Abstract
Background
The objective of this study is to develop a predictive model for the assessment of cervical lymph node metastasis risk in papillary thyroid carcinoma (PTC).
Methods
A retrospective study was conducted on 212 patients with PTC who underwent initial surgical treatment from August 2022 to April 2023 in 2 hospitals. Data were randomly split into 7:3 training-validation sets. Logistic regression was used for feature selection and predictive model creation. Model performance was assessed using receiver operating characteristic (ROC) and calibration curves. Clinical utility was determined using decision curves.
Results
Among the 212 patients with PTC, 104 cases (49.1%) exhibited cervical lymph node metastasis, while 108 cases (50.9%) did not. Multivariate logistic regression analysis revealed that age (OR = 0.95), FT3 (OR = 0.41), tumor maximum diameter ≥0.9 cm (OR = 1.85), intratumoral microcalcifications (OR = 1.84), and suspicious lymph node on ultrasound (OR = 2.96) were independent risk factors for lymph node metastasis in PTC patients (P < 0.05). The constructed model for predicting the risk of cervical lymph node metastasis demonstrated a training set ROC curve area under the curve (AUC) of 0.742 (95% CI: 0.664 - 0.821), with a cut-off value of 0.615, specificity of 87.8%, and sensitivity of 51.4%. The validation set exhibited an AUC of 0.648 (95% CI: 0.501 - 0.788), with a cut-off value of 0.644, specificity of 91.2%, and sensitivity of 43.3%. Including the BRAF V600 E mutation did not improve the model’s diagnostic performance significantly. Decision curve analysis indicated clinical feasibility of the model.
Conclusion
The predictive model developed in this study effectively predicts lymph node metastasis risk in PTC patients by incorporating ultrasound features, demographic characteristics, and serum parameters. However, including the BRAF V600 E mutation does not significantly improve the model’s diagnostic performance.
Plain language summary
Background
The objective of this study is to develop a predictive model for the assessment of cervical lymph node metastasis risk in papillary thyroid carcinoma.
Methods
A retrospective study was conducted on 212 patients with papillary thyroid carcinoma who underwent initial surgical treatment at the First Affiliated Hospital of Fujian Medical University from August 2022 to April 2023. Data randomly split into 7:3 training-validation sets. Logistic regression used for feature selection and predictive model creation. Model performance assessed using ROC and calibration curves. Clinical utility determined using decision curves.
Results
Among the 212 patients with PTC, 104 cases (49.1%) exhibited cervical lymph node metastasis, while 108 cases (50.9%) did not. Multivariate logistic regression analysis revealed that age (OR = 0.95), FT3 (OR = 0.41), tumor maximum diameter ≠¥ 0.9cm(OR = 1.85), intratumoral microcalcifications (OR = 1.84), and suspicious lymph node on ultrasound (OR = 2.96) were independent risk factors for LNM in PTC patients (P < 0.05). The constructed model for predicting the risk of cervical lymph node metastasis demonstrated a training set ROC curve area under the curve (AUC) of 0.742 (95% CI: 0.664 - 0.821), with a cut-off value of 0.615, specificity of 87.8%, and sensitivity of 51.4%. The validation set exhibited an AUC of 0.648 (95% CI: 0.501 - 0.788), with a cut-off value of 0.644, specificity of 91.2%, and sensitivity of 43.3%. Including BRAF V600E gene did not improve model's diagnostic performance significantly. Decision curve analysis indicated clinical feasibility of the model.
Conclusion
The predictive model developed in this study effectively predicts lymph node metastasis risk in PTC patients by incorporating ultrasound features, demographic characteristics, and serum parameters.However, Including the BRAF V600E mutation does not significantly improve diagnosis performance.
Introduction
Thyroid cancer has become 1 of the top ten common cancers worldwide. 1 In 2020, the global incidence rate was 10.1 per 100,000. 2 The most common pathological type is Papillary thyroid carcinoma (PTC). Although PTC progresses relatively slowly, with a 10 year survival rate of over 90%, 3 it has a high propensity to metastasize to surrounding lymph nodes, with a metastasis rate ranging from 50% to 90%.4,5 Moreover, within a certain range, as the number of lymph node metastases increases, the risk of recurrence for patients also rises. 6 Such patients require aggressive treatment modalities, such as unilateral or total thyroidectomy and therapeutic neck lymph node dissection. In cases where clinical indications are met, patients with cN1 (clinical lymph node positive) routinely undergo therapeutic neck lymph node dissection. 7 For clinically lymph node-negative (cN0) patients, a relatively conservative approach may be taken, including thyroid lobectomy, thyroid cancer ablation therapy, or follow-up observation. However, studies have found that approximately 20% to 80% of cN0 patients may have occult lymph node metastases identified in postoperative pathology. 8 Failure to perform prophylactic lymph node dissection in such patients may increase the risk of secondary surgery. Nevertheless, if prophylactic central compartment neck lymph node dissection is performed for all cN0 patients, it may elevate the occurrence of postoperative complications (such as recurrent laryngeal nerve injury and permanent hypoparathyroidism) without significant improvement in patient prognosis. 9 Therefore, accurately determining the cervical lymph node status of PTC patients is crucial for making rational and individualized surgical decisions.
Currently, imaging examinations such as ultrasound and computed tomography scans, as well as cytopathological examinations like fine-needle aspiration (FNA) or FNA with thyroglobulin measurement (FNA-Tg), are the main methods for preoperatively assessing cervical lymph nodes in PTC patients. 10 However, routine ultrasound examination has limited sensitivity in detecting cervical lymph nodes, and a single preoperative examination may not provide sufficient evidence for surgical decision-making. 11 Thus, preoperative assessment of lymph node metastasis (LNM) in PTC patients remains a challenging clinical issue, and there is a need for further exploration of more effective predictive methods.
Research on the relationship between changes in thyroid function and the risk of PTC suggests a correlation between the 2. 12 Thyroid-stimulating hormone (TSH) plays a crucial role in regulating thyroid function, and the Gαs-adenylyl cyclase-protein kinase A-cyclic adenosine monophosphate (cAMP) pathway is the classical pathway of TSH action. 13 Constitutive activation of the cAMP pathway may be associated with increased carcinogenic potential and decreased TSH levels. Lower TSH levels may lead to less differentiation of thyroid epithelial cells, increasing the tendency for malignant cell transformation. 14 In addition, thyroid hormones may have an independent impact on the occurrence of thyroid cancer, and lower levels of FT3 and FT4 have been associated with thyroid cancer diagnosis. 15 Some researchers have proposed TSH 16 and serum thyroid hormones 17 as new indicators for predicting PTC, but there are fewer studies using them to predict LNM in PTC patients. Their predictive value for LNM in PTC patients needs further investigation to be validated. 18 Therefore, in this retrospective study, we combine demographic characteristics, serum parameters, and conventional ultrasound features to construct a predictive model, aiming to improve the accuracy of preoperative LNM diagnosis for patients and provide a basis for selecting personalized treatment modalities.
Methods
Patients
A retrospective collection of patients diagnosed with thyroid cancer was conducted from August 2022 to April 2023 from the First Affiliated Hospital of Fujian Medical University and National Regional Medical Center, Binhai Campus of the First Affiliated Hospital, Fujian Medical University . All patients underwent either total or near-total thyroidectomy, unilateral lobectomy with isthmusectomy, and at least ipsilateral central neck lymph node dissection. Inclusion criteria were as follows: ① Initial surgery with postoperative pathological confirmation of PTC (excluding other pathological types of thyroid cancer, such as follicular carcinoma, medullary carcinoma, undifferentiated carcinoma, etc.); ② Complete availability of ultrasound images, laboratory data, and clinical pathological information. Exclusion criteria included: ① Concurrent presence of other malignant tumors; ② History of multiple surgeries in the thyroid region; ③ No neck lymph node dissection performed; ④ Prior use of thyroid hormone medication. This retrospective study utilized existing medical records and has been reviewed and approved by the Ethics Committee of the First Affiliated Hospital, Fujian Medical University (approval number: Minimally Invasive Ethical Review [2015]084-2). The requirement for informed consent was waived due to the retrospective nature of the study. This study complies with the TRIPOD guidelines. 19
Data Collection
(1) Clinical basic information: Gender, age, Body Mass Index (BMI: weight/height2). (2) Conventional ultrasound features of the primary lesion
10
: Multiplicity of nodules (selecting the largest nodule observed on ultrasound); Nodule size (maximum diameter of the largest nodule on ultrasound); Nodule location (left lobe, isthmus, right lobe, multiple locations); Nodule composition (solid or other), hypoechoic/very hypoechoic nodules (hypoechoic nodules defined by contrast with thyroid parenchyma, very hypoechoic nodules defined by contrast with adjacent neck muscles, indicating decreased echogenicity), longitudinal-to-transverse ratio greater than 1 (height greater than width), shape (irregular defined as serrated or lobulated), margin (indistinct defined as difficulty distinguishing the nodule edge from thyroid tissue), intranodular microcalcifications (point-like strong echoes with diameter ≤1 mm) and perinodular halo; Blood flow distribution within the nodule; Hashimoto’s thyroiditis. (3) Any of the following features detected during routine ultrasound examination are suspected to be cervical lymph node metastasis
20
: Rounded morphology (long diameter: short diameter <2:1), microcalcifications, cystic changes, hypoechoic areas, disappearance of the hilum structure, abundant or irregular blood flow within the lymph node (peripheral vascular formation). (4) BRAF V600 E gene (determined by pathological department cell pathology gene testing). (5) FT3, FT4, sTSH, PTH. (6) Blood immune indicators: Neutrophil-to-lymphocyte ratio (NLR), lymphocyte-to-monocyte ratio (LMR), preoperative systemic immune-inflammation index (SII), platelet-to-lymphocyte ratio (PLR).
Statistical Analysis
The PTC patients were divided into a 70% training set and a 30% validation set to ensure that the outcome events were randomly distributed between the 2 sets. The training set was used for variable selection and model construction, while the validation set was used to verify the results obtained from the training set. Descriptive statistics were used to analyze baseline data. Normally distributed data were expressed as mean ± standard deviation, while non-normally distributed data were presented as median (interquartile range). The consistency between the 2 randomly grouped datasets was validated.
For univariate analysis, categorical variables were analyzed using chi-square tests, and continuous variables were assessed using t-tests or rank-sum tests. To achieve the analysis objectives, tumor size (continuous variable) was converted to a categorical variable using the optimal cut-off value. Univariate and multivariate logistic regression analyses were performed for all variables, with variables demonstrating P < 0.05 considered as independent risk factors.
A predictive model for predicting LNM in PTC patients was constructed using stepwise regression based on the minimum Akaike information criterion value, and a nomogram was created. The predictive performance of the model was assessed using receiver operating characteristic (ROC) curves by calculating the area under the curve (AUC). Calibration curves were plotted, and the Hosmer-Lemeshow goodness-of-fit test was conducted to evaluate the difference between predicted and actual outcomes, confirming the accuracy of the predictions. Net reclassification improvement (NRI) and integrated discrimination improvement (IDI) were used to assess the improvement in risk prediction and measure the utility of the new model. Decision curve analysis (DCA) quantified the net benefits of different threshold probabilities, reflecting the feasibility of using the predictive model in clinical practice. Statistical analysis was performed using RStudio 4.2.2 software, with a significance level set at P < 0.05.
Results
Basic Information
A total of 212 PTC patients were included and randomly divided into a training set (n = 148) and a validation set (n = 64) using a 70:30 split (Figure 1). Based on postoperative histopathological examination results, the positive rates of LNM in the training and validation sets were 50.0% and 46.8%, respectively, and the difference in LNM positive rates between the 2 sets was not statistically significant (P = 0.677). There were no statistically significant differences in gender (P = 0.960) or age (P = 0.278) between the 2 datasets, confirming the reliability of the data used (Table 1). For ease of logistic regression analysis, tumor size (continuous variable) was categorized. ROC curve analysis was performed, and a cutoff value of <0.9 cm (119 cases) and ≥0.9 cm (92 cases) was determined. The odds ratio (OR) was 2.333 (95% CI: 1.345-4.094), categorizing tumor size as <0.9 cm or ≥0.9 cm. Flowchart of patient selection in this study. Clinicopathological and Ultrasonic Characteristics of Patients in the Training and Validation Cohorts. P value is derived from the univariable association analyses between each of the variables and CLNM status. Fisher’s exact test. SD, standard deviation; US, ultrasound; LNM, lymph node metastasis. Qualitative date were expressed as mean ± standard deviation or number percentages (%);quantitative date were expressed as median (25%-75% quantiles).

Variable Selection and Model Construction
Univariate and Multivariate Analyses of the Preoperative Predictors of LNM of PTCs in the Training Cohort.
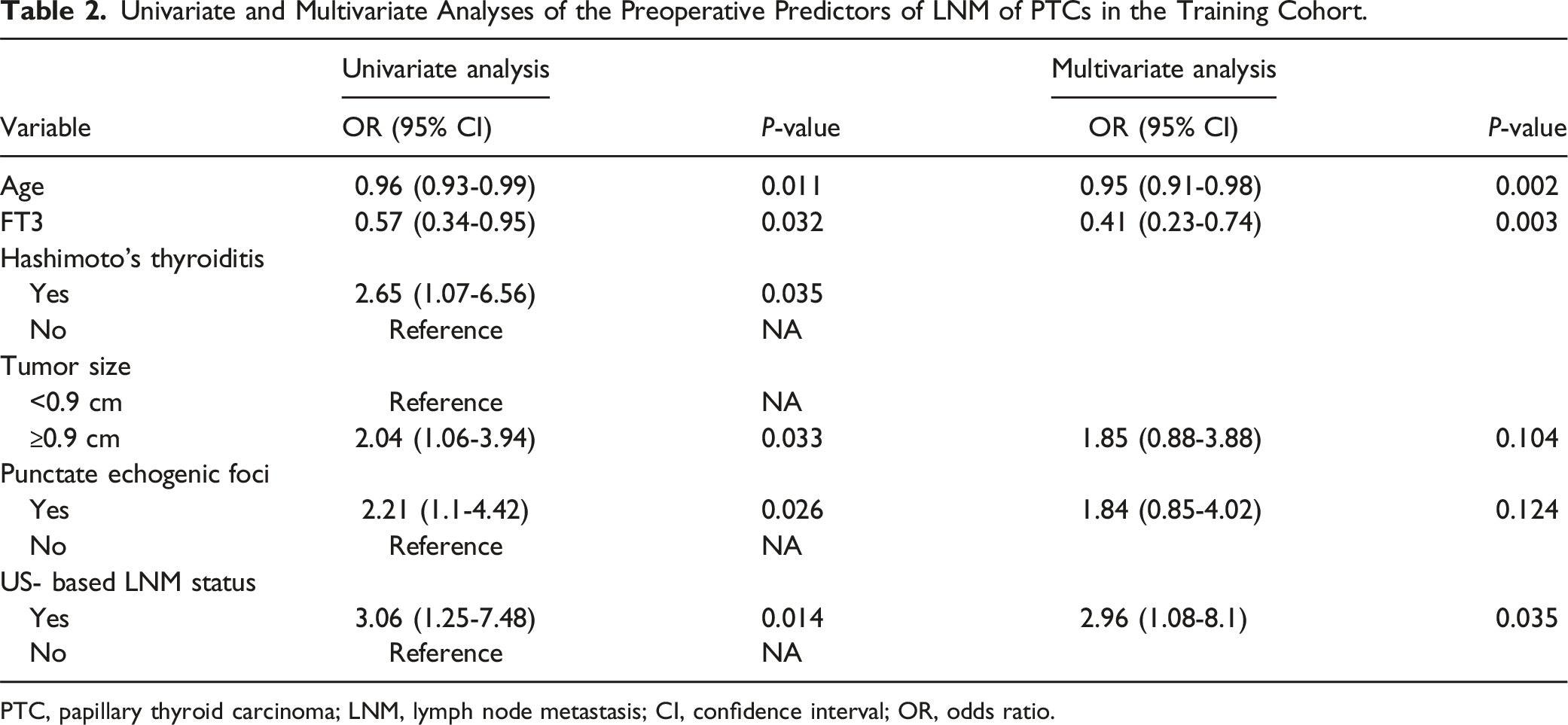
PTC, papillary thyroid carcinoma; LNM, lymph node metastasis; CI, confidence interval; OR, odds ratio.

Performance of the nomogram. (a) ROC curves of US-reported LNM status and BRAF V600 E the combined nomogram for predicting lateral LNM in the training cohort; (b) in the validation cohort. DeLong’s test showed that the AUCs were not significantly different between the model 1 and the model 2 in both cohorts (P > 0.05).
Model Construction and Validation
Risk Factors for LNM in PTC.

US-reported LNM status:model 1; US with BEAF V600 E:model 2.
Two Model Performances.

PPV, positive predictive value; NPV, negative predictive value.

Calibration curve of the combined nomogram. (a) Calibration curve of the nomogram in the training cohort, the Hosmer-Lemeshow test yield a nonsignificant statistic (P = 0.773); (b) calibration curve of the nomogram in the validation cohort, the Hosmer-Lemeshow test yield a onsignificant statistic (P = 0.070). The calibration curve illustrates the calibration of the nomogram in terms of the agreement between the predicted risk of LNM and the observed outcomes of LNM.

Decision curve analysis of the clinical multimodal nomogram. The blue and gray lines are the decision curves of our nomogram and all PTC patients with LNM who received treatment, respectively. The black line is the decision curve of all PTC patients without LNM who did not receive treatment. The decision curves show that the nomogram to predict LNM in patients with PTC provides a greater benefit than does the all or no treatment.
Comparison with the Ultrasound Features Combined with BRAF V600E Mutation Model (Model 2)
NRI and IDI.

NRI, net reclassification improvement, IDI, integrated discrimination improvement . P value is derived from the comparative analysis of the training and validation sets in Model 1 and Model 2.
Discussion
Currently, the surgical consensus for PTC follows the principle of “minimal effective treatment”. 21 Routine therapeutic lymph node dissection is performed for cN + patients. However, for cN0 patients, individualized treatment is considered, and there is still no consensus among countries on whether to undergo prophylactic central compartment lymph node dissection (pCND). 7 The controversy revolves around the limited improvement in prognosis for pCND patients and the higher risk of postoperative complications. Accurately assessing the status of neck lymph nodes in PTC patients before surgery is crucial. Therefore, this study aimed to integrate demographic characteristics, serum parameters, and conventional ultrasound features to construct a predictive model for preoperative assessment of LNM in PTC patients.
In this study, a total of 15 ultrasound features and 11 basic clinical data and laboratory indicators were included as potential predictors of LNM in PTC. Utilizing a stepwise regression based on the minimum value of Akaike’s information criterion, a predictive model was constructed. This model incorporated 5 variables: patient age, FT3 levels, maximum tumor diameter, presence of microcalcifications within the tumor, and suspicion of lymph node metastasis based on ultrasound characteristics. The model, based on ultrasound features, demonstrated excellent discrimination and calibration upon validation. The personalized treatment approach for patients holds clinical significance. For young PTC patients with low FT3 levels, tumor diameter ≥0.9 cm, presence of microcalcifications within the tumor, and suspected lymph node metastasis on ultrasound, a proactive treatment strategy is recommended. Similarly, for elderly cN0 patients with higher FT3 levels and smaller primary tumors, conservative follow-up observation or ablative therapy for the primary lesion may be considered.
Current research suggests that the most common genetic mutation in PTC is the BRAF V600 E gene mutation. 22 However, its association with LNM remains inconclusive. Some studies23,24 have reported a close correlation between the BRAF V600 E mutation and the prognosis, recurrence, and lymph node metastasis (LNM) of PTC patients. On the contrary, other studies25,26 have suggested that the BRAF V600 E mutation may not be predictive of lymph node metastasis. The results of our univariate logistic regression analysis indicate that the BRAF V600 E mutation showed no statistically significant association with LNM (P = 0.085). It is possible that the influence of this important predictive factor may have been outweighed by subtle variations due to confounding factors. Therefore, we included the BRAF V600 E mutation in model 2; however, model 2 did not significantly improve the predictive performance of the nomogram.
Some PTC patients appear to have a relative state of thyroid dysfunction. 12 However, the relationship between serum thyroid hormones and TSH requires further validation. Some researchers 15 have suggested that by excluding patients with thyroid function abnormalities, potential influences on PTC can be eliminated. They argue that both FT3 and FT4 have a direct impact on PTC. Additionally, certain studies have indicated that a decrease in FT3 levels may be a sign of dedifferentiation or malignant transformation of thyroid follicular cells, 27 suggesting that serum thyroid hormones may be helpful in predicting the malignant tumor risk in patients. The results of our multifactorial logistic regression analysis in this study demonstrate that FT3 is an independent risk factor for LNM. The nomogram illustrates that a decrease in FT3 levels is associated with an elevated risk of LNM. However, some studies have not found an association between serum thyroid hormones and LNM. 28 Further research is needed to explore the relationship between serum thyroid hormones and LNM.
One of the reference indicators for formulating individualized treatment and lymph node dissection plans for PTC patients is the maximum diameter of the tumor lesion. 29 Some researchers 30 believe that larger tumors are more likely to invade the capsule and spread to the surrounding area, potentially increasing the incidence of LNM. However, there is no unified standard for the diagnostic threshold of tumor size in existing research. 31 Some researchers have defined the threshold as 1 centimeter, 32 considering it may be a clinically significant cancer. However, if only nodules larger than 1 centimeter are evaluated, cancers within this critical range may go undetected. The results of this study indicate that a maximum nodule diameter of ≥0.9 cm holds significance in predicting LNM in PTC patients (OR: 2.35, 95% CI: 1.07-5.17, P = 0.033). This provides different options for patients with suspicious risks within the critical range, such as close observation or fine-needle aspiration (FNA).
Currently, suspected or confirmed thyroid cancer patients are required to undergo a comprehensive neck ultrasound examination, including both the thyroid gland and lymph nodes. 33 While ultrasound is a convenient, non-invasive, and rapid method for diagnosing lymph node metastasis, a meta-analysis based on retrospective studies suggests that routine ultrasound has limited capability in detecting LNM. 34 Any single ultrasound feature is not sensitive enough for detecting metastatic lymph nodes in PTC patients. 35 In this study, compared to a single conventional ultrasound examination, the Nomogram demonstrated an improvement in the accuracy, sensitivity, and specificity in diagnosing cervical lymph node metastasis. Additionally, combining suspicious features with other methods may be an effective approach. 36 The results of this study indicate that ultrasound suspicion of lymph node involvement is an independent risk factor for LNM (P < 0.05) and holds significance in predicting LNM in PTC patients (OR: 2.96, 95% CI: 1.05-8.31, P = 0.040).
This study reveals an increased risk of lymph node metastasis (LNM) in younger patients with PTC, aligning with previous research findings. 37 Some researchers have proposed that in PTC patients under 45 years of age, the occurrence of LNM is associated with a decreased overall survival rate, advocating for a rigorous assessment of lymph node involvement. 6 Conversely, another relevant prognostic study has indicated that LNM in young patients under 45 years of age has a relatively minor impact on prognosis. 38 However, this study lacks patient survival data and did not conduct a survival prognosis analysis, which should be addressed in future experiments. Furthermore, the occurrence and development of PTC are closely related to the immune microenvironment. 39 In the cellular ecosystem of PTC, immune-related factors significantly influence the tumor microenvironment. 40 Research indicates that PTC associated with neutrophil infiltration and (or) elevated platelet counts tends to exhibit increased aggressiveness.41,42 Neutrophils can suppress apoptosis and induce tumor angiogenesis by releasing inflammatory cytokines and phagocytic mediators, thereby altering the tumor microenvironment. 43 Additionally, platelet-derived growth factor receptor alpha (PDGFRα) induces cytoskeletal reorganization, which drives an invasive cell phenotype, promotes the formation of functional invasive pseudopodia in PTC cells, and facilitates lymph node metastasis.44,45 Preoperative blood immune indicators may serve as predictive markers for PTC. Some researchers have identified high NRL 46 and high SII 47 as adverse prognostic indicators for PTC patients. This study incorporated blood immune indicators such as LMR, PLR, NLR, and SII, but the results indicate that these indicators lack statistical significance in relation to LNM (P > 0.05). Future research with larger sample sizes is still needed to validate these findings.
Limitations
This study has several limitations. The sample size is relatively small, and the data for analysis all originate from a single institution. Additionally, retrospective analysis may introduce certain sample biases. Therefore, further research on the prediction of cervical lymph node metastasis in PTC would benefit from multi-center studies or larger clinical datasets.
Conclusions
We conducted a comprehensive analysis and identified age, FT3 levels, maximum tumor diameter, intratumoral microcalcifications, and ultrasound suspicion of lymph node metastasis as independent predictive factors for cervical lymph node metastasis (LNM) in patients with PTC. By integrating demographic characteristics, serological indicators, and conventional ultrasound features, we can guide personalized treatment for PTC patients, thereby avoiding unnecessary prophylactic lymph node dissection.
Supplemental Material
Supplemental Material - Prediction Model of Cervical Lymph Node Metastasis in Papillary Thyroid Carcinoma
Supplemental Material for Prediction Model of Cervical Lymph Node Metastasis in Papillary Thyroid Carcinoma by Huiting Chen, Li Zhu, Yong Zhuang, Xiaojian Ye, Fang Chen, and Jinshu Zeng in Cancer Control
Supplemental Material
Supplemental Material - Prediction Model of Cervical Lymph Node Metastasis in Papillary Thyroid Carcinoma
Supplemental Material for Prediction Model of Cervical Lymph Node Metastasis in Papillary Thyroid Carcinoma by Huiting Chen, Li Zhu, Yong Zhuang, Xiaojian Ye, Fang Chen, and Jinshu Zeng in Cancer Control
Footnotes
Authors’ Contributions
All authors have contributed significantly to the work described in the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Fujian Province under grant no. 2021J02028X.
Ethical Statement
Data Availability Statement
All data generated or analyzed during this study are included in this published article.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
