Abstract
Objective
To investigate the correlations between multigene alterations and clinicopathological features in papillary thyroid carcinoma (PTC) samples.
Methods
In this retrospective study, 111 cytological specimens of thyroid nodules, including 74 PTC samples and 37 benign samples, were analyzed using a 22-gene mutation assay employing next-generation sequencing. Clinicopathological information was retrospectively collected and analyzed.
Results
Gene alterations were associated with a higher rate of lymph node metastasis (LNM) and thyroid capsular invasion, a lower rate of coexisting Hashimoto's thyroiditis, the classical PTC subtype, and younger age (<45 years). Among the 22 genes tested, the
Conclusions
Our findings will provide a more comprehensive understanding of the relationship between gene mutations and PTC and may contribute to improved PTC management.
Keywords
Introduction
Papillary thyroid carcinoma (PTC) accounts for the majority of thyroid cancer cases; 80% of thyroid cancer cases are papillary carcinoma. 1 In recent years, a considerable increase in the incidence of PTC has occurred in many parts of the world.2,3 Even after excluding factors such as the expanded use of ultrasonic technology and the more general acceptance of early cancer screening,4,5 the true PTC incidence rate has still increased in recent decades. 6 PTC is generally associated with a good prognosis and a high survival rate when diagnosed early, 7 with a 10-year survival rate of over 95%. 8 However, the risk of relapse or persistent disease is still approximately 30%.8–11 Currently, an increasing number of researchers are focusing on the possibility of selecting patient groups with different risks for unfavorable outcomes to match a particular therapeutic approach and improve PTC outcomes.12,13 Clinicopathologic factors correlated with unfavorable outcomes include male sex, older age, large tumor size, extrathyroid invasion, lymph node metastasis (LNM), and distant metastasis.9,14
Molecular characteristics are also potential prognostic factors for PTC patients. PTC and its molecular features have been widely researched in recent decades. The
Genetic alterations in PTC may be correlated with different clinicopathological features. The relationship between genetic alterations and clinicopathological factors can also indicate the relationship between genetic alterations and prognosis to some extent when prognosis-related information is unavailable. However, the correlations between gene mutations and clinicopathological features in PTC prognosis are still controversial. For example,
Methods
Patients and specimens
Cytologic samples of thyroid nodules obtained by fine-needle aspiration biopsy (FNAB) were collected from the First People’s Hospital of Jiangxia District, Wuhan City, between June 2021 and October 2022. All data were collected and analyzed retrospectively. Our study was approved by the ethics committee of the First People’s Hospital of Jiangxia District, Wuhan on 5 March 2021 (No. 2021019) and was conducted in accordance with the declaration of Helsinki and the National Guidelines for Diagnosis and Treatment of Thyroid Cancer 2022 in China. 21 Written informed consent was obtained from all individual participants included in the study. All patients with thyroid nodules were diagnosed by ultrasonography and underwent FNAB or thyroidectomy to determine the final diagnosis. The reporting of this study conforms to the STROBE guidelines.22,23 The procedures used complied with the relevant guidelines and regulations. All patient details were de-identified.
Pathological review
Medical records, pathologic evaluations, and surgical pathology reports were reviewed to collect the demographic parameters and clinicopathologic characteristics of tumors. FNAB cytologic sample slides were examined and confirmed by two experienced pathologists, and if necessary, a third pathologist provided final confirmation of the different pathological tumors. Seven tumor pathological features were evaluated and recorded, including 1) tumor size (≤1 or >1 cm), 2) LNM, 3) thyroid capsular invasion, 4) multifocality, 5) presence or absence of Hashimoto's thyroiditis (HT), 6) presence or absence of nodular goiter, and 7) PTC subtype.
DNA extraction
Genomic DNA was extracted from cytological sample sections with at least 20% tumor cells using the QIAmp DNA Mini Kit (Qiagen, Hilden, Germany) following the manufacturer’s instructions. Then, 1% agarose gel electrophoresis was performed for DNA qualification. The concentrations of all samples were quantitated using a NanoDrop system (Invitrogen Life Technologies, Carlsbad, CA, USA) and a Qubit Fluorometer (Invitrogen Life Technologies).
Next-generation sequencing (NGS)
NGS was performed using the OncoAim® thyroid cancer multigene assay kit (Singlera Genomics, Inc., Shanghai, China). The OncoAim kit included 22 genes, as shown in Table 1, Supplementary Table 1, and Supplementary Table 2. The multigene assay kit was designed to simultaneously detect hot spot variations in these 22 genes, including single nucleotide variations, short insertions and deletions, and gene rearrangements. All 22 genes had been previously detected in tumor tissue of PTC patients and shown to include “pathogenic” or “likely pathogenic” variations, such as
Gene list of the OncoAim® thyroid cancer multigene assay kit.

Sequencing data were processed following the guidelines of the OncoAim® kit (Singlera). Sequencing reads were first quality-filtered using Trim-Glore software (version 0.4.0, https://www.babraham.ac.uk/science-facilities/bioinformatics) and then mapped to the reference human genome (hg19) using BWA software (version 0.7.12, http://sourceforge.net/projects/bio-bwa/files/bwakit/). After mapping, FreeBayes software (version 1.0.2, https://travis-ci.org/ekg/freebayes) was used to call single nucleotide variations and short insertions and deletions, and Blastall software (version 2.2.20, https://blast.ncbi.nlm.nih.gov/Blast.cgi) was used to call gene fusion. Hgvs annotation for single nucleotide variations and short insertions and deletions was performed using Ensembl Variant Effect Predictor (release-84, http://www.ensembl.org/info/docs/tools/vep/script/index.html). The minimum confidence threshold for variant calling was set to 2%. The gene variation result was classified as pathogenic, likely pathogenic, inconclusive, uncertain significance, likely benign, or benign according to the information from the ClinVar database. In this study, “pathogenic” or “likely pathogenic” results were considered positive.
Statistical analysis
SPSS statistical software (version 27.0, IBM Corp., Armonk, NY, USA) was used for all statistical analyses. R software (www.r-project.org) was used to create all figures. Continuous data are expressed as the mean ± standard deviation and were analyzed via an independent Student’s t-test or the Wilcoxon–Mann–Whitney test. Categorical data are expressed as quantities and percentages (n, %) and were analyzed via Pearson’s
Results
Detection of mutations
In total, 142 thyroid nodule cytologic samples were collected from 135 enrolled patients at the First People’s Hospital of Jiangxia District, Wuhan City, between June 2021 and October 2022. Of the 142 samples, 111 were available for analysis (Figure 1), including 74 PTC nodule samples and 37 benign nodule samples. Seventy-four malignant thyroid nodule samples were obtained from 71 PTC patients, including 22 men and 49 women, with ages ranging from 21 to 71 years and an average age of 46 years. The remaining 37 benign samples were from 37 patients with benign thyroid disease, including 7 men and 30 women, with ages ranging from 21 to 77 years and an average age of 55 years. All malignant thyroid nodules were histologically diagnosed as PTC after initial surgeries. Of the patients with benign nodules, 21 underwent thyroidectomy and were diagnosed with benign results, while the other 16 patients underwent FNAB and received non-malignancy results based on the Bethesda class 24 [class I (n = 1); class II (n = 11); class III (n = 4)]. Information regarding the samples is shown in Table 2 and Supplementary Table 3.

Consort diagram.
Clinicopathologic characteristics and mutation information of PTC and benign nodules.

LNM, lymph node metastasis; HT, Hashimoto’s thyroiditis; PTC, papillary thyroid carcinoma; SD, standard deviation; NG, nodular goiter.
Age and sex calculations were based on patient information; other data calculations were based on nodule information.
Among the 74 PTC cytological samples, 79.7% (59/74) exhibited at least one mutation of the 22 genes tested. Specifically, 62.2% (46/74) showed the presence of a

Landscape of mutation profiles in the PTC and benign groups. The waterfall diagram shows the cases with gene mutation only. PTC: papillary thyroid carcinoma. BTN: benign thyroid nodules.
The mutation rates were significantly different between the PTC group and the benign group (79.7% vs. 21.6%
Comparison of gene mutation rates between benign thyroid nodule patients and papillary thyroid carcinoma patients.
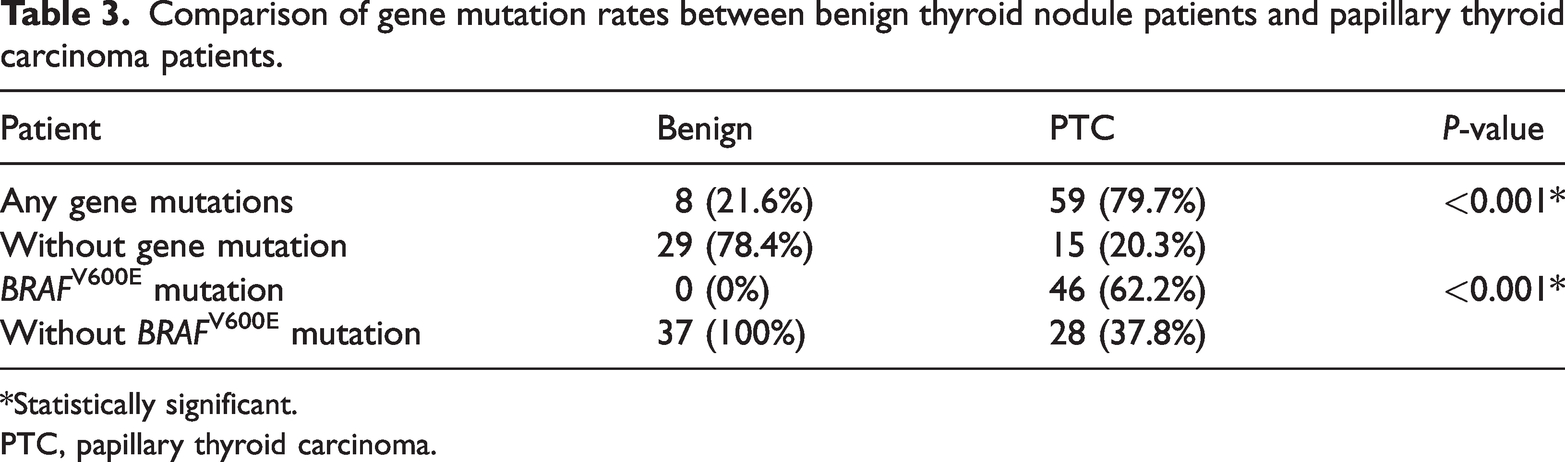
*Statistically significant.
PTC, papillary thyroid carcinoma.

Proportion of different gene alteration types in the PTC group and benign group (%). PTC: papillary thyroid carcinoma. BTN: benign thyroid nodules.
Correlation between gene mutations and clinicopathological features
Patient demographic characteristics, pathologic features of papillary carcinomas, and gene mutation information of the PTC group were analyzed and are summarized in Table 4. Gene mutations in tumors were associated with a higher rate of LNM (
Comparison of clinicopathological characteristics between patients with or without gene mutation and between patients with or without

LNM, lymph node metastasis; HT, Hashimoto’s thyroiditis, PTC, papillary thyroid carcinoma.
Statistically significant.
Association between LNM and clinicopathological characteristics or gene mutations
We performed univariate and multivariate analyses to identify risk factors for LNM. In the univariate analysis, LNM was associated with having gene mutations [odds ratio (OR) = 4.185, 95% confidence interval (CI): 1.188–14.746,
The association between LNM and clinicopathological characteristics or gene mutations in PTC.

LNM, lymph node metastasis; HT, Hashimoto’s thyroiditis; PTC, papillary thyroid carcinoma; OR, odds ratio; CI, confidence interval; NA, not applicable.
*Statistically significant.

Box and scatter plot showing the median and quartiles of age distribution of the LNM and non-LNM groups with papillary thyroid carcinoma. The horizontal axis indicates the groups with or without LNM; the vertical axis indicates age. The orange spots represent LNM samples and blue spots represent non-LNM samples. LNM: lymph node metastasis. Non-LNM: non-lymph node metastasis. *** indicates
Performance of a multigene assay in diagnosing cytologically indeterminate nodules
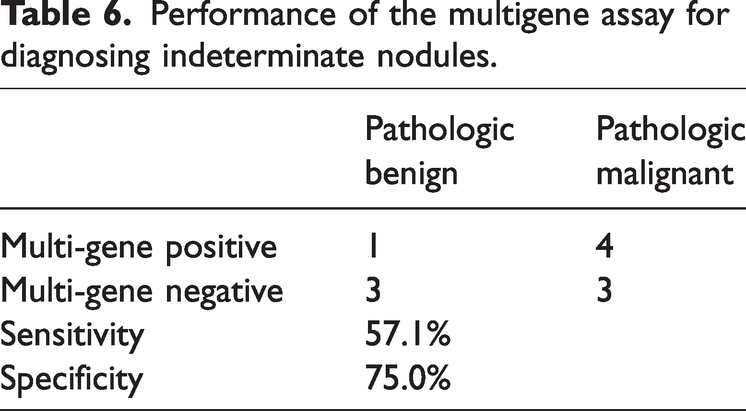
Among all cytological diagnosis samples, we identified 15 nodules classified as Bethesda class III or class IV, indicating indeterminate nodules. Among these 15 nodules, 11 underwent diagnostic surgical procedures to obtain the final pathological diagnoses; 7 were identified as positive and 4 were identified as negative. The sensitivity and specificity of the multigene test for these 11 nodules were 57.1% and 75.0%, respectively (Table 6 and Supplementary Table 6).
Performance of the multigene assay for diagnosing indeterminate nodules.
Discussion
PTC has a relatively high mutation rate, particularly for genes in the MAPK pathway, such as
There are also conflicting findings in previous studies regarding the relationship between mutations and clinicopathological factors; for example, some meta-analyses reported associations between
In this study, the correlation between mutation and younger age was a rare finding. In many tissue types, mutations accumulate steadily over time under normal circumstances,
36
and
FNAB is commonly used as a preoperative diagnostic tool for thyroid nodules. However, approximately 20% of nodules remain indeterminate even after FNAB diagnosis, with a malignant risk ranging from 10% to 40%.41,42 Enhancing the diagnostic accuracy of indeterminate nodules is crucial. Molecular testing has emerged as an important clinical tool, with approximately one-third of indeterminate thyroid nodule patients opting for molecular testing in the United States.43,44 Studies have shown that molecular tests, such as DNA-RNA tests (Thyroseq V3, multigene genomic classifier) and RNA tests (GSC, Afirma genomic sequencing classifier), can help a significant number of patients with indeterminate nodules avoid unnecessary diagnostic surgery.
41
However, these tests have limitations such as low specificity, high overdiagnosis rates, limited detection capabilities, and high costs. The diagnostic performance of the 22-gene test used in our study may require further verification, but it holds promise in providing useful information to improve subsequent management of indeterminate nodules. For example, the presence of
Our study has some limitations. The sample size of the nodules analyzed was limited, which could impact the evaluation of the relationship between multigene alterations and clinicopathological features. Additionally, some benign nodules in our study only underwent FNAB cytological diagnosis and lacked tissue histopathologic confirmation, which may have reduced the accuracy of our findings to some extent.
In conclusion, the correlations between multigene alterations and clinicopathological features in PTC were investigated and the diagnostic potential of gene mutation detection in uncertain thyroid nodules was explored in this study, which may be very helpful for the investigation of driver genes in PTC and contribute to improved PTC management.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241233166 - Supplemental material for Correlation between genetic alterations and clinicopathological features of papillary thyroid carcinomas
Supplemental material, sj-pdf-1-imr-10.1177_03000605241233166 for Correlation between genetic alterations and clinicopathological features of papillary thyroid carcinomas by Shixuan Shang, Huimin Yang, Meixiang Chen, Jin Wu, Xianjun Shi, Xiangqin Li, Ningning Feng, Zhaoqing Zheng, Hongmei Liu and Yunzhi Zhang in Journal of International Medical Research
Footnotes
Acknowledgements
The authors are grateful to Shunlin Zhao, Qinfu Wang, Ling Wang, and Rong Zhou for their help with the enrollment of patients in this study.
Author contributions
Conception and design: S Shang, H Yang, M Chen Administrative support: S Shang Provision of study materials or patients: M Chen, J Wu, X Shi Collection and assembly of data: M Chen, J Wu, X Shi, X Li Data analysis and interpretation: S Shang, H Yang, N Feng, Z Zheng, H Liu, Y Zhang Manuscript writing: All authors Final approval of the manuscript: All authors
Declaration of conflicting interests
Huimin Yang, Ningning Feng, Zhaoqing Zheng, Hongmei Liu, and Yunzhi Zhang declare their employment relationship with the NGS company Shanghai Singlera Medical Laboratory, which performed the sequencing work for this study. The remaining authors have no potential conflicts of interest to declare.
Ethics approval and consent to participate
Approval for this study was obtained from the ethics committee of The First People’s Hospital of Jiangxia District (No. 2021019). Written informed consent was obtained from all individual participants included in the study.
Funding
This work was supported by the Research Program of Wuhan Municipal Health and Family Planning Commission [WH21Z40].
Supplementary Material
Supplementary Table 1. Hot spot gene mutations in the OncoAim® kit
Supplementary Table 2. Information on gene fusions in the OncoAim® kit
Supplementary Table 3. Information on mutations and clinicopathologic features in papillary thyroid carcinoma and benign nodules
Supplementary Table 4. Comparison of the clinicopathologic features of BRAFV600E-mutated papillary thyroid carcinoma with those of RAS-mutated papillary thyroid carcinoma
Supplementary Table 5. Correlation analysis of mutations and clinicopathological features in the older age (≥45 years) subgroup
Supplementary Table 6. Gene mutation list of indeterminate nodules
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
