Abstract
Background
Conflicting results have been reported regarding the potential preventive effects of statins on the risk of cancer. This study investigated the associations of statin use with the incidence and mortality of kidney cancer in South Korea.
Methods
In this retrospective population-based cohort study using the National Health Insurance claims database, we compared patients aged 45-70 years who had used statins for at least 6 months to non-statin users matched by age and sex from 2005 to June 2013. The main outcomes were kidney cancer incidence and mortality according to statin use. Cox proportional hazard regression was used to calculate the adjusted hazard ratios (aHRs) and 95% confidence intervals (95% CIs).
Results
In the cohort of 1 008 101 people, the aHRs for the association between statin use and the outcomes were .84 (95% CI: 0.71-.99) for kidney cancer incidence and .65 (95% CI: 0.41-.98) for kidney cancer mortality. In the matched cohort of 337 578, the risk per 1000 people of cancer incidence and mortality was 1.63, 1.07, and .24, .17 in statin users and non-users, respectively. In matched cohort, the risk of kidney cancer incidence and mortality decreased, but it is not statistically significant. Also, there was no linear relationship with increased doses.
Conclusion
Statin use might be associated with a decreased risk of kidney cancer incidence and mortality, but it showed no statistical significance. This study was a large-scale analysis, however, further studies that are larger and multinational in scope are needed to confirm the beneficial effects of statins on survival.
Introduction
Global cancer incidence and death have risen to 18.1 million cases and 9.6 million cancer deaths in 2018, 1 and cancer was the second leading cause of death worldwide. 2 In 2020, the type of cancer that caused the most new cases was breast cancer, followed by cancers of the lung, colon/rectum, prostate, skin, stomach, liver, cervix uteri, esophagus, thyroid, and bladder; non-Hodgkin lymphoma; pancreatic cancer; leukemia; and then kidney cancer. According to GLOCAN estimates in 185 countries, kidney cancer accounted for 2.2% (431 288 cases) of the 18.1 million new cancer cases and 1.8% (179 368 deaths) of the 9.9 million cancer deaths in 2020. 1 Kidney cancer is the third most common urological malignancy and has the highest mortality among all urological cancers.
In South Korea (hereafter, Korea), kidney cancer was the second leading type of urological cancer in 2018, with 5456 new cases (age-standardized incidence rate, 6.7 per 100 000), while the most common type of urological cancer was prostate cancer with 14 857 new cases (14.3 per 100 000) and the third most common type was bladder cancer, with 4577 cases (4.4 per 100 000). 3 Whereas the overall cancer incidence rate has remained steady, the incidence rate of kidney cancer has steadily increased from 3.1 per 100 000 in 1999 to 5.3 in 2008 and 6.7 in 2018.
Statins lower blood cholesterol levels by inhibiting 3-hydroxy-3methylglutaryl-coenzyme A (HMG-CoA) reductase, which converts HMG-CoA to mevalonate. Inhibition of the mevalonate pathway leads to apoptotic cell death. 4 and controlling this pathway modulates the production of dolichol, geranyl pyrophosphate, and farnesyl pyrophosphate.5-8
Statins have been recognized for their preventive effect against various types of cancer, although studies have yielded mixed results.9-13 For kidney cancer, several systematic reviews have reported a non-significant reduction in cancer incidence and reduced cancer mortality among statin users.11,12,14 Nonetheless, the findings in the literature regarding the effect of statins on the risk of kidney cancer are inconsistent. Many studies on kidney cancer epidemiology have also reported cigarette smoking, obesity, and hypertension as well-established risk factors for renal cancer, and genetic factors might also influence renal cancer risk.15-20 Previous studies also reported that patients with hypertension were at higher risk for kidney cancer than those without hypertension.21-24 However, little research has examined the effects of statins against renal cancer according to the presence of hypertension.
Therefore, the aim of the current study was to investigate the associations of statin use with kidney cancer risk and mortality in patients treated with statins compared to the general population considering the presence of hypertension.
Methods
Data Source
This study was a retrospective population-based cohort study using National Health Insurance claims data and national statistical data on causes of death in Korea. Data from the Health Insurance Review and Assessment Service included information on healthcare utilization; patients’ diagnoses according to the International Classification of Disease and Related Health Problems, 10th Revision (ICD-10); and drug use history for the entire population of 50 million people. In order to determine patient mortality, the claims data of deceased patients were compared against Statistics Korea data, which included official government records of the causes of death for all deceased persons as determined at the time of death by a physician, and all the causes of all deaths were recorded according to ICD-10 codes. This study was reported following the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline. 25 The protocol was approved by the Institutional Review Board of Korea University (IRB KUIRB-2020-012), and an exemption was received for using anonymous data.
Study Population and Drug Exposure
The subjects of this study were individuals aged 45-70 years old who visited a medical institution between January 1 and December 31, 2005. The study included all of new statin users whose prescriptions for statins had been filled for at least 6 months at the first index date in 2005 and who had not been prescribed statins within the previous year. Non-statin users—consisting of 802 541 people—were extracted at a proportion of 1:4 from the general population and consisted of people who had not filled a prescription for statins with matching for age and sex based on the greedy nearest neighbor matching method. Also, to adjust the difference in comorbidities, we conducted matching between statin users and non-statin users with a 1:1 ratio by propensity scores for statin use without regard to outcomes by multiple logistic regression analysis using age, Charlson comorbidity index, and comorbidities.
26
The total study population was 1 008 101 people and propensity-matched cohort was 337 578 (Figure 1). Inclusion and exclusion criteria of the study population.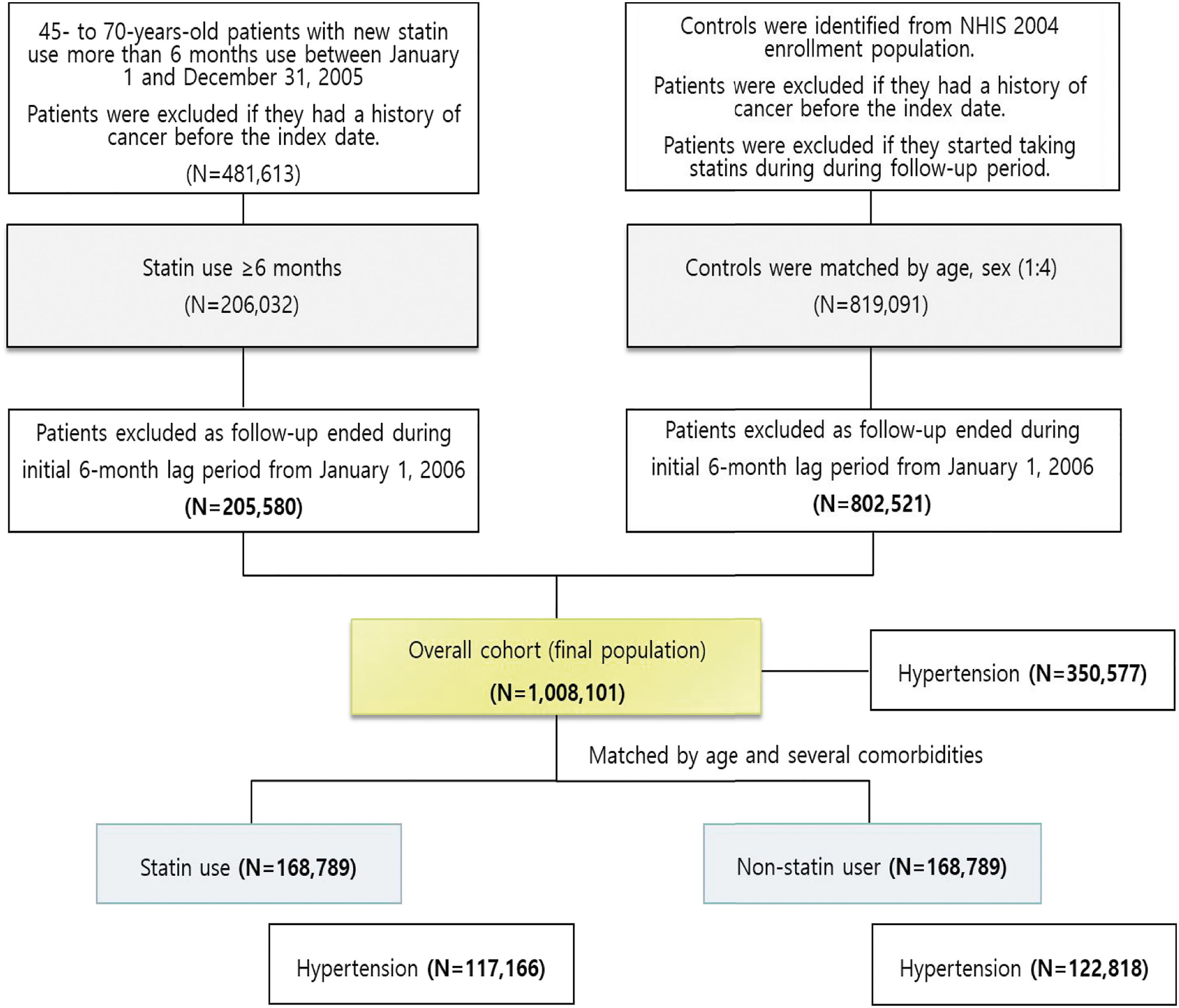
Statins were defined according to the Anatomic Therapeutic Chemical (ATC) Classification System of drugs as follows: atorvastatin, fluvastatin, lovastatin, pitavastatin, pravastatin, rosuvastatin, and simvastatin. 27 Drug exposure was calculated using the cumulative usage period from the index date in 2005 until the occurrence of the outcome. To calculate cumulative statin use, we used the date on which the drugs were prescribed, the daily dose of drugs, the number of pills per prescription, and the number of days of therapy to determine the defined daily dose (DDD) for each patient. We classified patients according to statin use (users vs non-users), and cumulative DDDs (cDDDs) of statins (less than 730 cDDDs, 730-1459 cDDDs, or 1460 or more cDDDs).
Outcome Measures
The outcomes of interest considered during the follow-up period were cancer incidence and cancer-related mortality. Cancer incidence was defined as the patient’s first hospitalization for a kidney cancer diagnosis (C64 and C65, according to the ICD-10) between January 1, 2006 (ie, 1 year after the index date) and June 30, 2013, referring to previous literature. 28 The follow-up period began 1 year after the index date, and data on the study population were recorded until cancer occurrence, death, or the end of the follow-up period (June 30, 2013). Causes of death as secondary outcomes were determined using information from Statistics Korea. We classified all causes of death, including deaths from cancer.
Potential Confounders
All possible confounders of the association between statin use and cancer according to hypertension were included, as follows: age, sex, comorbidities (Charlson comorbidity index [CCI], cardiovascular disease, diabetes mellitus, hyperlipidemia, liver dysfunction, renal failure), the number of physician visits during the baseline period, the number of hospitalizations during the baseline period, and the use of angiotensin-converting enzyme inhibitors and aspirin during the baseline period. The CCI was computed based on ICD-10 codes and was used as a summary measure. 29 CCI scores were classified as low (index score = 0), moderate (index score = 1-2), or high (index score ≥3), based on definitions from previous studies and to increase the statistical power.
Statistical Analysis
We used the chi-square test and the
We used the Cox proportional hazards regression model to calculate the hazard ratios (HRs) and 95% confidence intervals (95% CIs) for cancer incidence and mortality according to statin use after adjustment for the confounding factors. The confounding variables were age, sex, CCI, and comorbidities (hypertension, cardiovascular disease, diabetes mellitus, liver disease, renal disease), the number of physician visits during the baseline period, the number of hospitalizations during the baseline period, and the use of angiotensin-converting enzyme (ACE) inhibitors and aspirin during the baseline period. We also conducted a sub-group analysis according to the presence of hypertension.
Two-tailed
Results
General Characteristics
Baseline Characteristics of the Study Cohort and Propensity-Matched Cohort (%).

Note:
Among the matched 337 578 patients, the proportion of men was higher (39.20%) and the average age was younger (58.8 years) in statin users than in non-users. The proportion of statin users with comorbidities was similar to non-statin users, however, it showed a statistically significant difference. In total, 35.99% of statin users had received treatment for less than 4 years, 23.53% for greater than or equal to 4 and less than 6 years, and 40.48% for more than 6 years. Furthermore, 27.21% of statin users took fewer than 730 cDDDs of statins, 36.69% took 730-1459 cDDDs, and 35.10% took more than 1460 cDDDs (Table 1).
Cancer Incidence and Cancer Mortality According to Statin Use
Figure 2 shows the associations of statin use with cancer incidence and mortality. During a mean follow-up time of 7.6 (SD 1.2) person-years, there were 1133 cases of kidney cancer in the total population (1.12 per 1000 non-users vs 1.14 per 1000 statin users), and 547 cases in patients with hypertension (1.79 per 1000 non-users vs 1.27 per 1000 statin users). In the total population, 200 kidney cancer deaths were recorded (.21 per 1000 non-users vs .16 per 1000 statin users), of which 91 took place in patients with hypertension (.35 per 1000 non-users vs .15 per 1000 statin users). There was an association between statin use for at least 6 months and a reduced risk of cancer incidence and mortality. Statin use was significantly associated with a reduced risk of kidney cancer incidence (aHR = .84, 95% CI: 0.71-.99), and a reduced kidney cancer mortality (aHR = .64, 95% CI: 0.41-.98). In hypertension, there was a significant association between statin use and mortality related to kidney cancer (aHR = .60, 95% CI: 0.36-.98). Associations of statin use with the risk of kidney cancer incidence and mortality. aHR, adjusted hazard ratio. Adjusted for age, sex, Charlson comorbidity index (CCI), comorbidities (congestive heart failure, hypertension, diabetes mellitus, renal failure, liver dysfunction), and co-medication (aspirin, ACE inhibitors).
However, in matched cohort, statin use was not significantly associated with a reduced risk of kidney cancer incidence (aHR = .86, 95% CI: 0.71-1.04), and a reduced kidney cancer mortality (aHR = .80, 95% CI: 0.49-1.29). In hypertension patient among matched cohort, there was not significant association between statin use and kidney cancer or kidney cancer mortality (aHR = .86, 95% CI: 0.70-1.07 and aHR = .80, 95% CI: 0.49-1.29).
Kaplan-Meier analysis showed improvements in cancer mortality related to statin use; however, statistical significance was only found for kidney cancer survival, with statin users having a mean overall survival of 8.49 years as opposed to 8.00 years for statin non-users in both the overall population and patients with hypertension ( Kidney cancer mortality according to statin use, January 2006 to June 2013 in (A) total population, and (B) hypertension patients of overall cohort and (C) matched population, (D) hypertension patients of matched cohort. OS, overall survival.
Figure 4 shows cancer incidence and mortality according to cumulative statin use. In our analysis of the association between cDDD groups and the risk of cancer and cancer mortality, a significant dose-response relationship was not found between statin use and the risk of cancer. Compared with non-statin users, statin use of ≥1460 cDDDs was significantly associated with a reduced risk of kidney cancer incidence, with an aHR of .28 (95% CI: 0.20-.40), whereas statin use of <1460 cDDDs had a significantly higher risk (aHR = 1.23, 95% CI: 1.04-1.47). For cancer mortality, statin use of ≥1460 cDDDs was significantly associated with a reduced risk of kidney cancer mortality, with an aHR of .20 (95% CI: 0.07-.55), while statin use of <1460 cDDDs was not associated with a significantly decreased risk of cancer mortality. In patients with hypertension, the trends were similar. Statin use of ≥1460 cDDDs was associated with a reduced risk of kidney cancer incidence and mortality (aHR = .27, 95% CI: 0.18-.40, and aHR = .19, 95% CI: .06-.62, respectively) compared to non-users. However, statin use of <1460 cDDDs was associated with an increased risk of kidney cancer incidence and a non-significant risk of kidney cancer death. In the matched cohort, the trends were similar. Associations of cumulative statin use with the risk of kidney cancer incidence and mortality. aHR, adjusted hazard ratio; cDDD, cumulative defined daily dose. Adjusted for age, sex, Charlson comorbidity index (CCI), comorbidity (congestive heart failure, hypertension, diabetes mellitus, renal failure, liver dysfunction), and co-medication (aspirin, ACE inhibitors).
Discussion
This study examined the associations between statin use and cancer risk among the entire Korean population. We found that statin users had a lower risk of kidney cancer incidence and mortality than non-users, as well as improved cancer survival in total and hypertension patients. However, after matching using sex, age, and comorbidities, the reduced risk of cancer incidence and cancer mortality was not significant in patients due to small size. Additionally, cancer mortality according to cumulative statin use (in cDDDs) was examined, and a dose-response relationship was not confirmed with regard to kidney cancer incidence and mortality.
Previous studies on statin use for cancer prevention have reported conflicting results regarding cancer survival. A meta-analysis of 12 observational studies and randomized trials found a non-significant reduction of total kidney cancer risk among all statin users. 12 Another meta-analysis of 12 studies showed that statin use was associated with a significant 33% improvement in cancer-specific survival and a 26% improvement in overall survival. 11 However, a meta-analysis and umbrella meta-analysis found no effect of statins on the overall survival of patients with renal cell carcinoma.14,30 A study using 16 Veteran Affairs databases showed that statin use was significantly associated with a 48% risk reduction of renal cell carcinoma, 31 and another study found no evidence in all Danish renal cancer patients. 32 However, those previous studies differ from the present study in that they were case-control studies.
A study analyzing survival in patients undergoing surgery at a single institution reported that statins were associated with improved survival for renal cell carcinoma 33 ; in contrast, another study with patients undergoing nephrectomy found no significant association between statin use and kidney cancer survival. 10 However, those previous studies were limited to patients undergoing surgery, whereas the current study covered the total population of statin users and randomly selected age- and sex-matched individuals from the general population.
Our analysis suggests that the risk of kidney cancer incidence and mortality might be lower in statin users than in non-users, whereas previous research has found mixed evidence of risk reduction for both kidney cancer incidence and mortality among statin users compared to non-users. We used one of the largest population datasets analyzed to date, and the study design was robust to confounding. Although most systematic reviews investigating the potential effects of statins on kidney cancer reported no associations, many previous studies did not have sufficient statistical power due to the low number of events. The lack of findings regarding the risk of cancer in randomized controlled trials might be influenced by the limited long-term continuing use of statins and discontinuation of participation by those who experience symptoms of cancer. An accurate assessment of timely detection of cancer events could be conducted using a routinely collected database. In the context of these practical considerations, the design of our study might give meaningful results.
This study had several strengths. Our study included the entire population and used the National Health Insurance claims database for all statin users, with extraction of individuals from the general population at a proportion of 1:4 matching for age and sex. To date, few large-scale in-depth analyses spanning the entire national population—either for Korea or other countries—have explored the impact of statin use on kidney cancer incidence and mortality. These results, which cover the total population in a real-world setting, can be generalized to other Asian countries.
Secondly, despite the increasing incidence of kidney cancer, research on the novel cancer therapeutics for kidney cancer has remained relatively slow compared to research on other cancer types. Ethnic disparities have also been a recurrent problem in developed countries. There is a lack of evidence on the association between statin use and kidney cancer in Asian countries. Therefore, the results of this study are important, and the topic warrants further research.
Thirdly, a distinctive aspect of our study is that we classified the population according to the presence of hypertension. Although hypertension has been considered to be a predisposing factor for kidney cancer, no previous studies have investigated these factors (hypertension and statin use) simultaneously.
Fourthly, we collected data on statin dosage and stratified subjects accordingly. Although numerous studies have examined the association between kidney cancer incidence and statin use, relatively few studies have examined the effects of cumulative statin use on kidney cancer incidence and mortality. We computed and classified cumulative statin use. On this basis, we investigated the dose-response relationship between cancer risk and mortality and statin use, measured in cDDDs, which were calculated by multiplying daily dosages by the duration of statin use. This calculation is advantageous since it takes into account variable statin dosages and durations for which statins were taken.
Fifthly, we used data on individuals’ causes of death from Statistics Korea for our analysis of cancer mortality and its association with statin use.
Lastly, we defined statin users as patients who took statins for at least 6 months in order to avoid bias resulting from short-term statin users being included in the analysis. In addition, we excluded patients who died or had new cancer diagnoses within 1 year during the follow-up period to eliminate the effects of other potential underlying diseases on the analysis.
However, there also are several limitations to the study and our finding should be interpreted with caution. Given that this is an observational study, non-adherence to prescriptions could have influenced our results. The continued use of statins could potentially explain the reduced risk of death. Second, since we used claims data, we might not have fully captured other clinical risks. Moreover, confounding factors related to disease progression that may have occurred were not recorded. Lastly, although our dataset covers the entire population, these results might have been caused by racial differences or specific aspects of the Korean medical system. Therefore, these results should be interpreted with caution.
Conclusion
We found that statin use might be associated with a reduced risk of kidney cancer incidence and mortality, but it showed no statistical significance. Further studies that are larger and multinational in scope are needed to confirm the beneficial effects of statins on survival.
Footnotes
Author Contributions
HA: conceptualization and writing––review and editing. D-SK: formal analysis and methodology. D-SK and HK: writing––original draft.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
The protocol was approved by Institutional Review Board of Korea University (IRB KUIRB-2020-012), and waiver was got for using anonymous data based on the retrospective study.
