Abstract
Objective
The incidences of papillary thyroid carcinoma (PTC) and Hashimoto’s thyroiditis (HT) have shown increasing trends. Numerous studies have shown a close relationship between the two diseases, but the exact mechanism linking PTC with HT is still unclear. Interleukin-17 (IL-17) plays an important role in the development of malignant tumors. However, information on the association between IL-17 and thyroid disease is lacking.
Methods
Tissue samples were collected from patients with thyroid diseases admitted to the thyroid surgery department of our hospital between May 2015 and December 2017. The characteristics of the thyroid were observed by ultrasonography, hematoxylin-eosin staining, enzyme-linked immunosorbent assays, and immunohistochemistry.
Results
We found that HT with carcinoma (HTC) showed unique characteristics in two-dimensional ultrasound images. Moreover, IL-17 and vascular endothelial growth factor (VEGF) levels showed gradually increasing trends during the process of HT malignant transformation, with a significant positive correlation between the two cytokines. Serum IL-17 and VEGF levels could distinguish between HTC and HT with benign adenoma.
Conclusion
Our data suggest that serum IL-17 and VEGF levels may represent novel biomarkers for the diagnosis of HT malignant nodules.
Keywords
Introduction
Papillary thyroid carcinoma (PTC) is the most common pathological type of differentiated thyroid carcinoma, with an increasing incidence in many parts of the world in recent years. 1 Hashimoto’s thyroiditis (HT), an autoimmune disease accounting for approximately 22.5% of all thyroid diseases, has also demonstrated an increasing incidence. Interestingly, some studies found a higher incidence of PTC in patients with HT compared with non-HT patients. 2 Moreover, patients with PTC complicated with HT had a younger age of onset, a higher proportion of females, higher levels of thyrotropin, smaller tumor diameter, and higher probabilities of lymph node metastasis and membrane invasion, consistent with the poorer prognosis in patients with PTC complicated with HT.3,4 Importantly, HT may affect the efficacy of postoperative risk-adapted radioiodine therapy in PTC patients. 5 There is thus a well-recognized close relationship between the pathogeneses of PTC and HT; however, the exact mechanism is not clear.
Interleukin-17 (IL-17), first reported by Yao in 1995, 6 is a proinflammatory cytokine that is mainly secreted by T helper-17 (Th17) cells in the initial CD4+ T cell subset. IL-17 secretion contributes to epithelial homeostasis, acute inflammatory responses, and B cell stimulation after appropriate stimuli, acting as a bridge between the innate and acquired immune responses. 7 Notably, numerous studies have revealed that IL-17 plays important roles in various diseases, including infectious and autoimmune diseases, cardiovascular disorders, nonalcoholic fatty liver disease, and hematological and solid cancers. 8
Emerging evidence has indicated that IL-17 is highly expressed in malignant tumors, such as gastric carcinoma, colorectal cancer, non-small cell lung cancer, and hematologic cancers.9–12 Importantly, IL-17 may serve as a potential diagnostic biomarker for various malignant tumors, such as oral squamous cell carcinoma, breast cancer, stomach cancer, and lung cancer.13,14
Several studies have shown that IL-17 is highly expressed in both HT and PTC.15,16 Vascular endothelial growth factor (VEGF) plays an important role in the progression of various tumors.17,18 To better distinguish HT with PTC (HTC) from HT with benign nodules (HTB), we observed the ultrasound (US) characteristics of the thyroid during the malignant transformation of HT and analyzed the pathological changes using hematoxylin and eosin (HE) staining. We measured serum IL-17 and VEGF levels by enzyme-linked immunosorbent assays (ELISAs) and tissue levels by immunohistochemistry (IHC). We used receiver operating characteristic (ROC) curves to distinguish between these patients, and analyzed the areas under the curves (AUCs) to determine the efficacies of these markers for distinguishing between HTB and HTC.
Materials and methods
Patients and specimens
Tissue samples were collected from patients with thyroid diseases admitted to the thyroid surgery department of Longyan First Affiliated Hospital of Fujian Medical University between May 2015 and December 2017. Patients with other diseases, such as tumors, infection, hypertension, myocardial disease, and desmosis, were excluded. Patients were divided into HT, HTB, PTC, and HTC groups according to the pathologic results of US-guided puncture biopsy. Healthy volunteers without any disease were recruited from the Health Management Division of Longyan First Affiliated Hospital of Fujian Medical University between May 2015 and December 2017 as a control (HC) group.
All participants signed the study protocol and informed consent for voluntary enrollment. The study was approved by the Institutional Review Board of Longyan First Affiliated Hospital of Fujian Medical University.
US
The characteristics of the thyroid gland were observed by US according to the peak systolic velocity of the superior thyroid artery (PSV) and resistance index (RI). The detailed classification was as follows: grade 0: no blood flow signal in the thyroid parenchyma indicating normal thyroid blood flow; grade I: thyroid parenchyma dotted with strips and patches of color signals, with no fusion, and colored area <1/3 of the thyroid area; grade II: thyroid parenchyma scattered with patchy blood flow signals, partially fused into a large color mosaic, with the color area 1/3 to 1/2 of the thyroid area; and grade III: thyroid gland filled with color blood flow signals, forming a large fused mosaic with a color area >2/3 of the thyroid area. Focal changes in thyroid blood flow were classified as follows: type 0: sparse blood flow signal, star-like or no blood flow display; type I: medium blood flow signal, showing more dots and short strips; type II: abundant blood flow signals, showing streaks and small patches; and type III: rich blood flow signal.
HE staining
HE staining was performed according to a previous study. 19 Briefly, thyroid tissues were fixed with 10% buffered formalin for 24 hours, dehydrated in a graded ethanol series, embedded in paraffin wax, cut, stained with HE, and examined under a light microscope.
ELISA
Serum was extracted and stored at −80°C. The samples were then analyzed using ELISA kits for IL-17 and VEGF (eBioscience, San Diego, CA, USA) in accordance with the manufacturer’s instructions.
IHC
IHC staining of puncture biopsy specimens was performed according to a previous study. 20 Briefly, slides were blocked sequentially with 0.3% hydrogen peroxide and goat serum, followed by incubation with rabbit anti-human polyclonal antibodies against CD34 (1:500 dilution, Signalway Antibody, Ontario, Canada) and VEGF (1:500 dilution, Signalway Antibody) at 4°C overnight, and then probed with biotinylated goat anti-rabbit secondary antibody (Vector Laboratories, CA, USA) and high-sensitivity streptavidin–horseradish peroxidase conjugate (Vector Laboratories). To visualize staining, slides were incubated in 3, 3′-diaminobenzidine in 0.1% hydrogen peroxide in Tris–HCl buffer and subsequently counterstained with hematoxylin QS (Vector Laboratories).
The intensities of VEGF staining were scored as the proportion of positive cells as follows: 0: <5%; 1+: 6%–25%, 2+: 26%–50%; and 3+: >50%. The staining intensity was evaluated as follows: 0: none; 1: weak; 2: medium; and 3: strong. The total score was calculated by multiplying the two subscores. Scores >6 were strongly positive (+++), 4–6 were moderately positive (++), 2–3 were weakly positive (+), and 0–1 were negative (−).
Statistical analysis
All data were presented as the mean ± standard deviation. Statistical analysis was carried out using SPSS Statistics for Windows, Version 17.0 (SPSS Inc., Chicago, IL, USA). Parametric data were compared using one-way ANOVA followed by
Results
Characteristics and clinical information for patients and controls
The patients were divided into four groups according to the pathological results of US-guided puncture biopsies: the HT group (n = 50), HTB group (n = 30), PTC group (n = 60), and HTC group (n = 30). Thirty healthy volunteers were also included (HC group). Detailed clinical information is given in

Typical pathological features of patients. Typical pathological features of patients with Hashimoto’s thyroiditis (HT), HT with benign adenoma (HTB), papillary thyroid carcinoma (PTC), and HT with carcinoma (HTC). Hematoxylin and eosin stain. Original magnification: 100× and 400×.
We compared the clinical characteristics among the five groups. There was no difference in age among the groups based on variance analysis (F = 0.579) and no difference in sex based on χ2 test results (χ2 = 1.031).
US characteristics of HTC
We also analyzed the US characteristics of the thyroid in patients with HT, HTB, and HTC. Normal thyroid parenchyma showed even echoes and a smooth capsule (Figure 2a). In contrast, the thyroid gland in patients with HT showed thickened parenchymal echoes, uneven distributions, and a rougher capsule (Figure 2b). No blood flow signals were observed around and inside the nodules in the thyroid in HTB patients (Figure 2c), while HTC patients showed type-1 blood flow signals in the periphery, but no obvious blood flow signals in the interior of the thyroid. More obviously, the thyroid nodules in HTC patients showed an unclear boundary, irregular shape, high aspect ratio, more visible microcalcification or large calcification, partial calcification, solid hypoechoicity, non-uniform internal echo, and echo attenuation (Figure 2d).

Thyroid ultrasound features of patients. Thyroid ultrasound features of (a) healthy controls, and patients with (b) Hashimoto’s thyroiditis (HT), (c) HT with benign adenoma (HTB), and (d) HT with carcinoma (HTC). (e) Low-resistance blood flow signals measured by Doppler ultrasound in the peripheral spectrum of thyroid nodules in HTB patients; (f) high-resistance blood flow signals measured by spectral Doppler ultrasound in thyroid nodules of HTC patients.
We further focused on the differences between HTB and HTC patients. The artery blood flow PSV was significantly higher in thyroid US images in patients with HTC compared with HTB (43.2 ± 11.2 vs. 20.2 ± 10.2, P < 0.001), accompanied by a higher blood flow RI (0.80 ± 0.11 vs. 0.58 ± 0.10, P < 0.001). Moreover, the US incidences of microcalcification (73.3% vs. 16.7%, P = 0.022), a thyroid nodule aspect ratio >1 (80.0% vs. 6.7%, P = 0.003), and a low thyroid echo ratio (90.0% vs. 43.3%, P = 0.004) were all significantly higher in patients with HTC compared with HTB. Patients with HTC also had a significantly higher rate of suspicious abnormal lymph nodes in the neck (76.7% vs. 0%, P < 0.001) (Supplementary Table 2).
We detected low-resistance and high-resistance blood flow signals of the thyroid nodules in patients with HTC and HTB by spectral Doppler US (Figure 2e and f). High-resistance blood flow signals were mainly observed in thyroid nodules in HTC patients, while low-resistance blood flow signals were mainly observed in the periphery of thyroid nodules in HTB patients.
We compared the blood flow distribution of thyroid nodules between patients with HTB and HTC. The main blood flow type in HTC nodules was type I, while the main blood flow types in HTB nodules were types II and III. The blood flow grades of HTC nodules were mainly grades I and III, while HTB nodules were mainly grades I and II (Supplementary Table 3).
We further compared the blood flow parameters of nodules in HTB and HTC patients. The internal artery PSV of nodules in HTC patients was significantly increased compared with nodules in HTB patients (50.1 ± 2.2 vs. 42.1 ± 5.01, P < 0.05), and the RI was also significantly increased (0.72 ± 0.02 vs. 0.67 ± 0.06, P < 0.05) (Supplementary Table 4).
High serum levels of IL-17 and VEGF
We compared serum IL-17 levels among the five groups by ELISA. Serum IL-17 levels increased gradually from the HC group to the HT, HTB, PTC, and HTC groups. Compared with the HC group (5.96 ± 1.37 pg/mL), IL-17 levels were significantly higher in the HT (8.08 ± 1.19 pg/mL, P < 0.001) and HTB groups (8.52 ± 0.91 pg/mL, P < 0.001), and compared with the HTB group (8.52 0.91 pg/mL), levels were significantly higher in the PTC (9.20 ± 1.33 pg/mL) and HTC groups (10.18 ± 1.12 pg/mL) (both P < 0.001) (Figure 3a and Table 1). A previous study indicated that the combination of IL-17 and VEGF could serve as a potential diagnostic biomarker for oral squamous cell carcinoma, 10 and we therefore detected serum VEGF levels in the five groups and observed similar trends (Figure 3b). Compared with the HC group (165.67 ± 15.88 ng/mL), serum VEGF levels were significantly higher in the HT group (188.00 ± 26.29 ng/mL, P < 0.001), and compared with the HTB group (198.67 ± 23.35 ng/mL), serum levels were significantly higher in both the PTC (227.08 ± 27.51 ng/mL) and HTC groups (233.70 ± 9.21 ng/mL) (both P < 0.001) (Figure 3b and Table 1).

Serum levels of interleukin (IL)-17 and vascular endothelial growth factor (VEGF). (a) Serum IL-17 levels and (b) serum VEGF levels in the healthy control (HC), Hashimoto’s thyroiditis (HT), HT with benign adenoma (HTB), papillary thyroid carcinoma (PTC), and HT with carcinoma (HTC) groups. (c) Positive relationship between serum IL-17 and VEGF levels in patients. (d) Receiver operating characteristic curve analysis of serum IL-17 and VEGF levels from HTC and HTB patients.
Serum levels of interleukin-17 and vascular endothelial growth factor in each group.

*HC vs. HT, P < 0.001; **HTC vs. HTB, P < 0.001.
HC, healthy control; HT, Hashimoto’s thyroiditis; HT with benign adenoma; PTC, papillary thyroid carcinoma; HTC, HT with carcinoma.
We further explored the relationship between serum IL-17 and VEGF by Pearson’s correlation analysis. Serum VEGF levels were positively correlated with IL-17 levels (r = 0.456, P < 0.0001, 95% confidence interval (CI): 0.341–0.561) (Figure 3c). These data showed that serum VEGF and IL-17 levels were significantly correlated in patients with thyroid diseases.
The clinical prognosis of HTC is relatively poor; however, it is important to differentiate between HTC and HTB in terms of the clinical diagnosis and treatment. We therefore analyzed the abilities of IL-17 and VEGF to differentiate between HTB and HTC patients based on ROC curves. The AUC of serum IL-17 level was 0.904 (95% CI: 0.831–0.976, specificity 80.4%, sensitivity 80.2%, cut-off value of 9.395 pg/mL, Youden’s index: 0.606) (Figure 3d), and the AUC of serum VEGF was 0.885 (95% CI: 0.80–0.969, specificity 70.5%, sensitivity 99.1%, cut-off value of 216.5 ng/mL, Youden’s index: 0.696) (Figure 3d). These two markers showed good abilities to differentiate between HTC and HTB, indicating that serum levels of IL-17 and VEGF might serve as new tools for the clinical diagnosis of HTC.
High expression of VEGF in PTC and HTC
We confirmed the protein expression of VEGF in thyroid tissues by IHC staining of thyroid biopsy samples. VEGF protein was mainly expressed in follicular epithelial cells and vascular endothelial cells. We quantified and graded the IHC results according to the intensity and range of VEGF staining (Figure 4a and b). Consistent with the serum levels VEGF, tissue expression of VEGF was mainly negative and weakly positive in the HT and HTB groups, but mainly positive and strongly positive in the PTC and HTC groups. Tissue expression levels differed significantly among the four groups according to χ2 test (P < 0.001) (Table 2).
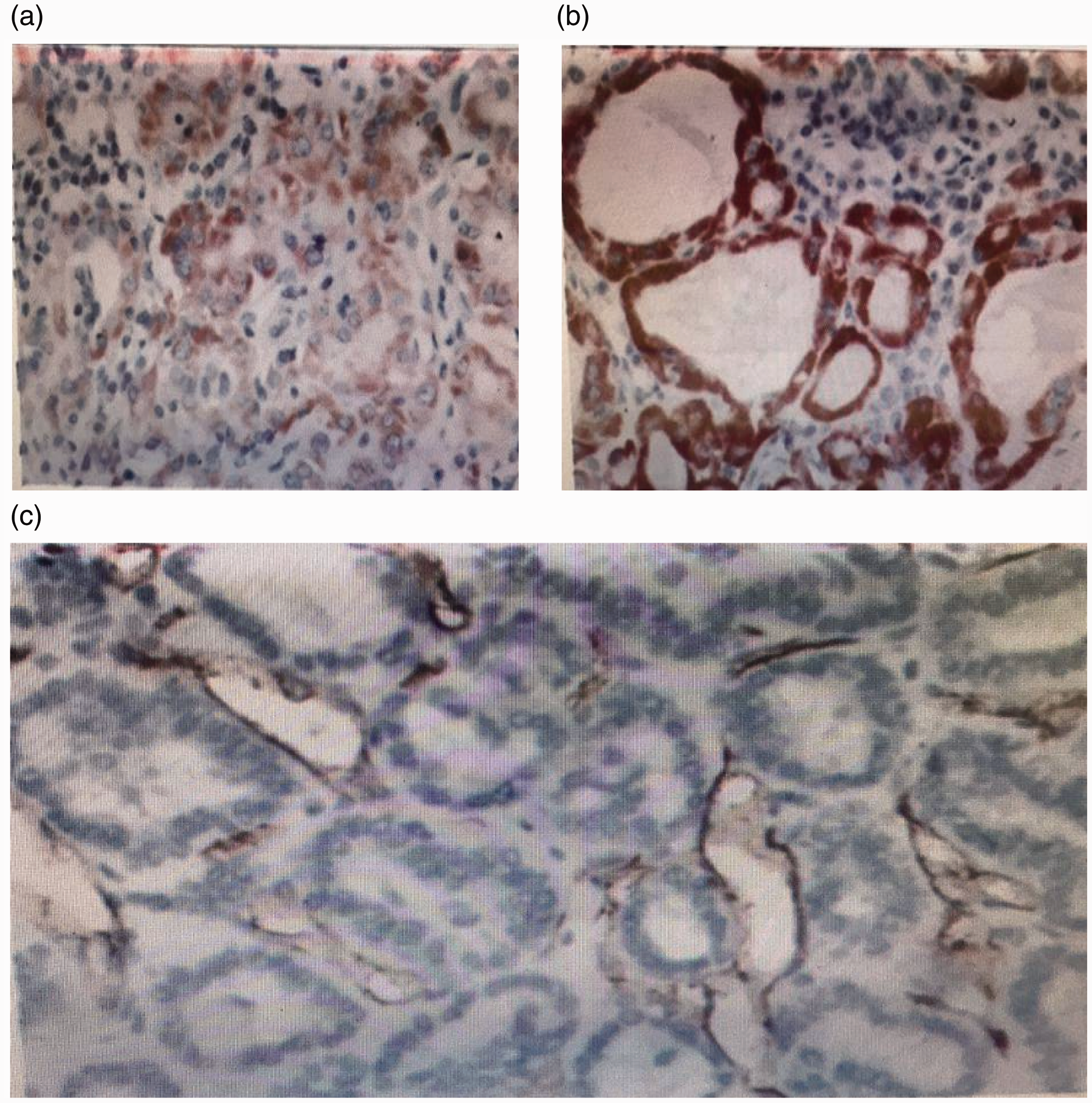
Vascular endothelial growth factor (VEGF) protein expression and distribution in thyroid follicular epithelial cells demonstrated by immunostaining. (a) Typical weakly positive and (b) positive pathological features of VEGF expression in thyroid tissue. (c) CD34 was positively expressed in vascular endothelial cells in thyroid tissue. Original magnification: 100×.
Expression of vascular endothelial growth factor in thyroid tissues.

VEGF, vascular endothelial growth factor; HT, Hashimoto’s thyroiditis; HT with benign adenoma; PTC, papillary thyroid carcinoma; HTC, HT with carcinoma.
High microvascular density (MVD) in PTC and HTC patients
Based on the high expression of VEGF in thyroid tissues from HTC patients, we determined the levels of angiogenesis by analyzing CD34-labeled microvessel density (MVD). 21 MVD was similar in HT and HTB patients and in PTC and HTC patients. However, MVD was significantly higher in patients with PTC and HTC compared with HTB (both P < 0.0001) (Figure 4c and Table 3). These data were consistent with the trend in VEGF expression.
Microvascular density in thyroid tissues in different groups.

*Comparison of HTC and HTB P < 0.001.
MVD, microvascular density; HT, Hashimoto’s thyroiditis; HT with benign adenoma; PTC, papillary thyroid carcinoma; HTC, HT with carcinoma.
Discussion
Some previous studies found that the incidence of PTC was significantly higher in HT than in non-HT patients.2,22 Moreover, patients with PTC complicated with HT have a poorer prognosis than PTC patients without HT.3,4 There is thus a close relationship between the pathogeneses of these two diseases. Although the exact mechanism linking these diseases is not clear, there are currently three main hypotheses: 1) HT is a precancerous lesion of PTC;2,23 2) the hypothesis of chronic inflammation; and 3) HT and PTC have common causes, potentially including immune factors (both thyroid globulin antibody and microsomal antibody have been found in the blood of HT and PTC patients),24–26 endocrine factors (HT can cause hypothyroidism and increase reactive thyrotropin, which can in turn cause carcinogenesis),27–31 and radioactive factors (exposure to X-rays can lead to thyroid inflammation and carcinogenesis, via the same mechanism).32–37
In this study, we explored the possible relationships among HT, HTB, HTC, and PTC by focusing on their US characteristics and expression of IL-17 and VEGF. We showed that blood flow PSV, blood flow RI, rate of microcalcification, a thyroid nodule aspect ratio >1, low thyroid echo ratio, and rate of suspicious abnormal lymph nodes in the neck were all significantly increased in thyroid US manifestations in patients with HTC compared with HTB. High-resistance blood flow signals were mainly observed in thyroid nodules in HTC patients, while low-resistance signals were mainly observed in the periphery of thyroid nodules in HTB patients. Moreover, HTC nodules mainly showed blood flow type I and blood flow grades I and III, while HTB nodules mainly showed blood flow types II and III and blood flow grades I and II. In addition, the internal artery PSV and RI were significantly increased in nodules in HTC patients compared with nodules in HTB patients. Meanwhile, RI, PSV and spectral morphology differed significantly between PTC and benign nodules. The US morphology of HTC nodules was consistent with that of PTC,38,39 and the morphology and number of blood flow distributions differed significantly between benign and malignant HT nodules. The difference between HTB and HTC may aid the diagnosis of HTC.
Previous studies found that IL-17 was secreted by Th17 cells, γδ T cells, CD8+ memory cells, eosinophils, neutrophils, and monocytes, but mainly by the Th17 cell population.7,40 Some studies found high IL-17 expression and an increased Th17/Treg cell ratio in patients with HT, and a positive correlation with autoantibodies (anti-thyroid globulin antibody).15,41,42 The Th17/Treg balance contributes to the pathogenesis of HT.43–45 The current results showed that IL-17 was highly expressed in HT, suggesting that IL-17 may be related to the pathogenesis of HT and may participate in its occurrence and development.
In this study, serum IL-17 levels were significantly higher in the HTC compared with the HC group. However, there was no significant difference between the HTC and PTC groups, suggesting that IL-17 was closely related to PTC and HTC, possibly indicating a common mechanism, as noted above. A previous study indicated that VEGF could serve as a potential diagnostic biomarker for oral squamous cell carcinoma. 13 In this study, we found similar expression trends in VEGF and IL-17 in serum and thyroid tissues, and both markers showed good abilities for differentiating between HTC and HTB. These results therefore suggest that serum levels of IL-17 and VEGF might serve as new tools for the clinical diagnosis of HTC. Moreover, we also showed that MVD was higher in PTC and HTC patients compared with HTB patients, which was consistent with the trend in VEGF expression. There are also limitations to our study. The mechanisms of IL-17 and VEGF involved in HTC remain unclear. Future research will focus on this point.
Conclusion
Serum IL-17 and VEGF levels increased gradually during the malignant transformation of HT, suggesting that they may be involved in the progression from HTB to HTC. In addition, serum VEGF levels were positively correlated with IL-17 levels during the malignant transformation of HT. Serum IL-17 and VEGF levels can significantly distinguish between HTB and HTC, and may thus be useful new biomarkers for the diagnosis of HT malignant nodules.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211067121 - Supplemental material for Interleukin-17 and vascular endothelial growth factor: new biomarkers for the diagnosis of papillary thyroid carcinoma in patients with Hashimoto’s thyroiditis
Supplemental material, sj-pdf-1-imr-10.1177_03000605211067121 for Interleukin-17 and vascular endothelial growth factor: new biomarkers for the diagnosis of papillary thyroid carcinoma in patients with Hashimoto’s thyroiditis by Shuiping Li, Shilin Li, Min Lin, Zuolin Li, Jinghua He, Jincheng Qiu and Jiantang Zhang in Journal of International Medical Research
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
