Abstract
Introduction
The national epidemiology of lung cancer (LC) in Finland, subdivided by histology and regional data, has not yet been studied in detail. This study examines the incidence, mortality, and mortality-to-incidence ratios (MIR) of LC and its subtypes in men and women during 2000-2019 in different regions of Finland.
Methods
An observational register-based study was conducted using nationwide data from the Finnish Cancer Registry (FCR). The cases were classified into adenocarcinoma (ADC), squamous cell carcinoma (SQC), small cell lung carcinoma (SCLC), and other and unknown histological types. The incidence and mortality were explored nationwide and separately in the 5 collaborative areas for healthcare and social welfare. MIR were used to estimate survival.
Results
The incidence and mortality of LC declined in men (78.6 to 25.9/100 000) but increased in women (5.0 to 15.1). The incidence of ADC increased from 4.4 to 7.2, while SQC and SCLC decreased from 5.0 to 3.6 and 3.5 to 3.0. The incidence of ADC increased in men and women, surpassing SQC as the most common histology with varying regional pace. The incidence of SQC and SCLC decreased in men but increased in women. MIR showed similar regional improvements to LC survival for ADC and SQC, but not for SCLC.
Conclusion
The Finnish LC rates changed slowly from a predominantly SQC and SCLC histology to ADC being the most common LC type. Sex-specific LC rates are converging nationwide. This study reports detailed Finnish LC data to inform future research and public health efforts.
Plain Language Summary
This study examined how lung cancer rates and types have changed across different regions of Finland. Using national cancer registry data, we found that adenocarcinoma has become the most common type of lung cancer, while overall patterns and survival rates differ by region and between men and women.
Introduction
In 2022, lung cancer (LC) was globally the most commonly diagnosed cancer (12.4% of total cases) and the leading cause of cancer deaths (18.7% of total cases). 1 In 2022, the age standardized incidence and mortality rates of LC were 23.6 and 16.8, respectively. Historically, men have always had higher LC incidence rates compared to women.1-3 In the late 20th century, a new downward trend emerged among men’s LC rates while women’s rates have continued increasing, bringing the sex-specific rates closer. Main reason for this shift is explained by women’s increasing and men’s decreasing tobacco consumption.1-3 It’s approximated that over ¾ of LC cases were attributable to smoking. Other suggested risk factors include environmental variables such as air pollution, occupational exposure, biomass fuels, household cooking and heating fuels. 1
LCs are divided into small cell (SCLC) and non-small cell lung cancers (NSCLC), of which adenocarcinoma (ADC) and squamous cell carcinoma (SQC) are the most common subtypes. 4 Previously in the United States, most LCs among men were SQC and among women ADC. Since 1990, the trend among men has also shifted towards a rising incidence of ADC.2,5 During the same timeframe, a similar change has been observed in Canada, Australia and in the majority of European countries. 2 Theories behind this shift include changes to smoking habits and the introduction of filtered cigarettes.6,7
LC incidence rate among Finnish men was twice the Nordic average in 1970 (135 vs 65/100 000), after which it declined to a more similar level the year 2000 onwards (80 vs 65). In contrast, Finnish women had markedly lower LC incidence rates compared to the Nordic average (20 vs 33) in the year 2000. 8 In the CONCORD-3 cancer survival study, LC survival in Finland was the lowest among the Nordic countries. 9 This result has brought forward the need to examine Finland’s practices on LC diagnostics and treatment. In another recent study comparing LC survival and incidence among the Nordics, the histological profile of LC in Finland was observed to differ from others. 10
Finland has a national cancer registry dating back to 1953. 11 Cancer diagnoses are reported automatically from most hospital registries and laboratory reports and completed from death certificates and manual clinician reports.11,12 The Finnish Cancer Registry (FCR) is considered very complete with up to 96% of solid and 86% of hematological malignancies cases being morphologically confirmed. 11 FCR provides yearly cancer reports and has an online open electronic data tool for cancer statistics but does not provide open data on cancer histology. 13
No previous studies have systematically investigated the changes in LC epidemiology in Finland stratified by histology, sex, and different Finnish regions. We aim to explore the Finnish national and regional LC incidence and mortality in men and women during 1968-2019. Moreover, our aim is to study LC incidence, mortality and mortality-to-incidence ratio (MIR) in relation to the histology, distinguishing between ADC, SQC, SCLC, other LC types, and unknown cases in men and women during 2000-2019. This study aims to clarify recent epidemiological trends in Finland and provide a foundation for future research and cancer control efforts.
Materials and Methods
Study Material and Methods
From 2023 onwards, Finland has had 5 collaborative areas (previously named university hospital districts) for healthcare and social welfare, which we are going to refer to them as their previous names for clarity. These areas are Helsinki (Southern Finland collaborative area), Turku (Western Finland collaborative area), Tampere (Inland Finland collaborative area), Kuopio (Eastern Finland collaborative area), and Oulu (Northern Finland collaborative area) (Supplemental Material 1).
This study is an observational, register-based, population-level study based on data from the FCR. The data FCR receives is reported automatically from most hospital registries and laboratory reports and completed from death certificates and manual clinician reports.11,12 The FCR follows the coding rules (ICD-0-3) set by the International Agency for Research on Cancer (IARC) and the International Association of the Cancer Registries (IACR), providing internationally comparable data and a completeness of up to 96% in solid malignancies.11,12
FCR’s open electronic data provides information on the LC prevalence, incidence, and mortality from 1953 to 2022. 13 Additionally, two specific data requests were made from the FCR in 2017 and 2023. A complete population-wide dataset of Finnish LC cases (topography C34 and morphology M8000/3-M9589/3) between 2000 and 2019 was acquired, including 49 760 incident cases and 41 469 mortality records during the study period. Main histological subgroups are ADC, SQC and SCLC. The fourth group includes the other, minor subtypes, for example large cell carcinoma, and the fifth group represents the cases with unknown histology or unclassified carcinomas. All the datasets included information from all 5 Finnish collaborative areas for healthcare and social welfare.
The FCR registered only histologically confirmed cases until 2012. Before 2012, LC cases diagnosed by cytological investigation were included in the unknown group. Approximately 250 cytologically confirmed LC cases are included annually; these constituted around 30% of the unknown category earlier, as seen in Supplemental Table 2. Minor biases may arise from regional differences in diagnostic practices and the classification of cytologically confirmed lung cancers before 2012, although these are expected to be minimal due to the registry’s standardized coding and high completeness.
All the data was received as aggregated and anonymized by the FCR and used with its permission. Ethical review and patient consent were waived due to the anonymized, registry-based nature of the study. The reporting of this study conforms to STROBE guidelines. 14
Statistical Methods
The incidence and mortality rates were age-standardized per 100 000 person-years using the world standard population (1966). The percentage changes in the incidence and mortality rates were calculated using the following formula:
For statistical analysis, linear mixed-effects models (LMMs) and paired t-test were used to examine temporal changes across study periods while accounting for regional variation. Separate LMMs were fitted for each histological subtype, with year period (2000-2004, 2005-2009, 2010-2014, and 2015-2019) as a fixed effect and region (Helsinki, Kuopio, Oulu, Tampere, and Turku) as a random effect, using restricted maximum likelihood estimation and a compound symmetry covariance structure. The earliest period (2000-2004) served as the reference category. Paired t-tests were used to compare pooled incidence rates between the first and last decades. Results are presented as mean differences with 95% confidence intervals (CI) and
The FCR mortality data is very complete and as a result mortality-to-incidence ratios (MIR) have been shown to have a high coherence with 1-year survival data.
11
For this reason, MIR were used to assess changes to survival in our data. For a clearer presentation, MIR were calculated as described by Vostakolaei et al, using the following formula:
Results
LC Incidence and Mortality Rates According to Sex in Finland and in Different Collaborative Areas
LC incidence rate among men declined by 67% from 78.6 to 25.9 per 100 000 person-years during the study period. On the contrary, among women the rate increased +205% from 5.0 to 15.1. Mortality followed closely these trends with a −69% decline from 68.5 to 21.0 among men and an +137% increase from 4.1 to 9.7 among women. In both sexes, the incidence and mortality rates began stabilizing during 2013-2019. Although women’s incidence rates increased yearly throughout the whole study period, mortality rates began declining after 2014 (Figure 1A–B). The Incidence (A) and Mortality (B) of Lung Cancer in Men and Women in Collaborative Areas of Finland in the Time Period 1968-2019
All collaborative areas followed closely national changes in incidence and mortality. There were some notable differences among women: At the start of the study period, the Kuopio area (3.4/100 000) had only half of the incidence of the Helsinki area (6.9). This difference was reduced at the end of our study period in 2019 (12.6 vs 16.5). Among men the incidence and mortality rates were very similar between districts, except for the Oulu area with constantly higher rates to others (Figure 1A–B).
LC Incidence and Mortality According to Histology, Sex and Areas
A complete, population-wide dataset of Finnish LC incidence and mortality records from 2000 to 2019 was obtained. Over this period, a total of 49 760 incident cases and 41 469 deaths attributable to LC were recorded. Among the incident cases, 32.5% were women and 67.5% men (not shown). 26.9% were diagnosed as ADC, 19.5% SQC, 13.0% SCLC, 10.5% other types and 30.1% unknown or unclassified carcinomas (Supplemental Table 3A). Among the mortality cases, 30.8% were women and 69.2% men (not shown). 23.9% were diagnosed as ADC, 18.8% SQC, 14.4% SCLC, 9.6% other types and 33.3% unknown or unclassified carcinomas (Supplemental Table 3B).
The Incidence and Mortality of ADC Increased in Both Sexes, But the Pace Varied Between Areas
The incidence of ADC increased nationally from 4.4 per 100 000 person-years in 2000 to 7.2 in 2019 (+64%). The rate was very stable until 2007 (4.2-4.4), after which it nearly doubled to 8.1 in 2015. Following this peak a new declining trend emerged (Figure 2A). From all the LC cases, the proportion of ADC increased from 18% to 35%. An increase was evident in both sexes respectively, from 16% to 31% in men and from 25% to 42% in women (Figure 2D–F). The Regional Incidence (Column Chart) and the National Incidence and Mortality (Line Chart) of (A) Adenocarcinoma (ADC), (B) Squamous Cell Carcinoma (SQC) and (C) Small Cell Lung Carcinoma (SCLC) in 2000-2019. The Histological Proportions of Lung Cancer (D) in Both Sexes, (E) Men and (F) Women in 2000 and 2019
The Average Incidence of ADC, SQC and SCLC in Men and Women in the Five Collaborative Areas in Finland in 2000-2019, Age-Standardized Per 100 000 Person-Years and Pooled Into Five-Year Intervals
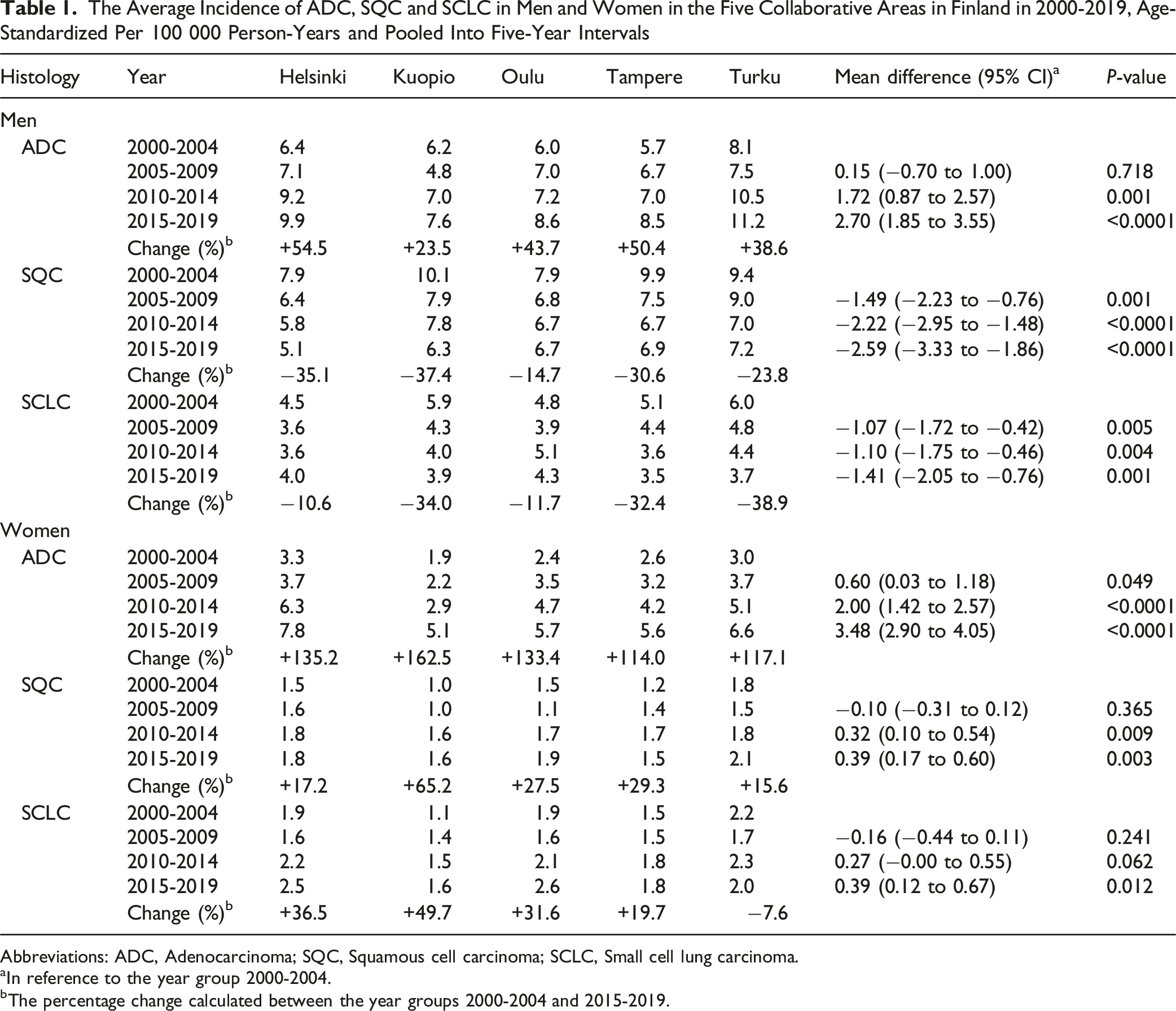
Abbreviations: ADC, Adenocarcinoma; SQC, Squamous cell carcinoma; SCLC, Small cell lung carcinoma.
aIn reference to the year group 2000-2004.
bThe percentage change calculated between the year groups 2000-2004 and 2015-2019.
The Average Mortality of ADC, SQC and SCLC in Men and Women in the Five Collaborative Areas in Finland in 2000-2019, Age-Standardized Per 100 000 Person-Years and Pooled Into Five-Year Intervals

Abbreviations: ADC, Adenocarcinoma; SQC, Squamous cell carcinom; SCLC, Small cell lung carcinoma.
aIn reference to the year group 2000-2004.
bThe percentage change calculated between the year groups 2000-2004 and 2015-2019.
The Incidence and Mortality Rates of SQC Decreased Among Men, But Increased Among Women
The incidence of SQC decreased nationally from 5.0 per 100 000 person-years in 2000 to 3.6 in 2019 (−28%). At the start of the study period there was a clear declining trend with a sudden drop in numbers (4.2-3.3) in 2007-2010. After an increase of up to 4.3 in 2012, the rates have levelled again to 3.6-3.9 in 2015-2019 (Figure 2B). From all the LC cases, the proportion of SQC slightly decreased from 22% to 19% nationally. There was a decrease in proportion from 26% to 23% in men and a slight increase from 11% to 12% in women (Figure 2D–F).
The number of SQC cases greatly decreased among men in every collaborative area (−2.59/100 000 [–3.33 to −1.86],
The mortality rate of SQC has declined on a national level. The mortality rate followed that of the incidence closely, with a decline from 4.2/100 000 in 2000 to 2.6 in 2019 (−38%). Interestingly, while incidence rates showed a sharp decline in 2010, mortality briefly spiked upward before realigning with the declining incidence trend thereafter (Figure 2B). A decrease was observed among men in all regions (−2.88 [–3.35 to −2.40],
The Incidence and Mortality Rates of SCLC Are Similar in Different Areas
The incidence of SCLC showed a declining trend from 3.5 per 100 000 person-years in 2000 to 2.5 in 2006 (−29%) but has since increased to 3.0 in 2019 (−14%). In the past 10 years, the incidence rate has remained stable at around 2.8-3.1, with very little annual variation (Figure 2C). From all the LC cases, the proportion of SCLC slightly decreased from 15% to 13% nationally, in both men and women (Figure 2D–F).
The number of SCLC cases decreased among men in every collaborative area (−1.41/100 000 [–2.05 to −0.76],
On a national level, the mortality rate of SCLC has declined. The mortality rate mirrored closely that of the incidence, decreasing from 3.3/100 000 in 2000 to 2.6 in 2019 (−21%) (Figure 2C). In the last 5-year period, mortality rates for SCLC and SQC have been very similar, despite the markedly lower incidence of SCLC (Figure 2B–C). A decrease in mortality was observed in all collaborative areas (−1.25 [–1.83 to −0.68],
Mortality-to-Incidence Ratio Showed Improving Trends in ADC and SQC Survival, But Not in SCLC
Throughout the study period, ADC had the highest survival with SQC close second. On a national level, the MIR of ADC increased from 20.9% to 33.1%. An increase was visible in both men (11.4% [5.1% to 17.8%], The National (A) and Regional (B-D) Mortality-to-Incidence Ratio (MIR) of Adenocarcinoma (ADC), Squamous Cell Carcinoma (SQC) and Small Cell Lung Carcinoma (SCLC) With Pooled Five-Year and Ten-Year Intervals and the Pooled Interval Average (Avg.) for Men (M) and Women (W) in 2000-2019
During the years 2015-2019, the Oulu area had the highest 5-year survival rate in all examined histological subgroups among men (ADC 32.5%, SQC 30.0%, SCLC 11.5%) and the Tampere area among women (ADC 46.6%, SQC 38.5%, SCLC 23.7%). The 10-year periods were very similar between the regions. Interestingly, the Helsinki area showed the most favourable outcomes among women in the SQC 10-year period group but exhibited no improvement among men. In contrast, the Kuopio area performed comparably to other regions among men but had the poorest outcomes among women. In the SCLC subgroup, regional variation was minimal, and improvements in survival were similarly limited (Figure 3B–D, Supplemental Table 5B-E).
Discussion
We examined the epidemiology of different histological subtypes of LC in both sexes in all collaborative areas for the first time. In most Nordic countries, ADC surpassed SQC as the most common LC histology during the 1990’s or even earlier.2,10 In Finland, however, the change happened approximately 10 years later, with regions like Kuopio going as far as 2014 in men’s group. As SQC and SCLC are associated with worse prognosis compared to ADC, it’s a natural partial explanation for Finland’s worse overall LC survival among the Nordics. Yet when histology specific survival was examined, Finland had more comparable 1- and 5- year ADC specific survival results, but clearly worse SQC and SCLC specific results, requiring further investigation.10,16,17
In 2020, Finland and Denmark shared similar slightly above median smoking prevalence to other Nordics, yet the LC histological distribution and LC survival in Denmark resemble more those of Norway and Sweden.10,17,18 Men’s smoking rates in Finland have declined for over 50 years, while women’s rates first increased for 30 years, before starting to descend in the early 2000’s.19,20 In 2019, the absolute difference in proportional smoking rates between the sexes was only 5% (men 14% vs women 9%), compared to the 40% (men 51% vs women 11%) in 1972.20,21 Since the latency from smoking to LC is usually 20 to 30 years, we can argue that the sex-specific incidence rates in Finland might converge in the future. In Sweden, Iceland and Denmark, women’s (35-64 years) LC incidence has already surpassed that of men. 3
Smoking in the Nordics is more common in the population with low socioeconomical status (SES). 19 Low SES is an independent risk factor for LC and an estimated half of that risk was explained by higher smoking prevalence alone.22,23 Low education levels were associated with a doubled risk for the incidence and mortality of LC in Finland. 24 Curiously, education levels were very similar between the Nordics and the Finnish regions in 2000-2013.19,25,26 When the Nordic SES was examined through family wealth, Finland ranked last. 27 This could be a reflection of the deep economic crisis in the 1990s, where among the Nordics Finland had the highest unemployment rate and the slowest recovering. 28 Additionally, during the 20th century Finland experienced a slower transition in work force from the industrial sector to the service sector, theoretically having a greater number of people exposed to occupational chemicals. 19 Blue-collar work, smoking and low SES are all associated more strongly with SQC and SCLC compared to ADC, thus possibly contributing to the histological differences present in Finland.22,23
Although age is a well-known risk factor for LC, age trends in Finnish women seem paradoxically inversed with LC incidence. 29 For the last 20 years, the Helsinki area had the lowest and the Kuopio area the highest proportion of women aged over 65 years, yet contradictorily women’s LC incidence was highest in the Helsinki area and lowest in the Kuopio area. Among men, regional LC incidence behaved more linear with age. Inconsistent associations between LC and traditional risk factors among women have been reported previously, for example in Hungary, emphasizing the need for a deeper understanding of the disease in women to improve preventive work. 30
Since the histology specific survival in Finland is behind other Nordics, one may assume that either the Finnish LC patients have worse prognosis from the start (e.g., have higher staging or worse performance status), or Finland treats lung cancer patients differently to other Nordics. Unfortunately, no studies comparing LC patient’s staging or performance status between the Nordics exist. When the usage of new systemic anti-cancer therapies in the European countries was compared, Finland was found being more conservative. 31 The reimbursement process in Finland is slow, which can cause delays in implementing new treatments. 24 Once treatment is accepted, it is equally available regardless of the collaborative area. As per international recommendations, LC treatment decisions are usually made in multidisciplinary teams. Finland has an active Lung Cancer Committee which provides recommendations to LC treatment paradigms and a Finnish Lung Cancer Program, which works to improve treatment protocols, research possibilities and to study the implementing of LC screening in Finland. 24
Our study has its obvious limitations due to its retrospective nature and register-based data. The FCR collects variables only directly associated with the tumour in question. 12 Due to this limitation, the current study lacks data on important variables for example smoking rates and tumour stage, and we are only left able to discuss possible reasons behind temporal and regional changes to Finnish LC rates. The strength of the study is the excellent completeness of the FCR data, which reflects accurately the Finnish LC situation. 11 The FCR follows the reporting rules set by the International Agency for Research on Cancer (IARC) and the International Association of the Cancer Registries (IACR), providing internationally comparable data and allowing our results to be compared with those from other countries.
The number of cases which were classified into the unknown category decreased by the end of our research period. This was mainly because cytological diagnoses have only been registered from 2012 onwards, which reduced the number of unknown cases greatly (Supplemental Table 2). During our study period of 2000-2019, LC diagnostic methods such as computed tomography, biopsy techniques and immunohistochemistry have developed immensely, which also explain diminishing unknown category. We do not believe that the changes to unknown group affected the proportional rates of other histological subtypes greatly, nor that it is the phenomenon behind changing ADC/SQC ratio in Finland, since it is unlikely a certain histology would be disproportionately represented within the unknown group.
This study provides detailed epidemiological data on LC to contextualize Finland’s limited LC survival rates. Further research is needed to identify the underlying causes of these results. We propose that the FCR should include additional variables, such as tumour staging, smoking status, and treatment information. Incorporating these data would facilitate more effective regional and international comparisons in future studies and help direct resources where they can have the greatest impact on public health, such as intensified tobacco control programs or screening protocols.
Conclusion
In Finland, lung cancer histology has gradually shifted from predominantly SQC and SCLC to ADC as the most common subtype. MIR trends show improving survival in ADC and SQC but not in SCLC. Sex-specific LC rates are converging nationwide, with declining trends in men and increasing trends in women. These findings provide detailed epidemiological context for understanding Finland’s limited LC survival outcomes and support future health policy planning.
Supplemental Material
Supplemental Material - Changing Epidemiology of Lung Cancer in Finland: A Registry-Based Study by Histology and Region
Supplemental Material for Changing Epidemiology of Lung Cancer in Finland: A Registry-Based Study by Histology and Region by Laura Puuniemi, Johanna Mäkinen, Jussi Koivunen, Riitta Kaarteenaho in Cancer Control
Footnotes
Ethical Considerations
All the data was received as aggregated and anonymized by the Finnish Cancer Registry and used with its permission. Ethical review and patient consent were waived due to the anonymized, registry-based nature of the study.
Author Contributions
LP: Writing- Original draft, Conceptualization, Investigation, Methodology, Formal analysis, Visualization. JM: Investigation, Writing- Review and Editing. JK: Writing – Review and editing. RK: Project administration, Supervision, Writing- Original draft, Conceptualization, Methodology, Investigation, Resources.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: LP was supported by The Foundation of the Finnish Anti-Tuberculosis Association, The Research Foundation of the Pulmonary Diseases and the Foundation of Väinö ja Laina Kiven Säätiö. JM received no funding. JK received grants from Cancer Foundation Finland, and a state subsidy from Oulu University Hospital. RK received grants for the research group from the Foundation of the Finnish Anti-Tuberculosis Association, The Research Foundation of the Pulmonary Diseases, The Research Foundation of North Finland, The Jalmari and Rauha Ahokas Foundation, and a state subsidy from Oulu University Hospital.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LP has received a congress travel grant from MSD. JM has received a congress travel grant from Roche. JK reports personal fees from Roche, personal fees from AstraZeneca, personal fees from Janssen, personal fees from BMS, personal fees from Merck, personal fees from Amgen, personal fees from Novartis, personal fees from Merck KgA, lecturing fees from Siemens Heatlhineers, personal fees from Sanofi, personal fees from Janssen, and Pfizer all outside the submitted work. JK is a former part-time employee at Faron Pharmaceuticals. RK reports consulting, lecture, and advisory board fees from Boehringer Ingelheim, a congress cost from Orion, an advisory board fee from Boehringer Ingelheim, outside the submitted work.
Data Availability Statement
The data used in this study were obtained from the Finnish Cancer Registry under a specific data permit. Access requires separate permission from the Finnish Cancer Registry.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
