Abstract
Background
Recent observations showed that systemic immune changes are detectable in case of breast cancer (BC). In this preliminary study, we investigated routinely measured peripheral blood (PB) parameters for malignant BC cases in comparison to benign breast conditions. Complete blood count, circulating lymphoid subpopulation, and serological carcinoembryonic antigen (CEA) and cancer antigen 15-3 (CA15-3) levels were considered.
Methods
A total of 127 female patients affected by malignant (n = 77, mean age = 63 years, min = 36, max = 90) BC at diagnosis (naïve patients) or benign breast conditions (n = 50, mean age = 33 years, min = 18, max = 60) were included in this study. For each patient, complete blood count and lymphoid subpopulations (T-helper, T-cytotoxic, B-, NK-, and NKT-cells) analysis on PB samples were performed. Hormonal receptor status, Ki-67 expression, and serological CEA and CA15-3 levels were assessed in the case of patients with malignant BC via statistical analysis.
Results
Women with malignant BC disclosed increased circulating T-helper lymphocytes and CD4/CD8 ratio in PB when compared to those affected by benign breast conditions (2.345 vs 1.894, P < .05 Wilcoxon rank-sum test). In the case of malignant BC patients, additive logistic regression method was able to identify malignant BC cases with increased CA15-3 levels (CA15-3 >25 UI/mL) via the hematocrit and neutrophils/lymphocytes ratio values. Moreover, in the case of women with aggressive malignant BC featured by high levels of Ki-67 proliferation marker, an increasing number of correlations were found among blood count parameters and lymphocytes subpopulations by performing a Spearman’s correlation analysis.
Conclusions
This preliminary study confirms the ability of malignant BC to determine systemic modifications. The stratification of malignant BC cases according to the Ki-67 proliferation marker highlighted increasing detectable alterations in the periphery of women with aggressive BC. The advent of novel and more sensitive biomarkers, as well as deep immunophenotyping technologies, will provide additional insights for describing the relationship between tumor onset and peripheral alterations.
Introduction
A growing number of scientific evidence points out that solid tumors must induce systemic immunological changes in peripheral blood and distant lymphoid organs to facilitate their progression and metastasis. 1 In the case of breast cancer (BC), it is known that tumor–host interactions are emerging as critical characteristics able to influence the spreading of disease and response to therapy. The long latency period between initial treatment and eventual recurrence in some patients suggests that a tumor may influence the host systemic environment to facilitate and sustain disease progression. In this context, the immune system is determinant and recent observations of Lei Wang et al 2 showed that tumor-induced systemic immune changes are detectable in the cytokine signaling of peripheral blood immune cells of women affected by malignant BC. In this way, the onset of BC can establish and promote an immunoregulatory environment/context which can produce detectable changes in the proportion and phenotype of effector and regulatory cell populations in the tumor and periphery. 3
The systemic alterations related to malignant BC onset are reflected by the altered neutrophil-to-lymphocyte ratio (NLR). Indeed, this feature can be used as an indicator of systemic inflammation, but it is recognized as a poor prognostic factor in various cancers, including BC. 4 Furthermore, it is currently accepted that an aberrant innate and/or adaptive immune response contributes to tumorigenesis selecting aggressive clones by inducing immunosuppression as well as stimulating cancer cell proliferation and metastasis. 3 As far as breast tissue is concerned, specific immune cells are resident in the breast tissue and contribute to the homeostasis of the mammary gland. 5 However, dysregulation of this equilibrium, due to still unknown causes, may lead to a state of chronic inflammation that favors cancer outgrowth. Despite the critical role played by immune cells in the tumor microenvironment, the effects on circulating immune cells of mediators are still not sufficiently understood. In this context, we decided to perform an in-depth analysis of circulating measurable parameters to evaluate whether the altered equilibrium due to cancer inflammation can influence the main parameters measurable in peripheral blood of patients affected by malignant BC in comparison to benign breast conditions. Thereby, we focused our attention on complete blood count results and flow cytometry determined immune subpopulations. Moreover, in the case of malignant BC, cancer antigen 15-3 (CA15-3) and carcinoembryonic antigen (CEA) levels, as well as Ki-67 expression, were included in the study for additional patient stratification. Finally, we tested whether BC subtypes or grading could correlate to these parameters alone or in combination with an automatic classification approach.
Materials and Methods
Study Population
The patients included in this study were enrolled at Ospedale Evangelico Betania (Naples, Italy) from 2016 to 2018. Each participant provided written informed consent prior to study enrollment. Ethical approval was provided by the Ethics Committee of IRCCS Pascale (Naples, Italy, Protocol n. 1/16 OSS SDN) and all experimental procedures performed in this study were in accordance with the principles of the Helsinki Declaration. From each participant, a total of 3mL of peripheral blood were collected in K3-EDTA vacutainer tubes (Becton–Dickinson, Ref. 368860). A total of 127 female patients affected by malignant (n = 77) or benign (n = 50) BC at diagnosis (naïve BC patients) were included in this study. Therefore, the samples were collected from naïve patients at the diagnosis. The patients were enrolled before they were treated with adjuvant therapy.
A statistical post hoc power analysis was performed to compute power values for given sample sizes, effect size, and alpha level. By selecting one-tailed Wilcoxon–Mann–Whitney test with 2 groups (i.e., Benign vs Malignant BC), N1 = 77 (malignant BC) and N2 = 50 (benign BC) participants, a medium effect size (f = .5), and an alpha level of .05, the power resulted around .84. Thus, our sample size will be more than adequate for the main objective of this study and should also allow for any subgroup analysis. G*Power software version 3.1 was used to compute the power analysis. 6
Blood Collection and Analysis
Peripheral blood was collected by venipuncture into EDTA-coated Vacutainer™ tubes (3 mL) and BD Vacutainer® SST™ II Plus Tubes (8 mL). Successively, complete blood count with leukocyte formula was performed using the CELL-DYN Ruby instrument (Abbott Laboratories, IL, USA) and serological levels of CA15.3 and CEA were tested using the Cobas® 6000 analyzer (Roche Diagnostics S.p.A, Basel, Switzerland).
Flow Cytometry Experiments
Blood samples were processed at the IRCCS SDN Biobank within 3–4 hours from collection. For the flow cytometry experiments, a sample aliquot of 200 μL of fresh EDTA-collected whole blood was used, while the remaining part was banked for plasma purification and collection of mononuclear cells as previously described. 7 Flow cytometry experiments were performed as follows: 100 μL of whole blood were distributed into 2 tubes containing the following antibody mixes:
Tube 1: 20 μL of CYTO-STAT tetraCHROME CD45-FITC/CD4-RD1/CD8-ECD/CD3-PC5 (Ref. 6607073, Beckman Coulter, Indianapolis, IN, United States); Tube 2: 20 μL of CYTO-STAT tetraCHROME CD45-FITC/CD56-RD1/CD19-ECD/CD3-PC5 (Ref. 6607013, Beckman Coulter, Indianapolis, IN, USA).
Antibody staining was performed at room temperature in the dark for 20 minutes, successively, the stained blood volume was diluted 1:20 with VersaLyse Lysing Solution (Ref. A09777, Beckman Coulter) and incubated for an additional 15 minutes in the dark. Finally, all tubes were centrifuged at 400g for 10 minutes, resuspended in 500 µL of Dulbecco’s phosphate-buffered saline solution (DPBS, Ref. 14190144, GIBCO, Thermo Fisher Scientific) supplemented with 2% of fetal bovine serum and acquired on an FC500 (Beckman Coulter) flow cytometer equipped with 488 nm (blue) laser. The CXP software (Beckman Coulter) and KALUZA 2.1 software (Beckman Coulter) were used for data acquisition and analysis, respectively.
Immunohistochemistry
The morphological and histological analyses were performed on formalin-fixed paraffin-embedded tissues at the Ospedale Evangelico Betania (Naples, Italy). Immediately after surgical excision, the tissue sample was totally immersed in 10% neutral buffered formalin for 48 hours. After fixation, the tissues were rinsed in 70% ethanol at 4°C before dissection by the pathologist. The tissue sections (at least 3 mm thickness) were inserted in embedding cassettes and were introduced into Thermo Scientific™ Excelsior™ AS Tissue Processor (Thermo Fisher Scientific, Waltham, MA, USA), where a series of passages in ethanol at different increasing concentrations ensured dehydration and penetration with wax paraffin. Then, the dehydrated tissues were embedded into wax blocks using a Leica EG1160 Embedding Center Dispenser. Tumors formalin-fixed and paraffin-embedded specimens were sectioned using a Thermo Scientific™ HM 340E Electronic Microtome and stained with hematoxylin and eosin Thermo Scientific™ Gemini AS Automated Slide Stainer instrument to assess tissue morphology.
The subtype classification of invasive BC was performed based on the surrogate definition of St Gallen 2013 consensus meeting guidelines. 8 For immunohistochemistry assay, the Ventana BenchMark ULTRA immunostainer (Roche Diagnostics, Basel, Switzerland) was used and diaminobenzidine was selected as chromogen according to the manufacturer’s protocol (UltraView Universal DAB detection kit; Ventana, Tucson, AZ). The utilized antibodies were: Estrogen Receptor (SP1 Rabbit Monoclonal Primary Antibody, confirm clone SP1 catalog number 790-4324), HER-2/neu (4B5 Rabbit Monoclonal Primary Antibody, pathway, num clone 4B5, cat. number 790-2991), Progesterone Receptor (1E2 Rabbit Monoclonal Primary Antibody, confirm clone 1E2 cat. number 790–4296), and Ki-67 (30-9 Rabbit Monoclonal Primary Antibody, confirm clone 30-9, cat. number 790–4286). Antigens expression was evaluated by 2 expert pathologists using a direct light microscope in bright fields at 20× and 40× magnification.
Statistical Analysis
Statistical analysis was performed using R Core Team (version 3.03, Austria). Continuous variables were expressed as mean and standard deviation (SD) or median and median absolute deviation. Data distribution was tested for normality through the Shapiro–Wilk test. Unpaired Student’s t-test or Wilcoxon rank-sum test, as required, was used for comparison between 2 groups. For comparison among more than 2 groups, ANOVA or the Kruskal–Wallis test was used. Categorical variables were expressed as a percentage and were compared using the chi-square test or the Fisher’s exact test. A P-value (P) of < .05 was considered significant. Bonferroni’s correction was used for multiple hypothesis correction, if necessary. A Spearman’s correlation analysis was run to investigate whether there was an association among variables in 2 or more groups separately. A Spearman’s ρ greater than .8 and significant P-value (P < .05) was set as a threshold to identify strong agreement between variables, whereas a Spearman’s ρ between .5 and .8 was set to identify moderate agreement between variables. Statistical analyses were carried out to compare benign and malignant BC groups and to investigate the association between aggressive and non-aggressive tumors in malignant BC groups. Among malignant BC patients, the population was stratified in tumor histotype and per 2 biomarkers (i.e., CA15-3 and Ki-67) characterizing the aggressiveness of the tumor.
First, patients were stratified by tumor histotype in 4 classes: in situ, Luminal A, Luminal B like HER2(+), and Luminal B like HER2(−) cases. Basal-like and HER2 (+) tumor cases were excluded at this stage due to the very low number of patients in those 2 groups. Moreover, 4 biomarkers associated with the immune response (CD4/CD8, Ki-67, N/L, e CA15-3) were investigated in patients with vascular infiltration, stromal reaction and tumor-infiltrating lymphocytes (TILS) in order to evaluate the immune response in infiltrating tumors.
Second, among malignant BC patients, the population was stratified into 2 classes to compare aggressive vs non-aggressive tumor conditions based on CA15-3, which is used to monitor response to BC treatment and disease recurrence. The threshold value was fixed to 25 U/mL. 9 Therefore, patients presenting values of CA15-3 greater than 25 U/mL were included in the aggressive tumor class, whereas patients presenting values of CA15-3 less than 25 U/mL were included in the non-aggressive tumor class.
Lastly, the malignant BC group was stratified via an unsupervised learning algorithm (k-means clustering with k = 3) in clusters presenting low, intermediate, or high values of the nuclear protein Ki-67, which is considered a good indicator of cellular proliferation. Therefore, the commonly used cut-off was not used to define the groups based on Ki-67 levels but an unsupervised method (i.e., clustering) was employed to discriminate the malignant BC patients in 3 groups characterized by low, intermediate, and high values of the nuclear protein Ki-67.
Machine Learning Classification
Machine learning (ML) approaches were considered to develop classifiers aiming to automatically classify aggressively vs non-aggressive malignant tumor cases stratified via CA15-3, based on the patient’s characteristics. Three most commonly used ML methods were investigated: Additive Logistic Regression (LogitBoost), which is a boosting algorithm as an approximation to additive modeling on the logistic scale using maximum Bernoulli likelihood as a criterion 10 ; Quadratic Discriminant Analysis (QDA) is a classifier with a quadratic decision boundary, generated by fitting class conditional densities to the data and using Bayes’ rule 11 ; Naïve Bayesian classifier (NB), a family of simple “probabilistic classifiers” based on applying Bayes’ theorem with strong (naïve) independence assumptions between the features. 12 The 3-fold person-independent cross-validation approach was used to validate the models. Due to the unbalanced nature of the dataset, SMOTE technique was used to attenuate the bias towards the classification in the majority class. 13 Binary classification performance measures were adopted according to standard formulae. 14 Due to the low number of patients included in the study, a combination of maximum 3 statistically significant and non-correlated features 15 were considered to train and validate the models to reduce overfitting problems and simplify the clinical interpretation of the results, by pointing the attention only on the most informative features. The best set of features was chosen for each model during training and validation as the one achieving the best performance.
Among the 3 different ML methods used to train and validate the classifiers (LogitBoost, QDA, NB), the best-performing model was chosen as the classifier achieving the highest area under the curve (AUC), which is a reliable estimator of both sensitivity and specificity rates.
Results
Study Population Description
Study Population.

ER, estrogen; PgR, progesterone; SD, standard deviation; Tis, tumor in situ; TILS, tumor-infiltrating lymphocytes.
aThose parameters were determined for malignant cases only.
Complete Blood Count Analysis
Blood Count Comparison Between Benign and Malignant Breast Neoplasia.
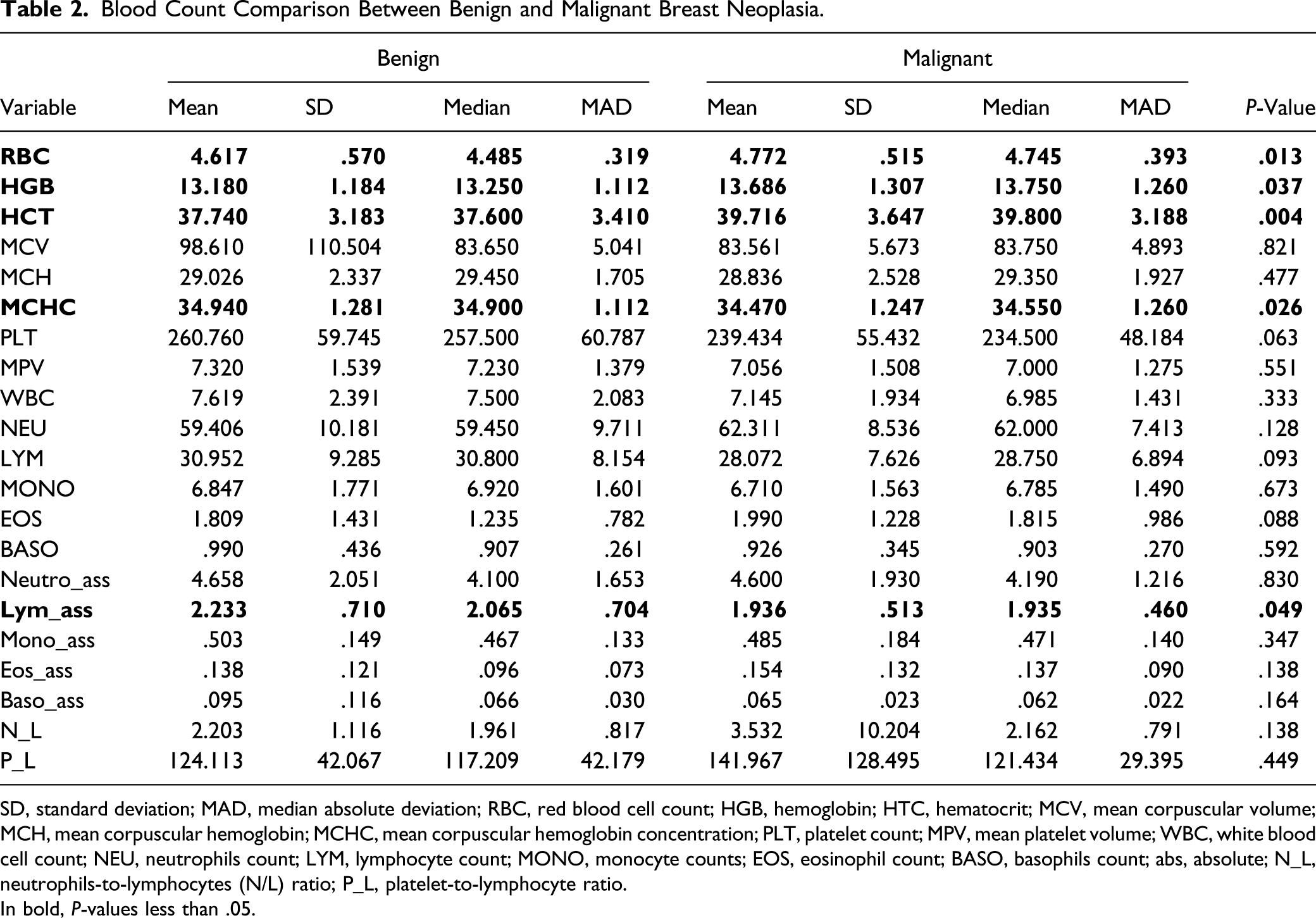
SD, standard deviation; MAD, median absolute deviation; RBC, red blood cell count; HGB, hemoglobin; HTC, hematocrit; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; PLT, platelet count; MPV, mean platelet volume; WBC, white blood cell count; NEU, neutrophils count; LYM, lymphocyte count; MONO, monocyte counts; EOS, eosinophil count; BASO, basophils count; abs, absolute; N_L, neutrophils-to-lymphocytes (N/L) ratio; P_L, platelet-to-lymphocyte ratio.
In bold, P-values less than .05.

Whole blood and lymphoid subpopulations analysis. Boxplots and P-values between malignant (M) and benign (B) breast tumor. The Wilcoxon rank-sum test was used for comparison between the 2 groups. A P-value (P) <.05 was considered significant.
Finally, regarding the white blood count data, no significant differences were detectable except in the case of the absolute number of lymphocytes that slightly increased (P = .05, Wilcoxon rank-sum test) in the benign group when compared to the malignant one (Figure 1, panel E). However, we performed an in-depth analysis of lymphoid subpopulations in order to better explore the lymphoid compartment.
Lymphocyte Subsets Analysis
The study of circulating lymphoid cells was performed using a 4-color FCM analysis panel as described in Figure 2. In particular, lymphocytes were initially selected in a CD45 vs SSC dot-plot (Figure 2, panel A). Then, T-cells were gated as CD45brightSSClowCD3pos (Figure 2, panel B) and subdivided into helper and cytotoxic T-cells as CD45brightSSClowCD3pos CD4pos and CD45brightSSClow CD3pos CD8pos events, respectively (Figure 2, panel C). NKT-cells were also included in the gating strategy and defined as CD45brightSSClow CDpos CD56pos (Figure 2, panel D). Finally, NK cells and B cells were defined as CD45brightSSClowCD56pos and CD45brightSSClowCD19pos (Figure 2, panel E), respectively. The results regarding the FCM lymphoid subpopulations studies are presented in Table 3. Interestingly, we found a significant (P < .05, Wilcoxon rank-sum test) increase of the circulating helper T-cells among malignant and benign BC cases. This data was associated with a reduced percentage of the cytotoxic T-cells and a consequent increase in the CD4/CD8 ratio when comparing the 2 groups (benign vs malignant BC). Peripheral blood lymphocytes selection by flow cytometry. Panel A displays the white blood cells distribution according to the CD45 vs SSC density plot, elliptic gate selects total lymphocytes, and CD45brightSSClow events. Panel B displays T-cells selection as CD45brightSSClowCD3pos population. Panel C discloses the percentage of Helper T-cells as CD45brightSSClowCD3posCD4pos and Cytotoxic T-cells as CD45brightSSClowCD3posCD8pos subpopulations. Panel D displays the identification of NKT-cells defined as CD45brightSSClowCD3posCD56pos events. Panel E shows NK cell and B cells identification in terms of CD45brightSSClowCD56pos and CD45brightSSClowCD19pos subgroups, respectively. Lymphocyte Subpopulations Identified by Flow Cytometry. SD, standard deviation; MAD, median absolute deviation; NK, natural killer. In bold, P-values less than .05.

PB Parameters in Relationship With Histotype Stratification for Malignant BC Cases
To evaluate whether peripheral blood (PB) parameters were influenced by histotype stratification in the case of malignant BC, we performed the Kruskal–Wallis test for the BC subtypes classification. Among malignant breast cases (n = 77), 4 patients presenting Basal-Like and HER2+ tumor type were excluded from the following analysis due to the small sample size. The remaining patients (n = 73) were stratified in 4 classes: in situ (n = 11), Luminal A (n = 25), LUMINAL B like HER2-Negative (n = 24), and LUMINAL B like HER2-Positive (n = 13). As shown in Supplementary Figure 1, the percentage of neutrophils (panel A) was significantly higher in Luminal B HER2-negative BC subtypes when compared to the other listed subtypes. Consequently, an opposite result was found when considering the percentage of lymphocytes (Supplementary Figure 1, panel B) that was significantly (P < .05, Kruskal–Wallis test) lower in the case of Luminal B HER2 like than in other BC subtypes. Interestingly, we found that in the case of Luminal B HER2-positive an increase in the percentage of Basophils (Supplementary Figure 1, panel C) was detectable in comparison to Luminal A and Luminal B HER2-negative. Furthermore, regarding the CA15-3 levels (Supplementary Figure 1, panel D), we found that this tumor marker was significantly higher in Luminal A and Luminal B cases than in situ subtypes. No significant differences were detectable when comparing BC subtypes for red blood cell parameters (RBC, HGB, MCHC, and MCV) platelet count and volume as well as lymphoid subpopulations. Only the CD4/CD8 ratio showed a significant increase in HER2-positive Luminal B subtype when compared to the HER2-negative counterpart (Supplementary Figure 1, panel E).
PB Parameters and Ki-67 Levels in Relationship to Tumor Microenvironment
To evaluate the relationship between PB derived parameters (CD4/CD8, N/L ratio, and CA15-3) as well as Ki-67 cancer proliferation index with tumor microenvironment in terms of vascular infiltration, stromal reaction, and TILS (tumor-infiltrating lymphocytes), the statistical analysis using the Kruskal–Wallis test was carried out in the malignant BC group. Regarding vascular infiltration, patients were divided in 3 categories (absent, focal and prevalent). No significant changes with respect to vascular infiltration were observed in the case of CD4/CD8 and N/L ratio, as well as CA15-3 levels. Only in the case of Ki-67 a significant increase from absent to focal (P = .002) and from absent to prevalent (P = .009) vascular infiltration was observed as shown in Supplementary Figure 2, panel A. As far as stromal reaction, patients presenting weak reaction were excluded due to low cases (N = 2). The biomarkers were, therefore, investigated among the classes: intense and prevalent. However, no statistical changes were observed for any biomarker (data not shown). Lastly, TILS were categorized in 3 classes (intense, prevalent, and weak) but the patients presenting intense tumor infiltration were excluded due to low number of cases (N = 4). CD4/CD8, CA15-3, and Ki-67 kept a constant trend among prevalent and weak classes (data not shown). Whereas N/L ratio (Supplementary Figure 2, panel B) was shown to be significantly lower in the weak class than the intense one (P = .028).
Aggressiveness Biomarkers Trends
To evaluate the relationship between the tumor and circulating markers described in this paper in the case of malignant BC, we performed a Kruskal–Wallis test as reported in Supplementary Table 2. The Ki-67 and CA15-3 showed an overall P-value less than .05, among tumor histotypes. Ki-67 showed a statistically significant decreased trend (P < .01) from in situ to Luminal A classes, whereas a statistically significant increasing trend was shown from Luminal A to Luminal B like HER2(+) (P < .001) and from Luminal A to Luminal B like HER2(−) (P < .001). No statistical differences were shown among Luminal B like HER2(+) and Luminal B like HER2(−) classes (P = .63), where the trend was kept constant. Regarding CA15-3, a statistically increased trend was shown from in situ to Luminal A (P = .012), and from in situ to Luminal B like HER2(−) (P < .01), and Luminal B like HER2(+) (P = .016). No statistically significant differences were shown among Luminal A and Luminal B like HER2(+) (P = .62) and from Luminal A and Luminal B like HER2(−) (P = .12) classes, where the trends were kept constant. CEA biomarker did not show any significant changes among tumor histotypes and it kept a constant trend among the classes (data not shown).
The Analysis Based on the Serological Levels of CA15-3
As previously discussed, the CA15-3 circulating biomarker increased in the case of invasive BC in comparison to the in situ counterpart. To better evaluate this finding, we decided to discriminate the malignant BC subpopulation into 2 groups: patients with CA15-3 levels higher than the cut-off value of 25 UI/mL (binary class: 1; n = 20), and those with CA15-3 below the cut-off (binary class: 0; n = 53). Among malignant BC patients (n = 77), 4 cases were excluded due to missing value in the evaluation of the antigen CA15-3. Among the measurable PB parameters (blood count, lymphocytes, and circulating tumor markers), HTC and N/L ratio were significantly (P < .05, Wilcoxon rank-sum test) different when comparing the 2 groups. Specifically, patients with increasing CA15-3 levels (>25 UI/mL) disclosed a lower HCT and a higher neutrophils/lymphocytes (N/L) ratio as shown in Figure 3 (panel A). Being the increasing levels of CA15-3 at diagnosis associated with advanced disease and poor outcome
16
we decided to test if HCT and N/L values were able to classify malignant BC cases in aggressive (CA15-3 >25 UI/mL) and non-aggressive classes (CA15-3 < 25 UI/mL) via ML algorithms. As reported in Figure 3 (panel B and C), the LogistBoost method outperformed the other ML methods (i.e., QDA and NB) achieving an AUC of 91%. This method showed high sensitivity (88%), specificity (85%), and overall accuracy of 87% to automatically detect aggressive malignant breast cases via HTC and N/L ratio values. Aggressive versus non-aggressive breast tumor via cancer antigen 15-3. (A) Boxplots and P-values of statistically significant variables (P-values [P] <.05, Wilcoxon rank-sum test) between aggressive (Group 1) and non-aggressive tumors (Group 0). (B) Performance Table of ML methods. (C) ROC curves for the best ML methods. LogitBoost, additive logistic regression; QDA, quadratic discriminant analysis; NB, naïve Bayesian classifier; SEN, sensitivity; SPE, specificity; ACC, accuracy; AUC, area under the ROC curve.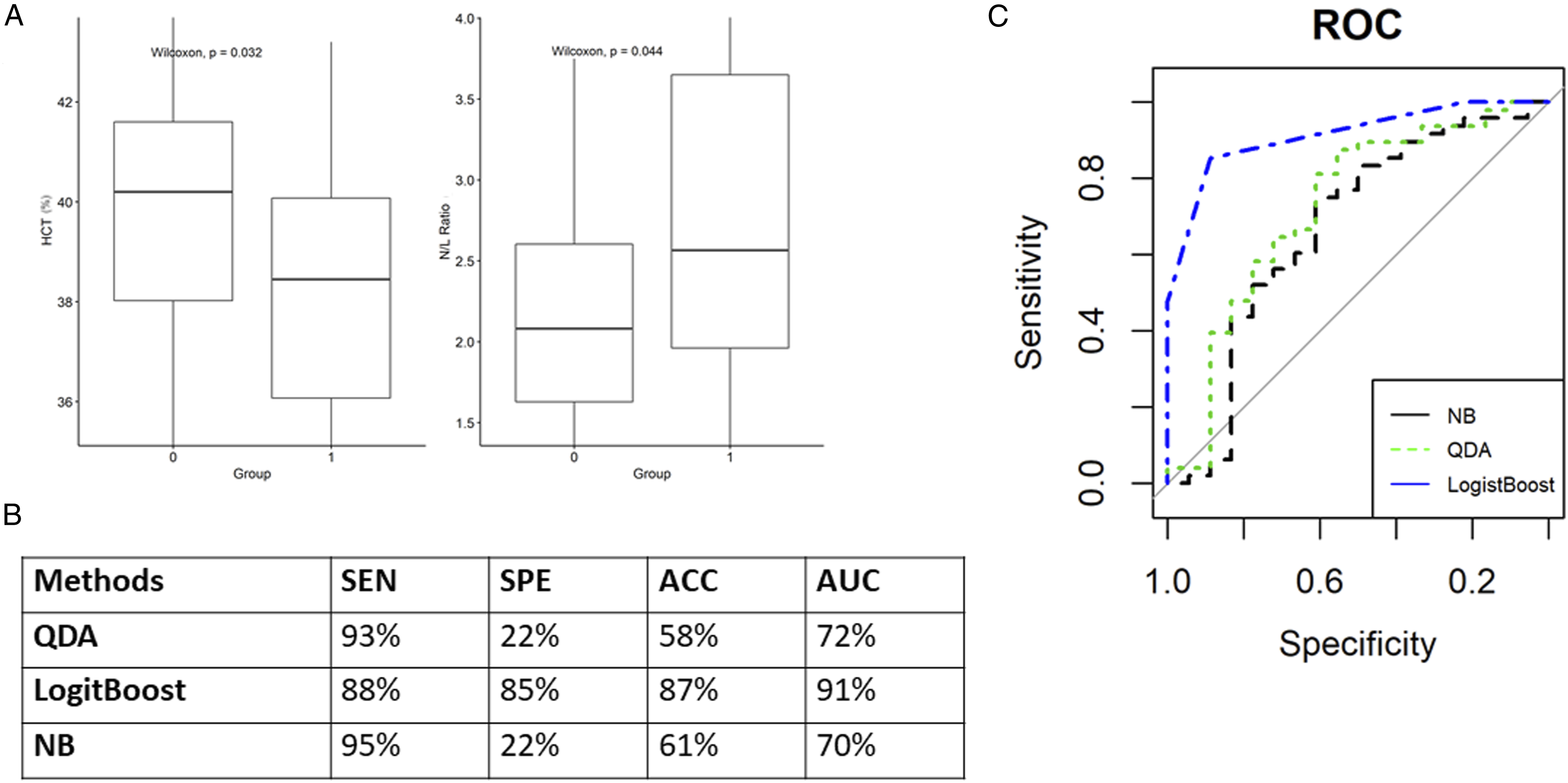
Automatically Detection of Malignant Phenotypes Associated with Ki-67 Proliferation Biomarker
The proliferation marker Ki-67 showed to be a good discriminator (P < .05) among tumor grading (G1, G2, and G3) as shown in Figure 4 (panel A), this finding is in line with previous scientific literature.17-19 Furthermore, considering that increased Ki-67 level is an important adverse prognostic factor, we decided to perform unsupervised k-means clustering to automatically separate malignant BC cases in 3 (k = 3) subgroups via Ki-67 values. Figure 4 (panel B–C) shows 3 different clusters identified as cases with low values of Ki-67 (n = 44) with a mean of 13% (SD = 4.6%), cases with intermediate values (n = 24) with a mean of 29% (SD = 6%), and cases with high values (n = 8) with a mean of 67% (SD = 7%). The unsupervised clustering segregated the malignant breast cases into 3 classes of aggressiveness based on Ki-67 values. These 3 groups were successively studied using a Spearman’s correlation analysis to evaluate how the onset of aggressive proliferating BC influences the periphery. Detection of malignant phenotypes associated with Ki-67 biomarker. (A) Boxplot and P-values (Kruskal–Wallis test) of Ki-67 among tumor grading (G1, G2, and G3). (B) Cluster Plot of Ki-67 (low, intermediate, and high clusters). (C) Boxplot and overall P-value (Kruskal–Wallis test) of the 3 Ki-67 clusters. (D) Correlation plot (Spearman’s analysis) of Ki-67 low cluster. (E) Correlation plot (Spearman’s analysis) of Ki-67 intermediate cluster. (F) Correlation plot (Spearman’s analysis) of Ki-67 high cluster.
Correlation Analysis to Identify Malignant Phenotype Characteristics via Ki-67
The Spearman’s correlation in all 3 groups separately showed an increased number of moderate to strong statistically significant correlations when moving from the Ki-67 low to the Ki-67 high cluster for most of the included parameters (Figure 4D-F). Supplementary Table 3 shows in detail the values obtained by the Spearman’s correlation analysis for each parameter in the Ki-67 defined clusters. We found that the Ki-67 high cluster displays the highest number of correlations among the included parameters. This finding highlighted that the occurrence of aggressive BC could alter the periphery at the hematological and immunological levels. The linkage between high Ki-67 expression and aggressiveness of the disease was confirmed in our patient’s group being the Ki-67-high tumors associated with low expression levels of estrogen and progesterone receptors (Supplementary Figure 3, panel A and B) as well as increasing levels of CA15-3 and neutrophils/lymphocytes ratio (Supplementary Figure 3, panel C and D). In this way, despite these preliminary findings, our study was able to confirm that the onset of aggressive disease is a pathological event not limited to breast tissue, but it can impact the entire organism.
Discussion
This preliminary work aimed to evaluate if the onset of malignant BC could impair circulating peripheral blood parameters routinely measured in diagnostics laboratories. Specifically, we focused our attention on complete blood count, lymphoid cell populations, serological CEA, and CA15-3 levels. This integrated approach is new; indeed, few works explored the BC onset influence on blood count as well as lymphoid subpopulations. Recently, Yang et al 20 explored the prognostic value of circulating lymphocytes on metastatic BC, but their observations were limited to lymphoid cells and did not include blood count parameters. The novelty of our work lies in the detection of BC by systemic measurements that could be of great aid in cancer diagnostics and surveillance of treatment response. In this initial study, the use of advanced automatic classification approaches, such as machine learning methods, helped to classify malignant BC cases in aggressive and non-aggressive classes based on the level of CA15-3 by only evaluating the HTC and N/L ratio. Moreover, we tested whether unsupervised automatic approach, such as k-means clustering, could separate malignant BC cases in 3 subgroups via Ki-67 values.
In our paper, the overall data analysis revealed that complete blood count alone was unable to discriminate between malignant or benign breast conditions. Indeed, despite the significant increase of HGB and RBC in the malignant group, we believe that this could be due to the different mean ages between the 2 groups. Specifically, most women with malignant BC are in the premenopausal period have an absence of monthly blood loss, so it is presumed that iron deficiency is prevalent in younger women with benign breast conditions and normal menstrual cycles. Then, we decided to investigate circulating white blood cells and lymphocyte subsets. In fact, recent observations by Kresovich et al 21 highlighted that shifts in circulating leukocyte profiles appear to precede a BC diagnosis. The flow cytometry analysis of lymphoid subpopulations in our patients' group revealed that the CD4/CD8 ratio (Helper T-cells /Cytotoxic T-cells) was higher in the PB of women with malignant BC than those affected by benign breast conditions. The CD4/CD8 ratio is a parameter frequently used for monitoring the immune system’s health. Generally, viral infections, tuberculosis, and corticosteroid use are some of the factors that influence the CD4 count and subsequently the CD4/CD8 ratio. In the case of BC, our preliminary observation is in line with Yang et al 22 showing that women with increased CD4/CD8 ratio (≥ 1.7) had significantly poor progression-free survival and with Aronov et al 23 proving in murine models that high circulating T-helper cells distinguished females with developed metastatic BC from the other groups. In this context, our data can be explained considering that helper T-cells influence innate immunity by modulating the character and magnitude of the inflammatory response. Indeed, they play critical roles in recruiting, activating, and regulating many facets of the adaptive immune response, with their helper functions for B cell– and CD8+ cytotoxic T cell-mediated responses. 24 In the case of the BC microenvironment, the presence of infiltrating IFN-γ–producing CD4+ Th1 and CD8+ T-cells, along with mature DCs, NK cells, M1 macrophages, and type 1 NKT-cells can generate effective and frequently attenuated antitumor responses. Conversely, CD4+ Th2 cells and type 2 NKT-cells in cooperation with CD4+ Tregs (regulatory), myeloid-derived suppressor cells, immature DCs, or M2 macrophages suppress antitumor immunity and promote tumor progression.25-27 The role of helper T-cells subsets is critical for malignant BC onset and progression. In the present paper, we were unable to describe the T-helper subsets (i.e., Th1, Th2, and T-helper); however, we believe that the local tumor-induced immune tolerance alters the distribution of PB circulating immune cells. Despite the limitation of our preliminary observation due to the low patient number and the missing in-depth immunophenotyping of helper T-cells subpopulations, we believe that the patients with an immunotolerant profile would be at risk of experiencing distant metastases of poor prognosis.
In the second part of this work, we focused our attention on women with malignant BC where CEA and CA15-3 circulating biomarkers were also taken into consideration. In the case of CEA, no significant trends among BC subtypes or presence/absence of nodal metastases were shown. These results are in agreement with the literature findings; indeed, the CEA marker is expected to rise when either cancer is not responding to treatment, or recurrence after treatment. 28 Moreover, while CA15-3 levels at diagnosis are unable to distinguish malignant from benign BC alone, we used it to define 2 patients’ groups: (i) those with elevated CA15-3 (> 25 IU/mL) and (ii) those with low (< 25 IU/mL) CA15-3 levels. This discrimination was made since elevated CA15-3 levels are frequently considered as a negative prognostic factor in the case of patients with luminal BC.29,30 Interestingly, we found that CA15-3 elevated cases were characterized by reduced HCT value and increased neutrophils/lymphocytes ratio. Different reports in the scientific literature confirmed that the high concentration of blood neutrophils is associated with advanced cancer and poor survival. Gago-Dominguez et al 31 showed that elevated NLR can be associated with increased risk of BC, principally Luminal A and HER2-negative BC. In our study, despite a small patient cohort, we found that the association of increased N/L ratio with reduced HCT value was associated with increased CA15-3 levels. The LogistBoost machine learning method showed a high sensitivity (88%), specificity (85%), and accuracy of 87% to automatically detect aggressive malignant breast cases (CA15-3 > 25) via HTC and N/L ratio values. Since several reports highlight that CA15-3 levels are associated with reduced disease-free survival, we believe that our findings can be of interest to discriminate against women at risk of aggressive cancer.
Another important consideration emerged when considering the BC stratification using the Ki-67 disease marker. The stratification of the malignant BC cases with Ki-67 antigen showed that several moderate and strong statistically significant correlations were detectable when moving from the Ki-67 low to the Ki-67 high clusters for most of the included parameters. Importantly, BC with higher Ki-67 levels disclosed increasing vascular infiltration, confirming the aggressiveness of the disease being able to induce neo-angiogenesis. This occurrence is necessary for supporting the rapid metabolic needs of rapidly growing tumors as well as allowing the generation of distant metastases. 32 Overall, these findings confirm that the occurrence of aggressive BC (featured by increasing Ki-67 and serological CA15-3 levels) could be more perturbative in the periphery at the hematological and immunological levels than in less aggressive BC cases. However, despite these initial observations, additional studies are needed. Indeed, our study was primarily focused on routine measurable medical laboratory parameters. Consequently, we have to consider some important limitations in the present study: (i) this is a preliminary report with a limited number of cases and without a control group composed of healthy subjects with a similar mean age to avoid age-related difference and (ii) no long term follow up data are available since we focused our interest on the initial diagnosis of BC with regard to disease stage and nodal metastases; surely, to better address the relationship of circulating blood parameters with the patient outcome as well as recurrence or metastases occurrence, future studies are needed. Furthermore, additional studies evaluating innovative circulating disease markers such as circulating DNA, miRNA or exosomes, and extracellular vesicles should be also considered especially regarding deep immunophenotyping studies, which are able to better evaluate significant changes in systemic immune functions.
Conclusions
The present paper aimed to evaluate changes in PB parameters in the case of malignant and benign breast conditions. According to other findings reported in the scientific literature, adaptations at immune cell levels were detectable in PB. Specifically, women with aggressive malignant BC disclosed increased CD4/CD8 ratio probably due to BC-induced immune tolerance. Additionally, in the case of malignant BC, increased N/L ratio with reduced HCT featured in the cases with increased CA15-3 levels, which were then considered at risk of reduced disease-free survival. Finally, the stratification of malignant BC cases with the Ki-67 proliferation marker highlighted the increasing changes occurring in the periphery in comparison to cases featured by low Ki-67 expression. In conclusion, this preliminary study confirms the ability of aggressive malignant BC to determine systemic immune changes. The advent of novel and more sensitive biomarkers, as well as deep immunophenotyping technologies, will provide additional insights for describing the relationship between tumor onset and peripheral alterations.
Supplemental Material
sj-pdf-1-ccx-10.1177_10732748211048612 – Supplemental Material for Impact of Breast Tumor Onset on Blood Count, Carcinoembryonic Antigen, Cancer Antigen 15-3 and Lymphoid Subpopulations Supported by Automatic Classification Approach: A Pilot Study
Supplemental Material, sj-pdf-1-ccx-10.1177_10732748211048612 for Impact of Breast Tumor Onset on Blood Count, Carcinoembryonic Antigen, Cancer Antigen 15-3 and Lymphoid Subpopulations Supported by Automatic Classification Approach: A Pilot Study by Simona Baselice, Rossana Castaldo, Rosa Giannatiempo, Giovanni Casaretta, Monica Franzese, Marco Salvatore and Peppino Mirabelli in Cancer Control
Supplemental Material
sj-xlsx-2-ccx-10.1177_10732748211048612 – Supplemental Material for Impact of Breast Tumor Onset on Blood Count, Carcinoembryonic Antigen, Cancer Antigen 15-3 and Lymphoid Subpopulations Supported by Automatic Classification Approach: A Pilot Study
Supplemental Material, sj-xlsx-2-ccx-10.1177_10732748211048612 for Impact of Breast Tumor Onset on Blood Count, Carcinoembryonic Antigen, Cancer Antigen 15-3 and Lymphoid Subpopulations Supported by Automatic Classification Approach: A Pilot Study by Simona Baselice, Rossana Castaldo, Rosa Giannatiempo, Giovanni Casaretta, Monica Franzese, Marco Salvatore and Peppino Mirabelli in Cancer Control
Supplemental Material
sj-xlsx-3-ccx-10.1177_10732748211048612 – Supplemental Material for Impact of Breast Tumor Onset on Blood Count, Carcinoembryonic Antigen, Cancer Antigen 15-3 and Lymphoid Subpopulations Supported by Automatic Classification Approach: A Pilot Study
Supplemental Material, sj-xlsx-3-ccx-10.1177_10732748211048612 for Impact of Breast Tumor Onset on Blood Count, Carcinoembryonic Antigen, Cancer Antigen 15-3 and Lymphoid Subpopulations Supported by Automatic Classification Approach: A Pilot Study by Simona Baselice, Rossana Castaldo, Rosa Giannatiempo, Giovanni Casaretta, Monica Franzese, Marco Salvatore and Peppino Mirabelli in Cancer Control
Supplemental Material
sj-xlsx-4-ccx-10.1177_10732748211048612 – Supplemental Material for Impact of Breast Tumor Onset on Blood Count, Carcinoembryonic Antigen, Cancer Antigen 15-3 and Lymphoid Subpopulations Supported by Automatic Classification Approach: A Pilot Study
Supplemental Material, sj-xlsx-4-ccx-10.1177_10732748211048612 for Impact of Breast Tumor Onset on Blood Count, Carcinoembryonic Antigen, Cancer Antigen 15-3 and Lymphoid Subpopulations Supported by Automatic Classification Approach: A Pilot Study by Simona Baselice, Rossana Castaldo, Rosa Giannatiempo, Giovanni Casaretta, Monica Franzese, Marco Salvatore and Peppino Mirabelli in Cancer Control
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by “Progetti di Ricerca Corrente” from the Italian Ministry of Health in part from 5x1000 SDN SpA for the 2017 year.
Ethical Approval
Ethical approval was granted by the Ethics Committee of IRCCS Pascale (Naples, Italy, Protocol n. 1/16 OSS SDN) and all experimental procedures performed in this study were in accordance with the principles of the Helsinki Declaration. This article does not contain any studies with animals performed by any of the authors.
Informed Consent
Written informed consent was obtained from all individual participants included in the study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
