the second recurrence events into second IBTR, LRR, and DM and provided detailed information on the entire clinical course of these patients.
The present study had some limitations. First, tumors with equivocal IHC HER2 expression patterns were not reexamined for gene amplification by FISH in the early study period. The cases graded as 2+ (equivocal) were classified as HER2 unknown status. In addition, because Korea’s health insurance has covered targeted therapy for patients with HER2-positive tumors since 2009, information on initial targeted therapy is lacking in this study. Second, this study included patients who did not receive RT, the standard treatment for early-stage breast cancer, after their first surgery for reasons such as old age, underlying comorbidity, refusal of treatment, skin lesions, and pregnancy. Some of the previous studies also included patients who had not received RT,22,23 and patients who do not receive standard treatment are also part of the current clinical practice. Third, this study had a retrospective single-institution study design. However, prospective studies or randomized trials are difficult to implement because of the heterogeneity of prognostic factors and the patients’ history of previous treatments.
In the absence of large prospective randomized studies, we believe that our study still has value as it highlights the course and prognosis of the long-term outcomes of IBTR patients. Identification of the factors affecting the prognosis may be helpful in stratifying high-risk IBTR patients, and clinical trials on the appropriate treatment for these patients should be conducted in the future.
Conclusion
In conclusion, our study delineated the clinical course and long-term outcome of patients with IBTR after BCS and identified the prognostic factors of recurrence after IBTR. IBTR patients had a favorable prognosis at long-term follow-up with a 10-year survival rate after IBTR of 70.3%; however, patients with hormone receptor-negative tumors, short DFIs, and tumors with LVI of IBTR had a higher risk of further recurrence and poorer survival. Treatment of IBTR patients is a complex and challenging task in modern breast cancer treatment. We expect that the results of this study would be helpful in understanding the course and prognosis of IBTR patients and establishing a management strategy.
Abbreviations
AC, anthracycline and cyclophosphamide
BCS, breast-conserving surgery
CI, confidence interval
CMF, cyclophosphamide, methotrexate, and fluorouracil
DFI, disease-free interval
DM, distant metastasis
DMFS, distant metastasis-free survival
DSS, disease-specific survival
ER, estrogen receptor
FISH, fluorescence in situ hybridization
HER2, human epidermal growth factor receptor 2
HR, hazard ratio
IBTR, ipsilateral breast tumor recurrence
IBTRFS, ipsilateral breast tumor recurrence-free survival
IHC, immunohistochemical
LRR, locoregional recurrence
LRRFS, locoregional recurrence-free survival
LVI, lymphovascular invasion
OS, overall survival
PR, progesterone receptor
RFS, recurrence-free survival
RT, radiotherapy
SD, standard deviation
TC, taxane and cyclophosphamide
Supplemental Material
Supplemental Material - Clinical Course and Predictors of Subsequent Recurrence and Survival of Patients With Ipsilateral Breast Tumor Recurrence
Supplemental Material for Clinical Course and Predictors of Subsequent Recurrence and Survival of Patients With Ipsilateral Breast Tumor Recurrence by Soo Yeon Baek, MD, Jisun Kim, MD, PhD, Il Yong Chung MD, PhD, Beom Seok Ko, MD, PhD, Hee Jeong Kim, MD, PhD, Jong Won Lee, MD, PhD, Byung Ho Son, MD, PhD, Sei-Hyun Ahn, MD, PhD, and Sae Byul Lee, MD, PhD in Cancer Control.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grant (Elimination of Cancer Project Fund) from the Asan Cancer Institute of Asan Medical Center in Seoul, Korea.
Ethical Approval
This study was conducted in accordance with the Declaration of Helsinki. This study was reviewed and approved by the Institutional Review Board of Asan Medical Center (2017-1341).
Statement of Informed Consent
Informed consent was waived because the study was based on retrospective clinical data.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Institutional Review Board of Asan Medical Center (2017-1341) approved protocols.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
