Abstract
Introduction
Triple-negative breast cancer (TNBC) represents approximately 10-20% of all breast cancer cases and is frequently associated with BRCA1 mutations. Numerous studies from Western populations have investigated the prevalence of germline BRCA mutations in individuals with TNBC; however, the prevalence of BRCA1/2 mutations in TNBC patients varies widely between countries and from study to study. Evidence from Asian populations, particularly Vietnamese patients, remains limited. In this study, we determined the prevalence of germline BRCA1/2 mutations among unselected Vietnamese patients with TNBC and analyzed the clinicopathological features.
Methods
We conducted a single-center retrospective study of 68 women diagnosed with TNBC at the Vietnam National Cancer Hospital. Germline BRCA1/2 testing was performed by next-generation sequencing.
Results
Overall, 19 Vietnamese patients (27.9%) had BRCA1/2 mutations, with 14 (20.6%) in BRCA1 and 5 (7.4%) in BRCA2. Three patients (4.4%) had variants of uncertain significance (2 BRCA1 mutations and 1 BRCA2 mutation). Thirteen distinct pathogenic or likely pathogenic variants (8 BRCA1 and 5 BRCA2) were found. Among patients diagnosed at ≤60 years, the prevalence of BRCA1/2 mutations was 32.0%. The average age at diagnosis for BRCA1/2 mutation carriers was notably lower than that observed in non-carriers (43.1 vs 51.7 years, P = .021). BRCA1/2 mutation carriers were also more frequently premenopausal (78.6% vs 43.9%, P = .025).
Conclusions
There is a high prevalence of BRCA1/2 mutations among TNBC patients in Vietnam. Women with TNBC in Vietnam should be screened for mutations in BRCA1/2.
Introduction
Triple-negative breast cancer (TNBC) is a subtype of breast cancer (BC) defined by the lack of expression of estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2). TNBC accounts for approximately 10-20% of all breast cancer cases and is more commonly diagnosed in younger patients. This subtype is frequently associated with germline mutations in the breast cancer susceptibility genes BRCA1 and BRCA2. 1 Accumulating evidence from Western countries indicates that over 10% of TNBC patients diagnosed before age 60 carry BRCA1/2 mutations, regardless of their family history (FHx) of BC and/or ovarian cancer (OC).2,3 Patients with metastatic breast cancer who carry BRCA1/2 mutations may benefit from therapies involving poly ADP-ribose polymerase (PARP) inhibitors alone 4 or in combination with cisplatin. 5 Therefore, genetic counseling and BRCA1/2 genetic testing are essential components of managing hereditary breast cancer. Specifically, the National Comprehensive Cancer Network recommends genetic testing for BRCA1/2 in TNBC patients with an initial diagnosis age of ≤60 years, regardless of FHx. 6
BRCA1/2 genetic testing is not widely accessible in Vietnam, and services for assessing hereditary cancer risk are rarely offered. Significant barriers to the implementation of genetic counseling and testing for Vietnamese women include the high cost of testing and the absence of public insurance coverage for genetic services. Healthcare providers often have limited awareness about the benefits of genetic risk assessment, and few clinicians possess expertise in genetic counseling. Currently, BRCA1/2 testing in Vietnam is primarily available only at certain major public hospitals and select private healthcare laboratories.
Consequently, there is currently no available data regarding the incidence of BRCA1/2 mutations, the proportion of pathogenic variant carriers, or the specific mutation profiles of germline BRCA1/2 mutations among Vietnamese patients diagnosed with TNBC. This data gap further contributes to limited awareness among the public and healthcare providers about the significance of genetic testing and effective risk management strategies for individuals who carry mutations.
In this study, we used next-generation sequencing (NGS) to determine the prevalence of germline BRCA1/2 mutations among unselected Vietnamese patients with TNBC and analyzed clinicopathological characteristics according to BRCA1/2 mutation status.
Methods
Our retrospective study included consecutively selected patients with TNBC who underwent germline BRCA1/2 testing using NGS at the Vietnam National Cancer Hospital between October 2021 and December 2023, based on predefined institutional clinical criteria, rather than through random or universal testing of all TNBC cases. The reporting of this study conforms to the STROBE guidelines. 7 The inclusion criteria for this study were as follows: (1) Patients with primary BC who were diagnosed with TNBC by immunohistochemistry on biopsy or surgical specimens and had not received neoadjuvant chemotherapy; (2) Patients who had undergone germline BRCA1/2 testing using NGS; (3) Complete clinical records. The exclusion criteria were: (1) Specimens of insufficient quality for histopathological or immunohistochemical evaluation; (2) Extracted DNA that was inadequate for BRCA1/2 mutation analysis by NGS.
Data Collection
We collected the following variables: genetic data (BRCA1/2 genetic test results, age at diagnosis, FHx of BC and/or OC, number of relatives with BC and/or OC within third-degree relatives), clinicopathological data (sex, menopause status, bilateral BC, tumor size, lymph node involvement, pN and pT stages), and personal cancer history of OC. Histopathologic features, including histologic types (according to WHO criteria), histologic grade (as determined by the Nottingham histologic score), lymphovascular invasion (LVI), necrosis, and stromal tumor-infiltrating lymphocytes (sTILs), were assessed for each case by reviewing the archival hematoxylin and eosin stained slides.
ER and PR status were assessed through immunohistochemistry (IHC), with positivity defined as staining in at least 1% of tumor cells. 8 For HER2 evaluation, only the intensity and pattern of membrane staining were considered, following the guidelines outlined in the 2023 recommendations of the American Society of Clinical Oncology and the College of American Pathologists. 9 Evaluation of HER2 expression status on IHC with the HER2-low group, including IHC 1+ or IHC 2+ and negative in situ hybridization (ISH) results (IHC 2+/ISH−), was performed. Based on immunohistochemical evaluation, TNBC was identified by negative ER/PR expression and the absence of HER2 overexpression.
BRCA1/2 Mutation Analysis
Peripheral blood samples were collected from the patient, and genomic DNA was extracted from the blood using the QIAamp® DNA Blood Mini Kit (Qiagen, Hilden, Germany) following the manufacturer’s instructions. We used BRCAaccuTest ™Plus (CE-IVD, NgenneBio, Seoul, Republic of Korea) for DNA fragmentation and library preparation. The final library was normalized to a concentration of 4 nM and prepared for sequencing using the Illumina MiSeqDx platform with MiseqDx Reagent Kit v3 (300-cycles) (Illumina, San Diego, CA, USA) according to the manufacturer’s instructions to generate paired-end reads. Sequenced reads of each sample were aligned to the reference sequence hg19 using NgeneAnalySys™ (CE-IVD, NgenneBio, Seoul, Republic of Korea).
The sequencing results were compared with the BRCA1 (NC_000017.9, NM_007294.3) and BRCA2 (NC_000013.9, NM_000059.3) reference sequences for variant detection. Variant annotation and classification were conducted according to the Human Genome Variation Society (HGVS) nomenclature guidelines. 10 Variants were interpreted following the 2015 American College of Medical Genetics and Genomics/Association for Molecular Pathology standards and guidelines for sequence variant interpretation, supported by multiple curated databases including ClinVar, BRCA Exchange, NCBI, and ENIGMA. Based on these criteria, variants were classified as pathogenic, likely pathogenic, variants of uncertain significance (VUS), likely benign, or benign. 11
This study divided TNBC patients who had been tested for BRCA1/2 gene variants into two groups: BRCA1/2 mutation carriers with pathogenic and likely pathogenic variants, and non-BRCA1/2 mutation carriers (likely benign and benign variants). Clinicopathological features were tabulated and compared between groups.
Statistical Analysis
Statistical analyses were performed using IBM SPSS Statistics for Windows, Version 20.0 (IBM Corporation, Armonk, NY, USA) and R, Version 4.2.2 (R Core Team, https://www.r-project.org/). Continuous variables are presented as mean ± standard deviation (SD) or median (range), and categorical variables as n (%). Differences in continuous variables were assessed using the independent-samples t-test, while categorical variables were compared using the χ2 test or Fisher’s exact test, as appropriate. All tests were two-sided, and P < .05 was considered statistically significant.
Ethical Consideration
All methods were carried out following relevant guidelines and regulations. Our study was approved by the Hanoi Medical University Ethics Committee, number 804/GCN-HĐĐĐNCYSH-ĐHYHN, March 3, 2023. Peripheral patient blood samples were acquired with informed consent following the local institutional review and the Declaration of Helsinki. All patient details were fully de-identified to protect patient confidentiality before data processing and analysis.
Results
Baseline Clinicopathological Features and BRCA1/2 Mutations
Among 280 TNBC patients who were treated at Vietnam National Cancer Hospital between October 2021 and December 2023, 68 patients were tested for BRCA1/2 mutations by NGS. All patients were female and Vietnamese. The median age was 49.3 (range 28-89 years). Fifty-two patients (76.5%) were 60 years old or younger. Nine patients (13.2%) had a FHx of at least one relative with BC or OC within their third-degree relatives. Of these, 6 cases had a FHx of BC (8.8%); 1 case had a FHx of OC (1.5%); 2 cases had a FHx of both BC and OC (2.9%).
We observed 19 BRCA1/2 mutation carriers (27.9%) and 19 BRCA1/2 pathogenic or likely pathogenic variants in the 68 TNBC patients. Of the mutation carriers, 73.7% (14 patients) and 26.3% (5 patients) showed a mutation in BRCA1 and BRCA2, respectively. Three patients (4.4%) have a VUS (Figure 1). Schematic diagram of patient selection. TNBC, triple-negative breast cancer; NGS, next-generation sequencing; +, patients carrying pathogenic or likely pathogenic variants; −, patients carrying benign or likely benign variants; VUS, variant of uncertain significance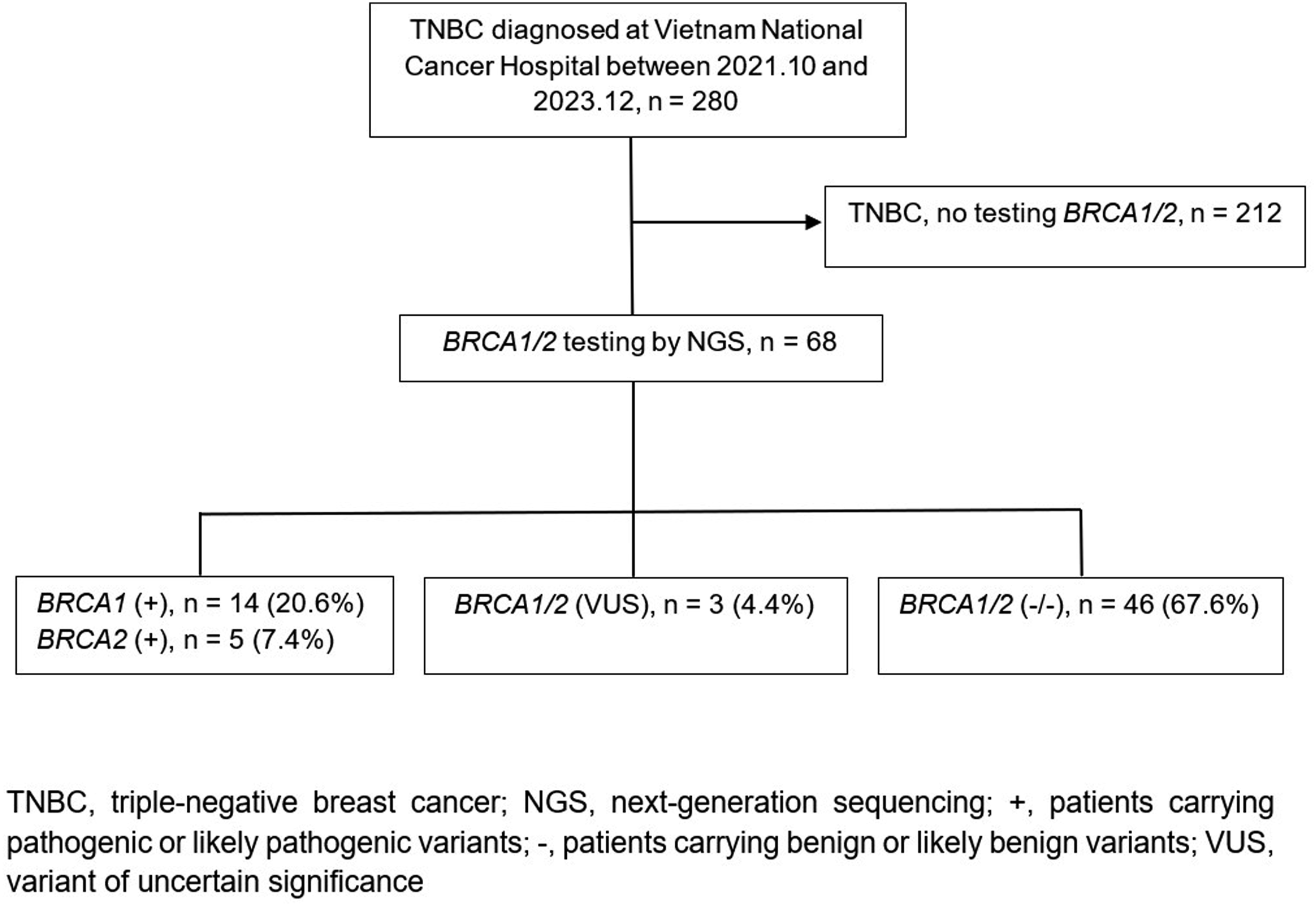
Baseline Clinicopathological Features and BRCA1/2 Mutations in Patients With TNBC (n = 65 a )

Abbreviations: SD, standard deviation; FHx, family history; BC, breast cancer; OC, ovarian cancer; LVI, lymphovascular invasion; NST, no special type; DCIS, ductal carcinoma in situ; sTILs, stromal tumor-infiltrating lymphocytes.
P < .05 for statistically significant differences are in bold.
aThree cases with variants of uncertain significance were excluded; only BRCA1/2 mutation carriers and non-carriers were compared.
bDifferences between BRCA1/2 mutation carriers and non-carriers were assessed using the independent samples t-test for continuous variables and the χ2 test or Fisher’s exact test, as appropriate, for categorical variables.
BRCA1/2 mutation carriers were more frequently premenopausal (78.6% vs 43.9%, P = .025 < .05). FHx of BC and/or OC, tumor site, LVI, histopathology, tumor size, tumor grade, lymph node status, ductal carcinoma in situ (DCIS), necrosis, sTILs, Ki67, and HER2 were not significantly different in those with or without BRCA1/2 mutation (Table 1).
Spectrum of BRCA1/2 Mutations
The Spectrum of BRCA1 Mutations (n = 14)

Abbreviations: BIC, breast cancer information core; HGVS, human genome variation society.
The Spectrum of BRCA2 Mutations (n = 5)

Abbreviations: BIC, breast cancer information core; HGVS, human genome variation society.
The Spectrum of BRCA1/2 Variant of Uncertain Significance (n = 3)

Abbreviations: BIC, breast cancer information core; HGVS, human genome variation society.
In this cohort, P and LP variants in the BRCA1 gene were most frequently identified in exon 11 and exon 20 (5 cases each, 35.7%). For the BRCA2 gene, the majority of P/LP variants were detected in exon 11 (4 cases, 80%).
BRCA1/2 Prevalence According to Age
BRCA1/2 Mutation Rates Across Age Groups (n = 65 a )

aThree cases with variants of uncertain significance were excluded.
Age at Diagnosis of Breast Cancer and Family History of Breast and/or Ovarian Cancer
The Relation Between Age and Family History

Abbreviations: SD, standard deviation; FHx, family history.
aP-value from independent samples t-test.
Discussion
In Vietnam, research on BRCA1/2 mutations in breast cancer has historically been limited by the lack of advanced molecular biology techniques. A study by Ginsburg OM et al reported a very low prevalence of BRCA1/2 mutations (0.8%) among unselected breast cancer cases; however, only 17 founder mutations previously identified in other Asian populations were tested, without comprehensive sequencing of BRCA1 and BRCA2. 12 In the past 5 years, NGS has become available and has been applied to BRCA1/2 testing in Vietnam. Nevertheless, existing studies using NGS have primarily focused on patients with hereditary breast and ovarian cancer syndrome, ovarian cancer, or isolated case reports.13–15 To the best of our knowledge, this study represents the first report on the prevalence of BRCA1/2 mutation among unselected TNBC patients in Vietnam. BRCA1/2 mutations were identified in 19 of 68 (27.9%) unselected Vietnamese patients with TNBC in our cohort. Among patients diagnosed at age ≤60 years, the prevalence of BRCA1/2 mutations was 32.0%, whereas it was 20.0% among those diagnosed at age >60 years (n = 65; 3 cases with VUS were excluded from the analysis).
Numerous studies from Western countries have examined the prevalence of BRCA1/2 mutations among unselected TNBC patients, with reported rates ranging from 9.4% to 18.2%. 16 Wong-Brown et al reported that in both Poland and Australia, approximately 10% of patients with TNBC harbored a germline BRCA1 or BRCA2 mutation. 17 However, data from Asia remains limited. In China, Zhong et al reported a prevalence of 20.3% for germline BRCA1/2 mutations (11 BRCA1 and 3 BRCA2) among 69 unselected TNBC patients. 18 Similarly, Fujisawa et al observed a prevalence of 20.0% in a cohort of 65 Japanese patients with TNBC. 19 In Korea, Jai Min Ryu et al reported a prevalence of 13.1%, identifying 97 BRCA1 and 35 BRCA2 mutations among 999 unselected TNBC patients. 20 Taken together, our findings indicate a higher prevalence compared with reports from Western populations and from several Asian cohorts. Several factors may account for the higher prevalence observed in our study. Genetic background and ethnic differences could partially explain the variation in BRCA1/2 mutation rates between populations. In addition, the relatively young age at diagnosis and the high proportion of patients diagnosed at ≤60 years (76.5% of cases) in our cohort may have contributed to the elevated prevalence. Differences in study design, sample size, and genetic testing methods may also influence the reported rates across studies.
Among patients with BRCA1/2 mutations in our cohort, the majority had BRCA1 mutations (73.7%), whereas BRCA2 mutations were less frequent (26.3%). We reaffirm the previously reported predominance of BRCA1 mutations among TNBC patients with BRCA1/2 mutations. Our findings align with those of Jai Min Ryu et al, who observed BRCA1 and BRCA2 mutation rates of 73.5% and 26.5%, respectively, in this population. 20 Similarly, our results are in concordance with the study by Gonzalez-Angulo et al, which demonstrated a higher frequency of BRCA1 mutations (12 out of 15 cases, 80.0%) compared to BRCA2 mutations (20.0%). 21
Our cohort data demonstrate that individuals with BRCA1/2 mutations are generally diagnosed with BC at a younger age than those without these mutations. This tendency is especially pronounced in BRCA1 mutation carriers, whose mean age at diagnosis was significantly lower than that of non-carriers. In contrast, the age of onset among BRCA2 mutation carriers closely resembled that of non-carriers, consistent with findings from prior studies.17,20
In our cohort, P/LP variants in the BRCA1 gene were most frequently detected in exons 11 and 20, whereas in the BRCA2 gene, the majority of variants were clustered in exon 11. Our findings are consistent with those of Ryu et al, who reported that most BRCA1 variants (51 of 97 cases, 52.6%) and BRCA2 variants (13 of 35 cases, 37.1%) were located in exon 11. 20 Exon 11 represents the largest coding region of both BRCA1 and BRCA2, encoding domains essential for DNA repair, which may explain why it is a mutational hotspot across diverse populations.22,23
A total of 5 patients (35.7% of the BRCA1 mutation group) were identified as carrying the same BRCA1 mutation. This recurrent variant (c.5251C>T, p.Arg1751Ter) has previously been reported in 3 Vietnamese patients in a cohort of 200 Asian-origin BC cases residing in the United States. 24 More recently, Hoang Anh Vu et al reported 4 Vietnamese OC patients (50% of the mutation-positive group) harboring the same BRCA1:c.5251C>T variant. 14 Whether this is a founder mutation predisposing breast and ovarian cancer in the Vietnamese cohort, this needs to be further studied with a larger patient sample size.
This study had a few limitations. First, the sample size in our study was limited because NGS testing is a newly implemented test in Vietnam, the testing cost is still high and not covered by health insurance, so not many TNBC patients can access this test. On the other hand, this study was performed by retrospective review of clinical information, and there was a possibility of incomplete information for FHx and recall bias. Despite these limitations, this study’s results are valuable because this is the first report on the prevalence of BRCA1/2 mutations among unselected TNBC in a relatively large and homogeneous Vietnamese population. In the future, predictive models for BRCA1/2 mutations should be developed that incorporate age, FHx, and additional risk factors associated with hereditary breast and/or ovarian cancer syndromes.
Conclusions
In conclusion, the prevalence of BRCA1/2 mutations among Vietnamese TNBC in our cohort was 27.9%, which is higher than that reported in studies from Asia, Europe, or North America with large cohorts. We demonstrated that the prevalence of BRCA1/2 mutations in unselected TNBC patients aged ≤60 years is 32.0%. Our findings support the recommendation of BRCA1/2 genetic testing for Vietnamese women with TNBC.
Footnotes
Acknowledgments
Genetic testing for BRCA1/2 was provided free of charge to patients under a sponsorship program supported by AstraZeneca. The sponsor had no role in study design, data collection, data analysis, interpretation, or manuscript preparation.
Ethical Considerations
The study was approved by the Hanoi Medical University Ethics Committee, number 804/GCN-HĐĐĐNCYSH-ĐHYHN, March 3, 2023.
Consent to Participate
Peripheral patient blood samples were acquired with informed consent following the local institutional review and the Declaration of Helsinki.
Author Contributions
Clinicopathological and genetic testing analysis, Tran Trung Toan and Ta Van To; Follow up data collection, Tran Trung Toan, Truong Thi Hoang Lan, Nguyen Van Thinh, and Duong Thi Ngoc Anh; Statistical analysis, Tran Trung Toan and Ta Van To; Writing-original draft, Tran Trung Toan and Ta Van To; Writing-review and editing, Tran Trung Toan, Ta Van To, and Bui Thi My Hanh; Study design, Tran Trung Toan and Ta Van To. All authors have accepted responsibility for the entire content of this manuscript and approved its submission.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data underlying this study’s findings are included in the article and can be obtained from the corresponding author upon reasonable request.
