Abstract
Introduction
Breast cancer is the leading cause of cancer-related deaths among women worldwide. In Costa Rica, it ranks first in incidence and fourth in terms of mortality. However, there is a lack of comprehensive information on treatment patterns and outcomes for breast cancer patients in Costa Rica.
Methods
This study utilized data from the National Tumor Registry, which was merged with the Costa Rica Social Security Fund (CCSS) to ensure comprehensive access to clinical information. The study is prospective and focused on patients diagnosed with breast cancer between January 2008 and December 2012. This combined dataset allowed for a more comprehensive analysis of patient characteristics, treatment patterns, and outcomes related to breast cancer in Costa Rica.
Results
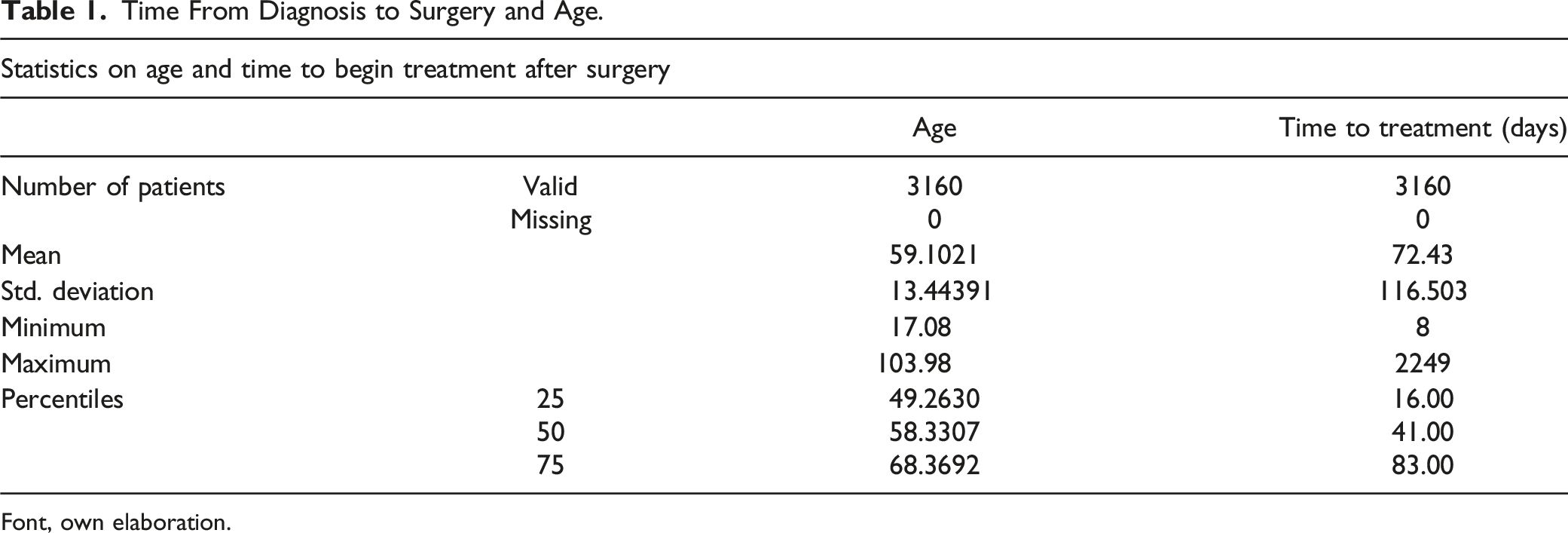
Among the 4775 patients diagnosed during this period, 3160 met the inclusion criteria for our study. The average age at diagnosis was 59.1 years, with 32.5% of patients being over the age of 65. Most of the patients (55.4%) identified themselves as homemakers, while 46.5% underwent core needle biopsy for diagnosis. Approximately 60% of women were diagnosed with early-stage disease (IA, IIA, and IIB), while 1.7% had metastatic disease, mainly affecting the bone. The mean interval between diagnosis and surgery was 72 days. Most patients (88.7%) received surgery as their initial treatment, and over half (54.4%) received some form of adjuvant therapy. Additionally, 85.6% of patients completed their prescribed treatment.
Conclusion
This study provides a comprehensive and detailed description of the characteristics and treatment patterns among breast cancer patients in Costa Rica. The findings contribute to our understanding of the disease in this population and can serve as a foundation for further research and improvement in breast cancer management and care.
Introduction
Cancer is a global public health challenge, responsible for nearly 10 million deaths each year. 1 Of particular concern is the fact that approximately 70% of these deaths occur in low- and middle-income countries. 1 Among all cancers, breast cancer has the highest mortality rate. 2
In Costa Rica, cancer is the second leading cause of death after cardiovascular diseases, accounting for 21% of all deaths. 3 Breast cancer is the most common cancer among women in Costa Rica, representing 24.5% of all cancer cases and 7.2% of all cancer-related deaths. 3
The Costa Rican health care system, managed by the Ministry of Health, includes the National Tumor Registry (NTR), which collects data on cancer patients’ sex, age, place of residence, incidence, and mortality. The Caja Costarricense del Seguro Social (CCSS) is responsible for providing medical care, diagnosis, treatment, and follow-up of patients. Nearly 80% of cancer diagnoses in Costa Rica are made at the CCSS. 4
Despite the high burden of breast cancer in Costa Rica, few studies have provided a comprehensive description of the characteristics of the patient population and their tumors. Therefore, the purpose of this study is to provide a detailed description of the clinicopathological features of the entire population of breast cancer patients in Costa Rica during the study period, with a focus on characteristics that have not been previously reported. By addressing this gap in knowledge, this study has the potential to inform public health strategies and improve breast cancer outcomes in Costa Rica.
Methods
Data Sources
The study is prospective and utilized administrative data on breast cancer patients reported between 2008 and 2012, provided by the statistics department of the Caja Costarricense del Seguro Social (CCSS). The CCSS is the national social security institution responsible for providing universal health care coverage in Costa Rica. The National Tumor Registry (NTR) database belonging to the Ministry of Health independent of CCSS was used to identify women aged 16 years and older who were registered with invasive breast cancer between January 1st, 2008, and December 31st, 2012. It should be noted that the most recent data available in the NTR was from 2014 and 2016 for cancer, as we did not have access to more recent information due to the limitations of the available database. The study protocol was evaluated by the Central Scientific Ethics Committee (CSEC), registered with the National Council for Health Research (NCHR) with code R015-SABI-00074.
Population and Data Collection
The study included all women aged 16 years and older who were registered in the NTR database and diagnosed with invasive breast cancer during the study period. A total of 4775 women were identified, and 3160 patients were included in the final cohort after excluding those diagnosed in private establishments, those with an unknown place of diagnosis, those treated in health centers that diagnosed less than 50 cases, and those without a file or clinical history. Written consents were obtained from each participant.
Data collection involved a comprehensive review of paper files for each individual patient. Data were collected from multiple sources, including public hospitals, by completing a standardized form that was specifically designed for this purpose. The data collection process ensured that information on the same individuals was gathered from all relevant databases and sources.
The collected data included various aspects of patient characteristics, such as age at diagnosis, time to treatment, hospital where patients were treated, occupation, age group, family cancer history, mammography, and ultrasound. Additionally, breast characteristics were recorded, including laterality, location of the lesion within the gland, and the diagnostic procedure performed. Tumor characteristics, such as the clinical stage of the tumor according to the AJCC classification seventh edition, were also documented, along with the clinical stage (early, late, and metastasis), number of positive nodes or cancer, histological pattern, degree of differentiation, tumor phenotype, site of metastasis (if any), invasion, Ki-67%, and HER2 status.
Furthermore, treatment characteristics were captured, including the type of surgery performed (conservative or radical) and the receipt of adjuvant therapies such as chemotherapy (QT), hormone therapy (HT), and radiotherapy (RT). Information regarding palliative or neoadjuvant therapies, as well as any combinations of treatment regimens, was also collected. Finally, the main medications used in the treatment of breast cancer were documented.
Dates of death were obtained from the NTR, medical centers, digital records, and death certificates and were further validated using the database of the Supreme Electoral Tribunal (SET).
Data Analysis
Continuous variables were summarized using the arithmetic mean with standard deviation and median with range, while categorical variables were summarized using relative frequencies, ratios, and 95% confidence intervals (CIs) for descriptive purposes. Descriptive statistical analysis was conducted using SPSS version 26 (IBM Corp., Armonk, NY, USA) and STATA version 17 (Stata Corp LLC, College Station, TX, USA) programs. Results were presented in tables and figures.
Results
General and Clinical Characteristics
Time From Diagnosis to Surgery and Age.
Font, own elaboration.
Description Analysis of the Cohort.

Font, own elaboration; CCSS, Costa Rican Social Security Fund; H., hospital.
Regarding occupation, the majority of patients (55.4%) described themselves as homemakers, followed by 10.7% as inactive at that time (n = 338), 7.8% as informal manual workers (n = 246), 5.2% as informal office workers (n = 164), and 4% as professionals (n = 126). Additionally, 32.5% of patients were over 65 years of age, and 14.2% were under 46 years. Furthermore, 22.3% reported having a family member with a history of cancer. For diagnosis, 37.3% received mammograms (MMGs), and 36.4% received ultrasounds (USs) for their cancer diagnosis in the CCSS. It is noteworthy that 35% of mammograms and 31.1% of ultrasounds were conducted in the private sector. Moreover, 47.6% of findings in mammograms and 40.7% of findings in ultrasounds were suspected of malignancy (see Table 2).
Breast Characteristics.

Font, own elaboration.
bFNB, fine needle biopsy.
aCore needle biopsy.
The most commonly used diagnostic procedure was core needle biopsy (CNB) in 46.5% of cases, followed by incisional biopsy (IB) in 24.6%, excisional (open) biopsy in 12.2%, and harpoon biopsy in 4.7%. Only 5.2% of patients underwent fine needle biopsy (FNB) (see Table 3).
Tumor Stage.

Font, own elaboration.
Pathological Characteristics of the Tumor.

Font, own elaboration.
The most frequent sites of metastasis were bone (3.0%), lungs (2.1%), and liver (1.4%). Only 4.5% of the patients reported attending the clinic due to “orange skin” in the breast. Vascular invasion was absent in 63.0% of cases, while 52.5% of cases had no lymphatic invasion. Vascular invasion was present in 24.9% of cases, and lymphatic invasion was observed in 35% of cases. Overall, 17.3% had positive lymphatic and vascular invasion. Specifically, 80.0% of reported Ki-67% patients were negative, and only 12.9% had elevated Ki-67%. 69.3% of women presented HER2 positive. Among patients reporting HER2 positive (+++), 76.9% were Ki-67 negative, and only 14.6% of patients had HER2 overexpression and elevated Ki-67 (>20%) (see Table 5).
Treatments of the Cohort
The average waiting time between diagnosis and surgery was 72 days. In terms of surgical treatment, 77.9% of patients underwent surgery within 90 days after diagnosis, while 22.1% waited more than 90 days. The majority (88.7%) underwent surgery as the first treatment. Among the surgical options, conservative surgery was performed in 46.6% of cases, radical surgery in 41.8% of cases, and the status was unknown in 11.6% of cases. At least one distant metastasis was observed in 11.3% of cases.
Treatments of the Cohort.

Font, own elaboration.
Concerning neoadjuvant therapies, 17.7% of patients received neoadjuvant QT, 1.3% received neoadjuvant RT, and 8.4% received neoadjuvant HT. Palliative QT was administered to 7.7% of patients, palliative RT to 2.5%, and palliative HT to 5.6% (see Table 6).
Combination treatments were reported in 58.3% of the patients. The most frequently used combination was QT, HT, and RT, accounting for 24.0% of cases, followed by HT and RT in 15.3% of cases, and QT with HT in 9.9% of patients. Monotherapy after surgery was low for all 3 available therapies, with QT used in 2.7% of cases, RT in 3.0% of cases, and HT in 9.8% of cases (see Table 6).
Type of Medication (Chemotherapy) Given to Patients.

Font, own elaboration.
Discussion
Breast cancer is a significant public health issue worldwide, with variations in its incidence and mortality rates across different countries and regions. In Costa Rica, breast cancer stands as the leading cause of cancer deaths among women, with an incidence rate of 41.3 per 100,000 women per year and a mortality rate of 13.7 per 100,000 women per year. 5 The current study aimed to provide valuable insights into the general and clinical characteristics of breast cancer patients in Costa Rica and the treatments they received.
Consistent with global patterns, the study findings revealed that the majority of patients were middle-aged women. The prevalence of invasive ductal carcinoma as the most frequent histological type aligns with the global prevalence as well. 6 Similarly, the predominance of the luminal type A subtype is in accordance with global patterns of distribution. 7
Interestingly, the study observed a different distribution of affected quadrants compared to the global pattern. In this cohort, the Superior External Quadrant (SEQ) was the most frequently affected, followed by the Superior Internal Quadrant (SIQ), according to the commonly reported SEQ involvement. 8
Metastasis patterns in this study were consistent with the literature, with bone, lungs, and liver being the most frequent sites of metastasis. 9 The absence of vascular invasion and lymphatic invasion in the majority of patients may indicate an early stage of the disease at the time of diagnosis. However, it is important to note that missing values in the data may have impacted the accuracy of these findings.
In terms of treatments, the study revealed a longer average waiting time between diagnosis and surgery (72 days) compared to the recommended waiting time of 30 days by the World Health Organization. 10 This finding suggests the need for improved access to timely surgical care for breast cancer patients in Costa Rica. The preference for surgery as the first treatment aligns with global treatment patterns. 11 Approximately half of the patients received adjuvant therapies, and combination therapies were frequently reported. The most commonly used medications included tamoxifen, aromatase inhibitors, anthracyclines, and taxanes. 12
It is important to acknowledge that this study has some limitations. The missing data points may have influenced the accuracy of certain results. Additionally, the absence of direct comparisons with other health care systems or references to global patterns limits our ability to fully contextualize these findings. Further research and studies with comparative analyses are warranted to gain a more comprehensive understanding of breast cancer characteristics and treatments in Costa Rica.
Conclusion
In conclusion, this study has provided a comprehensive overview of the general and clinical characteristics, as well as the treatment patterns, of breast cancer patients in Costa Rica. However, it is important to address the concerns raised regarding the practical implications of these findings for improving patient outcomes.
While the study offers valuable insights into the epidemiology and management of breast cancer in Costa Rica, it is essential to bridge this gap and highlight the potential impact of these findings on patient care and outcomes.
By utilizing the data generated in this study, health care providers can gain a better understanding of the specific needs and characteristics of breast cancer patients in Costa Rica. This knowledge can inform the development of targeted interventions aimed at improving early detection, access to treatment, and overall quality of care. For example, the identification of longer waiting times between diagnosis and surgery suggests a need to prioritize efforts to reduce delays and ensure timely surgical interventions.
Furthermore, the study’s findings regarding treatment patterns and medication usage can guide clinicians in selecting evidence-based treatment options that have shown effectiveness in the local context. This can lead to improved treatment outcomes and tailored therapeutic approaches for individual patients.
Moreover, policymakers can utilize these data to inform health care policies and resource allocation strategies. Understanding the prevalence of different histological types, stages, and sites of metastasis can aid in the development and implementation of targeted screening programs, specialized treatment centers, and support services for breast cancer patients.
In summary, by bridging the gap between the study’s findings and their practical application, this research has the potential to directly benefit breast cancer patients in Costa Rica. It can inform health care providers in delivering more personalized and effective care, guide policymakers in implementing evidence-based interventions, and ultimately contribute to improved patient outcomes and enhanced quality of life.
Strengths and Limitations
Strengths
• The study provides a comprehensive overview of the general and clinical characteristics and treatments of breast cancer patients in Costa Rica, which can inform health care providers and policymakers in improving breast cancer care and outcomes. • The study had a large sample size, including patients from multiple public hospitals across the country, which increases the generalizability of the findings. • The data were collected through a standardized process and analyzed using appropriate statistical methods.
Limitations
• The study only included patients from public hospitals, which may not be representative of the entire population. Patients from private hospitals may have different demographic and clinical characteristics and access to different treatments. • The study did not explore the reasons for the observed patterns of diagnosis and treatment, which could be the subject of further research. • The study did not collect data on the socioeconomic status of the patients, which may have an impact on the disease burden and health care utilization patterns. • The study did not collect data on patient outcomes, such as survival rates and quality of life, which are important for assessing the effectiveness of the health care system.
Footnotes
Acknowledgments
The authors would like to thank Lymarie Maldonado-Baez (Deputy Director, Cancer Prevention Program, NCI) for her help with guidance in the process. The authors gratefully thank Michael Halpern (Health Assessment Research Branch) for his support and guidance. The authors would like to thank Dera Tompkins (NIH Library Editing Service) for reviewing the manuscript and the NIH Fellows Editorial Board (FEB) for assistance with manuscript editing.
Author’s Note
The content of this paper is the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or the National Cancer Institute.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Costa Rica Social Security Fund.
Ethical Approval
The protocol was evaluated by the Central Scientific Ethics Committee (CSEC), located in the Directorate of the Center for Strategic Development and Information on Health and Social Security of Costa Rica, and registered with the National Council for Health Research (NCHR) under code R015-SABI-00074.
