Abstract
Objective
We compared the long-term prognosis of surgery and endoscopic treatment (ET) in patients diagnosed with Siewert Type II pT1N0M0 adenocarcinoma of the esophagogastric junction (AEG).
Methods
Using the Surveillance, Epidemiology, and End Results (SEER) database, we performed a real-world retrospective cohort study and enrolled patients with Siewert Type II pT1N0M0 AEG who underwent surgery or endoscopic treatment (ET) from 2010 to 2018. Matched cohorts were generated using propensity score matching Competing-risk analysis was applied. The cumulative incidence function was used to calculate cancer-specific death and other causes of death (OCD) at different time points. Univariate and multivariate analyses were performed to identify significant prognostic factors by using the subdistribution hazard ratio
Results
We enrolled 725 patients: 462 underwent surgery and 263 received ET. The 5 year cumulative CSD incidence significantly differed between surgery and ET cohorts (16.87% vs 11.08%, P = .01). Following PSM, 2 balanced groups (n = 219 patients each) were analyzed. No significant difference in the 5 year cumulative incidences of CSD was noted between cohorts (17.61% vs. 12.16%, P = .14). In multivariable analysis, the CSD incidence was high among patients with aged ≥65 (SHR 2.29, 95%CI 0.99-5.33, P = .05) and T1b-stage (SHR 1.92, 95%CI 1.03-3.57, P = .04); treatment (surgery or ET) was not significantly associated with cancer survival (SHR 1.51, 95% CI 0.81-2.81, P = .20).
Conclusion
Long-term survival did not significantly differ among patients with Siewert Type II pT1N0M0 AEG adenocarcinoma undergoing surgery or ET. ET may be considered in patients >65 years old or those with submucosal (T1b-stage) cancer of AEG.
Keywords
Introduction
Over the last few decades, there has been a sharp increase in the incidence of adenocarcinoma of the esophagogastric junction (AEG) in Western countries.1,2 This growing incidence may be associated with 2 main risk factors: obesity and gastroesophageal reflux disease, both more common in non-Asian countries. 3 The esophagogastric junction (EGJ), the meeting point of the esophageal squamous epithelium and gastric columnar mucosa, forms the demarcation line of the stomach and esophagus. 4 Given the anatomical location, the classification of AEG and optimal treatment strategies remain controversial.
Clinically, the Siewert classification is the most widely accepted classification of EGJ adenocarcinoma, categorized as follows: Siewert I (lower esophageal cancer, the epicenter within 1-5 cm above the EGJ), Siewert II (true cardia cancer, the center within +1 to −2 cm of the EGJ), and Siewert III (subcardial cancer, the epicenter within 2-5 cm below the EGJ). 5 However, this schema has not yet been universally employed. The Surveillance, Epidemiology, and End Results (SEER) database utilizes the International Classification of Diseases for Oncology, third edition (ICD-O-3) topography classification. In this system, tumors of the gastric cardia are identified by the “C16.0 Cardia” of ICD-O-3, which can be categorized as the Siewert II EGJ cancers (the true cardia cancer).6,7
Considering the 3 AEG subtypes, it has been reported that the incidence of Siewert II AEG has dramatically increased by appropriately 2-fold in recent decades. 8 However, no consensus has been reached regarding the optimal treatment of Siewert II AEG, particularly regarding the optimal treatment approaches, the extent of lymph node dissection, or reconstruction methods. 9 With the help of advances in endoscopic technology, EGJ tumors can be detected at an early stage. Considering patients with T1-stage superficial Siewert II AEG, endoscopic treatment (ET) has been developed as a minimally invasive alternative, although surgical resection remains the traditional option. Several authoritative guidelines recommend ET and surgery as 2 major treatment options for Siewert II AEG with T1a and T1b stage.10–12 Compared with surgery approaches, endoscopic therapies allow the preservation of gastric cardia and have been associated with a reduction in postoperative morbidity and mortality.13,14 Accordingly, both treatment strategies can be considered for patients with superficial Siewert II AEG.
Several studies have reported the comparable short- or long-term efficacies of surgery and ET in patients with AEG; however, these findings mostly pertained to Asian populations.15–17 Compared with these studies focusing on Asia populations, few studies specifically compared the long-term survival among Western populations. Furthermore, no independent analyses are available to compare differences in survival between surgery and ET, particularly for T1-stage AEG, limited to mucosa and submucosa. Therefore, to the best of our knowledge, the present study was the first to compare the long-term prognosis between surgery and ET in patients with Siewert Type II pT1N0M0 AEG using the national SEER data among the United States population.
Methods
This study was conducted in accordance with the Declaration of Helsinki. Institutional review board approval and informed consent were not required in the current study because SEER research data are publicly available and all patients data are de-identified.
Herein, we collected population-based data using the SEER program. The SEER database named “SEER research Plus Data, 18 registries, Nov 2020 sub (2000-2018)” was searched by using the SEER*Stat program version 8.3.9. The topography code C16.0 (ICD-O-3) was used to primarily identify qualified patients with Siewert Type II EGJ cancer.
Inclusion Criteria Were as Follows
(1) Siewert Type II EGJ cancer (the true gastric cardia cancer); (2) pT1N0M0 stage carcinoma (no tumor invasion beyond the submucosa); (3) histopathological diagnosis of adenocarcinoma on post-treatment specimens, including the ICD-O-3 codes: 8140-8389 for adenocarcinoma; (4) surgical resection or ET without additional adjuvant therapies. Exclusion criteria were as follows: (1) histopathological diagnosis of other types of carcinoma (such as squamous cell carcinoma, neuroendocrine tumor, gastric stromal tumor, lymphoma, and other specific or mixed types); (2) tumor with the undefined pathological origin and metastatic cancer; (3) age <18 years; (4) adjuvant therapies such as radiotherapy or chemotherapy before or after the primary treatment; (5) T2/T3/T4 stage with positive lymph node metastasis; (6) tumor diagnosis was solely on autopsy or based on the death certificate; (7) missed follow-up (survival at 0 months). The eighth TNM staging system of the American Joint Committee on Cancer (AJCC) was applied to enroll the largest possible number of patients. 18 The tumor stage was categorized as I, II, and III according to the post-treatment pathological status. The invasion depth was classified as musocal (T1a), submucosal (T1b). The tumor size was categorized as <3 cm and >3 cm. Histologic grade was categorized as well-differentiated, moderately differentiated, poorly differentiated, and undifferentiated type. Based on the surgery code for the stomach in the SEER program coding and staging manual: surgery included the following subgroups: partial or subtotal gastrectomy; near-total or total gastrectomy; ET including both excisions (polypectomy, excisional biopsy, laser excision) and local tumor destruction (photodynamic therapy, cryosurgery, laser). The survival time was defined as the duration from the date of diagnosis to the death or the last follow-up. The baseline and clinicopathologic characteristics (age at diagnosis, sex, race, year of diagnosis, tumor size, stage, histology and grade), cancer incidence, treatment strategies (surgery or ET), and survival-related information (survival months, status, cancer-specific death (CSD), and other causes of death (OCD)) were collected accordingly.
Statistical Analysis
Statistical analysis was performed using SPSS Statistics for Windows, Version 25.0 (IBM Corporation, Armonk, NY), and roentgen software (version 4.1.0). Pearson chi-square test or Fisher’s exact tests were applied to compare categorical variables. The propensity score matching (PSM) algorithm was used to generate matched cohorts by controlling confounding bias. 19 The match ratio of patients in both surgery and ET groups was 1:1, with a .01 of match tolerance. The sample size calculation was performed considering log-rank analysis accounting for competing risks in PASS 15 version, 20 based on the following parameters: α = .05 (2-sided); power: 1-β = .8; event of interest rate in ET group, Fev1 = .15 and event rate in surgery group, Fev2 = .10; competing risks rate in the ET group, Fcr1 = .17 and competing risks rate in the surgery group, Fcr2 = .12. The resulting total sample size was 201 for each group.
The primary outcomes were CSD and OCD in each group at different time points. Competing-risk regression analysis was applied using the ‘survival’ and ‘cmprsk’ packages in R. 21 The cumulative incidence function (CIF) was used to calculate the incidence of CSDs and OCDs at the 1-, 3-, and 5- year time points. The CIF differences between surgery and ET groups were compared using the Fine-Gray test. Univariate and multivariate completing regression was performed to identify significant prognostic factors using the subdistribution hazard ratio (SHR). The reported statistical significance levels were two-sided, and P ≤ .05 was deemed statistically significant.
Results
Baseline Characteristics
Based on the established inclusion and exclusion criteria, 725 patients were enrolled, among whom 462 underwent surgery and 263 received ET (Figure 1). The number of patients receiving ET increased by 15.6% annually (95% CI: −11.4-42.6%) from 2010 to 2018. Conversely, we detected a decreasing trend in the number of patients undergoing surgery (annual percent change: −12.5%, 95% CI: −28.5-3.5%) during the same period. Figure 2 shows the distribution of patients who underwent surgery or ET from 2010 to 2018. Flowchart of generating the study groups. The distribution of patients who received surgery or ET from 2010 to 2018. The solid histograms and y-axis on the left represents the number of patients, and the dotted lines and y-axis on the right represent the ratio of patients who received ET or surgery in different years.

Considering the ET group, the median age was 71 years (interquartile range [IQR], 55-87 years), comprising 74.5% males, and 6.8% Hispanic subjects. In the surgery group, the median age was 67 years (IQR, 53-81 years), with 78.1% males, and 7.4% Hispanic subjects. The proportions of patients >65 years of age was higher in the ET group than that in the surgery group (72.2% vs. 61.0%, P<.01). Moreover, the ET groups comprised a greater number of patients in the T1a stage than the surgery group (70.3% vs. 38.5%, P < .01).
PSM
Baseline Characteristics of Patients.

Note: Each P value was derived from the Chi-square or Fisher’s exact analyses between the single variables in ET or surgery groups. *statistical significance was considered as P ≤ .05.
Survival Analysis
Survival Data of CSD and NCD in this Study.

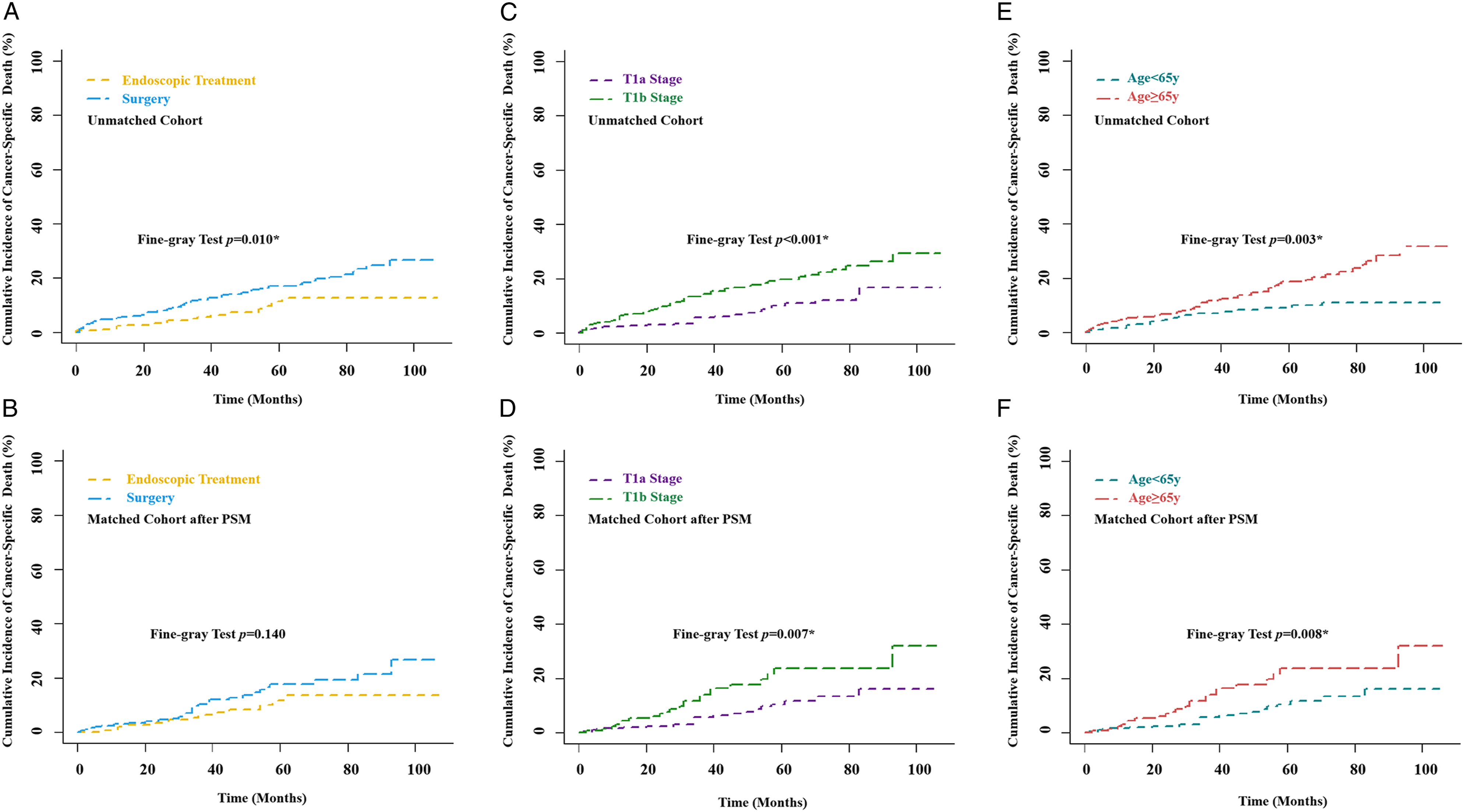
The cumulative curves for the long-term cancer-specific death in surgery vs Endoscopic treatment (ET) group before (A) and after (B) PSM, in T1a vs T1b-stage group before (C) and after (D) PSM, and in age≥65 y vs age<65 y group before (E) and after (F) PSM.

Forest plot of the long-term cancer-specific death in different subgroups.
Univariate and Multivariate Analyses of SEGC Cancer-Specific Survival.
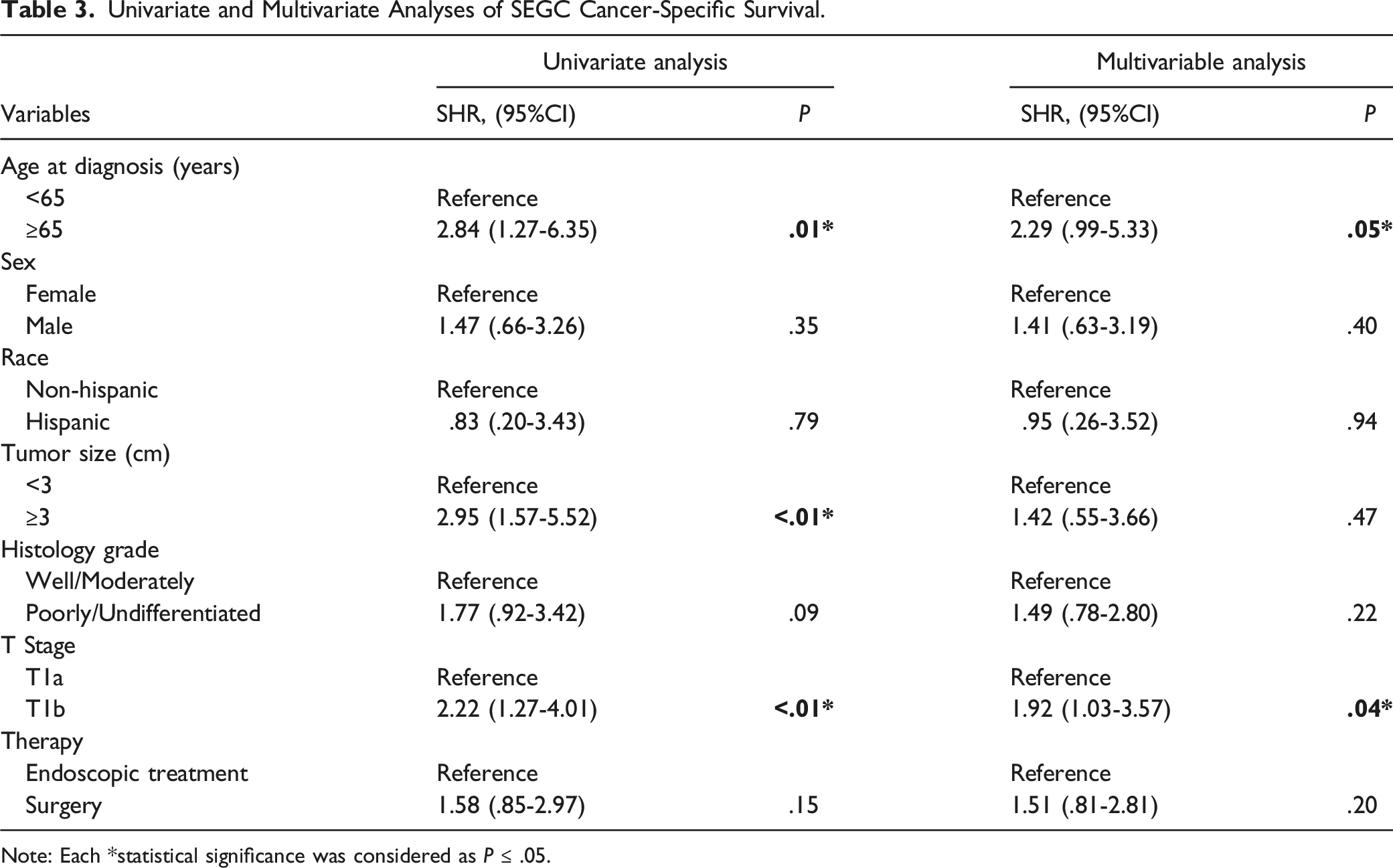
Note: Each *statistical significance was considered as P ≤ .05.

The companion of the long-term cancer-specific death in subgroups: (A) detailed surgical methods; (B) detailed ET methods; (C) Age≥65 years; (D) Age<65 years.
Distribution of Patients Receiving Surgery Or Endoscopic Treatment.

Univariate and Multivariate Analyses of SEGC Other Cause of Death.

Note: *statistical significance was considered as P ≤ .05.
Discussion
Given the unique location on the boundary between the esophagus and the stomach, no consensus has been reached regarding an optimal therapeutic strategy for EGJ adenocarcinomas. 22 ET and surgery are 2 therapeutic options available for patients with Siewert Type II T1M0N0 AEG. In the present study, we documented a decreasing trend of surgical resection and an increasing trend for ET among patients undergoing treatment from 2010 to 2018. These findings indicate that ET has been increasingly recognized as an effective strategy for early-stage AEG. However, few studies assessing Western populations have provided survival data for these patients post-treatment. To address this gap in knowledge, our study compared the long-term prognosis between ET and surgery using the SEER database. To reach a more precise statistical comparison: (1) we applied the PSM algorithm to decrease confounding factors such as age, tumor size, differentiation, histological type, and tumor invasion; (2) we applied the completing-risk regression analysis, which may be more precise by providing an alternative to Cox regression in the presence of 1 or more competing survival outcomes, such as tumor-specific mortality and other causes of mortality. 23
The standard Cox model is a well-established methodology for calculating survival outcomes. Completing-risk regression is an alternate statistical method for handling survival data and has been reported in several high-impact journals. 24 Wolbers et al 25 have reported that the standard Cox model overestimated the 10 year risk of coronary heart disease when compared with the estimate from the competing-risk regression. This finding could be attributed to the results of the traditional Cox survival function, with estimates of incidence biased upward, regardless of whether the competing events are independent of 1 another. It should be noted that several clinical survival studies lacking competing events. For instance, in our study, the primary outcome in the present study was time to death due to AEG. Death attributable to a non-cancer cause is considered a competing event. A subject who dies of a disease of other systems is no longer at risk of death attributable to cancer. Accordingly, the completing-risk model has greater relevance than Cox regression in analyzing survival data in our study, given the presence of competing survival events.
The most important findings of the present study were as follows: (1) there was no statistically significant difference in the long-term survival of patients undergoing surgery or ET; (2) the surgery group had a higher risk of death than the ET group, considering patients ≥65 years old or with T1a stage tumor; (3) Age and tumor invasion could be independent prognostic factors for patients with Siewert Type II early AEG. (4) In patients aged ≥65 years or with T1b tumor, ET could be a primary strategy, affording a comparable survival rate to surgical treatment.
Surgery is the primary treatment strategy for gastric cancer and AEG owing to its advantages in terms of complete tumor resection and lymph node dissection. 26 However, surgery is well-known to be associated with considerable postoperative morbidity and mortality. 27 Compared with surgical resection, ET is a minimally invasive procedure with low morbidity and mortality and improved quality of life owing to organ preservation. 28 Typically, the indications for ET include mucosal (T1a) and submucosal (T1b) cancer without lymph node metastasis.29,30 In the present study, the enrolled patients were node-negative T1 stage within the indication of ET. Our results detected no significant difference in the long-term survival outcome between surgery and ET among Western populations, consistent with the findings previously reported in Asian countries.15–17 Moreover, the CSD rates in ET or surgery groups did not significantly differ considering patients with submucosal (T1b) cancer. Accordingly, ET can be considered a primary treatment strategy for T1b tumors, given its minimally invasive nature and capacity to afford an improved quality of life. The ET choice may be more relevant in patients older than 65 years, given the significantly high CSD detected in the surgery group. Our study emphasized the application of ET in submucosal (T1b) cancer, as surgery remains the primary treatment strategy recommended by most Western guidelines. However, our results revealed that ET could afford a comparable survival rate to surgical treatment in patients with T1b tumors. Thus, we propose utilizing ET as a primary method in patients with T1b tumors. Although comparisons performed before PSM showed a higher CSD in the surgery group than that in the ET group, this could be attributed to confounding bias, given the presence of different pathological characteristics between the 2 groups. After adjusting confounding bias, no statistical difference was detected.
Compared with the survival data (more than 90%) reported in previous Asian studies, the 5 year disease-specific survival was reduced in both surgery (87.8%) and ET (82.4%) cohorts in the present study. This finding could be attributed to 2 possible factors. Firstly, the previously reported Asian studies are single-center studies with relatively small sample sizes, and their survival data includes all Siewert types of early-stage cancer. Our study specifically investigated the survival outcome in Siewert II type cancer of AEG. In addition, Asian studies mainly performed endoscopic submucosal dissection (ESD) as the primary endoscopic resection method. However, ET includes both local tumor excisions and destruction (photodynamic therapy, cryosurgery, or laser) in the SEER database. Furthermore, local tumor excisions were not described in detail to differentiate whether patients received exact endoscopic mucosal resection or ESD. Thus, survival data cannot be precisely compared between Asian and Western populations owing to different categories of ET. Secondly, Americans and Asians are 2 distinct ethnic groups, and different genetic factors may underlie the incidence of AEG cancers in these groups. Thus, the survival data may differ. Furthermore, we found that patients who underwent ET had a considerably higher incidence of OCD than those who received surgery. Patients ≥65 years had a significantly high risk of OCD. This may be attributed to the higher incidence of obesity, diabetes, and cardiovascular diseases among Western populations than among Asian populations.
To the best of our knowledge, few studies have investigated the long-term prognosis of AEG in Asian countries. Feng et al 17 have recently reported that adjuvant chemotherapy, the ratio of positive to negative lymph nodes, intraoperative blood transfusion, tumor size, perineural invasion, serum CEA, and duration of hospital stay post-surgery could be independent risk factors for survival in all stages of Siewert Type II AEG adenocarcinoma. Guo et al 31 have revealed that the age, tumor grade, T stage, and N stage are independently associated with the survival prognosis of AEG cancer with no distant metastasis. However, no report has independently investigated the long-term prognosis among Western populations with early-stage (T1) cancer. Our study identified age and tumor invasion as 2 independent prognostic factors in T1M0N0 AEG adenocarcinoma. These findings emphasized the role of age and tumor invasion in evaluating AEG in all stages.
Our study has some limitations that need to be addressed. Firstly, this was a retrospective cohort study using the SEER database. Variable selection bias may exist as some detailed clinicopathological factors were not accessible, such as margin, lymphovascular invasion, complete resection rate, and local or distal metastasis. Secondly, given the limitations of the SEER database, we were unable to access the survival data in Siewert type I and III of AEG adenocarcinoma. Thus, additional studies are needed to determine whether long-term survival outcomes differ among different Siewert types of AEG adenocarcinomas. However, the strengths of the present study should be noted. We, for the first time, independently compared the long-term prognosis of surgery and ET in patients with Siewert Type II pT1N0M0 AEG among the United States population. Secondly, the survival data in our study may be more precise owing to the application of the PSM methodology and competing-risk regression.
Conclusion
Long-term survival outcomes did not differ significantly between patients who received surgery or ET in the U.S. population. For Siewert Type II pT1N0M0 AEG adenocarcinoma, ET may be primarily considered in patients older than 65 years or those with sub-mucosal (T1b-stage) invasion.
Footnotes
Author Contribution
Han Chen: study concept and design, analysis and interpretation of data; drafting of the manuscript, authorship; Xin Yu: study concept and design, analysis and interpretation of data, authorship. Ruoyun Yang: data extraction, design and order the figures and tables, assessment of study quality, authorship. Shuo Li: data extraction, design and order the figures and tables, authorship. Guoxin Zhang: design and order the figures and tables, assessment of study quality. Xinmin Si and Xiaoying Zhou: critical revision of the manuscript for important intellectual content; obtain funding; study supervision, authorship.
Declaration of Conflicting Interests
The authors(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The raw data and programming code could be assessed by contracting the corresponding author. The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Natural Science Foundation of China (No. 82100594), Jiangsu province Hospital (the First Affiliated Hospital with Nanjing Medical University) Clinical Capacity Enhancement Project (JSPH-MC-2022-29) and Postgraduate Research and Practice Innovation Program of Jiangsu Province (SJCX22_0697). All the authors approved the submitted version of the manuscript.
Ethnic Approval
This study was conducted in accordance with the Declaration of Helsinki.
Informed Consent
Institutional review board approval and informed consent were not required in the current study because SEER research data are publicly available and all patients data are de-identified.
