Abstract
Objective
This study aims to evaluate the impact of economic status on the cause-specific survival (CSS) of patients with esophageal adenocarcinoma (EAC).
Methods
Using propensity score matching (PSM), we analyzed data from 4676 patients with EAC, categorized into low household income (LHI) and high household income (HHI), sourced from the Surveillance, Epidemiology, and End Results database. Significant pre-PSM differences in age, gender, race, and tumor stage (T stage) were observed, necessitating PSM adjustment. The PSM incorporated these variables along with radiotherapy to balance the groups effectively. Kaplan–Meier curves and accelerated failure time (AFT) and random survival forest (RSF) models were used to assess the prognostic impacts.
Results
According to the pre-PSM data, a significant difference in CSS was observed between the LHI and HHI groups (median CSS = 1.25 years vs 1.50 years, P < .05). Post-PSM analysis maintained the result (P < .05). Using the AFT model, HHI was found to significantly enhance CSS (pre-PSM time ratio [TR] = 1.145, post-PSM TR = 1.146). Male gender (pre-PSM TR = 0.762, post-PSM TR = 0.819) and certain non-single marital statuses (such as divorced, pre- and post-TRs <1.000) are also prognostic risk factors for EAC (P values <.05). Additional significant prognostic risk factors for EAC included advanced American Joint Committee on Cancer stages (III and IV), higher tumor stages (T3 and T4), higher node stages (N1–N3), without surgery, and without chemotherapy, as identified using the AFT and/or RSF mothods. The results derived from the RSF model are consistent with those of the AFT model.
Conclusion
Higher economic status is positively related to EAC CSS. Beyond standard clinical treatments, interventions such as enhancing socioeconomic support and preventive aspects, including intensive health education and EAC screening, could play a crucial role in improving EAC prognosis.
Keywords
Introduction
As one of the most common cancers, esophageal cancer (EC) ranks 10th in incidence among all tumors worldwide. 1 The main treatments for this cancer include surgical resection, radiotherapy, and chemotherapy.2-4 Despite current medical advancements, the prognosis for EC patients remains poor, with a 5-year survival rate of only 20%, slightly higher than that of pancreatic cancer (which has the lowest survival rate). 5 Thus, EC is one of the most lethal cancers. Additionally, the vast majority of patients with EC are diagnosed at an advanced stage, 6 further limiting the effectiveness of treatment. Esophageal adenocarcinoma (EAC), one of the two main subtypes of EC (the other being esophageal squamous cell carcinoma), has seen increasing incidence over the past decades, particularly in developed countries such as the United States.7,8 Despite advancements in treatment, the prognosis for patients with EAC remains suboptimal, and the factors influencing this prognosis are not yet fully understood. Therefore, this study aims to identify the key prognostic factors affecting patients with EAC, which could inform the development of more targeted and effective treatment and management strategies for EC.
In addition to the clinical and pathological characteristics of cancer, sociological factors affecting cancer prognosis are receiving increasing research attention.9,10 Among these factors, economic status is widely believed to have a significant impact on the prognosis of various cancers. As shown by many studies, the overall survival rate of low-income patients is significantly lower than that of high-income patients across various cancers due to potential differences between low-income and high-income patients’ access to diagnostic and treatment resources. 11 For example, a study of hepatocellular carcinoma revealed that patients from rural areas and low-income families were diagnosed more advanced tumor stages (T stages) at diagnosis and had significantly higher mortality rates. 12 Similarly, in patients with bone and soft tissue sarcomas, low income was significantly associated with lower 5-year overall survival rates. 13 This trend is also evident in EAC, where previous studies have also shown that lower median household income is associated with a worse prognosis in early-stage (T1aN0M0) patients with EAC. 14 However, early-stage patients represent only a small proportion of the EAC patient population, 6 and the incidence of early-onset EC is declining. 15 Therefore, it is crucial to investigate how socioeconomic factors affect the prognosis of the broader EAC population, rather than those at certain stages, to inform disease-specific interventions and aspects of prevention.
This study aims to evaluate the impact of economic status on the survival of patients with EAC in the United States using a large-scale retrospective cohort study based on the SEER (Surveillance, Epidemiology, and End Results) database, 16 which may provide valuable references for the management of EAC.
Materials and Methods
Study Design and Ethical Approval
This study uses retrospective data from the SEER program 16 and aims to evaluate the impact of economic status on the cause-specific survival (CSS) of patients with EAC in the United States. We use the propensity score matching (PSM) 17 method to rigorously control for confounding factors, ensuring comparability between the high household income (HHI) and low household income (LHI) groups, thereby more accurately assessing the impact of economic status on the prognosis of patients with EAC. The data used in this study have been anonymized in compliance with SEER data usage requirements 16 ; thus, ethical committee approval is not required. This study complies with the RECORD guidelines. 18
Study Subjects and Data Collection Criteria
Data for this study were sourced from the SEER*Stat 8.4.3.
19
The inclusion criteria were as follows: patients pathologically diagnosed with EAC between 2000 and 2020; aged 18 to 89 years; with clear information on gender, age, race, marital status, and American Joint Committee on Cancer (AJCC) staging; recorded information on surgery, radiotherapy, and chemotherapy; and clear CSS information and follow-up time. The exclusion criteria were as follows: CSS time <1 month (i.e., the interval from the diagnosis of EAC to death due to the disease is less than 1 month) and the presence of multiple lesions. The detailed process of patient selection is shown in Figure 1. Detailed process of patient selection in this study.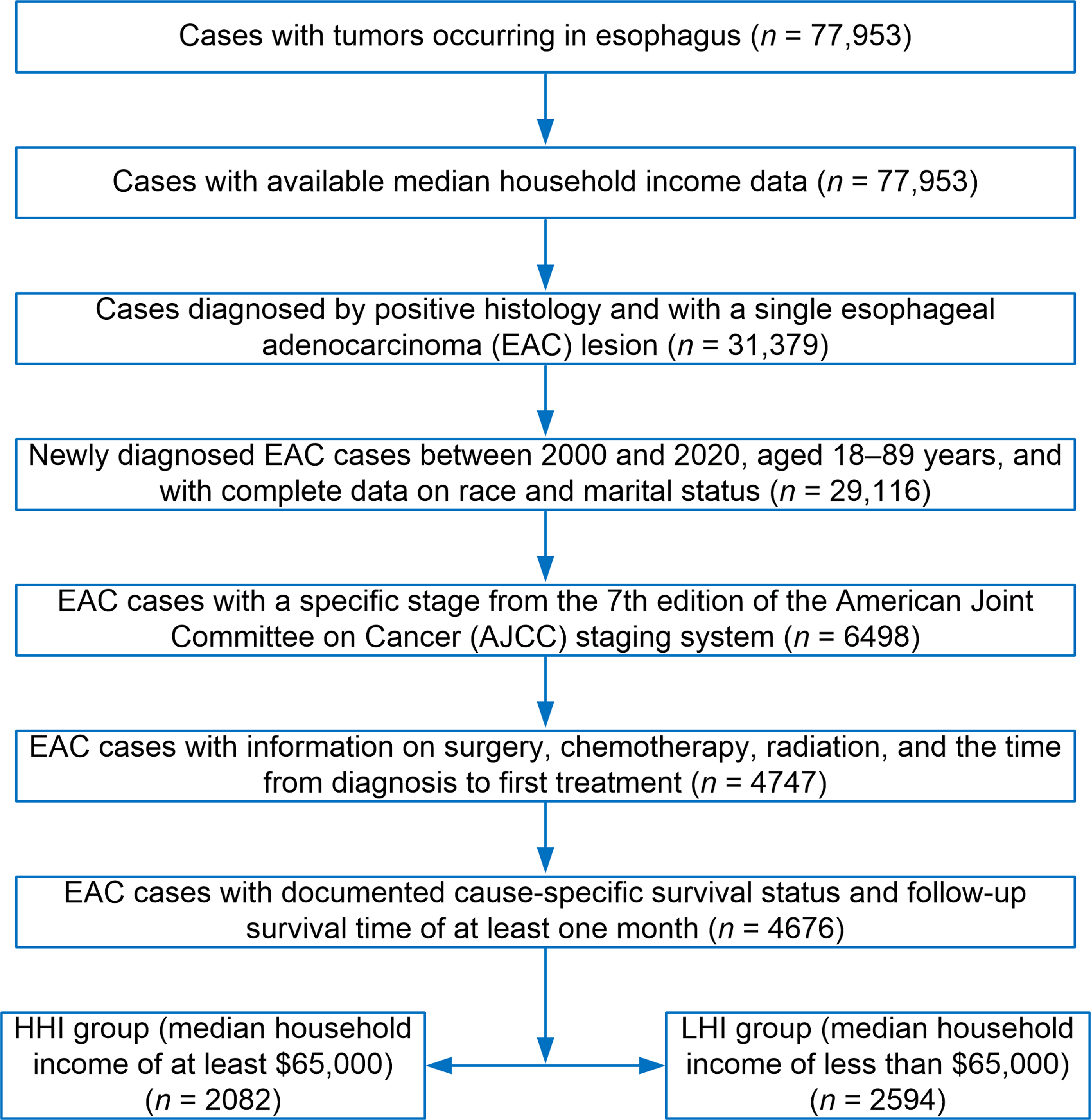
Variables and Endpoint
In this study, all independent variables were recorded as categorical variables. The variables collected in this study were demographic characteristics (economic status, gender, age, race, marital status), clinical characteristics (AJCC, tumor (T)-node (N)-metastasis (M) staging [T, N, M staging] information), treatment information (surgery, radiotherapy, chemotherapy, the time from diagnosis to first treatment), and follow-up information (follow-up time and CSS).
Specifically, economic status is defined as the median household income of the patient’s county in the past 12 months (adjusted to 2021 inflation dollars). Patients in the LHI group have a median household income of less than $65,000, while those in the HHI group have a median household income of at least $65,000. Gender, age, race, and marital status refer to the attributes of patients at the time of diagnosis. Race is categorized into four groups: White, Black, American Indian/Alaska Native, and Asian or Pacific Islander. Since White individuals constitute the majority of study subjects, race in this study is grouped as White and Others (American Indian/Alaska Native, Asian or Pacific Islander). Marital status includes the following categories: Single (never married), Widowed, Divorced, Unmarried or Domestic Partner, Married (including common law), and Separated.
For the AJCC stage and its T, N, M staging information, the 7th edition was used in this study. Information on whether the patient received the three treatment modalities—surgery, radiotherapy, and chemotherapy—was also collected for analysis. The primary endpoint of the study was CSS, defined as the interval from the diagnosis of EAC to death due to the disease. Follow-up time was converted into years in this study.
PSM Parameters
To reduce the sample differences between the LHI and HHI groups, this study used 1:1 matching and the “optimal” matching method. Matching variables included parameters that were unbalanced between the HHI and LHI groups before PSM, such as age. The matching effectiveness was evaluated using chi-square tests between the two groups, Love plots, and standardized mean differences (SMDs). An SMD less than 0.1 indicated good matching.
Statistical Analysis
All data analyses were performed using R 4.2.2 statistical software and its packages tableone, MatchIt, survival, survminer, randomForestSRC, and cobalt.20-22 Chi-square tests were used to detect the balance between the LHI and HHI groups and to test for collinearity among different variables. Kaplan–Meier survival analysis was used to evaluate CSS differences between the LHI and HHI groups. accelerated failure time (AFT) and random survival forest (RSF) models were employed to explore factors affecting CSS in patients with EAC. The statistical significance level was set at P < .05.
Results
Baseline Characteristics and PSM
The Characteristics of Patients Between the Low Household Income (LHI) and High Household Income (HHI) Groups Based on Data Before Propensity Score Matching.
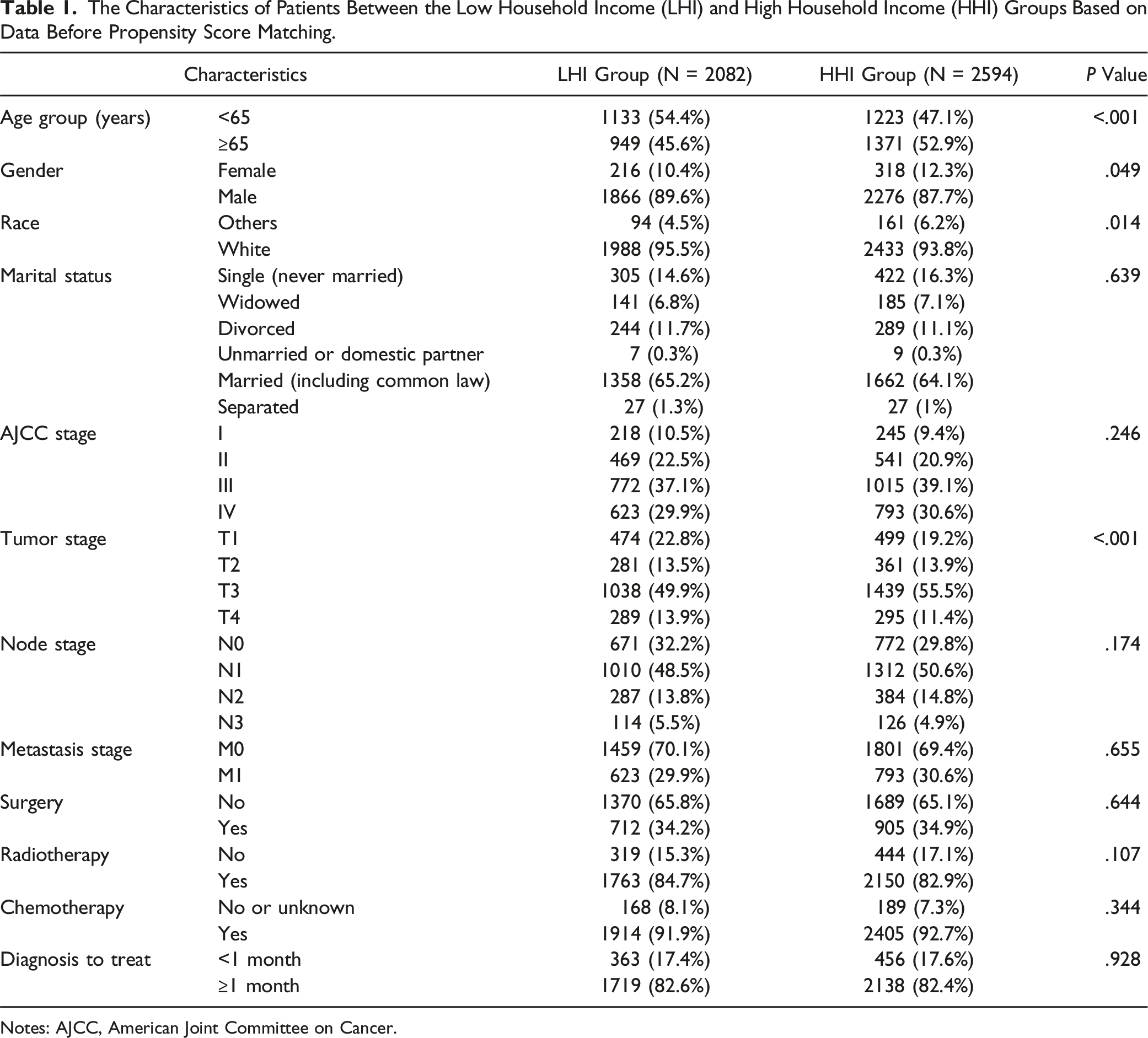
Notes: AJCC, American Joint Committee on Cancer.
The Characteristics of Patients Based on Data After Propensity Score Matching.

Notes: AJCC, American Joint Committee on Cancer.
Prognostic Differences Between the LHI and HHI Groups
To explore the prognostic differences between the LHI and HHI groups, Kaplan–Meier curves were plotted based on pre- and post-PSM data. According to the pre-PSM data, over a follow-up period exceeding 10 years, the LHI group exhibited shorter CSS at nearly every stage than the HHI group; moreover, a statistically significant difference in CSS was observed between the LHI (median CSS = 1.25 years) and HHI (median CSS = 1.50 years) groups (P < .05, Figure 2A). Similarly, post-PSM data showed that the LHI group had shorter CSS than the HHI group (median CSS: 1.25 years vs 1.50 years) (P < .05, Figure 2B). These results indicate that improving the economic conditions of patients with EAC may help enhance prognosis and increase survival rates. Kaplan–Meier curves before (A) and after (B) propensity score matching (PSM). CSS, cause-specific survival.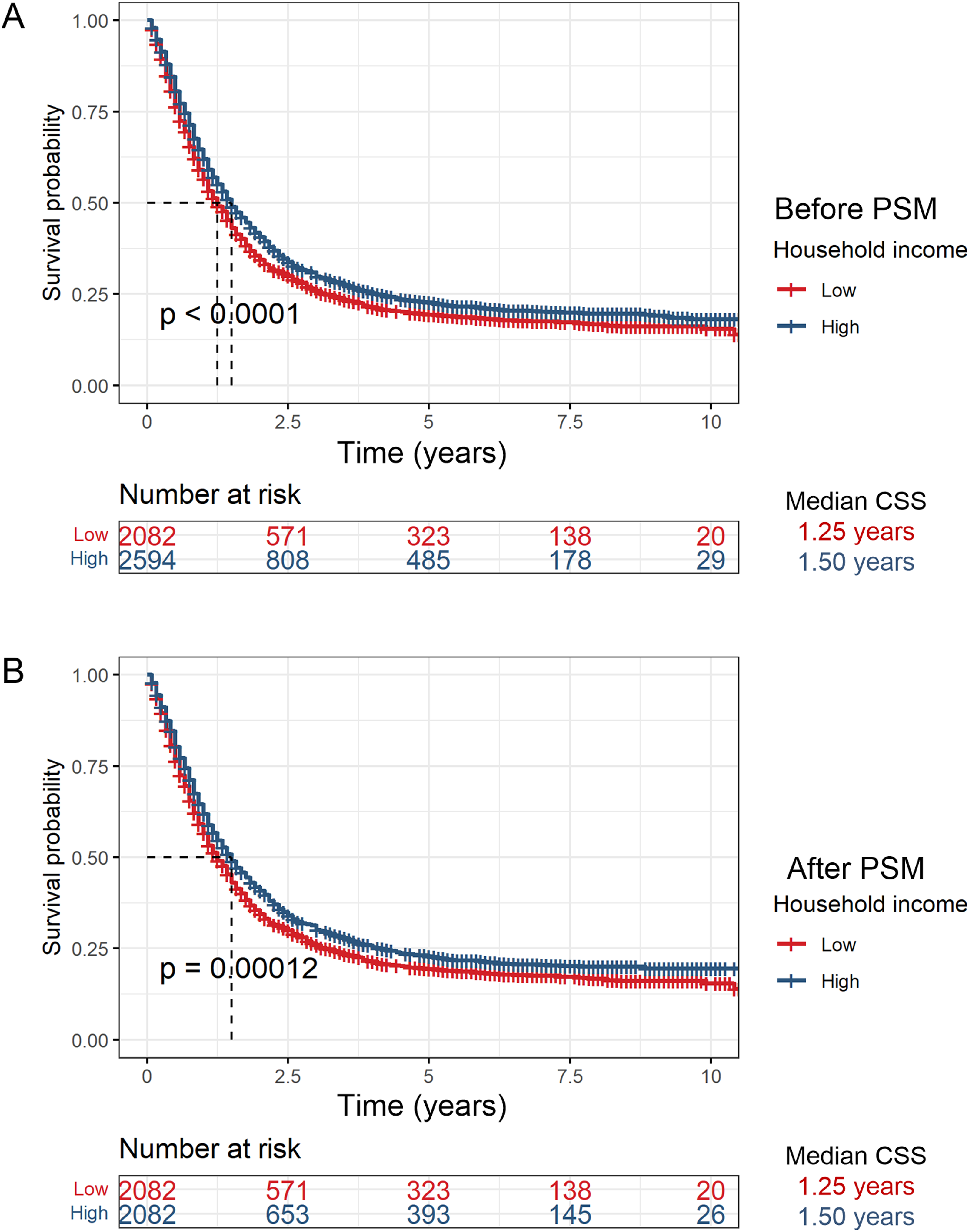
Factors Affecting the Prognosis of Patients with EAC
Factors Affecting the Cause-Specific Survival (CSS) of Patients Based on Data After Propensity Score Matching (PSM).

Notes: *The difference was statistically significant with a P value of <.05. Household income refers to the median household income.
Certain disease-related parameters also significantly influence the prognosis of patients with EAC. According to AJCC staging, patients with disease progression to stage III and IV had significantly worse prognoses, especially those at stage IV, who exhibited the lowest TR (pre-PSM: TR = 0.385, 95% CI: 0.323-0.460; post-PSM: TR = 0.370, 95% CI: 0.308-0.444; P values <.05, Table 3). Conversely, patients at stage T2 showed longer survival times than those at stage T1 (pre-PSM: TR = 1.338; post-PSM: TR = 1.310, 95% CI not including 1; P values <.05, Table 3), suggesting that in cases of smaller EAC (T1–T2 stages), prognosis is not always inversely proportional to tumor size.
Surgical treatment significantly improved survival rates, considerably extending survival times (pre-PSM: TR = 2.757, 95% CI: 2.525-3.010; post-PSM: TR = 2.769, 95% CI: 2.522-3.039; P values <.05, Table 3). Chemotherapy also emerged as a significant factor for improved prognosis, with TRs above 2 before and after matching (95% CI not including 1; P values <.05, Table 3). In contrast, radiotherapy showed no significant impact on prognosis. Pre-PSM data indicated that the interval from diagnosis to initial treatment affected prognosis, but post-PSM data showed that this variable did not significantly affect CSS (95% CI including 1; P values >.05, Table 3).
The above results suggest that eight factors—economic status, gender, marital status, AJCC stage, T stages, N stage, surgical treatment, and chemotherapy—can influence the prognoses of patients with EAC. An RSF model was used to verify the impact of these eight factors on the prognoses of patients with EAC in the United States. Pre-PSM data showed that patients with low income, male gender, certain non-single marital statuses (such as widowed, divorced, and unmarried or domestic partner), high AJCC stages (III and IV), high T stages (T3 and T4), high N stages (N1–N3), no surgical treatment, and no clear chemotherapy history had a higher relative frequency of mortality (Figure 3). The results derived from pre-PSM data were not only validated by post-PSM data (Figure 4) but also consistent with the AFT model results mentioned earlier. Random survival forests illustrating the impact of different variables on the relative mortality of patients with esophageal adenocarcinoma (based on pre-propensity score matching data). For marital status: SG, Single (never married); WD, Widowed; DD, Divorced; UD, Unmarried or Domestic Partner; MR, Married (including common law); SE, Separated. Random survival forests illustrating the impact of different variables on the relative mortality of patients with esophageal adenocarcinoma (based on post-propensity score matching data). For marital status: SG, Single (never married); WD, Widowed; DD, Divorced; UD, Unmarried or Domestic Partner; MR, Married (including common law); SE, Separated.

Discussion
The impact of economic status on cancer prognosis has attracted increasing research attention. 11 Numerous studies have highlighted the role of socioeconomic factors in determining survival outcomes across various cancers, such as soft tissue sarcoma 13 and melanoma. 23 However, the effect of economic status on EAC prognosis remains underexplored. This study evaluated the impact of economic status on the CSS of 4676 patients with EAC diagnosed in the United States, revealing that patients in the LHI group had significantly poorer CSS than those in the HHI group. PSM was employed to reduce baseline characteristic differences between the two groups, resulting in a more balanced sample. 17 Data both before and after PSM showed the same trend, highlighting the importance of economic status on CSS. Additionally, male gender, high AJCC stages (III and IV), high T stages (T3 and T4), high N stages (N1–N3), certain non-single marital statuses (such as widowed, divorced, and unmarried or domestic partner), absence of surgical treatment, and lack of a clear chemotherapy history were identified as risk factors for the prognosis of patients with EAC. These findings may provide valuable references for the clinical management of EAC.
Economic status is closely associated with the prognosis of various cancers, including EAC. Abdel-Rahman et al reported that patients with malignant melanoma of the skin and poor socioeconomic status had worse overall survival compared to those with better economic status. 23 They also also observed that lower socioeconomic status was associated with poorer melanoma-specific survival. 23 Similarly, in bone and soft tissue sarcoma, hepatocellular carcinoma, thyroid cancer, and limited-stage small cell lung cancer, low-income patients exhibited significantly lower survival rates than high-income patients.10,12,13,24 A same trend was found in EAC: both studies by Kramer et al. 25 and Geng et al., 14 and our own research have revealed the positive impact of HHI on CSS in patients with EAC. Unlike studies by Geng and Kramer,14,25 which focused on a limited subset of early-stage (T1aN0M0) or locally advanced (T4) patients, our analysis encompassed patients with EAC across all AJCC stage, thereby enhancing the generalizability of our findings that LHI is associated with poorer CSS in patients with EAC. Our study is notable for its larger sample size (n = 4676), longer time scale (up to 10 years), and the use of multiple statistical methods (Kaplan–Meier curves, AFT, and RSF) to corroborate the association between economic status and CSS.
The mechanisms that underpin the effect of lower socioeconomic status on EAC CSS are likely multifactorial. In certain cancers, late cancer diagnosis, differences in treatment methods, and greater physiological stress among LHI patients may contribute to the effect of economic status on prognosis.10,12,13 According to EAC-related reports, Kramer et al discovered that in T4-stage patients with EAC, an LHI of less than $46,000 was associated with a higher overall survival risk 25 ; however, they did not further discuss the potential mechanisms of this finding. Further, Geng et al assumed that the worse prognosis in LHI patients might be associated with more aggressive EC phenotypes, lower follow-up monitoring rates, and limited health care resources in rural areas (those with LHI tend to live in rural areas). 14 In our study, the disease severity (e.g., AJCC stages) and treatment modalities (surgery, radiotherapy, and chemotherapy) between the HHI and LHI groups were balanced after PSM, a robust method to reduce confounding factors. 26 Moreover, results from the AFT model showed that, even after adjusting for these covariates, economic status remained an independent prognostic factor for EAC CSS. Therefore, it is unlikely that disease severity and initial treatment modalities (at least for surgery, radiotherapy, and chemotherapy) are the primary reasons for the impact of socioeconomic status on CSS in EAC patients.
However, several factors, including treatment effectiveness, may influence the prognosis of EAC patients with lower economic status. One such factor is treatment adherence (e.g., continuity of chemotherapy), as low-income cancer individuals are more likely to exhibit poorer adherence due to the high costs of treatment. 27 Additionally, considering that the income levels in our study refer to the median household income of certain counties in the United States and that patients with EAC and LHI tend to reside in rural areas, the limited health care resources in and around these regions may affect the CSS of patients with EAC in the LHI group. Even among patients with a precancerous condition for EAC (i.e., Barrett’s esophagus), the inequity in health care access due to economic disparities has been documented. 28 Besides health care resources, poor health literacy may impact the prognoses of patients with EAC and lower economic status.29,30 Furthermore, EC patients often experience negative emotions, such as anxiety and depression, during radiotherapy, which can lead to complications such as malnutrition and worsen their condition. 31 In such cases, low-income individuals have fewer opportunities for specialized care and community support, potentially resulting in poorer quality of life and prognoses.31,32 In summary, lower socioeconomic status may affect EAC CSS through multiple factors, including limited health care resources, unhealth habits, and psychological factors. Therefore, it is recommended to allocate more health care and educational resources to economically disadvantaged regions, as doing so may help intervene in and prevent EAC.
Other social factors are also significant prognostic indicators for patients with EAC. Our study found that male patients with EAC had worse prognoses than female patients. Previous research has attributed this to biological differences, treatment choices, and lifestyle differences between the sexes.33,34 Certain non-single marital statuses (such as widowed, divorced, and unmarried or domestic partner) were identified as risk factors for poor EAC prognosis compared to being single. This situation may be related to differences in psychological support and lifestyle changes. 35 These findings underscore the importance of considering social factors into the clinical management of EAC.
Apart from social factors, the prognoses of patients with EAC are closely related to disease characteristics and treatment modalities. It is generally believed that higher stages of the same pathological type of cancer are associated with worse overall prognoses.36,37 Our study found that this holds true for EAC, with patients at advanced AJCC stages (III and IV), high T stages (T3 and T4), and high N stages (N1–N3) experiencing poorer prognoses. Interestingly, the AFT and RSF methods used in this study consistently showed that patients with T2-stage EAC had better prognoses than those with T1-stage EAC. This suggests that for early-stage patients, the depth of tumor invasion (propria and more superficial) may not be fully correlated with the EAC prognosis and may be influenced by other factors, such as tumor location and differentiation. 38 This observation may provide some reference value for prognosis assessment of early-stage (T1-2) cancers. Additionally, the three major treatments (surgery, radiotherapy, and chemotherapy) are important factors in achieving favorable prognoses in EAC.25,39,40 Our study also found that surgery and a clear chemotherapy history are important determinants of EAC prognosis, and radiotherapy is associated with relatively reduced mortality in patients with EAC. This highlights the importance of aggressive treatment in improving outcomes for patients with EAC.
This study emphasizes the importance of considering multiple factors in managing patients with EAC. However, there are certain limitations. First, as a retrospective study, it is subject to potential selection bias. Although PSM was used, it cannot eliminate all potential confounding factors. Second, the data were sourced from a single SEER database, which may limit the generalizability of the findings due to the lack validation from multicenter data. Lastly, the definition of income level in this study was relatively simplistic and did not account for more complex economic factors, such as household composition and the patients’ actual economic burden. As a result, this study could not analyze key aspects like equivalized income for each individual. Future research should expand sample sizes and incorporate multicenter data to address these limitations, providing more comprehensive intervention strategies to improve patient survival and quality of life.
Conclusion
Low-income patients have significantly poorer CSS than high-income patients and that economic status is an important independent factor for the prognoses of patients with EAC. Considering multiple social and demographic factors comprehensively is significant for improving the prognoses of patients with EAC.
Supplemental Material
Supplemental Material - The Impact of Economic Status on Cause-specific Survival in Patients With Esophageal Adenocarcinoma in the United States: A Retrospective Analysis
Supplemental Material for The Impact of Economic Status on Cause-Specific Survival in Patients with Esophageal Adenocarcinoma in the United States: A Retrospective Analysis by Xiu-Shun Xiang, Guo-Sheng Li, Jun Liu, Xiang Gao, Gui-Yu Feng, Jing-Xiao Li, Tao Huang, Jian-Ji Guo, Nuo Yang, and Hua-Fu Zhou in Cancer Control.
Footnotes
Acknowledgments
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by Guangxi medical and health appropriate technology development and popularization application project (S2022070), Self-financed scientific research of Guangxi Zhuang Autonomous Region Health Commission (Z20211032), National Key Clinical Specialty Construction Project of China, Guangxi Medical and Health Key Discipline Construction Project, Guangxi Key Clinical Specialty Construction Project.
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
