Abstract
Objective
We aimed to retrospectively investigate whether the neutrophil to lymphocyte ratio (NLR) and the monocyte to lymphocyte ratio (MLR) can predict the prognosis of patients with metastatic renal cell carcinoma (mRCC) treated with sunitinib or sorafenib.
Methods
We retrospectively identified 210 patients with mRCC treated with sunitinib or sorafenib from 2007 to 2017 at Fudan University- and Hexi University-affiliated hospitals. Overall survival (OS) and progression-free survival (PFS) were evaluated using the Kaplan–Meier method. Multivariate regression analysis was used to evaluate predictors of PFS and OS.
Results
Low NLR (<2.85) and MLR (<.30) were strongly associated with increased PFS and OS. Multivariable analyses verified that the NLR and MLR were both independent prognostic factors. Additionally, the NLR was negatively correlated with CD8+ and CD4+ T-cell infiltration in tumors.
Conclusion
In patients with mRCC treated with sunitinib and sorafenib, an NLR <2.85 and MLR <.30 was associated with superior PFS and OS, which may be related to the reduced lymphocytic infiltration of tumors.
Keywords
Introduction
In recent years, the treatment of patients with metastatic renal cell carcinoma (mRCC) has improved significantly with the development of targeted therapy and immunotherapy.1-3 However, a precise biomarker for accurate prognosis prediction of patients with mRCC has not been reported. RCC is closely related to host inflammation and immunity as they play important roles in tumor development and progression via promoting the proliferation and survival of cancer cells, angiogenesis, and influencing the response of tumors to systemic therapy.4-6 The inflammatory cells in the tumor microenvironment proliferate and produce inflammatory mediators.
Cancer development and progression are significantly modulated by the interactions between the different immune cell types (neutrophils, macrophages, lymphocytes, etc.). Recent studies have shown that inflammatory cells ratios are associated with the prognoses of patients with solid tumors. Specifically, the neutrophil to lymphocyte ratio (NLR) and monocyte to lymphocyte ratio (MLR) are easy-to-determine, cost-effective, and reliable systemic inflammatory biomarkers that are related to the prognosis of different cancer types.7-9 The NLR has been identified as an independent prognostic variable for gastric, breast, and colon cancers.10-12 It provides clinicians with easy-to-obtain, objective information via inexpensive routine blood examination. Thus, the aim of this study was to assess whether NLR and MLR can predict overall survival (OS) and progression-free survival (PFS) in patients with mRCC treated with first-line tyrosine kinase inhibitors (TKIs).
Materials and Methods
Patient Selection and Evaluation
Data from continuous selection 210 patients with mRCC who were treated with sunitinib or sorafenib at XXXXX (Double Anonymization) Hospital between 2007 and 2017 were retrospectively reviewed. Patients’ data and their individual NLRs and MLRs were collected from electronic medical records. NLR and MLR were calculated based on the latest hematological data of patients before receiving TKI treatment. We de-identified all patient details. All patients provided written informed consent, and the study design was approved by the research ethics committees of the above institutions. All patients were diagnosed with mRCC according to pathology and imaging examinations. The inclusion criteria were: (1) patients underwent radical or partial nephrectomy following confirmed pathological diagnosis of RCC; and (2) patients treated with sunitinib or sorafenib as first-line systemic therapy for metastatic lesions. The exclusion criteria were: (1) history of other malignancies or having previously received anti-cancer therapy; (2) insufficient tissue samples; (3) necrotic tumor area greater than 80%; (4) patients with incomplete data, loss of follow-up, medical history of glomerulonephritis or nephrotic syndrome, hepatic cirrhosis, chronic obstructive pulmonary disease, chronic infection, inflammatory bowel disease, or other malignancies. The pathological characteristics of the patients were evaluated, including histological subtypes, tumor size, TNM staging, and nuclear grading. The histological subtypes were classified according to the World Health Organization 2016 Classification, and the nuclear grading of tumors was performed according to the International Society of Urological Pathology grading system issued in 2013. PFS was defined as the time from the start of TKI treatment to the diagnosis of disease progression or death from any cause. The OS period was defined as the time from the start of TKI treatment to death from any cause. The reporting of this study conforms to the REMARK guidelines. 13
Immunohistochemistry for determining CD8+ T and CD4+ T Cell Infiltration in Tumors
The tissue samples were fixed with formalin, embedded in paraffin, and cut into sections. The sliced sections were dewaxed in xylene and dehydrated in alcohol. After blocking endogenous peroxidase activity with 3% (v/v) hydrogen peroxide and normal goat serum, the sections were incubated with the primary antibody, mouse anti-human CD8 (ab17147, Abcam; concentration 1:400) at 4°C overnight. The next day, the sections were incubated with the secondary antibody, rabbit anti-human CD4 (ab133616, Abcam; concentration 1:200) at 37°C for 20 minutes. Finally, 3,3’-diaminobenzidine was used as chromogen to detect immunoreactive bands and hematoxylin was used for counterstaining were used as described previously. 14 Five independent areas of each slide were examined under the microscope to calculate the total count of CD8+ and CD4+ T tumor-infiltrating lymphocytes, and the patients were stratified using the median value of positively stained cell count.
Statistical Analysis
Using univariate analysis, we analyzed whether clinicopathological factors, NLR, or MLR were independent predictors of OS and PFS in patients with mRCC. Factors with significant correlation were included in the multivariate Cox analysis. The Chi-square test was used to compare categorical variables, Kaplan–Meier method was used for survival analysis, and a log-rank test was used to assess differences between survival curves. All analyses were performed using SPSS (version 21.0; IBM Corporation, Armonk, NY, USA). P values <.05 were considered statistically significant.
Results
Neutrophil to Lymphocyte Ratio and Monocyte to Lymphocyte Ratio Significantly Correlated with Clinical Characteristics of Metastatic Renal Cell Carcinoma Patients
Clinical and Pathologic Characteristics of Patients According to NLR (n = 210).
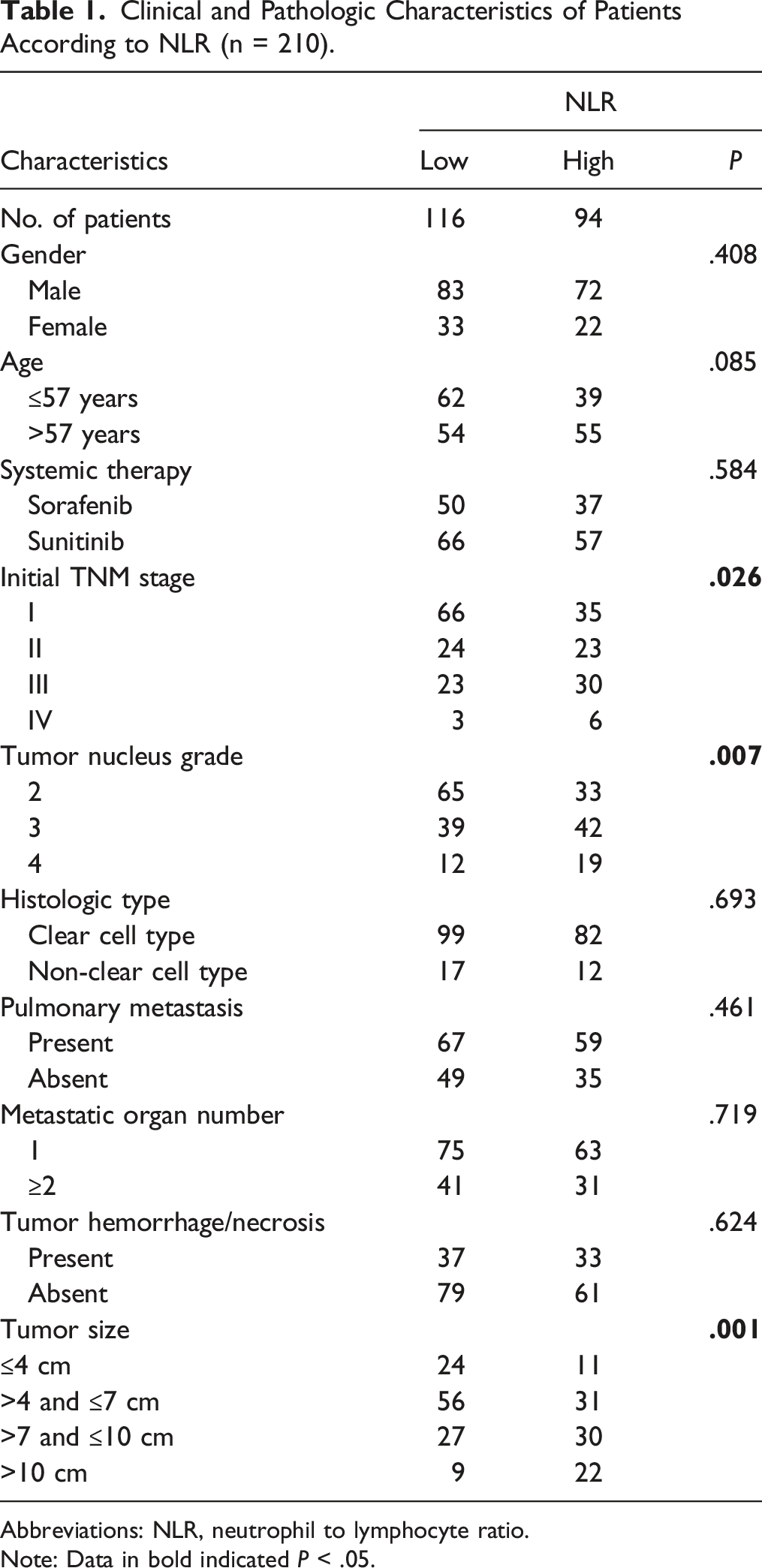
Abbreviations: NLR, neutrophil to lymphocyte ratio.
Note: Data in bold indicated P < .05.
High Neutrophil to Lymphocyte Ratio Correlated with Poor Patient Survival
To investigate whether a high NLR in patients with mRCC correlates with a worse prognosis, Kaplan–Meier survival curves were constructed to compare the OS and PFS of patients with high NLR score (>2.85) to those of patients with low NLR score (<2.85). The cut point level was determined using X-tile software.
15
Data revealed that a low NLR correlated with better OS and PFS outcomes in patients with mRCC (P = .0003 and P = .0044, Figure 1(A) and (B)). Moreover, we conducted subgroup survival analyses for patients receiving sorafenib and those receiving sunitinib. The median OS duration of patients treated with sunitinib was 60 months, higher than that of the sorafenib group at 37 months. The median PFS duration of patients treated with sunitinib was 31 months, higher than that of the sorafenib group at 20 months. Importantly, NLR could predict OS and PFS regardless of which TKI was used. In this context, a low NLR correlated with better OS and PFS in the sorafenib group (P = .0060 and P = .0246, Figure 1(C) and (D)). The same result was observed in the sunitinib group, where low NLR correlated with better OS and PFS in patients with mRCC (P = .0029 and P = .0178, Figure 1(E) and (F)). High NLR is associated with poor survival of patients with metastatic renal cell carcinoma (mRCC). Kaplan–Meier curves for overall survival (A) and progression-free survival (B). Kaplan–Meier curves for overall survival (C) and progression-free survival (D) in sorafenib subgroup. Kaplan–Meier curves for overall survival (E) and progression-free survival (F) in sunitinib subgroup. P-values were calculated using the log-rank test.
High Monocyte to Lymphocyte Ratio Correlated with Poor Patient Survival
To investigate whether a high MLR in patients with mRCC correlates with a worse prognosis, Kaplan–Meier survival curves were constructed to compare the OS and PFS of patients with high MLRs (>.30) to those of patients with low MLRs (<.30). The cut point level was determined using X-tile software.
15
Data revealed that a low MLR correlated with better OS and PFS in patients with mRCC (P = .0017 and P = .0189, Figure 2(A) and (B)). Similar to NLR, MLR can also predict OS and PFS in mRCC patients with different TKI drugs sunitinib and sorafenib; a low MLR correlated with better OS and PFS in the sorafenib group (P = .0086 and P = .0110, Figure 2(C) and (D)). The same result was observed in the sunitinib group, where low MLR correlated with better OS and PFS in patients with mRCC (P = .0446 and P = .0255, Figure 2(E) and (F)). High MLR is associated with poor survival of patients with metastatic renal cell carcinoma (mRCC). Kaplan–Meier curves for overall survival (A) and progression-free survival (B). Kaplan–Meier curves for overall survival (C) and progression-free survival (D) in the sorafenib subgroup. Kaplan–Meier curves for overall survival (E) and progression-free survival (F) in the sunitinib subgroup. P-values were calculated using the log-rank test.
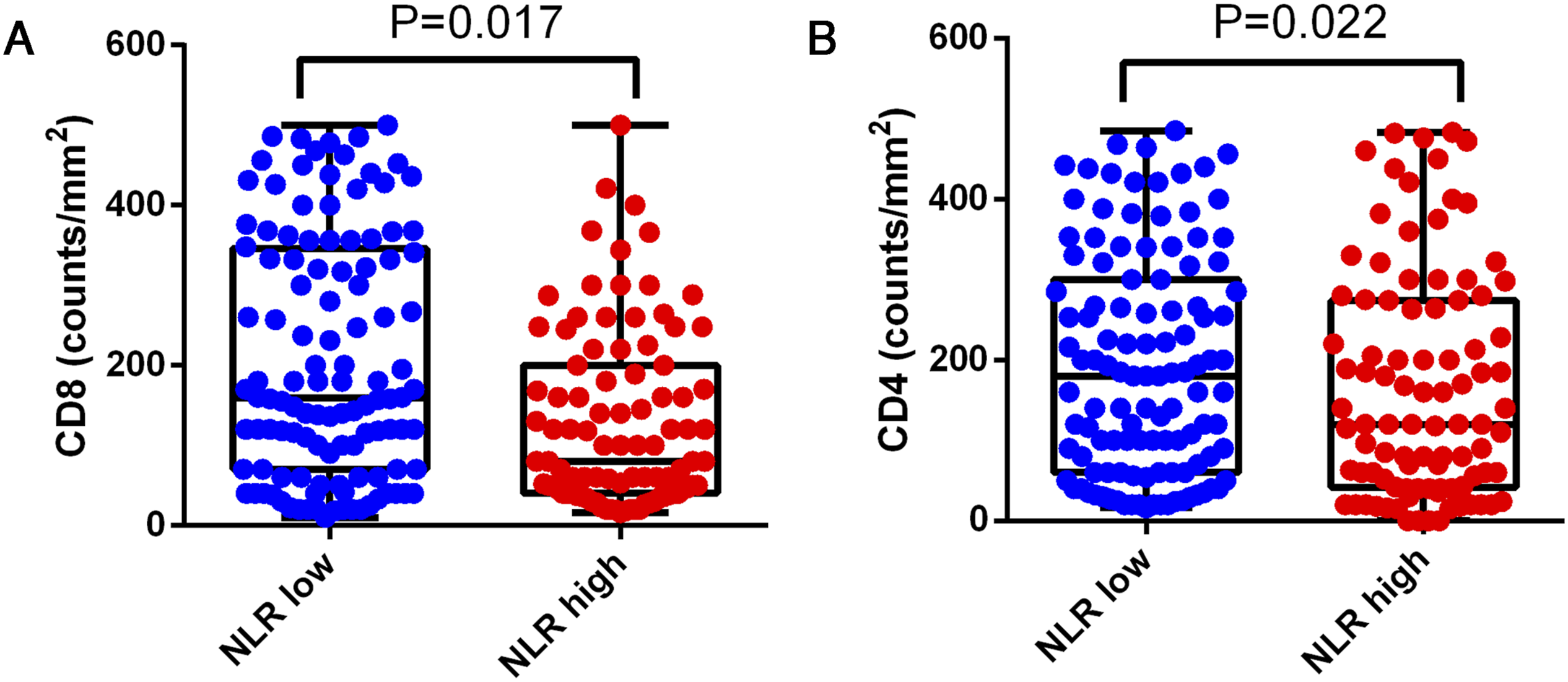
Association of Neutrophil to Lymphocyte Ratio and T Cell Infiltration in Metastatic Renal Cell Carcinoma
Representative images of tumor-infiltrating CD8+ and CD4+ T cells of NLR/MLR high and low groups are shown in Figure 3. The association between the NLR and the number of infiltrating CD8+ T cells was statistically significant (Figure 4(A); P = .017). The association between the NLR and the number of infiltrating CD4+ T cells was also statistically significant (Figure 4(B); P = .022). Our findings suggest that patients with low NLR have less infiltration of CD8+ and CD4+ T cells. Representative image of infiltrating CD8+ T cells and CD4+ T cells. Representative image of infiltrating CD8+ T cells in the high (A) and low (B) NLR groups. CD4+ T cells in the high (C) and low (D) NLR groups. CD8+ T cells in the high (E) and low (F) MLR groups. CD4+ T cells in the high (G) and low (H) MLR groups. Association between the NLR and T cell infiltration in mRCC. CD8+ T cell (A) and CD4+ T cell (B) density in the low and high NLR groups.
The Neutrophil to Lymphocyte Ratio Serves as an Independent Prognostic Factor for Metastatic Renal Cell Carcinoma
Univariate and Multivariate Cox Regression Analyses for OS of Patients (n = 210).
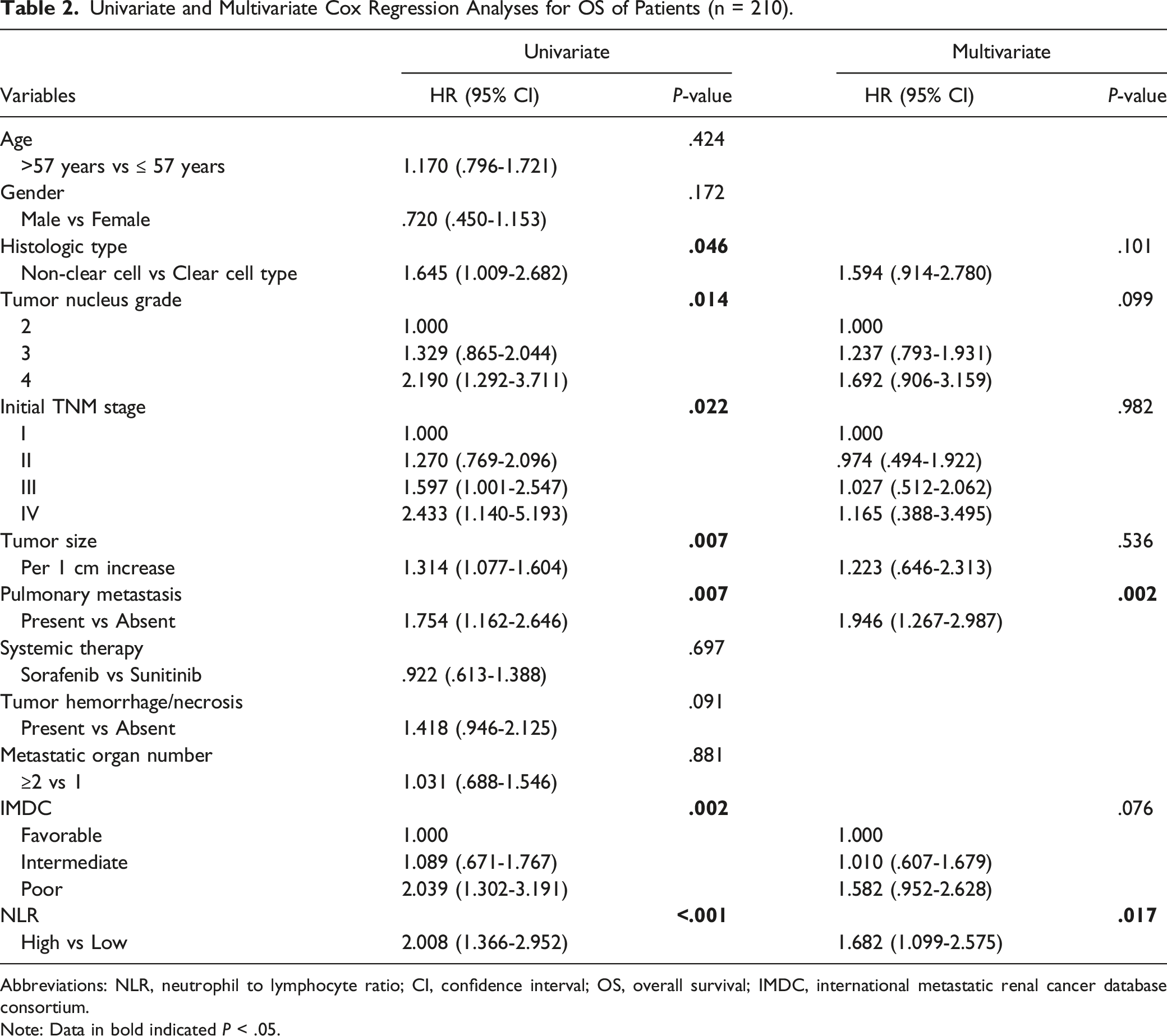
Abbreviations: NLR, neutrophil to lymphocyte ratio; CI, confidence interval; OS, overall survival; IMDC, international metastatic renal cancer database consortium.
Note: Data in bold indicated P < .05.
Univariate and Multivariate Cox Regression Analyses for OS of Patients (n = 210).

Abbreviations: MLR, monocyte to lymphocyte ratio; CI, confidence interval; OS, overall survival; IMDC, international metastatic renal cancer database consortium.
Note: Data in bold indicated P < .05.
Discussion
An increased NLR or MLR before treatment is a poor prognostic factor for different cancers, including gastrointestinal cancers, soft tissue sarcomas, nasopharyngeal carcinoma, and lung cancer.16-19 In our study, patients with low NLR and MLR values (NLR <2.85 and MLR <.30) had longer PFS and OS. The relative proportion of neutrophils is larger in patients with a high NLR and they tend to have worse prognosis. This reflects enhanced neutrophil-dependent inflammatory reactions in the tumor microenvironment and the relatively low proportion of lymphocytes, which reduces lymphocyte-mediated anti-tumor immune response, can facilitate cancer progression, and cause poor prognosis in patients with malignant tumors. Increasing evidence supports the involvement of systemic inflammation in the development and progression of cancer. 20 Pro-inflammatory factors are potential prognostic markers for multiple cancers because tumor-related inflammatory indicators significantly impact cancer development and drug response than conventional prognostic indicators. Hematology-based parameters commonly used in clinical practice, such as the neutrophil or lymphocyte count and the resulting NLR and MLR, are relatively easy to obtain and evaluate without additional costs; therefore, they can be used as reliable indicators to evaluate the prognosis of patients with mRCC.
High NLR and increased proportion of neutrophils in the circulatory system contribute to tumor invasion and progression, and are indicative of poor patient prognosis. Neutrophils produce a variety of cytokines, such as tumor necrosis factor (TNF), interleukin (IL)-1, IL-6, IL-8, and granulocyte-macrophage colony-stimulating factor, and secrete the pro-angiogenic vascular endothelial growth factor, which collectively contribute to tumor progression.21-23 The reduced lymphocyte count due to high NLR is unfavorable for anti-tumor immunity as CD8+ and CD4+ T cells are the most important lymphocytes for fighting cancer cells and their reduced count significantly impacts anti-tumor immune response.24-26 We noted decreased and increased counts of infiltrating CD8+ and CD4+ T lymphocytes in the tissue samples of patients with mRCC with a high NLR and those with a low NLR, respectively. This may explain why the NLR can predict the prognosis of cancer patients. It is widely known that CD8+ and CD4+ T lymphocytes are an important part of the tumor microenvironment and promote anti-tumor immunity via modifying cytokine milieu of the TME, exerting cytotoxic effects, and promoting the humoral-mediated immune response. However, further research is needed to elucidate how CD8+ and CD4+ T cell invasion can be induced into the tumor microenvironment for better anti-tumor immune response.
In our study, we found that the NLR and MLR are closely related to TNM stage, Fuhrman grade, and tumor size. The higher the TNM stage, Fuhrman grade, tumor size, MLR, and NLR, the worse the patient prognosis. Using Cox regression analysis, we further confirmed that increased NLR and MLR were independent predictors for OS and PFS in patients with mRCC. This indicates that the NLR and MLR can be used as a prognostic indicators for patients with mRCC, and provide a simple and effective reference value in clinical testing. In patients with low NLR, the median OS and PFS of patients using sunitinib were better than those of patients using sorafenib.
Nevertheless, our study had some limitations. First, it was a retrospective observational study. Therefore, we could not exclude the possibility of unequal distribution of unidentified clinicopathologic parameters in our patient cohort. Second, the total number of patients for whom we analyzed the NLR and MLR was relatively small. Finally, neutrophil, monocyte, and lymphocyte counts may be influenced by concurrent infections and intake of drugs that cannot be accounted for in this analysis. Thus, further studies should be designed to overcome these limitations and validate these findings.
Conclusions
High NLR and MLR are associated with worse PFS and OS in patients with mRCC. A high NLR is significantly associated with reduced infiltration of CD8+ and CD4+ T lymphocytes. Our results showed that the NLR and MLR may be associated with PFS and OS of patients treated with TKIs for mRCC, and may contribute to treatment decisions, patient selection, and future clinical trials design.
Supplemental Material
Supplemental Material - Systemic Inflammatory Biomarkers Predict Survival of Patients Treated With Tyrosine Kinase Inhibitors for Metastatic Renal Cell Carcinoma
Supplemental Material for Systemic Inflammatory Biomarkers Predict Survival of Patients Treated With Tyrosine Kinase Inhibitors for Metastatic Renal Cell Carcinoma by Zhaojuan Wang, Yujie Qin, Xuxia Chai, Lina Lu, Ping Xue, Runrun Lu, Chengrui Miao, Haimei Ma, Xiaoyi Hu, and Jiaxi Yao in Cancer Control.
Footnotes
Acknowledgments
Author Contributions
Z.W., X.H., J.Y. designed this work. X.C., Y.Q. wrote the manuscript and make statistical analysis. L.L., P.X., R.L., C.M. and H.M. performed the survival analysis, statistical analysis, and immunohistochemistry. X.H. and J.Y. performed the data review. All authors have read and approved the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a grant from the Hexi University President Fund Innovation Team Project (CXTD2022012), Hexi University 13th Science and Technology Innovation Project (No.127).
Ethical Statement
Ethical ratification was authorized by the Ethics Committee of Hexi University Affiliated Zhangye People’s Hospital (approval number B2017-018) and ethics committees of the Zhongshan Hospital, Fudan University (approval number B2016-030). Written informed consent was obtained from all the patients.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
