Abstract
Objectives
Although metastatic breast cancer (MBC) is considered incurable, a specific subset of patients exhibits prolonged survival and even achieve a “cure”. We retrospectively identified predictive prognostic factors and systemic therapy models to find this group of potentially cured patients.
Methods
Consecutive patients diagnosed with MBC from 1991-2016 in West China Hospital were included. Univariate and multivariate analyses were conducted to assess the association of clinical factors and systemic therapy models with overall survival (OS), breast cancer-specific survival (BCSS) and progression-free survival (PFS).
Results
The median OS was 63.4 months. Age, tumor size, lymph node metastasis, histologic grade, molecular subtype, site and number of metastases and metastasis-free interval (MFI) were related to the prognosis of MBC (P < .05). Patients with T1, N0-1, luminal A, bone metastasis, OMBC (oligometastatic breast cancer) or metastasis-free interval (MFI) ≥ 3 years showed the median OS more than 10 years (P < .001). Independent prognostic factors that correlated with OS and BCSS were residence, lymph node metastasis, histologic grade, molecular subtype, and site of metastasis (P < .05). The group of sequential chemo-endocrine therapy (ST) in hormone receptor (HR)-positive MBC patients showed the highest overall response rate (ORR) (P < .05). However, patients who received endocrine therapy (ET) showed the best OS, BCSS and PFS in the first two-line treatment, followed by ST and chemotherapy (CT) (P < .05).
Conclusions
Our study shows the predictive prognostic factors and systemic therapy models to facilitate patients likely to achieve long-term survival.
Keywords
Introduction
Breast cancer is the most common malignancy and the leading cause of cancer death among women in the world. 1 Approximately 6%-10% of newly diagnosed BC are metastatic, whereas 20%-50% of patients with early BC will eventually develop metastatic disease.2,3
Metastatic breast cancer (MBC) is a heterogeneous disease. It is characterized by different subtypes and different metastatic behaviors, including metastatic lesions ranging from solitary to diffuse and multiple organ involvement. 4 Previous studies have suggested that MBC with some special characteristics may result in significantly longer survival. OMBC (oligometastatic breast cancer), a subset of MBC with single or few metastases limited to a single organ, is considered to be significantly less aggressive and potentially curable with aggressive treatment.5,6 The metastasis-free interval (MFI) is another special prognostic factor associated with survival after metastasis, and patients with a long MFI have a favorable prognosis. 7 However, the time cut-off for MFI to prognosis evaluation was not identical and definitive. Some studies have shown that MFI <2 years is associated with a worse prognosis for MBC patients.7,8 Other suggests that there is no significant difference for patients with MFI of 5-10 years compared to <5 years and prolonged MFI greater than 10 years could improve recurrent MBC survival. 9 Other prognostic factors identified in MBC patients include age at metastasis, tumor size or lymph nodes in the primary lesion, histologic grade, molecular subtypes and metastatic site.10,11
Furthermore, modern systemic therapy with local options also improves the prognosis of MBC patients. MBC was once considered incurable. Recently, the promising results of MBC patients undergoing “curative” surgery or radiotherapy and the long-term survival achieved with modern systemic therapies renewed the discussion on this complex topic.12-14 Some researchers suggest a cure of breast cancer as a condition where cancer cells are eradicated, permitting a normal lifespan without recurrence. Others consider that cancer should be rendered harmless (without clinically significant adverse effects) for a long time and that there is no need to destroy all tumor cells.14-16 However, no matter which definition it is, the ultimate goal in MBC management should be to prolong the duration of life while maintaining a good quality of life. Once the definition for “cure” of MBC is established, the significance and strategy of treatment need to be renewed. Physicians will no longer think that treatment is palliative, even negative; some patients with MBC will deserve and actively seek more intensive treatment to seize the chance of achieving a “cure”. Therefore, we need to further optimize the treatment strategy for MBC patients.
Hormone receptor (HR)-positive MBC has been regarded as a chronic disease with a long survival for patients. Endocrine therapy (ET) is first recommended for the treatment of HR-positive MBC because of its exact curative effect and favorable adverse effect profile. 17 Chemotherapy (CT) may be performed for the presence of visceral crisis, severe symptoms, or endocrine resistance to rapidly alleviate clinical symptoms and control tumor development. However, some experts suggest that HR-positive patients may have preferred sequential therapy (ST) and rapidly obtained complete alleviation. 18
In China, BC is expected to account for 15% of all new cancers in women. Breast cancer is the leading cause of cancer death in women younger than 45 years. 19 The studies and data of prognosis and treatment for MBC are still deficient and unclear. Thus, a patient registry analysis of medical oncologists’ real-life daily practice in Chinese MBC patients was conducted. The primary data sources in the registry were mainly from the Breast Cancer Information Management System (BCIMS). 20 It contains over 15,600 BC patients’ demographic characteristics, clinical information and treatment modes. Relying on expert follow-up teams, the lost to follow-up rate was below .4% in 2021. We tried to identify predictive prognostic factors and systemic therapy models to facilitate the selection of patients likely to achieve long-term survival.
Materials and Methods
Study Population
This was an observational, population-based cohort study. All consecutive patients diagnosed with MBC were identified in the BCIMS at West China Hospital, Sichuan University, from June 1991 to December 2016. The BCIMS was built in 2008 accompanied by a specialized follow-up team and under professional supervision and maintenance. The staff working to collect patient data were trained and conducted regularly (usually twice a month) to ensure that data recording was performed correctly and completely. Every registered patient with pathologically confirmed addresses an ID and then enters the information recording process. Patient data are recorded manually by an information follow-up team rather than automatically collected from the Hospital Information System (HIS). 20 All inpatients were instructed to return to the hospital for re-examination and therapeutic evaluation based on their condition and treatment regimen.
MBC was defined as distant recurrence or metastatic breast cancer and pathologically confirmed or assumed based on radiologic findings, while patients with locoregional recurrence were excluded from this analysis.10,21 Similarly, due to different biological characteristics, 22 bilateral primary breast cancer, male patients, and patients without follow-up data were also excluded from this study. For patients who have finished regular evaluation with the recommended examinations, a new record including checking results and effect assessment will be created using the same ID during each follow-up. Follow-up was conducted via interviews at outpatient appointments or, if necessary, via telephone or postal contact.20,23
This study was approved by the Clinical Test and Biomedical Ethics Committee at West China Hospital, Sichuan University (reference number 2012-130). Written consent forms were obtained from all participants. The reporting of this study conforms to STROBE guidelines. 24 And we have de-identified all patient details in this study.
Patient Data Collection and Analysis
We collected the following variables retrospectively: age at primary diagnosis, residence, menopause status, family history, comorbidity, histological type, tumor stage, number of lymph node metastases, histologic grade, molecular subtype, HR and human epidermal growth factor receptor-2 (HER2) status, Ki-67 level, site and number of metastases. HR (including estrogen receptor and progesterone receptor) and Ki-67 level was accessed by immunohistochemistry (IHC). HER2 status was estimated by immunohistochemistry and fluorescence in situ hybridization (FISH), if needed. Both adjuvant and palliative treatment information were also recorded.
Among women with relapsed breast cancer, MFI was defined as the time from the diagnosis of primary nonmetastatic breast cancer to the date of the first distant metastases. Except for those with de novo stage IV, patients with recurrent MBC were divided into four groups: < 3 years and ≥3 years. OMBC was considered as a subset of MBC with single or two metastases limited to a single organ in the study. Overall survival (OS) was calculated from the time of the first diagnosis of metastatic disease and date of death (any cause) or censored to the date of latest contact. And breast cancer-specific survival (BCSS) was determined from the time of the first diagnosis of metastatic disease and date of death due to breast cancer. Progression-free survival (PFS) was defined as the interval from the start of the first diagnosis of distant metastases to disease progression or death. PFS was calculated for the first three lines of therapy and was divided successively into PFS1, PFS2, and PFS3. For example, PFS1 was defined as the time from the initiation of this first-line treatment as time (months) and the date of first disease progression or death or censored to date of latest contact. We first evaluated the PFS of the total population for the first three lines of therapy. Subset analyses were performed according to age at primary diagnosis, tumor stage, number of lymph node metastases, histologic grade, molecular subtype, site and number of metastases and MFI. Tumor response was calculated for the first three-line therapy, according to the RECIST criteria, 25 if possible. The overall response rate (ORR) was the proportion of patients who achieved a CR (complete response) or PR (partial response). For MBCs, radiographic evaluations to assess the extent of disease needs to be done every 2 to 4 chemotherapy cycles or every 2 to 3 months when using endocrine therapy. Effect evaluation is implemented by a BC physician. However, it was unlikely that all patients could be evaluated according to RECIST in a real-world setting.
Statistical Analysis
We next calculated and plotted the cumulative rates of breast cancer-specific and overall mortality using a competing risk model. 26 A Cox proportional hazards model was applied to determine the independent factors of survival in multivariable analysis. P values <.05 were regarded as significant. All statistical analyses were performed with SPSS 26.0 software (SPSS Inc., Chicago, IL, USA) and STATA statistical software (version 16; STATA, College Station, TX).
Results
Patient Characteristics
Characteristics of Patients With Metastatic Breast Cancer.

aAt primary diagnosis.
bInitial primary breast tumor.
Abbreviations: HER2, human epidermal growth factor receptor 2; IDC, invasive ductal carcinoma; ILC, invasive lobular carcinoma; MFI, metastasis-free interval.
Overall and Breast Cancer-specific Survival of Metastatic Breast Cancer Patients
The median follow-up time for the whole cohort was 61.22 months (range 1-327 months), with 697 patients (44.2%) dying at the end of the follow-up period. The median OS was 63.4 months. The OS rates were 81.47% at 1 year, 52.33% at 3 years, and 29.84% at 5 years. The cumulative rates of breast cancer-specific mortality were lower among young patients (≤45 years), primary lesion with a smaller tumor, lymph negative, lower histologic grade or luminal subtype. For metastasis sites, women with only bone or lung metastasis had a lower risk of breast cancer-specific mortality than those with brain or liver metastasis. Moreover, the mortality of OMBC patients was lower than that of those with three or more metastatic sites. Compared with those MFI <3 years, women with de novo stage IV disease had a relatively lower risk of breast cancer-specific mortality (Figure 1(A)-(H)). And Patients with T1, N0-1, luminal A, bone metastasis, OMBC or MFI ≥3 years showed a very low risk of breast cancer-specific mortality. Similar patterns were noticed for OS (Figure S1(A)-(H)). Kaplan–Meier estimates of breast cancer-specific survival by (A) age, (B) histologic grade, (C) tumor staging, (D) lymph node staging, (E) molecular subtype, (F) metastatic site, (G) number of metastases and (H) metastasis-free interval (MFI).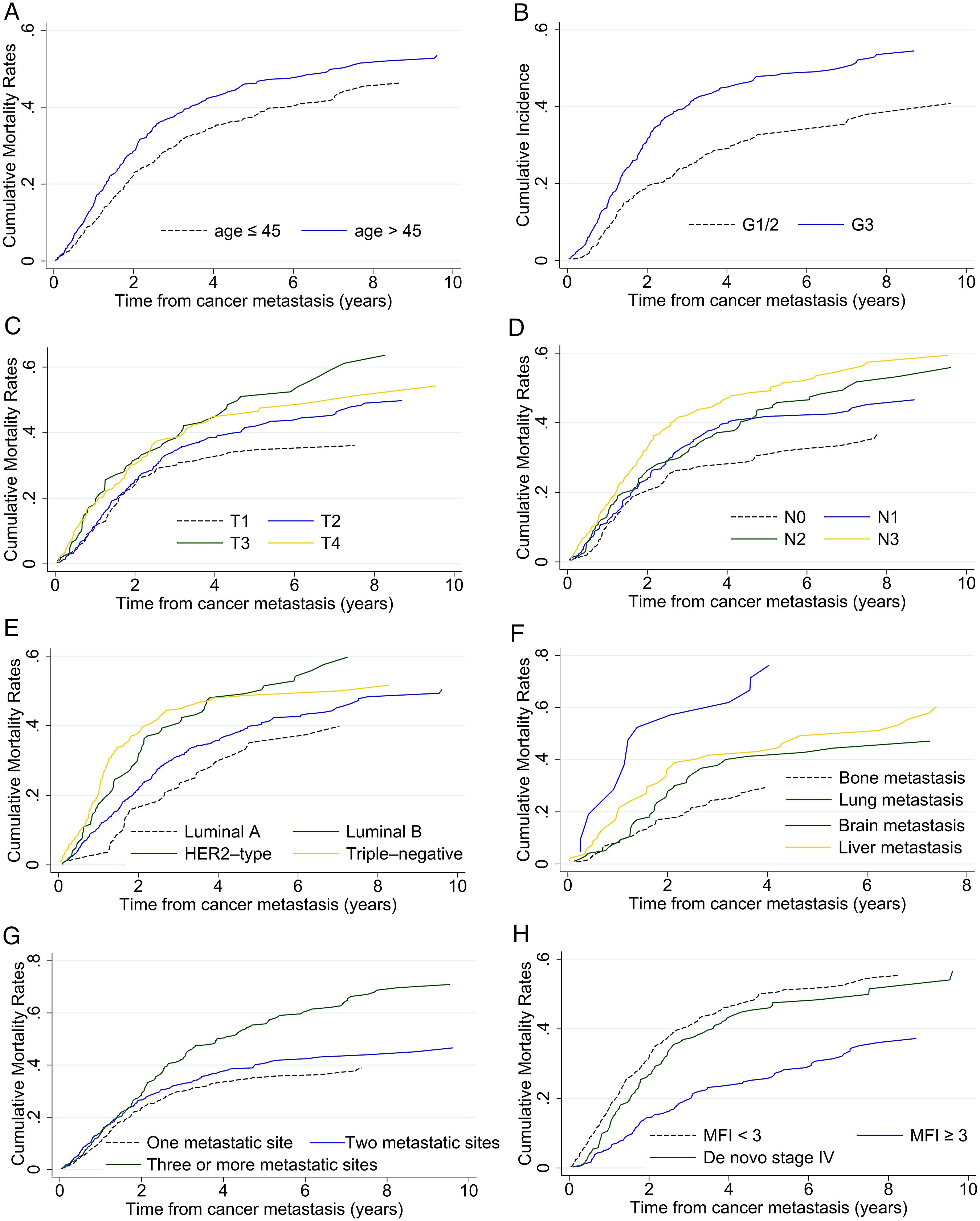
Progression-free Survival and Treatment Response in Metastatic Breast Cancer Patients
Progression-Free Survival According to the Line of Therapy in Metastatic Breast Cancer patients.
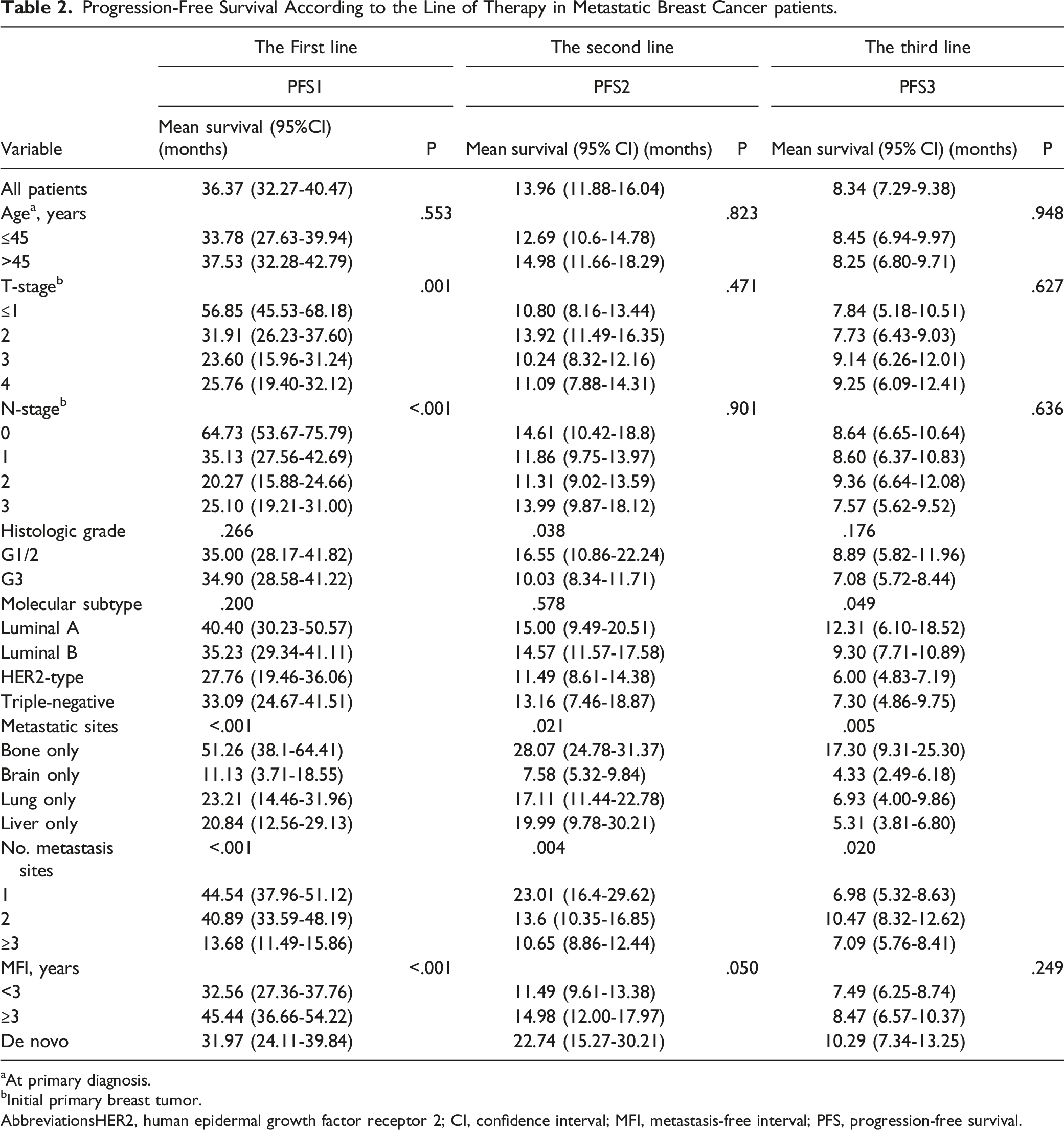
aAt primary diagnosis.
bInitial primary breast tumor.
AbbreviationsHER2, human epidermal growth factor receptor 2; CI, confidence interval; MFI, metastasis-free interval; PFS, progression-free survival.
Multivariable Analysis of Overall, Breast Cancer-specific and Progression-free Survival in Metastatic Breast Cancer Patients
Multivariable Analysis of Overall, Breast Cancer-Specific and Progression-Free Survival With Metastatic Breast Cancer Patients.
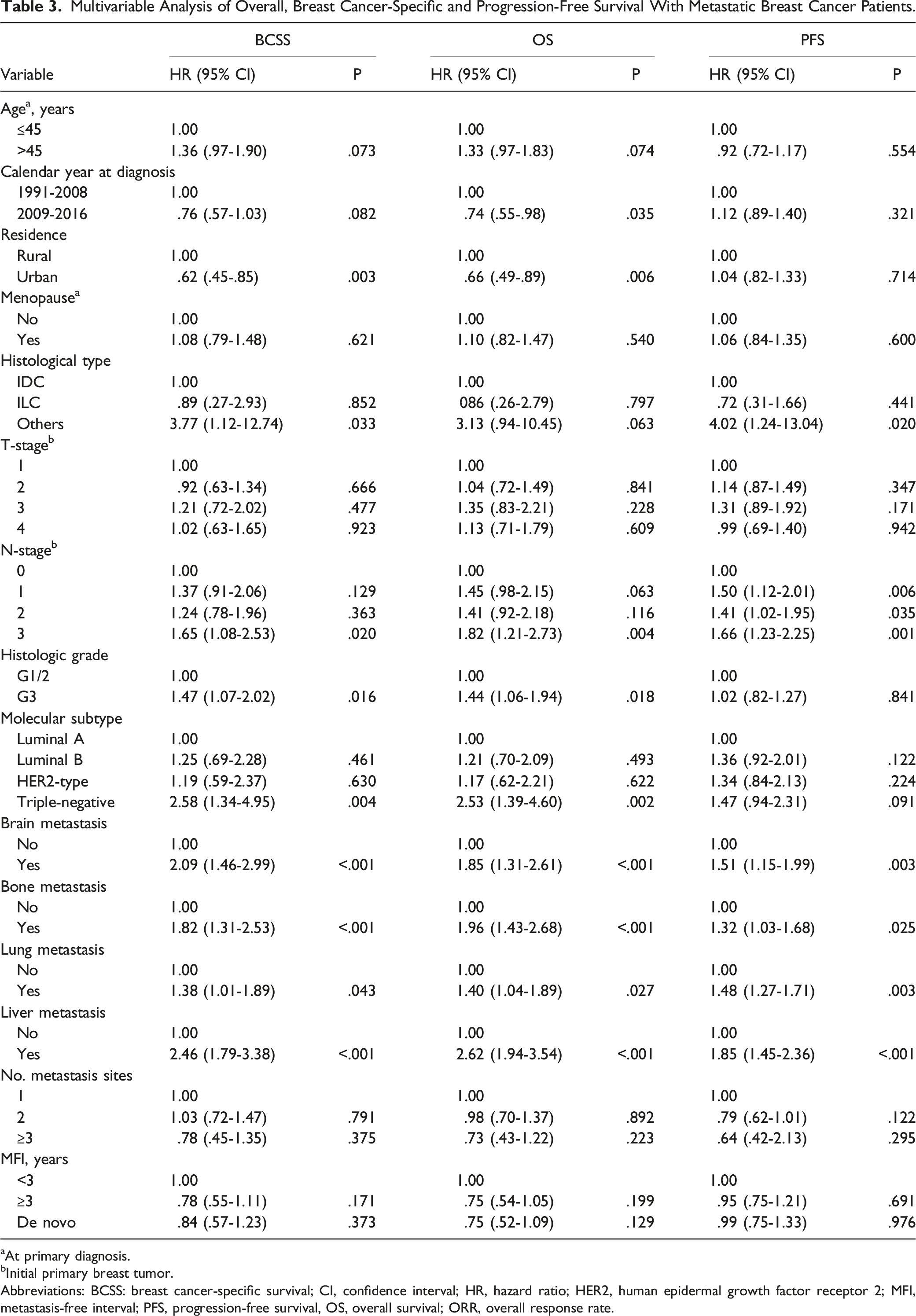
aAt primary diagnosis.
bInitial primary breast tumor.
Abbreviations: BCSS: breast cancer-specific survival; CI, confidence interval; HR, hazard ratio; HER2, human epidermal growth factor receptor 2; MFI, metastasis-free interval; PFS, progression-free survival, OS, overall survival; ORR, overall response rate.
Associations of Systemic Therapy Models With Prognosis and Treatment Response in HR-Positive Metastatic Breast Cancer Patients
Progression–Free Survival According to the Systemic Therapy Models in Hormonal Receptor-Positive Metastatic Breast Cancer Patients.

CT, chemotherapy; CI, confidence interval; ET, endocrine therapy; PFS, progression-free survival; ST, sequential chemo-endocrine therapy.
Associations of the Systemic Therapy Models With Overall, Breast Cancer-Specific or Progression-Free Survival in Hormonal Receptor–Positive Metastatic Breast Cancer Patients.

Abbreviations: BCSS, breast cancer-specific survival; CT, chemotherapy; CI, confidence interval; ET, endocrine therapy; HR, hazard ratio; OS, overall survival; PFS, progression-free survival; ST, sequential chemo–endocrine therapy.
aHRs were adjusted for age at diagnosis (≤45 years or >45 years), residence (rural or urban), T-stage (1, 2, 3 or 4), N-stage (0, 1, 2 or 3), histologic grade (G1/2 or G3), molecular subtype (luminal A, luminal B, HER2-type or triple-negative), bone metastasis (no or yes), brain metastasis (no or yes), lung metastasis (no or yes), liver metastasis (no or yes), no. metastasis sites (1, 2 or ≥3), MFI (<3, ≥3 or de novo).
Discussion
To the best of our knowledge, this is the first large single-center patient registry with long follow-up that focused on exploring the prognosis and treatment mode in consecutive unselected MBC patients from a real-world scenario in southwest China. In our study, the median OS was 63.4 months, which exceeded the survival, varying from 21.6 to 39.5 months mentioned in some European and American countries.7,10,11,27 Until then, the best prognosis for MBC was reported by the MD Anderson Cancer Center (MDACC), with the longest OS of 58 months, 28 and from Japan, with 55 months. 29 A very important fact for explanation is that the majority of the enrolled patients were from the age-old cohort studies (1974-2000 and 2000-2008, respectively). Our data were from a consecutive unselected population. The survival differences of MBC patients may be related to the follow-up time and the development and application of treatment technology.
Similar to primary breast cancer, clinical prognostic factors remain of paramount importance to guide further treatment decisions and provide outcome evaluation for MBC patients. Herein, we identified the predictive prognostic factors to distinguish and try to select potential patients who could achieve long survival. Residence, lymph node metastasis, histologic grade, molecular subtype, and site of metastasis were independent prognostic factors for OS and BCSS. As expected, most of them were confirmed as prognostic factors in MBC by other reports.7,10,30 What is encouraging is that our study showed MBC patients with smaller tumor size, negative initial lymph node metastasis, luminal A, OMBC, MFI ≥3 years or only bone metastasis, had an excellent OS, BCSS and PFS. Some studies have also suggested that the number of lymph node metastases is associated with reduced survival. In contrast, those studies did not show that patients with negative lymph node metastasis had such an amazing life period.31,32 Thus, these patients have great potential to be the next batch being cured. Aggressive application of curative treatment could be considered to reach clinical cure. 16
MFI is a known strong prognostic factor for the survival of patients with MBC. Our results also support the conclusion, but the difference was that the time cut-off was three years for our patients. In addition, patients with de novo MBC showed a relatively higher risk of breast cancer-specific mortality in comparison with those with MFI ≥3 years but better than those with recurrent MBC with MFI <3 years. However, for tumor response, patients with de novo metastatic disease showed the highest ORR in the first-line treatment. This might be because, on the one hand, de novo MBC is suggested to represent a poor prognostic group with early distant metastasis as a sign of more aggressive disease; on the other hand, the hypothesis shows that patients with de novo MBC may have a better prognosis due to its therapy-naïve status and less resistance to systemic therapy. 33 Moreover, our previous study showed that 79.4% of patients with de novo MBC underwent surgery for the primary tumor, and surgery had a significantly positive impact on OS. 34 Thus, these women could obtain a better curative effect in the early stages. Additionally, of note is that, although recurrent MBC of MFI ≥3 years had the best PFS in the first-line treatment, it showed a significant decrease in the follow-up. One of the crucial reasons may be that biomarker discrepancies were observed between primary and recurrent/metastatic breast cancer lesions and had a certain influence on treatment strategies after relapse and disease outcome.35,36
We further analyzed the PFS and tumor response according to the line of therapy in other subgroups. As expected, a major difference in PFS was seen in the subgroup analysis, including the highest survival rate for the luminal A subtype, followed by the luminal B and triple-negative subtypes. The HER2-positive subtype showed the worst outcome. However, previous studies showed that the HER2-positive subtype exhibited a higher median OS and PFS than the luminal B and triple-negative subtypes. 7 This finding may be explained by the effect of anti-HER2 therapy that is able to beneficially alter the natural history of HER2-positive disease.3,37,38 The anti-therapy in our HER2-positive patients was less than 30%, leading to a survival reduction. According to the site of metastasis, bone-only metastatic patients always maintain the highest survival, although the tumor response is inferior to that of patients with visceral metastasis. This may suggest that the effect of its intrinsic characteristics on prognosis is far greater than the effect of current treatment. More than anti-HER2 therapy or anti-visceral metastatic treatment, all these findings remind us that we can do more for our patients.
It is controversial whether ET or ST is more beneficial for MBC patients with HR positivity. In our study, some patients with HR-positive breast cancer who respond well to chemotherapy would use endocrine therapy as maintenance therapy. Compared with CT, ST was more effective in increasing objective remission, but ET remained the best effective treatment to increase short-term and long-term survival in the first two lines. However, ST could be regarded as a great decision in the follow-up medical treatment. A previous study also suggested that ST is effective in prolonging survival in MBC patients. 39 Recently, increasing evidence has shown significantly prolonged progression-free survival and a manageable toxicity profile for first-line cyclin-dependent kinase 4/6 inhibitor (CDK4/6i) plus AI in patients with HR-positive/HER2-negative advanced breast cancer.17,18 The US Food and Drug Administration (FDA) has approved CDK4/6i in combination with endocrine therapy for both first-line and second-line treatment of HR-positive/HER2-negative, advanced or metastatic breast cancer. 40 Unfortunately, few patients in our study used CDK4/6i.
Our study had several limitations. Although our study is the large-scale cohort design with virtually complete follow-up, largely limiting the common sources of bias. The rich information on demographic and clinical characteristics helped to identify the association of predictive prognostic factors and systemic therapy models with survival in MBC patients. However, the BCIMS for extracting patient information was built in 2008. Before 2008, it only retrospectively contains patient records dating back to 1989 and records limited information. And patients in this period of time could not be fully included in the study. In a real-world setting, we could not ensure that every tumor was evaluated according to RECIST and every patient was followed up punctually. Additionally, CDK4/6i plus endocrine therapy plays an important role in HR-positive MBC patients. Due to a lack of data about CDK4/6i combined endocrine therapy, we were incapable of analyzing this content. Similarly, anti-HER2 therapy in our HER2-positive patients urgently needs to be improved. We will continue to track and collect patients to obtain and analyze real-world data on such therapeutic schedules. Besides, as this cohort is based on a regional medical center, the findings may not be generalizable to the population worldwide. In conclusion, our findings support and add complete and comprehensive evidence to the critical concepts in the care of women with MBC.
Conclusion
To date, MBC is generally incurable. Our study shows the predictive prognostic factors and systemic therapy models to facilitate patients likely to achieve long-term survival. Endocrine therapy plays an important role and should be used in HR-positive MBC patients as soon as possible. In conclusion, our findings represent a large-scale real-life cohort of MBC in China and support complete and comprehensive evidence to the critical concepts in the care of women with MBC. A high medical is needed to move toward cure for a unique population of MBC patients.
Supplemental Material
Supplemental Material - Prognosis and Treatment of Metastatic Breast Cancer From A Real-World Scenario in China: A Retrospective Cohort Study
Supplemental Material for Prognosis and Treatment of Metastatic Breast Cancer From A Real-World Scenario in China: A Retrospective Cohort Study by Yuxin Xie, Ji Ma, Xueming Xia, Hong Zheng, and Qiheng Gou in Cancer Control
Footnotes
Acknowledgments
We thank all staff members working on the Breast Cancer Information Management System (BCIMS) for their contributions to data collection and management.
Authors Contributions
Yuxin Xie, Qiheng Gou, and Hong Zheng conceived the study concept and design. Yuxin Xie, Ji Ma and Xueming Xia collected data. Yuxin Xie and Qiheng Gou performed statistical analysis. Yuxin Xie drafted the manuscript. All authors significantly contributed to the critical revision of the manuscript, data interpretation, and approved the submission. The work reported in the paper has been performed by the authors, unless clearly specified in the text.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This Work Was Supported by the National Natural Science Foundation of China (Grant Number: 81902723).
Ethical Approval
This study was approved by the Clinical Test and Biomedical Ethics Committee at West China Hospital, Sichuan University (no. 2012-130). Written consent was obtained from all participants. And waiver was got for using anonymous data based on the retrospective study.
Data Availability
The data and other items supporting the results in the paper will be made available upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
