Abstract
Background
Local advanced rectal cancer (LARC) patients who achieved pathological complete response (pCR) after neoadjuvant chemoradiotherapy (NCRT) generally have a favorable prognosis. This retrospective study aimed to evaluate the prognostic value of magnetic resonance imaging (MRI) parameters and neutrophil-to-lymphocyte ratio (NLR) in LARC patients with pCR.
Methods
Between 2015 and 2019, 180 LARC patients who achieved pCR after NCRT and surgery were included. MRI parameters and NLR were evaluated as potential predictors for 5-year overall survival (OS) and disease-free survival (DFS) using the Kaplan–Meier and COX regression analysis.
Results
With a median follow-up time of 68.3 months, the 5-year OS and DFS rates were 94.2% and 91.4%, respectively. Thirteen patients (7.2%) died, 2 (1.1%) experienced local recurrence, and 15 (8.3%) experienced distant metastases. Pretreatment MRI parameters and NLR were correlated with 5-year OS and DFS in pCR patients in the univariate analysis. The multivariate analysis identified baseline EMVI and NLR as independent predictors for 5-year OS and DFS (all P < .05). Patients in the low-risk group (EMVI-negative and/or NLR ≤ 2.8, n = 159, 88.3%) had a more favorable 5-year DFS compared to those in the high-risk group (EMVI-positive and NLR > 2.8, n = 21, 11.7%) (95.6% vs 59.4%, P < .001), with similar findings for 5-year OS (97.4% vs 70.6%, P < .001).
Conclusions
This study showed that MRI parameters and NLR were associated with long-term prognosis in patients with pCR. These findings could aid in stratifying pCR patients and guide subsequent treatment and follow-up strategies.
Keywords
Introduction
Colorectal cancer is the third most common cancer and the second leading cause of cancer-related deaths worldwide. 1 Neoadjuvant chemoradiotherapy (NCRT) followed by total mesorectal excision (TME) is the standard treatment for locally advanced rectal cancer (LARC). 2 After NCRT, tumor cells exhibit varied responses to therapy, and the degree of response is closely related to prognosis, as demonstrated by several studies.3,4 Pathological complete response (pCR), the optimal treatment response, is characterized as the absence of residual tumor cells in the primary tumor and lymph nodes. Approximately 20% of patients achieved pCR following NCRT and surgery.3,4 A pooled analysis of 3105 LARC patients indicated that the 5-year disease-free survival (DFS) was 83.3% in the pCR group and 65.6% in the non-pCR group (P < .001). 3 Thus, pCR is frequently used as the primary endpoint in current clinical research of LARC. 5
Despite the association of pCR with favorable long-term prognosis, recurrence still occurs in some patients. Limited studies have investigated the prognostic factors of pCR patients. Furthermore, the benefits of adjuvant chemotherapy in pCR patients remain contentious. 6 In pCR patients, therapeutic and follow-up strategies may differ based on distinct prognostic factors. Therefore, identifying pCR patients at high risk of recurrence is crucial for optimizing clinical management.
Magnetic resonance imaging (MRI) offers unique advantages by providing accurate images of soft tissue structures in the mesorectum, establishing itself as the standard for clinical staging of LARC. 7 The current American Joint Committee on Cancer (AJCC) staging system for LARC encompasses a heterogeneous group of tumors in stages II-III, and the prognosis of LARC correlates significantly with parameters such as the depth of extramural invasion, extramural venous invasion (EMVI) and involvement of the mesorectal fascia (MRF). 8 These parameters have demonstrated prognostic relevance in LARC patients after NCRT, 8 guiding risk-adapted treatment strategies as recommended by the European Society for Medical Oncology (ESMO). 7 Previous data indicated that MRI-defined low-risk LARC patients exhibited a pCR rate exceeding 30% following NCRT, contrasting with lower rates observed in those categorized as MRI-defined high-risk LARC.9,10 However, few studies have focused on the prognostic value of baseline MRI parameters in LARC patients with pCR.
Cellular inflammation components, known for their dual roles in the promotion or containment of tumors, have been proven to correlate with prognosis in various solid malignancies, including rectal cancer. 11 The neutrophil-to-lymphocyte ratio (NLR) is a widely utilized and economical biomarker for predicting tumor prognosis 11 A meta-analysis reported that NLR was an independent clinical predictor of overall survival (OS) and DFS in patients with rectal cancer undergoing curative surgery. 12 Moreover, elevated baseline NLR was associated with poor prognosis in LARC patients treated with NCRT. 13 Nevertheless, the prognostic effect of NLR on LARC patients with a pCR status after NCRT remains unclear.
Therefore, we conducted a study to evaluate the prognostic value of MRI parameters and NLR in patients who achieve pCR after NCRT and surgery.
Materials and Methods
Patients Selection
Retrospectively, all patients diagnosed with histologically confirmed rectal adenocarcinoma who received NCRT at our center from January 2015 to November 2019 were eligible for inclusion in this study. The inclusion criteria were as follows: (1) patients who underwent pelvic MRI before NCRT, (2) clinical stage was cT3-4 or lymph node-positive, (3) lower edge of the tumor no more than 10 cm from the anal edge, (4) received NCRT in our center, (5) pathologically diagnosed ypT0N0 after surgery, (6) older than or equal to 18 years old, and (7) had an Eastern Cooperative Oncology Group score of less than 2. The exclusion criteria were as follows: (1) the presence of a second primary cancer that may affect the prognosis, and (2) loss to follow-up after surgery. Additionally, eight patients with ypT0 who underwent local excision to preserve anal function and remained regional recurrence-free within 2 years after surgery were also included in this analysis. Venous blood samples of patients were collected at baseline.
This study was approved by the ethics committee of Peking University Cancer Hospital (2025YJZ28), and the requirement for obtaining informed patient consent was waived. The reporting of this study conforms to the STROBE guidelines. 14 All patient details have been de-identified.
MRI Assessment
A pelvic diagnostic or simulation high-resolution MRI in the supine position before NCRT was necessary, requiring a three-dimensional T2-weighted sequence, especially in axial scanning. The slice thickness was 3-5 mm. Axial scanning should be angulated perpendicular to the rectal tumor axis. 15 The cT stage, T3 substage, cN stage, EMVI, MRF status, and tumor length and thickness were evaluated based on the ESMO and European Society of Gastrointestinal and Abdominal Radiology consensus meeting guidelines.7,15 The distance from the lower edge of the tumor to the anus less than 5 cm is defined as “low”, and 5-10 cm is defined as “middle”.
Evaluation of tumor regression using MRI is recommended 5-12 weeks after NCRT before surgery.
Neoadjuvant Treatment
Patients underwent computed tomography (CT) and MRI simulation using contrast-enhanced CT with a 5 mm slice thickness in the supine position, secured with a thermoplastic film. A full bladder and an empty rectum were maintained to ensure consistency in the positioning of the rectal tumor and to protect the intestine. Simultaneous integrated boost intensity-modulated radiation therapy (SIB-IMRT) was performed in all patients with prescription doses for the planning gross tumor volume (PGTV) of 50-50.6 Gy and planning target volume (PTV) of 41.8-45 Gy, respectively, in 22-25 fractions. Detailed contouring procedures have been previously described. 16
Patients received capecitabine with or without oxaliplatin as concurrent chemotherapy. With comprehensive informed consent regarding the benefits and risks, induction or consolidation chemotherapy (XELOX regimen) was implemented in some cases.
All patients were informed about the risks and benefits of NCRT and provided written informed consent to treatment.
Surgical and Postoperative Treatment
After NCRT, a multidisciplinary team developed the following treatment strategies. Patients were recommended for surgery, predominantly TME. Local excision was considered in specific cases to preserve organ function, based on mutual decision-making between physicians and patients.
A pCR was defined by the absence of tumor cells in the primary tumor and lymph nodes. The assessment of surgical specimens was conducted independently by two senior pathologists using a standardized protocol. 17
Adjuvant chemotherapy was recommended according to National Comprehensive Cancer Network guidelines. XELOX (oxaliplatin and capecitabine) and FOLFOX (oxaliplatin, leucovorin, and 5-FU) were preferred, while capecitabine monotherapy was an alternative.
Follow-up
After radical surgery, patients were advised to regular follow-up at 3-month intervals for the first 2 years, 6-month intervals for 3 to 5 years, and annually thereafter. Follow-up assessments included symptoms, physical examination, blood tests, and CT examinations of the thorax, abdomen, and pelvis.
Statistical Analysis
Data were collected and analyzed using the Statistical Package for the Social Sciences (IBM Corp. SPSS Statistics for Windows, v.22.0, Armonk, NY, USA). The optimal cut-off values for NLR, tumor length, and thickness were determined using the X-tile software version 3.6.1 (Yale University, New Haven CT, USA). 18 Student’s t-test was used to compare differences in continuous data, and the Chi-square or Fisher’s test for categorical data. OS was defined as the date from radical surgery to death from any cause. DFS was defined as the date from radical surgery to any type of locoregional recurrence, distant metastasis, or death for any reason. OS and DFS analyses were performed using the Kaplan–Meier method, and the results were compared using the log-rank test. The Cox regression model was used for univariate and multivariate analysis. Variables with a P-value of < 0.05 in univariate analyses were included in multivariate analyses.
Results
Patient Characteristics
Characteristics of Patients With Pathological Complete Response (n = 180).
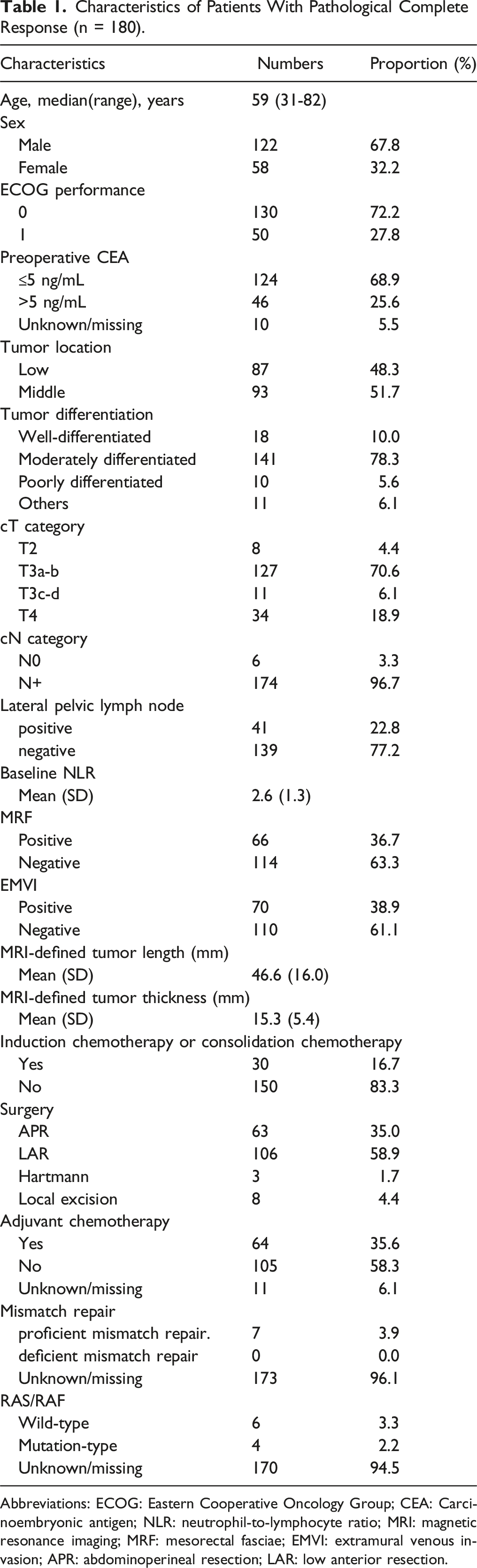
Abbreviations: ECOG: Eastern Cooperative Oncology Group; CEA: Carcinoembryonic antigen; NLR: neutrophil-to-lymphocyte ratio; MRI: magnetic resonance imaging; MRF: mesorectal fasciae; EMVI: extramural venous invasion; APR: abdominoperineal resection; LAR: low anterior resection.
In terms of pretreatment MRI parameters, 34 (18.9%) patients were diagnosed with clinical T4, 174 (96.7%) patients had clinical N+, 66 (36.7%) patients were MRF-positive, and 70 (38.9%) patients were EMVI-positive.
Outcomes in Patients with pCR
The median follow-up time was 68.3 months (range: 21.2-108.4 months). Among 180 patients, the 5-year OS was 94.2% (Figure 1A), the 5-year DFS was 91.4% (Figure 1B), the 5-year local recurrence-free survival was 93.7% (Figure 1C), and the 5-year distant metastasis-free survival (DMFS) was 91.4% (Figure 1D). During the follow-up period, 13 patients (7.2%) died and 15 patients (8.3%) experienced tumor recurrence. Local regional recurrence occurred in 2 patients (1.1%) in the left pelvic wall, both of whom were combined with distant metastases. Among 15 patients (8.3%) who suffered distant metastases, 5 had pulmonary metastases, 2 had liver metastases, 1 had brain metastases, 1 had peritoneal metastases, 1 had retroperitoneal lymph nodes, and 5 had multiple organ metastases (3 of 5 involved the lung, and 2 of 5 involved the liver) when they first diagnosed with distant metastases. Kaplan‒Meier Survival Curves for Overall Survival (A), Disease-free Survival (B), Local Recurrence-free Survival (C), and Distant Metastasis-Free Survival (D) in Patients With Pathological Complete Response.
Association Between MRI Parameter and Survival Outcomes
We analyzed the impact of MRI characteristics on 5-year OS and DFS. The X-Tile software identified the optimal cut-off value of MRI-defined tumor length as 64 mm for 5-year OS and DFS. Patients were classified into the group with tumor length ≤ 64 mm (n = 160, 88.9%) and the group with tumor length > 64 mm (n = 20, 11.1%). The tumor length ≤ 64 mm group had significantly better 5-year OS (95.4% vs 85.0%, P = .001) and DFS (93.0% vs 79.7%, P = .008) compared to the group with tumor length > 64 mm. Similarly, the tumor thickness > 20 mm (n = 20, 11.1%) patients had worse 5-year OS (85.0% vs 95.4%, P = .001) and DFS (79.7% vs 93.0%, P = .007) compared to those with tumor thickness ≤ 20 mm (n = 160, 88.9%).
In addition, patients with MRF-negative status had significantly better outcomes than those with MRF-positive status in terms of 5-year OS (96.3% vs 90.8%, P = .012,) and 5-year DFS (94.6% vs 85.8%, P = .023). EMVI-negative patients also had a better 5-year OS (99.0% vs 87.0%, P < .001) and DFS compared to EMVI-positive patients (96.3% vs 84.0%, P = .002).
The 5-year OS was 94.2% and 94.1% in patients with cT2-3 and cT4 (P = .081), and the corresponding DFS was 92.3% and 87.4% (P = .193). Patients with cN + had a similar 5-year OS (94.0% vs 100.0%, P = .513) and DFS (91.7% vs 83.3%, P = .424) compared to cN0.
Association Between NLR Level with Survival Outcomes
According to the X-Tile software, the optimal cut-off value for NLR level was 2.8 for 5-year OS and 2.8 for 5-year DFS. Therefore, patients were categorized into low-NLR (NLR ≤ 2.8, n = 121, 67.2%) and high-NLR (NLR > 2.8, n = 59, 32.8%) groups.
The 5-year OS rates in the low-NLR and high-NLR groups were 96.6% and 89.3% (P = .016). The 5-year DFS rates in the low-NLR and high-NLR groups were 95.0% and 83.7% (P = .029).
Univariate and Multivariate Analysis for Survival Outcomes
Univariate and Multivariate Analysis for Overall Survival in Patients With Pathological Complete Response.

Abbreviations: HR: hazard ratio; CI: confidence interval; ECOG: Eastern Cooperative Oncology Group; CEA: carcinoembryonic antigen; MRI: magnetic resonance imaging; MRF: mesorectal fasciae; EMVI: extramural venous invasion; APR: abdominoperineal resection; LAR: low anterior resection; NLR: neutrophil-to-lymphocyte ratio. The bold indicated statistical significance, with P < 0.05.
Univariate and Multivariate Analysis for Disease-free Survival in Patients With Pathological Complete Response.

Abbreviations: HR: hazard ratio; CI: confidence interval; ECOG: Eastern Cooperative Oncology Group; CEA: carcinoembryonic antigen; MRI: magnetic resonance imaging; MRF: mesorectal fasciae; EMVI: extramural venous invasion; APR: abdominoperineal resection; LAR: low anterior resection; NLR: neutrophil-to-lymphocyte ratio. The bold indicated statistical significance, with P < 0.05.
Risk Stratification in Patients with pCR
Based on the results of the multivariate analysis, patients were categorized into low-risk (EMVI-negative and/or NLR ≤ 2.8) and high-risk (EMVI-positive and NLR > 2.8). Therefore, patients were divided into a low-risk (n = 159, 88.3%) group and a high-risk (n = 21, 11.7%) group. The patients with pCR in the low-risk group demonstrated significantly better 5-year OS than those in the high-risk group (97.4% vs 70.6%, P < .001; Figure 2A), as well as in 5-year DFS (95.6% vs 59.4%, P < .001; Figure 2B). Kaplan‒Meier Survival Curves for Overall Survival (A) and Disease-Free Survival (B) in the Low-Risk and High-Risk Groups in Patients With Pathological Complete Response.
Discussion
While most previous research has focused on identifying clinical predictors associated with achieving pCR in LRAC patients after NCRT and has demonstrated that pCR predicted a favorable long-term prognosis, few studies have analyzed prognostic factors specifically in patients with pCR. Focusing on the pCR subgroup, our research revealed that pretreatment MRI parameters and baseline NLR were correlated with prognosis in pCR patients after NCRT in the univariate analysis. In the multivariate analysis, high NLR levels (> 2.8) and EMVI-positive status were independent predictors for poorer OS and DFS. pCR patients categorized in the low-risk group (EMVI-negative and/or NLR ≤2.8) exhibited a more favorable 5-year DFS compared to those in the high-risk group (EMVI-positive and NLR > 2.8) (95.6% vs 59.4%, P < .001), with consistent findings for 5-year OS (97.4% vs 70.6%, P < .001). Therefore, these findings underscore the potential utility of pretreatment MRI parameters and NLR in guiding treatment decisions and clinical follow-up strategies for patients who achieve pCR after NCRT.
After NCRT, patients achieving pCR often exhibit excellent oncological outcomes. In a study including 242 patients with pCR after NCRT, Jiang et al. 19 reported the 5-year OS and DMFS were 94.7% and 88.9%, respectively. A pooled analysis of 3105 LARC patients who achieved pCR after NCRT indicated that the 5-year crude DFS was 83.3%, higher than the 65.6% observed in those who did not achieve pCR, which highlights the significant benefit of achieving pCR. 3 Consistent with these findings, our study showed that patients who achieved pCR had an ideal 5-year DFS (91.4%) and OS (94.2%). However, a minority of pCR patients still develop tumor recurrence. Park et al. 4 found that the 5-year recurrence-free survival of pCR patients was 90.5%, with a 5-year distant metastasis rate of 7.0% and no local recurrence occurred within 5 years. These results revealed that the recurrence pattern in pCR patients was predominantly distant metastasis. In a study of 195 LARC patients with pCR, 3 had local recurrence and 15 had distant metastasis (including 7 lung metastases). 20 Our research corroborates these findings, emphasizing the necessity of routine monitoring of distant areas, especially the lungs, in pCR patients after NCRT.
The role of adjuvant chemotherapy in pCR patients after NCRT remains significantly controversial. Some studies have demonstrated that pCR patients can still benefit from adjuvant chemotherapy, with the OS of those who received adjuvant chemotherapy being superior to those who did not.6,21 Conversely, other studies proposed that adjuvant chemotherapy can be safely omitted in pCR patients, as it failed to confirm survival advantages.22,23 Therefore, the identification of reliable prognostic markers for stratifying pCR patients could potentially resolve this ongoing dispute, and serve to guide follow-up strategies. Based on the risk stratification of our findings, low-risk pCR patients may potentially benefit from the omission of chemotherapy to avoid treatment toxicity, and a reduction in the costs of follow-up examinations. In contrast, high-risk patients may still require adjuvant chemotherapy or at least more frequent follow-up. Furthermore, our study found pCR patients with only EMVI-positive or high NLR still exhibited a favorable prognosis. An interesting perspective emerging from our findings is that a single risk factor may be insufficient to accurately discriminate the prognosis of pCR patients and the presence of multiple adverse characteristics may lead to truly poor prognosis, which need large sample size study to prove. These findings not only enhance our comprehension of pCR patient management but also provide valuable insights for the direction of future research.
To the best of our knowledge, few studies have investigated the relationship between MRI parameters and long-term prognosis in LARC patients achieving pCR. MRI plays a crucial role in assessing tumors, and its parameters are associated with prognosis in LARC. Among MRI parameters, EMVI positivity has been associated with increased rates of distant metastases and poor treatment sensitivity. A meta-analysis including 2237 patients from 11 studies indicated that baseline EMVI positivity was associated with worse OS (P < .001). 24 Zhang et al. 25 analyzing 517 LARC patients undergoing NCRT, similarly identified EMVI as a predictor of metastasis-free survival (P < .010) and OS (P = .010). Our study demonstrated that even in patients with pCR, the 5-year DFS and OS of EMVI-positive patients were significantly lower than those of EMVI-negative.
In our study, MRF status, tumor length and thickness also demonstrated correlations with long-term prognosis in pCR patients in the univariate analysis. Previous studies had similar findings. The MERCURY study revealed that the 5-year OS was 62.2% in patients with MRF-positive compared with 42.2% in patients with MRI-negative (P < .01), and the 5-year DFS was 67.2% vs 47.3% (P < .05) in LARC patients. 26 As for tumor length, a sub-analysis of the TRIGGER feasibility study indicated a median tumor length of 43 mm in patients with a good response to NCRT, compared to 50 mm in those with poor response (P < .001). 27 Tumor thickness was a predictor for pCR in LARC patients after NCRT in a study. 28 Although the relationship between these three factors and prognosis was not significant in the multivariate analysis of our study, this is likely due to the limited sample size and number of events. The predictive values of MRF status, tumor length, and thickness in pCR patients warrant further research.
The mechanisms underlying the relationship between NLR and outcomes in LARC patients remain unclear. Tumor progression may be associated with an imbalance between protumor (inflammatory) and antitumor (immune) forces.12,29 Neutrophils are known to release inflammatory cytokines that promote angiogenesis and accelerate tumor progression, 30 and neutrophilia were linked to poor outcomes in LARC patients, as evidenced in the CAO/ARO/AIO-04 trial. 31 Conversely, lymphocytes are crucial for immunity and tumor control, with tumor-infiltrating lymphocytes playing a significant role in the antitumor immune response. 32 A high NLR may indicate an immune microenvironment that promotes tumor progression. A meta-analysis of thirty-one studies reported that pretreatment high-NLR was associated with worse OS and DFS in patients undergoing curative rectal cancer resection. 33 Similarly, a study including 199 LARC patients identified that a cut-off value of NLR ≥ 2.8 was an independent predictor for OS after NCRT. 34 Our results are consistent with previous studies, confirming that NLR level is correlated with long-term prognosis in patients with pCR.
As a retrospective study, this research inevitably has some limitations. First, due to the limitations of the screening criteria, the study population exhibited relative heterogeneity. Additionally, both the sample size of a single-center, and the number of events in the multivariate analysis were limited. Multicenter, prospective, and large-scale trials are needed to further confirm our findings.
Conclusion
In conclusion, pretreatment high NLR (> 2.8) and EMVI-positive status were independent prognostic factors of poor OS and DFS among LARC patients with pCR. The 5-year DFS and OS for pCR patients in the low-risk group (EMVI-negative and/or NLR ≤ 2.8) were better than those in the high-risk group (EMVI-positive and NLR > 2.8). A closer clinical follow-up strategy is recommended for pCR patients, especially routine chest CT examination. High-risk patients with pCR may warrant intensive adjuvant therapy.
Footnotes
Acknowledgments
The authors would like to thank all patients who participated in this study.
Statements and Declarations
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Beijing Hospitals Authority Clinical medicine Development of special funding support (No. ZLRK202327), Beijing Municipal Science &Technology Commission (No. Z181100001718192), Beijing Hospitals Authority’s Ascent Plan (No. DFL20220902), National Natural Science Foundation (No. 82073333), Capital’s Funds for Health Improvement and Research (No. 2020-2-1027), Science Foundation of Peking University Cancer Hospital (No. BJCH2024CZ09), Peking University Medicine Sailing Program for Young Scholars’ Scientific & Technological Innovation (No. BMU2023YFJHPY022), Clinical Research Fund For Distinguished Youth Scholars of Peking University Cancer Hospital (No.QNJJ202419), and Science Foundation of Peking University Cancer Hospital (No. ZY202506).
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets analyzed during the current study are available from the corresponding author upon reasonable request.
