Abstract
Background
Hashimoto’s thyroiditis (HT) is a common autoimmune disease, and an increasing number of papillary thyroid carcinoma (PTC) cases are being diagnosed in HT patients. The aim of this study was to investigate the proportion and function of different T cell subtypes in peripheral blood, especially CD4+Foxp3+ regulatory T cells (Tregs), in PTC with and without HT. We also studied the effect of HT on PTC.
Methods
Flow cytometry was used to detect the proportions of CD4+ T cells, CD8+ T cells, CD4+Foxp3+ Tregs, and the level of interleukin-10 (IL-10) secreted by CD4+ T cells in the peripheral blood of PTC patients with and without HT. AimPlex bead-based immunoassays were used to determine serum cytokine levels.
Results
No significant differences were observed in the proportion of CD4+ or CD8+ T cells in the two groups of PTC patients. The percentage of CD4+FoxP3+ Tregs and their levels of IL-10 were significantly lower in PTC patients with HT than in those without HT.
Conclusions
Our findings suggest that CD4+Foxp3+ Tregs have vital regulatory functions in the initiation and progression of PTC by secreting the inhibitory cytokine IL-10.
Keywords
Introduction
Papillary thyroid carcinoma (PTC) is the most common type of thyroid cancer, representing 80% to 95% of all pathological thyroid diagnoses. PTC frequently co-exists with Hashimoto’s thyroiditis (HT), which is the most prevalent organ-specific autoimmune disease. 1 HT is characterized by the presence of thyroid-specific autoantibodies, chronic lymphocytic infiltration of the thyroid gland, and eventually, hypothyroidism. 2 Noureldine et al. 3 reported that lymphocytic infiltration in HT is frequently associated with PTC and may be a risk factor for developing this type of cancer; their findings suggest that overlapping molecular mechanisms govern the early stages of oncogenesis and inflammation in the thyroid gland. Moon et al. 4 demonstrated by meta-analysis that PTC patients with HT were associated with better clinicopathologic features and clinical outcomes that those without HT. Although several experimental studies have suggested an immunological link between the synchronous appearance of HT and PTC, the underlying mechanism remains unclear.4,5
Regulatory T cells (Tregs), formerly known as suppressor T cells, are a subpopulation of CD4+T cells that modulate the immune system and are characterized by expression of the transcription factor Forkhead box P3 (FoxP3), which is a specific marker for Tregs. Tregs are important for maintaining immune homeostasis and protecting the host against autoimmune diseases. 6 Recent studies have shown that Tregs are associated with tumor progression and poor prognosis. 7 The aim of this work was to explore associations between T cell subgroups, especially Tregs, with the initiation and progression of PTC by examining the levels and functions of different T cell subsets in PTC patients with and without HT.
Materials and methods
Study participants
Peripheral blood samples were collected before operations from PTC patients in the General Surgery Department of Tianjin Medical University General Hospital between October 2015 and July 2016. All patients were selected randomly and had no other autoimmune diseases, other malignancies, or recurrent tumors at the time of the investigation. The study was planned according to the ethical guidelines outlined in the Declaration of Helsinki. This project was approved by the Ethical Committee of Tianjin Medical University General Hospital. All patients gave informed consent prior to enrollment in the investigation.
Isolating peripheral blood mononuclear cells (PBMCs)
We isolated PBMCs with lymphocyte isolation medium (Solarbio, Beijing, China). A mixture of heparin-anticoagulated blood and an equal volume of saline was gently layered on top of the lymphocyte isolation medium using a 1-mL pipette. Layering was done slowly so that the blood and lymphocyte isolation media were in two layers. We aspirated the whitish buffy coat PBMCs that formed at the interphase after centrifuging the tubes at 400
Antibodies and cell surface marker staining
To stain for cell-surface markers, cell samples were stained with the following fluorescent dye-conjugated monoclonal antibodies: FITC-conjugated anti-human CD4, PE-conjugated anti-human Foxp3, Percp-Cy5.5-conjugated anti-human CD3, APC-conjugated anti-human CD8, and PE-conjugated anti-human IL-10, which were all purchased from BioLegend (San Diego, CA, USA).
Briefly, we resuspended cell pellets in 100 µL PBS and incubated them with an appropriate volume of fluorescently labeled antibodies accompanied by FcR-block in the dark for 20 minutes. We then washed the samples twice with cold PBS, and then resuspended them in 300 µL PBS for immediate flow cytometry detection. A BD FACS Canto II flow cytometer (BD Biosciences, San Jose, CA, USA) was used to make cell counts, and CD4+ T (CD3+CD4+) and CD8+ T (CD3+CD8+) cells were analyzed with BD FACS Diva software (BD Biosciences).
Foxp3 staining for flow cytometry detection
A Foxp3 staining kit (Ebioscience, San Diego, CA, USA) was used for Foxp3 staining. Briefly, we added 1 mL of 1× Foxp3 Fix/Perm solution to resuspended cell pellets and incubated the mixture at room temperature in the dark for 20 minutes. After washing and resuspending the cell pellets in 1× Foxp3 Perm buffer, the cells were added to an appropriate amount of PE-conjugated anti-human Foxp3 antibody and incubated at room temperature in the dark for 30 minutes. We washed the samples twice with cell staining buffer and resuspended them in 300 µL of PBS for flow cytometry. The CD3+CD4+Foxp3+ strategy was used to calculate the proportion of Tregs.
Intracellular cellular cytokine staining
For interleukin (IL)-10 staining, we stimulated isolated PBMCs with PMA (50 ng/mL; Sigma Aldrich, St. Louis, MO, USA) and ionomycin (1 mg/mL; Sigma Aldrich) in the presence of GolgiPlug (BD Bio-sciences) for 4.5 hours. The cells were then stained with Percp-Cy5.5-conjugated anti-human CD3 and FITC-conjugated anti-human CD4 antibodies followed by fixation, permeabilization, and staining with intracellular PE-conjugated anti-human IL-10. We washed the samples twice with cold PBS and suspended cell pellets in 300 µL of PBS for immediate flow cytometry detection.
Serum cytokine detection
Peripheral venous blood (approximately 2 mL) was obtained from all patients in the morning after fasting. Serum was collected after centrifuging at 1000
Statistical analysis
All statistical analyses were performed using SPSS for Windows, version 16.0 (SPSS Inc., Chicago, IL, USA) or GraphPad Prism 6.0 (GraphPad Software, Inc., San Diego, CA, USA). Data are expressed as mean ± standard deviation and were analyzed using the independent t-test or non-parameter (Mann–Whitney) test after testing for normality (Shapiro–Wilk test). Spearman rank correlations were used to analyze correlations between tumor sizes and the immunological parameters. The comparison of patients’ sex and age were by the Fisher’s exact probability test. P values <0.05 were considered statistically significant.
Results
Among the 35 PTC patients included in this study, 10 were diagnosed with HT and 25 did not have HT. First, we found that all patients comorbid for PTC and HT were female. Therefore, we included the sex ratio in our analysis. Because our cohort had less than 40 participants, we used Fisher’s exact probability test to determine if the observed difference in the age composition between patients with/without HT was statistically significant and found that it was not (Table 1). Similarly, no significant difference was detected in the overall age composition between the two groups (Table 1). The reason for this result may be because PTC combined with HT most frequently occurs in middle-aged women. We also compared clinical measurement data for thyroid function and found that thyroglobulin (TG), thyroid stimulating hormone (TSH), TG antibody (TGAb), and thyroid peroxidase antibody (TPOAb) were increased markedly in PTC patients with concurrent HT. No significant differences were seen in serum free triiodothyronine (FT3) or free tetraiodothyronine (FT4) levels between the two groups (Table 2). These results conformed to the clinical diagnoses and physiological evaluations.
Characterization of the included patients with thyroid diseases.

Notes: PTC: papillary thyroid carcinoma; HT: Hashimoto’s thyroiditis.
Clinical hematology results (mean ± standard deviation).

Notes: PTC: papillary thyroid cancer; HT: Hashimoto’s thyroiditis; FT3: free triiodothyronine; FT4: free tetraiodothyronine; TSH: thyroid-stimulating hormone; TG: thyroglobulin; TGAb: thyroglobulin antibody; TPOAb: thyroid peroxidase antibody.
1Comparison between the two groups, P < 0.05; 2comparison between the two groups, P < 0.01.
We next analyzed the percentages of T cell subtypes (CD4+ T cells, CD8+ T cells, and CD4+Foxp3+ Tregs) in peripheral blood samples. No differences were observed in the proportions of peripheral CD4+ or CD8+ T cells in PTC patients with or without HT (Figure 1a–c). However, the proportion of CD4+Foxp3+ Tregs was significantly decreased in the PTC+HT group compared with the PTC-only group (Figure 1d,e). There were no significant differences in Treg levels in groups divided by clinicopathological features, including tumor size, multifocality, nodal metastasis, and TNM stage (Table 3). Furthermore, there were no correlations between tumor size and the proportions of CD4+ T cells, CD8+ T cells, or CD4+Foxp3+ Tregs (rs = 0.023, 0.066, and 0.010; P = 0.9002, 0.7256, and 0.9595, respectively). We also detected serum concentrations of IL-10 and IL-35, both of which are the primary effectors of Tregs. Serum IL-10 levels were significantly lower in PTC patients with HT, whereas no significant difference was found in IL-35 levels (Figure 2). These results indicate that CD4+ T cells are the main source of IL-10 and that their ability to secrete cytokines is decreased in PTC patients with HT (Figure 3).
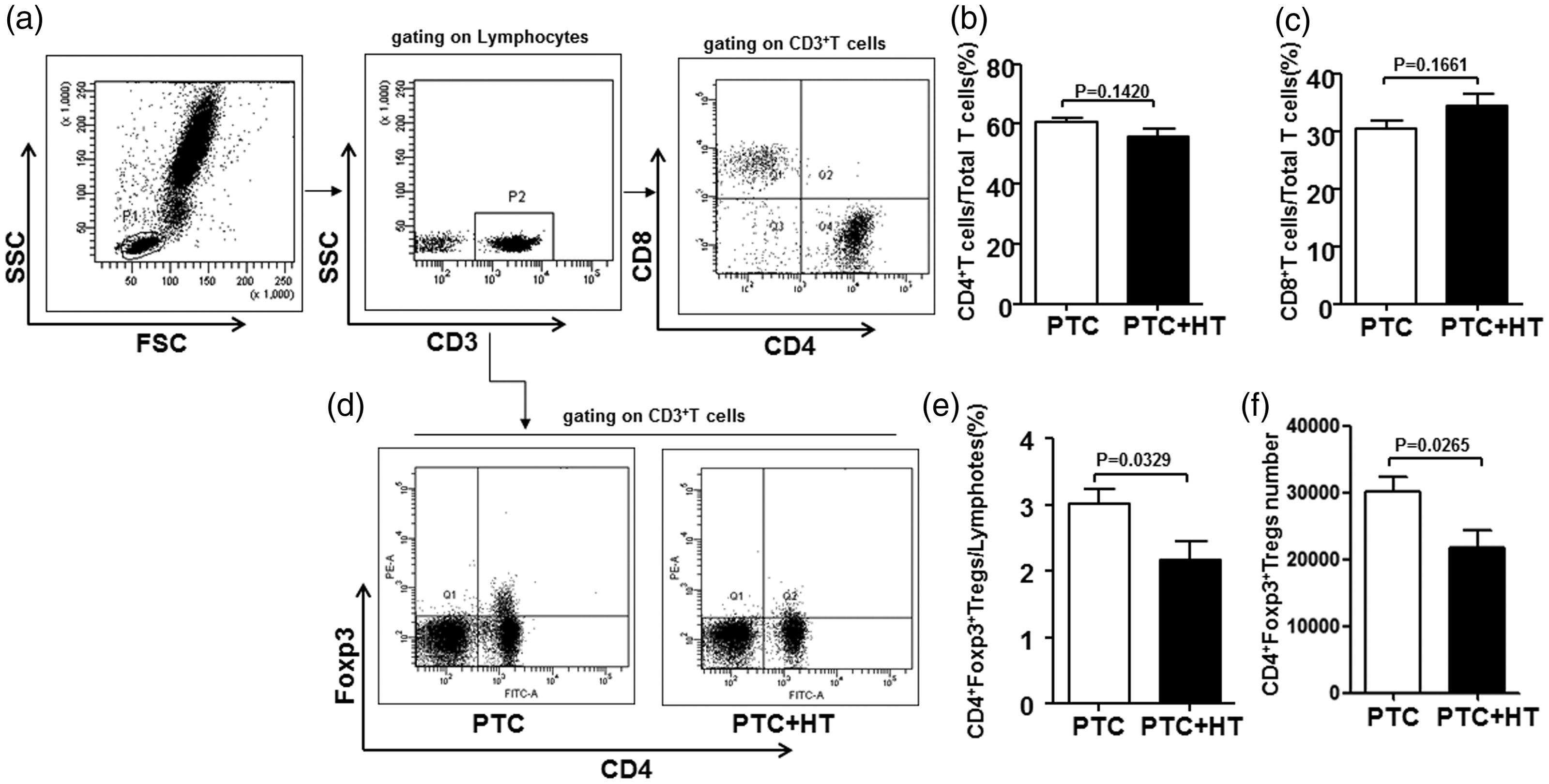
Decreased levels of Tregs in the peripheral blood of PTC patients with concurrent HT. Isolated lymphocytes from PTC patients with and without HT were labeled with FITC-anti-CD4, APC-anti-CD8, Percp-cy5.5-anti-CD3, or PE-anti-Foxp3 antibodies and analyzed by flow cytometry. (a) Representative staining of CD4+ and CD8+ T cells. All cells were gated on CD3 staining. (b) Mean and standard deviation values for CD4+ T cell proportions from all patients. (c) Mean and standard deviation values for CD8+ T cells proportions from all patients. (d) Representative staining of CD4+Foxp3+ Tregs. (e) Mean and standard deviation values for CD4+Foxp3+ Treg proportions from all patients. (f) Mean and standard deviation values for CD4+Foxp3+ Treg numbers from all patients. Tregs: regulatory T cells; PTC: papillary thyroid carcinoma; HT: Hashimoto’s thyroiditis.
The relationship between tumor invasiveness and regulatory T cells (Tregs).

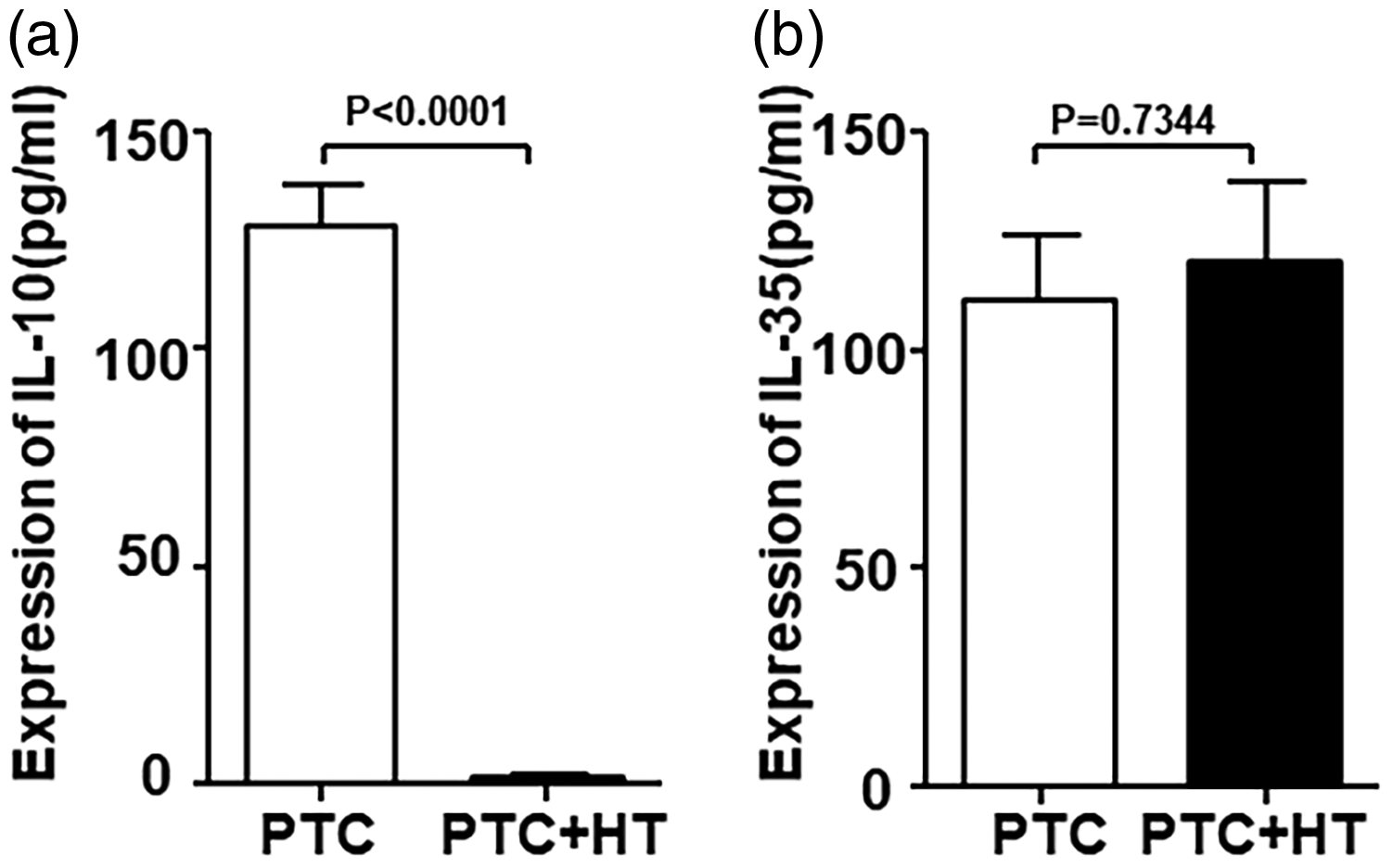
Decreased IL-10 levels in the peripheral blood of PTC patients with HT. Serum was collected from all patients to detect IL-10 and IL-35 concentrations. Graphs show the mean concentrations of (a) IL-10 and (b) IL-35 in the peripheral blood of PTC patients without HT (PTC,

CD4+ T cells from PTC patients with HT produced less IL-10. Isolated lymphocytes from PTC patients with and without HT were labeled with FITC-anti-CD4 and PE-anti-IL-10 antibodies and analyzed by flow cytometry. (a) Representative staining of IL-10+CD4+ T cells. All cells were gated on CD4 staining. (b) Mean and standard deviation values for IL-10+CD4+ T cell proportions.
Discussion
There is a functional relationship between chronic immune responses and cancer. HT is characterized by diffuse lymphocytic infiltration, fibrosis, and lymphoid follicles, also known as chronic lymphocytic thyroiditis. PTC is the most common condition that co-develops in primary HT patients. 8 In the presence of HT, PTC patients have more favorable clinical outcomes for recurrence and mortality. Dvorkin et al. 9 showed that HT is associated with smaller primary tumors and less lymph node metastasis. It was later shown that PTC patients with coexisting HT need less radioactive iodine treatment, indicating a more favorable prognosis. 10 Thus, HT may have a protective effect in PTC patients. This study aimed to determine the effect of Tregs on the development of PTC in HT patients. Although more female than male patients were included in our study, no significant differences were observed in the sex or age ratios. The reason for this finding might be that PTC mostly occurs in young middle-aged women and has an overall female-to-male ratio of 4:1. 11 This result suggested that estrogen and its receptors do not account for the difference in performance of PTC-only and PTC accompanied by HT patients.
Previous studies have shown that PTC leads to an increased proportion of Tregs. High Treg frequency in primary thyroid tumors is associated with aggressiveness, advanced tumor progression, and poor prognosis. 12 Elevated Treg levels in the tumor microenvironment can protect tumors and reduce the effect of immunological antitumor attacks.9,13 We detected the proportions of peripheral T lymphocyte subgroups and found no significant changes in the percentages of CD4+ or CD8+ T cells (Figure 1a–c). Conversely, both the absolute number and the proportion of CD4+Foxp3+ Tregs were significantly decreased in PTC+HT patients compared with PTC-only patients (Figure 1d–f). We found no significant correlation between the proportion of CD4+Foxp3+ Tregs and several clinical indexes of tumor aggressiveness, including tumor size, multifocality, and nodal metastasis (Table 3). These findings indicated that Tregs might be important in the concurrence of PTC with HT, but less associated with malignant behaviors. To further explore mechanistic changes in these Tregs, we detected serum levels of three anti-inflammatory cytokines that are predominantly produced by Foxp3+ Tregs (IL-10, IL-35, and transforming growth factor-β [TGF-β]). Our results showed that serum levels of IL-10 were significantly decreased in PTC+HT patients compared with PTC only patients (Figure 2a). No difference was observed in peripheral IL-35 (Figure 2b) levels between the two groups and no TGF-β was detected.
To determine the source of serum IL-10, we isolated PBMCs and analyzed IL-10 secretion levels by CD4+ and CD8+ cells. Flow cytometry showed that IL-10 was primarily expressed by CD4+ T cells and showed a significant decrease in PTC+HT patients compared with PTC-only patients. Because Foxp3+ Tregs are a major source of IL-10, we propose that the decrease in CD4+FoxP3+ Treg numbers might be the cause for reduced IL-10 levels in patients with PTC accompanied by HT.
In summary, these findings suggest that Tregs play a regulatory role in concurrent PTC and HT by having reduced IL-10 secretion, suggesting IL-10 could be an immune-based preventative therapy for PTC patients. However, many questions still need to be answered, including how the timing of IL-10 treatment would be scheduled and its exact molecular mechanism.
Abbreviations used in this paper
PTC: papillary thyroid carcinoma
HT: Hashimoto’s thyroiditis
FT3: free triiodothyronine
FT4: free tetraiodothyronine
TSH: thyroid-stimulating hormone
TG: thyroglobulin
TGAb: thyroglobulin antibody
TPOAb: thyroid peroxidase antibody
IL: interleukin
TGF-β: transforming growth factor-β.
