Abstract
Background
Papillary thyroid carcinoma (PTC) is the most common subtype of thyroid carcinoma, and Hashimoto’s thyroiditis (HT) has been postulated to have a relationship with PTC. This study aims to assess clinical and pathological characteristics of patients with papillary thyroid carcinoma coexisting with Hashimoto’s thyroiditis.
Methods
A retrospective study was conducted in a cohort of patients with thyroid carcinoma at the Department of Surgery, Shanghai General Hospital from January 2017 to December 2018. Medical records of patients who had PTC with or without HT were reviewed and clinical and histopathological characteristics of these patients were analyzed.
Results
A total of 632 patients with thyroid carcinoma were identified. Among them, 614 (97.15%) had PTC and 120/614 (19.0%) harbored PTC together with HT. PTC was significantly associated with HT, as compared with other histological subtypes (P < .001). Patients with coexisting PTC and HT (PTC + HT group) were significantly younger than patients with PTC alone (PTC group) (P = .008). There were more women in the PTC + HT group than in the PTC group (88.3% vs. 73.1%, P < .001). TSH, TGAb, and TPOAb levels were significantly higher in the PTC + HT group than in the PTC group (P ≤ .001). In addition, tumor diameter was smaller in the PTC + HT group than in the PTC group (P = .034). The PTC + HT group showed a significant better recurrence-free survival than the PTC group. Furthermore, immunohistochemical analysis revealed that patients in the PTC + HT group had a higher positive rate and higher expression intensity of Ki67 than patients in the PTC group.
Conclusions
Our study revealed that patients with coexisting PTC and HT were younger, had smaller tumor diameters, a better prognosis, and higher positive rates and expression intensity of Ki67, than did patients with PTC alone.
Keywords
Introduction
Thyroid carcinoma is the most common endocrine malignance tumor in the world, with a significant increase in global incidence over the past decades.1–3 Furthermore, its incidence has increased from 3.6/100,000 persons in 1973 to 13.8/100,000 persons in 2017. 4 It is true both in the South5,6 and East.7,8 Although the reason for this increase remains unclear, it has been associated with the increasing use of thyroid imaging, radiation, increased iodine intake, environmental pollutants, and chronic lymphocytic thyroiditis including Hashimoto’s thyroiditis (HT).
Hashimoto’s thyroiditis has been postulated to have a relationship with papillary thyroid carcinoma (PTC), the most common subtype of thyroid carcinoma; and it has been suggested that HT has a possible risk factor for developing PTC. 9 This relationship was first discovered in 1955, and several subsequent retrospective studies then suggested similar results.10,11 However, its exact mechanism remains uncertain. Some studies have shown that the presence of HT in patients with PTC is associated with less aggressive clinical characteristics and presentation,12,13 whereas other studies did not reveal similar effects.14,15 Thus, the influence of HT on the behavior of PTC remains a matter of debate.
The objective of this study was to investigate clinical, pathological, and immunohistochemical characteristics in a large cohort of patients with PTC coexisting with or without HT, in order to reveal the intrinsic relationship between the PTC and HT.
Materials and Methods
Patients and Study Design
A cohort of patients with thyroid carcinoma at the Department of General Surgery, Shanghai General Hospital from January 2017 to December 2018 was retrospectively analyzed. This study is reported according to the STORBE guidelines for cohort studies. 16 The patients were selected consecutively. All patients underwent clinical, ultrasonographic, and fine-needle aspiration biopsy (FNAC) evaluation in the preoperative period. If the tumor was confined to one lobe, a unilateral thyroidectomy + isthmus resection was performed, plus prophylactic ipsilateral central lymph node dissection. If it was multifocality in bilateral lobe or existed extracapsular invasion, a total thyroidectomy plus prophylactic bilateral central lymph node dissection was performed. All patients had a final histopathological diagnosis of thyroid carcinoma. Patients with a history of HT and hypothyroidism may have taken levothyroxine before surgery. After surgery, all patients received suppressive doses of levothyroxine, and 95 patients received radioactive iodine therapy (RAI therapy). RAI therapy was used in patients with extrathyroidal extension or tumor size > 4 cm, or more than 5 metastatic lymph nodes. The dose of RAI therapy was 150 mCi typically. All the patients were followed up in our institution every 3 months in the first year and then annually if disease-free. Follow-up examination included physical examination, neck ultrasound, serum thyroglobulin (TG) and antithyroglobulin antibodies (TGAb), and diagnostic whole body scan if necessary. Follow-up time was defined as the time from the initial surgery to the most recent follow-up record, otherwise for the deceased patients, it is the time of death, caused by incurable thyroid cancer. At the end of the study, 27 patients suffered recurrence diseases, and one patient died of metastatic PTC.
Medical records of patients who had thyroid carcinoma with or without HT were reviewed, and clinical and histopathological characteristics of these patients were analyzed. In current study, the patients were admitted for treating thyroid cancer. TgAb and antithyroid peroxidase antibody (TPOAb) were only examined once before operation. All patients underwent surgeries and have corresponding pathological diagnoses, including thyroid carcinoma and HT. So serum TgAb and TPOAb levels were not routine indicators for the diagnosis of HT in the current study, and diagnosis of HT was only based on histopathological findings. In our hospital, the normal values of TPOAb, TgAB, and TSH are presented as TPOAb (5.00–38.00 IU/mL), TgAb (10.00–125.00 IU/mL), and TSH(.35–4.75 uIU/mL), respectively. Pathologically, HT was defined as the presence of diffuse lymphocytic infiltration with formation of lymphoid follicles and reactive germinal centers; infiltration must occur in normal areas of the thyroid, distinct from the site of thyroid cancer. Peritumoral inflammatory response and small-area lymphocytic infiltration are not considered HT. Pathological and immunohistochemical detection was conducted and diagnosed by the same pathologist team.
The study was conducted in accordance with the Declaration of Helsinki. This study was conducted under the confidentiality agreement of Shanghai General Hospital institution’s Research Commission and approved by the ethical committee of Shanghai General Hospital (2016KY021). Informed consent was obtained from all patients who donated their specimens.
Definitions
In this study, tumors were considered multiple lesions when two or more tumor focuses were found in the same or different lobes of the gland. Papillary microcarcinoma was defined as tumors with a diameter ≤1.0 cm by histological examination. Lymph node status was defined by the pathological evidence of metastasis in lymph nodes that were removed. Extracapsular invasion was defined as both gross extrathyroidal extension (gETE) and microscopic extrathyroidal extension (mETE). Clinicopathological staging procedures were performed according to the American Joint Committee on Cancer TNM staging system (eighth Edition). 17
Immunohistochemical Analysis
Antihuman CK19, HBME-1, Galectin-3, TTF-1, Calcitonin, TG, CEA, and Ki67 antibodies were obtained from Gene Tech Company Limited, Shanghai, China (GM0888, GM3505, GT2267, GT2180, GT2066, GT2122, GT2108, and GT2094). All specimens were fixed in 10% neutral buffered formalin (pH 7.4), dehydrated, and embedded in paraffin according to conventional methods. Specimens were cut into 4 μm sections and stained with hematoxylin and eosin (H&E). Immunostaining was performed using monoclonal antibodies. Paraffin from paraffin sections was removed before they are immersed in distilled water. Paraffin sections were then rinsed in PBS and blocked with 3% hydrogen peroxide in methanol at room temperature for endogenous peroxidase ablation. Slides were incubated with primary monoclonal antibodies for one hour at room temperature. After washing with PBS, slides were incubated with biotinylated secondary antibodies for 30 min. The reaction became visible after staining with DAB and was kept in the dark for 10 minutes at room temperature.
In Ki67 immunohistochemical analysis, 10 high-power visual fields were randomized selected under the microscope and then the number of positive cells per 100 tumor cells was counted. The average value is expressed as the expression intensity of Ki67. Typical pictures are displayed in Figure 1, which showed the picture of Ki67 expression intensity of 1%, 5%, 10%, and 20%. Ki67 expression intensity in PTC (En Vision, ×200). A, B, C, D expression intensity is 1%, 5%, 10%, and 20% separately.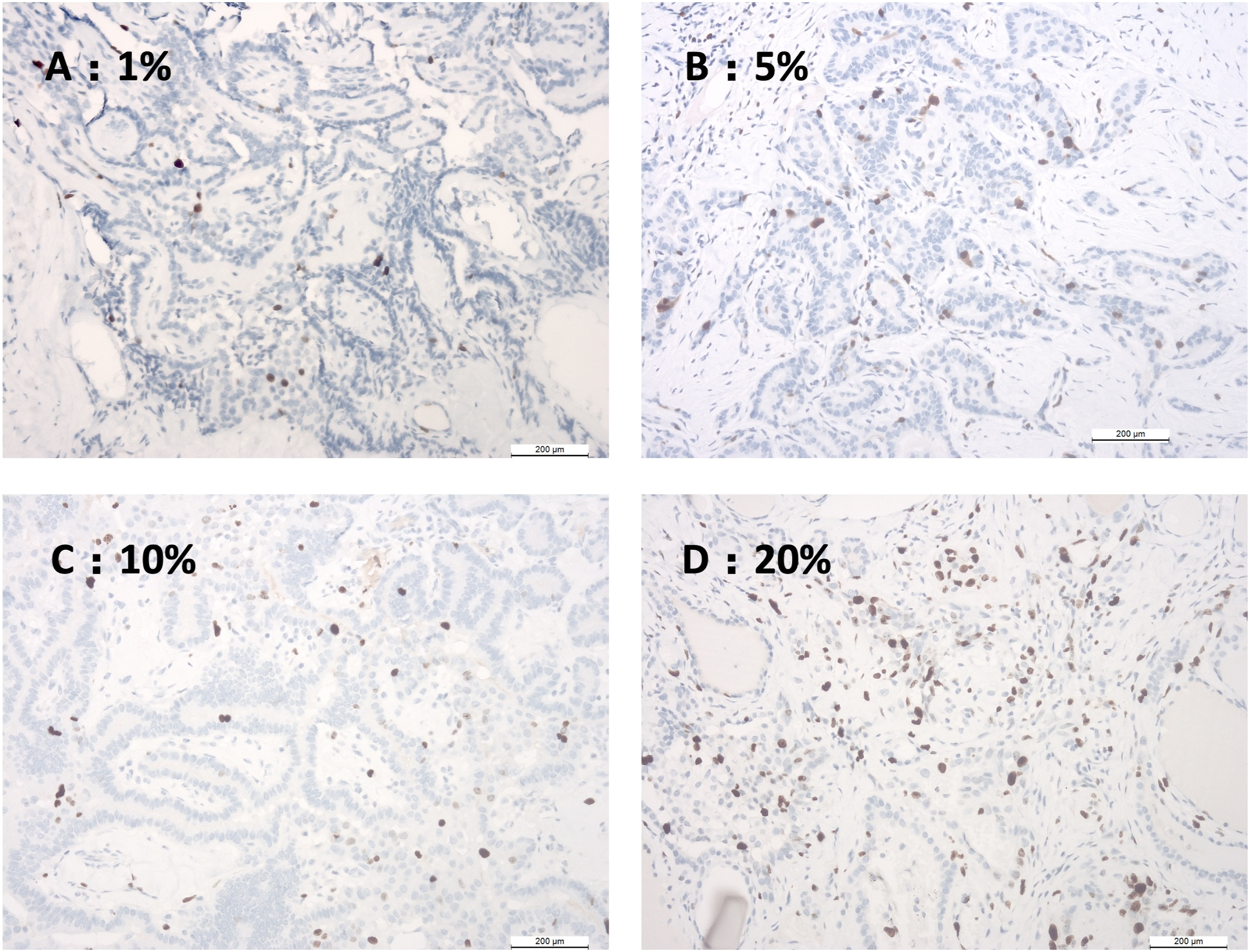
All the slides were examined by two pathologists under a confocal microscope (Leica TCS SPE, Germany).
Database
The following parameters of patients were entered into a dedicated database: age, gender, coexistent HT, lymph node status, histopathological description, nodule size, multifocality, TNM staging, clinicopathological staging, positive status of CK19, HBME-1, Galectin-3, TTF-1, Calcitonin, TG, CEA and Ki67, expression intensity of Ki67, and data of blood biochemical measurements of TSH, TGAb, and TPOAb.
Statistical Analysis
Statistical analysis was performed using SPSS V.20.0. Quantitative data are presented as mean ± SD, and t-test was used for comparisons. X 2 test and Fisher’s exact test were used for comparisons of continuous data. In order to identify risk factors for thyroid carcinoma, multiple logistic regression analysis with backward elimination was used. Overall survival and recurrence-free survival were investigated by Kaplan–Meier analyses. Independent poor survival risks were identified by Cox proportional hazards regression model analysis. Survival curves were calculated and Log-rank test was performed. Differences were considered significant when P < .05. Prism 8.0 was used to make the graphs.
Result
Patient Characteristics
A total of 632 patients who had thyroid carcinoma with or without HT were identified from January 2017 to December 2018. Among these patients, 481 (76.1%) patients were women; and the mean age of patients was 46.64 ± 12.38 years (range, 16–83 years).
Histological Subtypes of Thyroid Carcinoma.

Abbreviations: HT, Hashimoto’s thyroiditis.
Clinical and Pathological Characteristics of Patients with Papillary Thyroid Carcinoma
Clinical and Pathological Characteristics of Patients With Papillary Thyroid Carcinoma.

Abbreviations: TGAb, thyroglobulin antibody; TPOAb, thyroid peroxidase antibody; TSH, thyroid-stimulating hormone. Plus–minus values are expressed as mean ± SD.
Tumor diameter was smaller in the PTC + HT group than in the PTC group (P = .034), but there was no significant difference with respect to multifocality, extracapsular invasion, lymph node metastasis, papillary thyroid microcarcinoma, TNM stage, or clinicopathological stage between the two groups (Table 2).
Survival Analysis
The median follow-up time was 42 (12–60) months. We performed Kaplan–Meier analyses on overall survival (OS) and recurrence-free survival (DFS) among the PTC + HT group and PTC group. As revealed in Figure 2, the two groups differed significantly in DFS (log-rank test chi-square = 4.002, P = .045), but OS did not showed significant differences (log-rank test chi-square = .227, P = .633). In order to account for the influence of age and gender, a Cox proportional hazards regression model analysis was conducted. The results revealed that even after adjusting for age and gender, HT (−) remained a weak prognostic factor for PTC patients’ survival (Table 3). Kaplan–Meier curves of overall survival and recurrence-free survival in the PTC-coexisted HT group (HT (+) group) and PTC-alone group (HT (-) group). The OS rates for patients were 100% and 99.8%, respectively (log-rank chi-square = .227, P = .633). The RFS of the patients were 99.2% and 94.5%, respectively (log-rank chi-square = 4.002, P = .045), which showed a difference between two groups. Cox Proportional Hazards Regression Model Analysis of Recurrence-free Survival (RFS). Abbreviations: HT, Hashimoto’s thyroiditis.

Immunohistochemical Characteristics of Papillary Thyroid Carcinoma
Immunohistochemical analysis revealed that patients in the PTC + HT group had a higher positive rate of Ki67 (Figure 3) and higher expression intensity of Ki67 (Figure 4) than patients in the PTC group. However, no significant differences were observed in positive rates of CK19, HBME-1, Galectin-3, TTF-1, Calcitonin, TG, and CEA between the two groups (Table 4). Distribution of various proteins expressed in the PTC-coexisted HT group and PTC-alone group by immunohistochemical analysis. The vertical axis represents the case number. It is showed a higher positive rate of Ki67 in the PTC + HT group than the PTC-alone group (P = .023). However, no significant differences were observed in the positive rates of CK19, HBME-1, Galectin-3, TTF-1, Calcitonin, TG, and CEA between the two groups (P = Positive, N = Negative). Distribution of Ki67 expression intensity in the PTC-coexisted HT group and PTC-alone group by immunohistochemical analysis. The vertical axis is Ki67 expression intensity (%), and the red lines represent the mean value. T-test revealed a significant difference (P = .0005) between the PTC-alone group (HT (-) group) and PTC-coexisted HT group (HT (+) group) in the expression intensity of Ki67. Immunohistochemical Characteristics of Papillary Thyroid Carcinoma. Abbreviations: CK19, Cytokeratin-19; HBME-1, human bone marrow endothelial cell-1; TTF-1, thyroid transcription factor-1; TG, thyroglobulin; CEA, carcino-embryonic antigen; Ki67, Kiel nuclear-associated antigen. Plus-minus values are expressed as mean ± SD.

Discussion
In the present study, we retrospectively assessed clinical and histopathological characteristics in a patient cohort of thyroid carcinoma. We found that patients with coexisting PTC and HT were younger, had smaller tumor diameters, had a better survival than the PTC-alone group, and had higher positive rates and expression intensity of Ki67 than did patients with PTC alone.
The link between HT and PTC has long been a topic of controversy. The association between PTC and HT was first described in 1955. 18 The studies of Loh 13 and Girard 19 revealed a strong association between PTC and lymphocytic thyroiditis. They revealed that patients with PTC and lymphocytic thyroiditis had smaller mean tumor diameters, lower frequency of extra-thyroid extension, and earlier clinicopathological staging than did patients with PTC alone, suggesting that lymphocytic infiltration may contribute to the management of tumor growth and proliferation. Kim et al 20 found a higher frequency of women, lower mean tumor diameters, and lower mean age in patients who had PTC with HT, than patients without HT. Similar to PTC, the study of Dvorkin et al 21 found that differentiated thyroid cancer is also associated with less aggressive disease and better outcome in patients with coexisting HT. In our study, we found that patients with coexisting PTC and HT were younger and had smaller tumor than did patients with PTC alone, which is consistent with previous reports 19 in a certain degree. However, no significant differences were observed in multiple lesions, extracapsular invasion, and clinicopathological stage. This difference may be explained by Jankovic’s study, 22 whose meta-analysis revealed that there is no clear evidence to support the correlation between HT and PTC in population-based FNAB studies; although many thyroidectomy studies, which may be subject to selection bias, have reported a positive correlation. The average prevalence rate of PTC in patients with HT was 1.20% in 8 FNA studies of 18,023 specimens and 27.56% in 8 archival thyroidectomy studies of 9884 specimens. Therefore, more prospective studies with longer follow-ups are needed to further elucidate this relationship.
The expression of Ki67 is closely related to the proliferation and growth of tumor cells and is widely used as a proliferation biomarker in routine pathological examinations. Studies have shown that compared with normal tissues, the expression of Ki67 is significantly higher in poorly differentiated malignant tissues than in normal tissues.23,24 The Ki67 index was an independent prognostic factor for survival, including all stages and grades. It has been shown that there is a correlation between the ratio of Ki67-positive malignant cells and patient survival. 25 Interestingly, our study revealed a higher positive rate of Ki67 and higher expression intensity of Ki67 in PTC patients with HT than in patients without HT, while the PTC + HT group showed better prognosis based on survival analysis in our study. This result appeared paradoxical; however, considering HT as an autoimmune disease characterized by intrathyroidal lymphocytic infiltrate, several studies have found that Ki67 was more expressed in the lymphocytes of patients with HT than in normal ones.26,27 It may be the explanation for this seemingly contradictory phenomenon.
In our study, the PTC + HT group exhibited a better prognosis than the PTC- lone group, even after adjusting for age and gender, which showed the protective effect. This result was consistent with the majority of previous studies.19–21 However, some studies have indeed reported a worse prognosis among patients who had thyroid cancer with thyroiditis.28,29
The concept of chronic inflammation as a risk factor for the development of malignancies has been well-established for other tumors. However, with respect to these two entities, the association of cause and effect between them remains uncertain. PTC commonly develops in patients with autoimmune thyroiditis,30–32 which raises the question of how thyroid malignancies develop despite immune responses; or does autoimmune thyroiditis develop due to an antitumor immune response? Recently, research seems to confirm the important influence of the human immune system in responding to malignant diseases. While infection and immune responses are associated with an increased risk of malignant disease, it is clear that immune effects can also influence malignant progression. Moreover, it is not unreasonable to hypothesize that chronic thyroid inflammation may increase malignant transformation. Nicolson’ study revealed a strong correlation between immune lymphocytic infiltrates in the thyroid generates a DNA-damaging environment, and some DNA damage was instead repaired incorrectly leading to mutations and ultimately carcinogenesis. 33 Several studies suggested that stimulating TSH concentrations may be a potential factor for this relationship. 34 However, HT may also be a response to tumors in genetically predisposed subjects deferring cancer progression. The favorable outcome of these patients suggested that this association with thyroiditis may represent an anti-tumor response, 35 although it is not clear whether the coexistence of both diseases is simply a greater chance for a synchronous occurrence of two high-prevalence diseases. HT and PTC may also be the result of potential genetic or environmental exposure factors, rather than one leading to another.
Nonetheless, we acknowledge the limitations in our investigation. Our study was retrospective in nature and was carried out in a single institution. We also note that HT itself was not a thyroidectomy indication in our study.
Conclusion
In conclusion, our study has shown that the presence of HT was associated with younger age, smaller tumor diameters, a better prognosis, and higher expression intensity of Ki67 in patients with PTC.
Footnotes
Author Contributions
Ye Yang and Min Wang: conceived and designed the study and drafted the manuscript; Jun Liu and Min Wang participated in data collection; Xiaoqin Shi and Jun Liu performed the statistical analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Shanghai Songjiang District Science and Technology Research (Medical and Health) Project (grant number 20SJKJGG304) and Shanghai General Hospital Characteristic Research Project (grant number CTCCR-2021C19), Post-graduation Medical Education Project of Shanghai Jiao Tong University School of Medicine (BYH20220401), and Education Research Project of Nanjing Medical University (2021ZC096).
