Abstract
Introduction
Combined small cell lung cancer (C-SCLC) represents a rare subtype of all small cell lung cancer cases, with limited studies investigated its prognostic factors. The aim of this study was to construct a novel nomogram to predict the overall survival (OS) of patients with C-SCLC.
Methods
In this retrospective study, a total of 588 C-SCLC patients were selected from the Surveillance, Epidemiology, and End Results database. The univariate and multivariate Cox analyses were performed to identify optimal prognostic variables and construct the nomogram, with concordance index (C-index), receiver operating characteristic curves, and calibration curves being used to evaluate its discrimination and calibration abilities. Furthermore, decision curve analysis (DCA), integrated discrimination improvement (IDI), and net reclassification index (NRI) were also adopted to assess its clinical utility and predictive ability compared with the classic TNM staging system.
Results
Seven independent predictive factors were identified to construct the nomogram, including T stage, N stage, M stage, brain metastasis, liver metastasis, surgery, and chemotherapy. We observed a higher C-index in both the training (.751) and validation cohorts (.736). The nomogram has higher area under the curve in predicting 6-, 12-, 18-, 24-, and 36-month survival probability of patients with C-SCLC. Meanwhile, the calibration curves also revealed high consistencies between the actual and predicted OS. DCA revealed that the nomogram could provide greater clinical net benefits to these patients. We found that the NRI for 6- and 12-month OS were .196 and .225, and the IDI for 6- and 12-month OS were .217 and .156 in the training group, suggesting that the nomogram can predict a more accurate survival probability. Similar results were also observed in the validation cohort.
Conclusion
We developed and verified a novel nomogram that can help clinicians recognize high-risk patients with C-SCLC and predict their OS.
Introduction
Small cell lung cancer (SCLC) consists of 15% of lung cancer cases and is characterized by an exceptional aggressive, early occurrence and metastasis, and poor prognosis. 1 According to the 1999 World Health Organization classification of lung cancer, SCLC can be divided into pure small cell lung cancer (P-SCLC) and combined small cell lung cancer (C-SCLC).2,3 C-SCLC refers to a mixture of SCLC and non-small cell lung cancer (NSCLC) components, in which NSCLC components could be squamous cell carcinoma (SCC), adenocarcinoma (ADC), large-cell neuroendocrine carcinoma (LCNEC), spindle-cell carcinoma, and giant cell carcinoma.2,4 As a rare subtype of SCLC, it is reported that C-SCLC makes up about 10% of all SCLC cases. 5 In fact, the actual incidence of C-SCLC may be higher than this level because most C-SCLC patients were diagnosed through postoperative pathology. 5 Because of increased crush artifact and fewer cells in small sample biopsy, specimens from bronchoscopy and needle biopsy are challenging to make a precise diagnosis for C-SCLC.1,6,7
Although an increasing number of studies focused on the therapeutic progress and survival outcome of patients with SCLC, only limited studies investigated the clinical characteristics, prognosis, and relevant prognostic indicators of C-SCLC. According to some previously published studies, C-SCLC shared some common epidemiological and clinical characteristics with SCLC: they are prevalent in men and smokers, and most patients were diagnosed at the time of advanced disease stage.1,5 In a retrospective study conducted in China, Lei et al. revealed that surgery is still the optimal and effective treatment option for early-stage C-SCLC. 2 They also indicated that the subsequent adjuvant chemotherapy could improve the OS of these patients. 2 Recently, He and his colleagues reported that IA-IB stage C-SCLC could benefit from surgery. 5 However, adjuvant chemotherapy seemed to have few effects on improving the survival outcome of these patients. 5 Despite the fact that few studies have explored the prognostic factors of C-SCLC, their conclusions are inconclusive and based on the small sample size single cohort study.8-11 Hence, it is urgently needed to identify the prognostic factors of C-SCLC and develop a risk stratification system to recognize high-risk patients and initiate an early intervention for these patients.
To the best of our knowledge, there was no available nomogram constructed to predict the survival probability of C-SCLC so far. In this premier, we aimed to investigate the prognostic factors of patients with C-SCLC using a large C-SCLC cohort from the Surveillance, Epidemiology, and End Results (SEER) database and develop a novel risk stratification system to predict their overall survival (OS). Besides, we also verified the nomogram in a validation cohort and performed a series of tests to evaluate its performance and clinical utility.
Methods
Data Acquisition
All patients were obtained from the SEER database in this large population-based retrospective study, with SEER*Stat 8.35 used for data extraction. The SEER database collects data from 18 cancer registries of the National Cancer Institute and it includes data of nearly 30% of US population. 12 The latest information on follow-up and prognosis of the SEER database was released in December 31, 2016. We conducted this study under the requirement of the Declaration of Helsinki.
Patients Selection
Patients diagnosed with C-SCLC between 1975 and 2016 were initially identified from the SEER database. The detailed criteria for inclusion and exclusion are as follows: (1) malignancies that originated in main bronchus and lung (SEER primary site code: C340-C349); (2) the International Classification of Diseases code O-3 morphology was 8045 (for all SCLC patients: 8041-8045); and (3) patients without complete records for American Joint Committee on Cancer (AJCC)-TNM staging, treatment, OS, and other crucial clinical information were excluded. In the present study, we did not perform power calculation for estimation of sample size.
Cohort Establishment and Variable Selection
All eligible patients enrolled in the whole dataset were randomly divided into training and validation cohorts according to a ratio of 7:3 by exploiting the “createDataPartition” function in R software. In the current study, the training cohort was used to develop a predictive signature, with the validation cohort being adopted to verify its predictive ability and clinical utility. Seventeen variables were obtained from the SEER database, including age at diagnosis, gender, race, marital status at diagnosis, tumor location, tumor grade, AJCC-T stage, AJCC-N stage, AJCC-M stage, clinical stage, surgery type, radiation status, chemotherapy status, and location of distant metastasis. Then, we adopted univariate and multivariate Cox regression analyses to select optimal variables for predictive model construction.
Statistical Analysis
All categorical variables were summarized as count and percentage, with a Chi-square test being adopted to compare the difference between the training cohort and the validation cohort. The univariate and multivariate Cox regression analyses were used to identify independent prognostic factors for patients with C-SCLC. All variables with P-value <.05 in the univariate analysis were selected into multivariate Cox regression analysis. The optimal variables in the multivariate analysis were used to construct a nomogram. Besides, we calculated the concordance index (C-index) and generated receiver operating characteristic (ROC) curves and calibration curves to evaluate the discrimination ability and calibration ability of the nomogram in the 2 cohorts. Furthermore, we also performed decision curve analysis (DCA)13-15 and calculated net reclassification index (NRI) and integrated discrimination improvement (IDI) 16 to assess the clinical utility and net clinical benefits when the nomogram was adopted to guide clinical practice. In this study, R software version 3.6.3 and SPSS software version 23.0 for Windows were adopted for all statistical analyses.
Results
Clinical Characteristics of the Participants
Overall, 2329 cases were confirmed as C-SCLC in the SEER database according to the previously defined criteria. After excluding patients in accordance with the previously defined inclusion and exclusion criteria, 588 C-SCLC patients were included in this study, as presented in Figure 1. The mean age of patients in the whole cohort was 67.6±9.0 years old. There were 314 male patients and 274 female patients. White people were the most predominant ethnicity, accounting for 84.0% of cases. The vast majority of patients (69.9%) were diagnosed at the advanced disease stage. It showed that 66.7% of patients received chemotherapy and 46.3% received radiotherapy, while only 30.1% of patients underwent surgery. Regarding the detailed surgery type, 117 patients underwent lobectomy, 33 patients underwent wedge resection, and 16 patients received pneumonectomy, respectively. Besides, we observed that the liver was the most common distant metastasis organ, accounting for 14.3% of patients, followed by bone (13.9%) and lung (12.9%). The detailed demographical and clinicopathological characteristics were summarized in Table 1. Flow chart of the study. Demographic and Clinical Characteristics of Patients with C-SCLC. Abbreviations: C-SCLC, combined small cell lung cancer; AJCC, American Joint Committee on Cancer. * represents P value< .05.

Then, all patients were randomly divided into training cohort (412 patients) and validation cohort (176 patients) according to a ratio of 7:3, with a Chi-square test being adopted to examine whether there was a statistical difference between the two cohorts. It showed that except for tumor grade and AJCC-N stage, there was no significant statistical difference among other clinicopathological characteristics (Table 1).
Univariate and Multivariate Cox Regression Analysis
Univariate and Multivariate Cox Analyses on Variables for the Prediction of Overall Survival of Patients With C-SCLC.
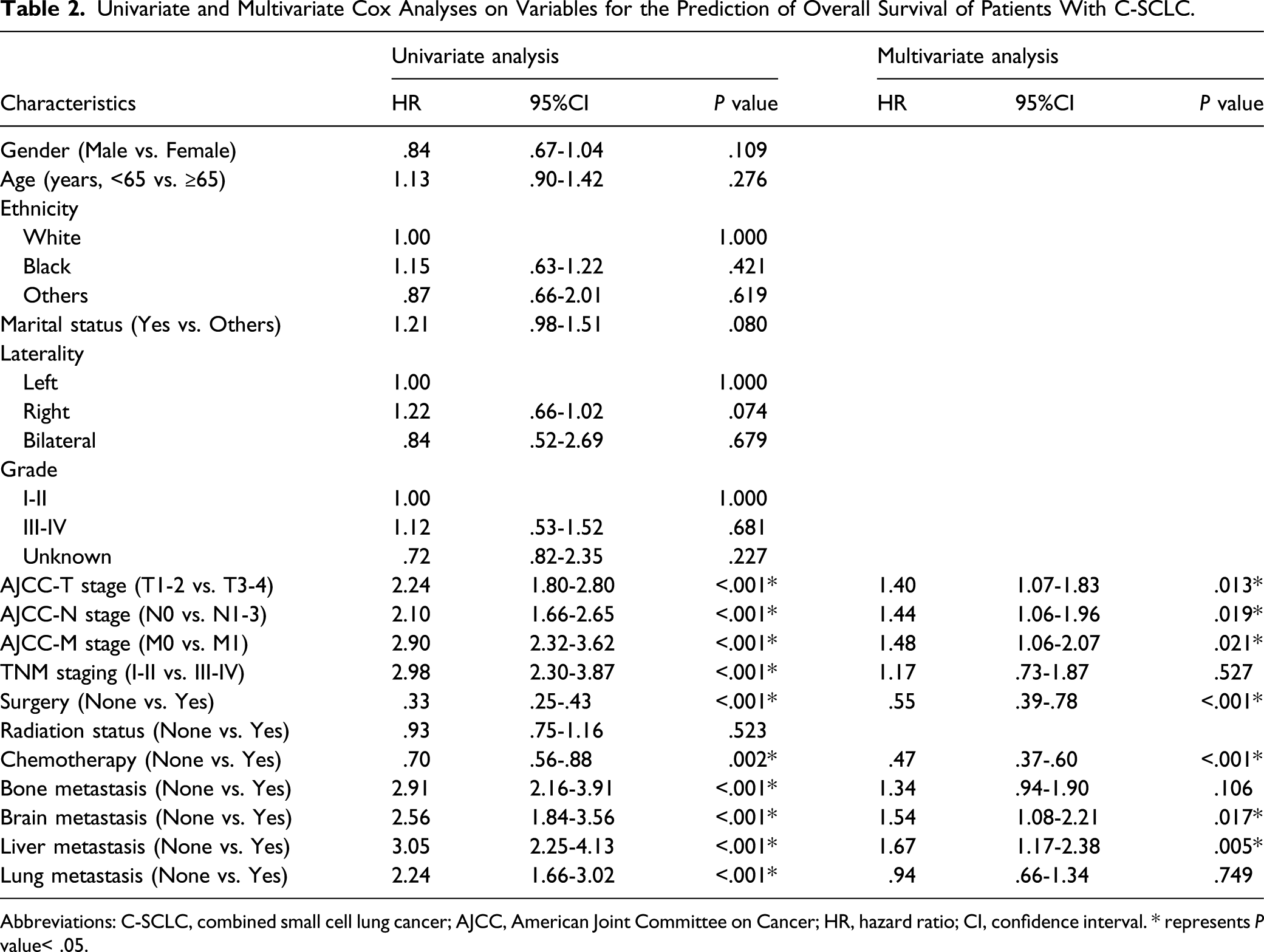
Abbreviations: C-SCLC, combined small cell lung cancer; AJCC, American Joint Committee on Cancer; HR, hazard ratio; CI, confidence interval. * represents P value< .05.
Nomogram Development and Validation
We constructed a nomogram to predict the survival probability of patients with C-SCLC via R software, “rms” and “regplot” packages. Figure 2 demonstrates an example of using the nomogram to predict the survival probability of a given patient. In this nomogram, the independent predictive factors identified through the multivariate analysis were employed to predict the total point of each patient, thus predicting the 6-, 12-, 18-, 24-, and 36-month survival probability of these patients (Figure 2). Besides, we calculated the C-index of this nomogram in the two cohorts to estimate its predictive power, suggesting the constructed predictive model had excellent performance in predicting the OS of C-SCLC (training cohort: .751; validation cohort: .736, respectively). Furthermore, we also generated ROC curves and calibration curves to assess the discrimination and calibration abilities of the nomogram in the two cohorts. It showed that no matter in the training cohort (Figure 3A) or validation cohort (Figure 3B), the constructed nomogram has higher area under the curve (AUC) in predicting 6- (.874 vs. .803), 12- (.824 vs. .783), 18- (.795 vs. .800), 24- (.800 vs. .808), and 36- (.795 vs. .807) month survival probability of patients with C-SCLC. Meanwhile, the calibration curves revealed high consistencies between the actual and predicted OS in the two cohorts (Figures 3C and D). To sum up, the above results elucidated that this nomogram has an excellent predictive ability for the survival probability of patients with C-SCLC. The constructed nomogram for predicting 6-,12-,18-,24-, and 36-month OS of patients with C-SCLC. The patient was a 67 years old married male diagnosed as C-SCLC with T2bN0M1b stage. He underwent chemotherapy and radiotherapy and did not receive surgery. This patient also combined brain metastasis. From the nomogram, we can easily calculate that his total point was 398, which belongs to the high-risk group. Besides, we also can calculate that the 6-,12-,18-,24-, and 36-month death probability for this patient were 31.7%, 56.5%, 71.2%, 81.3%, and 88.7%, respectively. Assessment of the discrimination and calibration abilities of the constructed nomogram using ROC curves and calibration curves. (A), (B) The ROC curves for predicting 6-,12-,18-,24-, and 36-month OS of C-SCLC patients in the training cohort and validation cohort based on the nomogram, (C), (D) The calibration curves for predicting 6-,12-,18-,24-, and 36-month OS of C-SCLC patients in the training cohort and validation cohort based on the nomogram. ROC, receiver operating characteristic curve; C-SCLC, combined small cell lung cancer.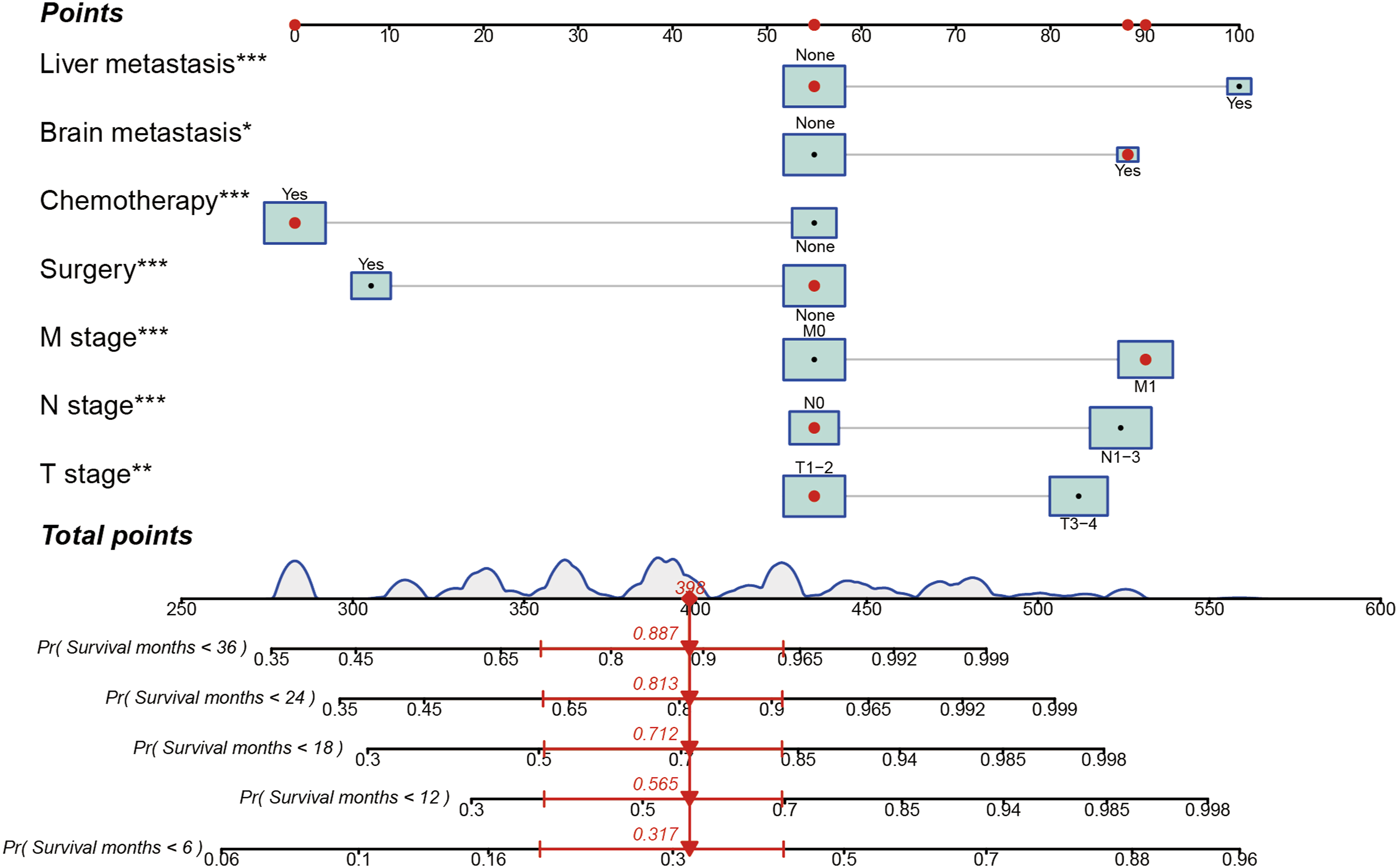

Clinical Utility Evaluation of the Nomogram
Because the ROC curve and calibration curve are based on the sensitivity and specificity of the predictive model, they cannot recognize false positive and false negative cases. Therefore, DCA was widely adopted to assess the clinical utility and net clinical benefits when the predictive model guides clinical practice. Therefore, we performed DCA to evaluate the net clinical benefits that the nomogram would bring to patients compared with the classic TNM staging system. We observed that the nomogram could predict better 6-month OS and add more clinical net benefits than the classic TNM staging system for a specific range of threshold probabilities in both the training cohort (range: .08-.83) and validation cohort (range: .12-.80) (Figures 4A and B). A similar result was also observed for the 12-month OS prediction (Figures 4C and D). Decision curve analysis of the nomogram and classic TNM staging system for predicting survival benefits of patients with C-SCLC. (A), (B) 6- and 12-month survival benefits in the training cohort, (C), (D) 6- and 12-month survival benefits in the validation cohort. C-SCLC, combined small cell lung cancer.
NRI and IDI of the Nomogram vs the TNM Staging System for Predicting OS of Patients with C-SCLC.

Abbreviations: NRI, net reclassification index; IDI, discrimination improvement; CI, confidence interval; C-SCLC, combined small cell lung cancer.
Risk Stratification Ability Assessment of the Nomogram
Ultimately, all patients were divided into low- and high-risk groups according to the median of total points in the training cohort (195) and the validation cohort (138) to evaluate the risk stratification ability of the constructed nomogram. Meanwhile, we also generated Kaplan–Meier survival curves to show the survival difference between different risk groups. We observed that the survival probability of patients in the high-risk groups was significantly lower than patients in the low-risk groups (Figures 5A and B), suggesting the constructed nomogram could accurately recognize high-risk patients. Kaplan–Meier survival analysis for evaluating the risk stratification ability of the nomogram in patients with C-SCLC. (A) Kaplan–Meier survival curve in the training cohort, (B) Kaplan–Meier survival curve in the validation cohort. C-SCLC, combined small cell lung cancer.
Discussion
C-SCLC represents a rare subtype in SCLC, with limited studies reported its clinical outcome and prognostic factors. In the present study, we explored the clinical characteristics, prognosis, and prognostic factors of these patients via a large C-SCLC dataset from the SEER database. Most importantly, we developed a nomogram based on 7 optimal prognostic variables to predict the survival probability of C-SCLC. We also performed a series of validations to evaluate its predictive ability and clinical utility. Ultimately, we found that the constructed nomogram has an excellent performance in predicting the OS of these individuals compared with the classic TNM staging system. Besides, by calculating NRI and IDI, we observed that if the nomogram were used to guide clinical practice, it would bring more incredible clinical net benefits to C-SCLC patients.
We identified that advanced AJCC-T stage, N stage, M stage, brain metastasis, and liver metastasis were correlated with unfavorable OS in C-SCLC in multivariate Cox regression analysis. Nevertheless, we found that patients can benefit from surgery and chemotherapy. Previous studies had proposed some factors that were potentially correlated with OS of C-SCLC, including smoking history, 10 extensive-stage disease, 11 lymph node metastasis,2,8 adjuvant treatment,2,10,11 and pathologically combined LCNEC 10 and SCC. 17 Lei et al. reported that lymph node metastasis was significantly correlated with decreased disease-free survival (DFS) and OS in surgically resected C-SCLC, consistent with our finding. 9 In addition, in a previously published study, Men et al. observed that positive lymph nodes ratio >10% was an independent risk factor of OS for these patients. 8 As far as we can see, no study reported the effect of distant organ metastasis on the OS of C-SCLC. In this study, we observed that liver, bone, and lung were the most predominantly distant metastatic organs. Only 10.9% of cases developed brain metastasis, which is similar to the biological behavior of P-SCLC.18,19 Furthermore, multivariate analysis revealed that brain metastasis and liver metastasis were correlated with unfavorable OS in C-SCLC. Therefore, consistent with P-SCLC, liver metastasis20-22 and brain metastasis 23 are also crucial negative prognostic factors of OS for patients with C-SCLC. The above results suggest that distant organ metastasis is not rare in C-SCLC, and detailed examination should be considered when we make a diagnosis the first time. Besides, precise and individualized management should also be given for them since this subtype of patients had limited survival time.
Adjuvant therapy is another important prognostic factor for patients with C-SCLC. Although the vast majority of studies elucidated that adjuvant therapy can provide survival benefits for these patients, they included patients with different characteristics from different research centers. Therefore, the prognostic role of some adjuvant treatments is still controversial in C-SCLC. This study found that patients who underwent surgery and chemotherapy were significantly associated with prolonged OS. Interestingly, we observed that radiotherapy did not improve the prognosis of these patients. In most retrospective studies, researchers revealed that surgery was not significantly correlated with the prognosis of C-SCLC, no matter what type of resection was adopted.2,8,10,17 On the contrary, Guo et al. indicated that receiving sublobectomy was correlated with decreased OS for patients with C-SCLC. 11 Besides, in a similar population-based study, He et al. investigated the treatment options for C-SCLC. 5 They reported that surgical treatment could improve the OS of IA-IB C-SCLC patients. 5 The possible reason for the above difference is that our study included both early and advanced-stage patients, while most of the published studies only enrolled surgically resected patients. Regarding the treatments for advanced-stage C-SCLC patients, chemotherapy with or without radiotherapy was the paramount consideration for these patients, similar to the treatment strategy for P-SCLC patients. Recently, He et al. indicated that chemotherapy-based treatment should be considered prior for advanced-stage patients, while adjuvant chemotherapy seemed to have few effects on early-stage patients. 5 On the contrary, Lei et al. revealed that postoperative adjuvant chemotherapy significantly prolonged the OS of patients with C-SCLC. 2 It can be attributed to the fact that the latter study only analyzed surgically resected patients. Therefore, large-scale and prospective studies are warranted to investigate the effect of chemotherapy on the prognosis of patients with different disease stages.
As we all know, P-SCLC is initially exceptionally responsive to cytotoxic therapy. Early-stage P-SCLC patients can achieve long-term disease control through concurrent chemoradiotherapy (CRT). 1 Numerous studies also explored the effect of postoperative radiotherapy on the OS of C-SCLC.2,8,11,17,24 In a study conducted by Men et al., they indicated that postoperative chemotherapy was not significantly correlated with improved OS of C-SCLC. 24 However, subgroup analysis revealed that postoperative chemotherapy significantly improved the survivals of patients with stage III or N2 disease. 24 No similar results were reported in other studies. Hence, it proves that C-SCLC is not very sensitive to chemotherapy and radiation compared with P-SCLC. A personalized treatment strategy should be considered for these patients. Although SCLC initially responds well to CRT, it is easy to develop brain metastasis. 25 Therefore, prophylactic cranial irradiation (PCI) is recommended as part of the standard management in most non-metastatic SCLC who respond well to initial cytotoxic treatment. 1 Wang et al. suggested that the risk of brain metastasis is relatively high in C-SCLC. 10 Besides, they also revealed that PCI could improve progression-free survival and OS of these patients and decrease the occurrence of brain metastasis in surgically resected C-SCLC patients. 10 On the contrary, in a study performed in China, Guo et al. aimed to compare the clinical characteristics and prognosis between P-SCLC and C-SCLC. 11 They indicated that PCI could only prolong OS of P-SCLC. 11 However, no statistical difference was observed when they analyzed the effect of PCI on OS of C-SCLC in multivariate analysis. 11
Finally, we identified 7 optimal variables via multivariate analysis and developed a nomogram to predict the survival probability of patients with C-SCLC. No matter in the training cohort or validation cohort, the nomogram showed excellent predictive ability for the clinical outcome of these patients. Due to the TNM staging system provides more precise lymph nodal staging and better anatomic discrimination for the measurement of outcome, it is more suitable for clinicians to acquire a piece of more accurate staging information instead of the previous Veterans Administration Lung Study Group staging system. 1 In the present study, we also compared the predictive ability of the constructed nomogram and classic TNM staging system for OS of patients with C-SCLC by conducting DCA and calculating NRI and IDI. DCA suggested that the constructed nomogram could provide more excellent clinical net benefits to patients with C-SCLC when it was adopted to clinical practice. Furthermore, the positive value of NRI and IDI also indicated that the constructed nomogram had a good predictive ability of the prognosis for these individuals compared with the classic TNM staging system. Subsequently, all patients were divided into low- and high-risk groups according to the median of total points. Besides, it also suggested that high-risk patients had shorter OS than low-risk patients through Kaplan–Meier survival analysis. Taken together, the constructed nomogram had excellent performance in predicting the survival probability of patients with C-SCLC. Besides, it will bring more significant net benefits to patients if we adopt the nomogram to support clinical practice.
To the best of our knowledge, this is the first study that constructed a novel nomogram to predict the OS of patients with C-SCLC. Although the constructed nomogram has a good performance and clinical utility, some inevitable disadvantages need to be discussed. First, although the SEER database provides a large dataset of C-SCLC, we did not perform sample size estimation in this study. Therefore, selection bias cannot be eliminated completely. Second, some crucial variables cannot be obtained from the SEER database, such as smoking history, comorbidity, detailed mixed pathological components, chemotherapy regimens, and information of PCI. According to previously published studies, SCLC combined with LCNEC, SCC, and ADC are common pathological types in these patients. Due to the lack of a large sample size study, the relationship between different types of combined components and the prognosis of patients with C-SCLC need to be further evaluated. Third, we all know that the application of immune checkpoint inhibitors (ICIs) in SCLC significantly improved the survival outcome of these patients. To our regret, there were no available records when we tried to evaluate the effect of immunotherapy on the prognosis of C-SCLC. Could this rare subtype of patients also benefit from immunotherapy? Maybe we need more relevant studies to answer this question. Last but not least, despite that we verified our results in the validation cohort and observed good performance of the nomogram, validating the predictive model in an independent external dataset is necessary in the future.
Conclusions
In summary, C-SCLC is a rare subtype in all SCLC cases. In this study, we investigated the potential predictive factors of prognosis for patients with C-SCLC. Ultimately, we constructed a novel nomogram that can accurately predict the OS of patients with C-SCLC. Given its potential clinical utility and good performance, our nomogram will provide potential survival benefits for these individuals if it is adopted to guide clinical practice. Furthermore, large-scale and prospective studies are also warranted in the future to verify our findings.
Footnotes
Abbreviations
SCLC, Small cell lung cancer; WHO, World Health Organization; P-SCLC, pure small cell lung cancer; C-SCLC, combined small cell lung cancer; NSCLC, non-small cell lung cancer; SCC, squamous cell carcinoma; ADC, adenocarcinoma; LCNEC, large-cell neuroendocrine carcinoma; SEER, Surveillance, Epidemiology and End Results; OS, overall survival; AJCC, American Joint Committee on Cancer; AIC, Akaike information criterion; C-index, Concordance index; ROC, receiver operating characteristic; DCA, decision curve analysis; NRI, net reclassification index; IDI, integrated discrimination improvement; AUC, area under the curve; CRT, concurrent chemoradiotherapy; PCI, prophylactic cranial irradiation; PFS, progression-free survival; VALSG, Veterans Administration Lung Study Group; ICIs, immune checkpoint inhibitors.
Author Contributions
Conception/design: Y. Y., Z. R., X. F., T. T., and X. L.; Provision of study material: A. J., N. L., R. Z., and S. L.; Collection and/or assembly of data: A. J., H. G., J. W., X. Z., and M. R.; Data analysis and interpretation: A. J., N. L., R. Z., and S. L.; Manuscript writing: A. J.; Final approval of manuscript: Y. Y. and Z. R. All authors read and approved the final manuscript and agree to be accountable for all aspects of the research in ensuring that the accuracy or integrity of any part of the work is appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant from the Shaanxi Provincial Science and Technology Innovation Team [grant number 2021TD-44].
Ethics approval and consent to participate
Because the data extracted from the SEER database in this study did not contain personally identifiable information, informed consent and ethical proof were not required.
