Abstract
Introduction
Thymic epithelial tumors are the most common mediastinal tumors. Despite the high survival rate after surgery, some patients still require postoperative adjuvant therapy and closer follow-up. Hematological indicators such as biochemical routines and coagulation indicators have been reported to be independently associated with the prognosis of various malignancies. Therefore, we included hematological indicators in the analysis.
Methods
The data of 105 patients with thymic epithelial tumors were retrospectively collected from Sun Yat-sen University Cancer Center, and the patients with missing preoperative hematological indicators were excluded. X-tile software was used to obtain the best cutoff value of each preoperative hematological indicator, and COX regression analysis and Kaplan-Meier survival curves were used to demonstrate statistically significant results.
Results
COX univariate regression analysis of all patients showed that Masaoka stage, T stage, WHO histologic types, D-dimer, albumin-fibrinogen ratio (AFR), Fibrinogen (Fbg) were associated with postoperative overall survival (P < .05). T stage, WHO histologic types, D-dimer, and AFR were associated with postoperative recurrence-free survival (P < .05). Finally, multivariate regression analysis showed that T stage, D-dimer levels were independently associated with postoperative overall survival (OS) and recurrence-free survival (RFS) in patients with thymic epithelial tumors.
Conclusions
For thymic epithelial tumors, higher preoperative D-dimer levels predict poorer survival and shorter recurrence-free survival. This may help guide postoperative adjuvant therapy and follow-up patterns in patients with thymic epithelial tumors.
Introduction
Thymic epithelial tumors are mediastinal tumors that originate from thymic epithelial cells 1 ; they can be divided into thymomas (including the main subtypes A, AB, B1, B2, and B3) and thymic carcinomas. 2 Thymic carcinomas can be divided into more than 10 subtypes, including squamous cell carcinoma and neuroendocrine tumors. 3 Currently, surgical resection is the main treatment modality, and complete resection (R0) is one of the key prognostic factors affecting disease-free survival and overall survival.4-6 Postoperative adjuvant chemoradiotherapy is mainly used for patients with advanced thymic epithelial tumors7,8; moreover, a variety of clinical factors, such as tumor size, T stage, Masaoka stage, WHO histologic type, and tumor vascularity violation, have been found to correlate with the prognosis of thymic epithelial tumors.9-13
Routine biochemical indicators have been reported to be associated with prognosis in an increasing number of tumors, such as non-small cell lung cancer, 14 esophageal cancer, 15 and breast cancer. 16 Conventional biochemical markers, such as LDH and CRP, have been reported to be valuable prognostic biomarkers in thymic epithelial tumors17,18; however, the prognostic value of other biochemical markers related to liver function has not been demonstrated.
D-dimer and fibrinogen levels were the coagulation indicators that correlated with cancer prognosis; they also correlate with patient prognosis in non-small cell lung cancer, 19 esophageal cancer, 20 and gastrointestinal cancer. 21 However, this has not been adequately studied in thymic epithelial tumors.
Since thymoma are relatively indolent and thymic epithelial tumors have a low incidence, effective clinical prognostic biomarkers have not yet been identified. Therefore, we aimed to further determine possible prognostic indicators, such as D-dimer, albumin-to-fibrinogen ratio (AFR), alanine aminotransferase (ALT), aspartate aminotransferase (AST), ALT/AST, glutamyltransferase (GGT), and other biochemical and coagulation indicators, based on previous studies to explore biomarkers with prognostic value for more accurate individualized treatment.
Material and Methods
Ethics Statement
This study was reviewed by the ethics committee of the Sun Yat-sen University Cancer Center; ethical approval and informed consent exemption was granted (No. B2020-353-01) and we obtained the subject’s informed consent exemption, our study have de-identified all patient details. The data of this study were reviewed and registered in the Sun Yat-sen University Cancer Center database (RDDA2021002090), in compliance with the Declaration of Helsinki.
Study Population
This study retrospectively collected clinical information of patients with thymic epithelial tumors who underwent thymic tumor resection at the Sun Yat-sen University Cancer Center between May 2005 and August 2015. The patient inclusion criteria were as follows: (1) patients older than 18 years; (2) presence of histopathologically-confirmed thymic epithelial tumors, including thymoma and thymic carcinoma (TC); (3) complete relevant laboratory tests (hemostatic coagulation test, biochemical routine, etc) within 7 days before surgery; and (4) complete surgical resection (R0, no residual disease). Patients were excluded if (1) they underwent radiotherapy or chemotherapy prior to surgery or before and after surgery, or were given an unknown sequence of treatment with surgery; (2) they had more than one malignancy or a history of other malignancies; (3) follow-up was less than 5 years; (4) postoperative survival time was less than 3 months; (5) the surgical method was cryoablation; (6) they only underwent thymoma biopsy; or (7) follow-up information was incomplete. The reporting of this study conforms to STROBE guidelines. 22
Data Collection
Hematological-related indicators for patients, such as D-dimer (DD, μg/mL), AFR, alanine aminotransferase (ALT, U/L), aspartate aminotransferase (AST, U/L), AST/ALT, serum alkaline phosphatase (ALP, U/L), glutamyltransferase (GGT, U/L), blood glucose (GLU, mmol/L) (measured on an empty stomach in the morning), fibrinogen (Fbg, g/L), blood type, and basic clinical information of the patient, such as sex, age, smoking history, alcohol history (daily alcohol consumption, the specific alcohol consumption is unlimited and not described), tumor family history, tumor size (cm), T stage, Masaoka stage, WHO histologic type, underlying disease (patient with hypertension and/or diabetes), and myasthenia gravis (MG).
Follow Up
The follow-up strategy was specific for the first 2 years, and all postoperative patients were followed-up every 6-12 months, every 12 months in the third to fifth years, and then continued annually. The follow-up examination items were chest CT scan and hematological examination (blood routine, biochemical routine, tumor markers, etc). The last follow-up was conducted in August 2020. The primary endpoints of this study were OS and RFS. OS was defined as the time from randomization to death from any cause. RFS was defined as the length of time after primary treatment for a cancer that the patient survived without any signs or symptoms of that cancer.
Statistical Method
In this study, SPSS statistical software (version 25.0; IBM, Chicago, Illinois, USA) was mainly used for Kaplan-Meier survival curve analysis and COX regression analysis, and the log-rank test was used to test the survival difference between the 2 groups. The R statistical software packages “rms,” “foreign,” “survminer” were used to plot survival curve images (version 4.0.3; https://www.r-project.org/). Using X-tile software to find optimal cutoff values for all hematological continuous variables (http://www.tissuearray.org/rimmlab), all statistical tests were two-way and P values < .05 were considered statistically significant.
Result
Patient Characteristics
Basic Demographic Data, Disease Specific Characteristics (n = 105).
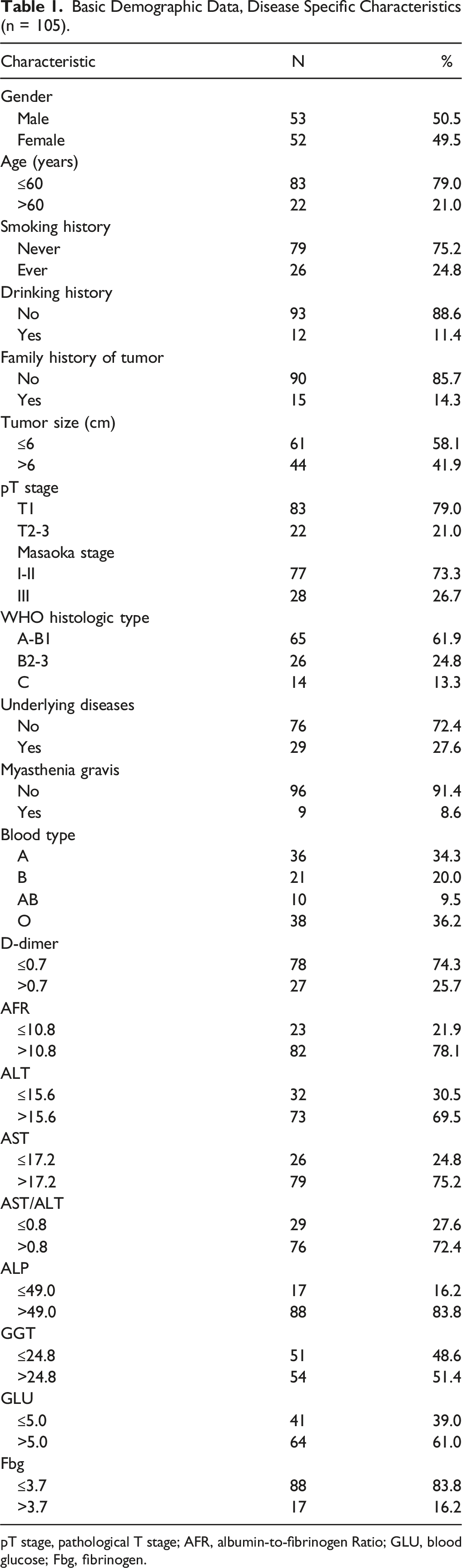
pT stage, pathological T stage; AFR, albumin-to-fibrinogen Ratio; GLU, blood glucose; Fbg, fibrinogen.
Optimal Cut-Off Values for Preoperative Hematological Parameters
To the best of our knowledge, no study has determined the optimal cut-off value of the preoperative biochemical routine and coagulation index for thymic epithelial tumors. In this study, OS was used as the endpoint, and preoperative hematological markers were determined using X-tile software. The optimal cutoff values of D-dimer, AFR, ALT, AST, AST/ALT, ALP, GGT, GLU, Fbg were .7 μg/mL, 10.8, 15.6 U/L, 17.2 U/L, .8, 49.0 U/L, 24.8 U/L, 5.0 mmol/L, 3.7 g/L.
Univariate and Multivariate Survival Analysis Based on Overall Survival
Univariate and Multivariate Analysis Results in Thymic Epithelial Tumor Based on Overall Survival (n = 105).

pT stage, pathological T stage; AFR, albumin-to-fibrinogen Ratio; GLU, blood glucose; Fbg, fibrinogen.

KM analysis of T stage (A) and D-dimer (B) based on overall survival.
Univariate and Multivariate Survival Analysis Based on Relapse-Free Survival
Univariate and Multivariate Analysis Results in Thymic Epithelial Tumor Based on Relapse-Free Survival (n = 105).

pT stage, pathological T stage; AFR, albumin-to-fibrinogen Ratio; GLU, blood glucose; Fbg, fibrinogen.

KM analysis of T stage (A) and D-dimer (B) based on relapse-free survival.
Discussion
The main endpoints of this study were OS and RFS. Statistical analysis of biochemical routines and blood coagulation indices showed that T stage and D levels were independently associated with the prognosis of patients with thymic epithelial tumors. Additionally, in univariate analysis, we found that AFR was associated with both OS and RFS in patients with tumors, which may be a prognostic marker worth investigating in the future.
For basic clinical information, a large number of studies showed T stage, WHO histologic types,23,24 and Masaoka stage6,25 to be correlated with the prognosis of thymic epithelial tumors, which is consistent with the results of the univariate analysis and Kaplan-Meier survival curve in our study. Although WHO histologic types and Masaoka staging did not show an independent prognostic effect in the final results, considering that this study’s results are limited by sample size, we believe that an increase in sample size will yield an independent prognostic value (Figures 3 and 4). KM analysis of Masaoka stage (A), WHO (B), AFR (C) and Fibrinogen (D) based on overall survival. KM analysis of Masaoka stage (A), WHO (B) and AFR (C) based on relapse-free survival.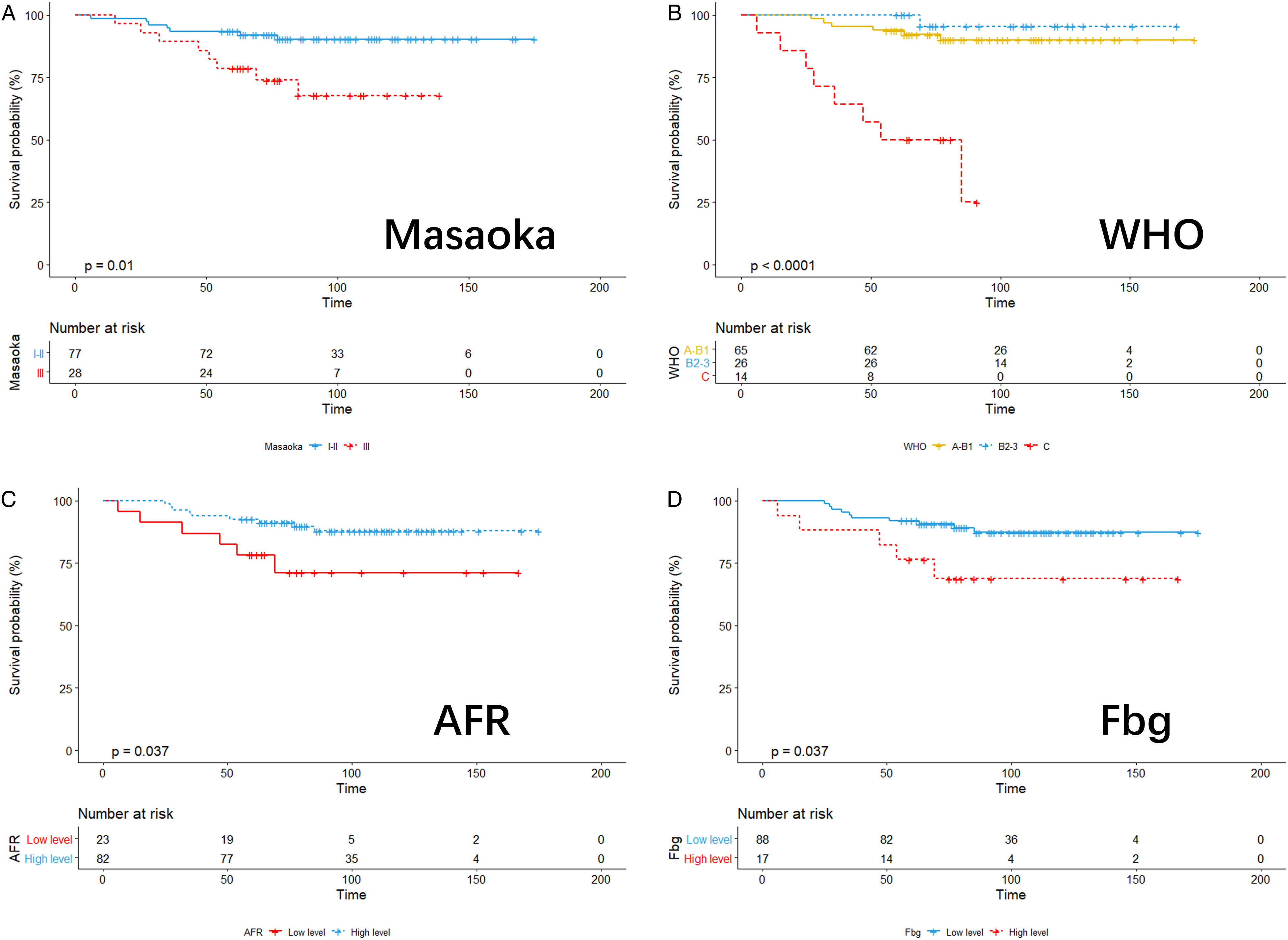

An increasing number of studies have used hematological indicators as prognostic factors in cancer patients. In previous studies on thymic epithelial tumors, Liu et al analyzed 60 postoperative patients with thymic carcinoma and found that pre-treatment serum LDH level was an independent prognostic factor affecting OS and DFS in patients with thymic carcinoma. 26 Valdivia et al also arrived at a similar conclusion. 27 Janik et al retrospectively postoperatively analyzed preoperative CRP levels in 128 patients with thymic epithelial tumors and found that elevated CRP levels were predictive of poor prognosis and tumor recurrence. 17 Although some valuable biochemical indicators have been identified, there are still a large number of biochemical-related indicators to be explored; therefore, this study aimed to discover more valuable prognostic indicators.
D-dimer is the simplest degradation product of fibrin, and its elevation indicates hypercoagulability and secondary hyperfibrinolysis in vivo. D-dimers have been found to be associated with prognosis in a variety of tumors, including non-small cell lung cancer, 28 gastrointestinal cancer, 29 pancreatic cancer 30 and so on, which may be related to the activation of coagulation function in cancer. 31 Components of the coagulation and fibrinolytic systems act on different stages of tumor growth, invasion, and metastasis by promoting the movement and invasion of tumor cells, thus supporting development of the tumor stroma, and inducing tumor angiogenesis. This ultimately leads to poor prognosis for cancer patients.32-34 The lack of specificity, which was considered a disadvantage of D-dimers in previous studies, 35 appears to translate into an advantage in prognostic assessment. However, to our knowledge, D-dimer’s association with thymic epithelial tumors has not been previously reported; we found that preoperative D-dimer levels were an independent prognostic factor for postoperative OS and RFS in patients with thymic epithelial tumors.
Albumin, produced by the liver, maintains intravascular osmotic pressure, promotes substance transport, and scavenges free radicals. It is now considered an indicator of nutritional status and has been previously shown in our group’s studies to be associated with the prognosis thymic epithelial tumor prognosis. 36 Fibrinogen is an acute-phase soluble plasma glycoprotein involved in blood coagulation, fibrinolysis, and inflammation, which is thought to play an important role in tumor metastasis, matrix formation, and angiogenesis. 37 Regarding thymic epithelial tumors, Janik et al reported that higher preoperative fibrinogen serum concentrations are associated with higher tumor stage, more aggressive tumor behavior, recurrence, and worse outcome. 38 Our results also confirmed an association between fibrinogen levels and overall survival. The prognostic value of the albumin-fibrinogen ratio has not been studied in thymic epithelial tumors; hence, this study included this index that may represent the level of nutritional and inflammatory responses in patients, and finally found it to be significantly associated with postoperative OS and RFS in tumor patients, in the univariate analysis. However, the mechanism underlying this association remains unknown, and further confirmation of our findings is needed in future with larger clinical sample sizes and experimental studies.
Glucose is required for tumor growth, likewise tumor metastasis and recurrence, which depend on energy 39 ; hence, this study included preoperative fasting blood glucose as an indicator. Biochemical routines contain a large number of relevant indicators that represent a patient’s liver and other organic functions. Although changes in these indicators have been studied in a variety of tumors as effective prognostic markers,40-43 they have not been explored in thymic epithelial tumors. In summary, other related indicators, such as ALT, AST, AST/ALT, GGT, ALP, and GLU were included in this study; unfortunately, no direct correlation was found with prognosis of thymic epithelial tumors in this study.
A growing research body recently identified novel prognostic factors of cancer. However, most studies have focused on biomarkers that require complicated molecular and genetic testing.44,45 The cost and complexity of these tests limit their practical application. In contrast, our study used laboratory test results as prognostic factors for routine clinical surveillance. Moreover, blood tests routinely used in clinical medicine do not require specialized equipment or expertise and are more reliable than most tests performed in biological laboratories.
Our study found a new valuable hematological marker for thymic epithelial tumors: D-dimer. It can predict the prognosis of patients with thymic epithelial tumors. The model is used as follows: if the patient has a lower D-dimer (≤.7 μg/mL) before surgery, it means that the patient may have a better prognosis, If the patient has a higher D-dimer (>.7 μg/mL) before surgery, it is considered that the patient may have a high risk of recurrence and a poor prognosis. It is recommended that clinicians should fully evaluate the value of postoperative adjuvant therapy to implement the best possible Individualized treatment strategies. At the same time, clinicians should fully assess the value of shortening postoperative patient follow-up periods and D-dimer monitoring at each follow-up visit.
This study had some limitations. First, this was a single-center, small-sized sample study. This study faces the possibility that the number of research samples may be insufficient. In future, multicenter, large-sample studies are needed to confirm this hypothesis. Second, this was a retrospective study; hence, prospective studies are necessary for validation of this study’s findings. Finally, the dynamic changes in postoperative biochemical and coagulation indices were not included in the research scope; thus, their values need to be further explored.
Conclusions
By retrospectively analyzing more than 10 years clinical data of patients in our center, we found that preoperative serum D-dimer levels and T stage could independently predict OS and RFS of patients with thymic epithelial tumors after R0 resection. Preoperative serum D-dimer levels may be a powerful biomarker for predicting postoperative prognosis of thymic epithelial tumors.
Footnotes
Acknowledgments
We would like to thank the Sun Yat-sen University Cancer Center for providing valuable clinical data, Thanks to our respected pioneer, Professor Tie-Hua Rong, who oriented us to surgical oncology. We thank our patients who silently provided data, Finally, we are grateful to Editage (![]() ) for its excellent polishing work.
) for its excellent polishing work.
Author Contributions
Conception, design of the work, and interpretation of data: G-WM and Y-YH. Provision of study materials or patients: G-WM and XL. Acquisition of data: Y-YH, XL, and YH. Analysis of data: S-HL, Y-YH, and YH. Drafted the manuscript: Y-YH. substantially revised the manuscript: G-WM, YH, and Y-YH. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Wu Jie-ping Medical Foundation (320.6750.2020-15-7).
Data Availability Statement
Ethics Approval and Consent to Participate
This study was approved by the Medical Ethics Committee of Sun Yat-Sen University Cancer Center (B2020-353-01) and complied with the Declaration of Helsinki. Data were recorded at the Sun Yat-sen University Cancer Center, under the record number: RDDA2021002090. At the same time, this study has obtained the exemption of informed consent application from the Ethics Committee of Sun Yat-sen University Cancer Center.
