Abstract
Breast cancer has surpassed lung cancer as the most common cause of cancer deaths, worldwide. Early breast cancers are treatment sensitive and patients under standardized treatment have prolonged. Breast cancer treatment has significantly evolved from the conventional surgical approach and radiotherapy to local and systemic adjuvant therapies. Though localized breast cancers are clinically manageable, distant recurrence is a cause of morbid concern. Adjuvant systemic therapy is effective in both distant and local recurrences and hence gained significant attention. Early breast cancer prognosis has greatly improved in the past 3 decades with reduced mortality rates due to the widespread use of adjuvant therapy. It can markedly increase the cure rate of breast cancers, and postoperative adjuvant therapy became a part of comprehensive breast cancer treatment. Further research to understand the early breast cancer characteristics could expand the treatment modalities that can improve the outcomes and survival benefits of breast cancer patients.
Keywords
Introduction
Breast cancer has now overtaken lung cancer as the most common malignancy worldwide, with high annual incidence of 11.7% and the fifth leading cause of cancer deaths worldwide in 2020. 1 Clinical presentation of breast cancer is localized tumor in 61% of patients, 32% of patients show regionally advanced tumor, and 7% show metastases. 2 Deeper understanding on clinical and pathogenic attributes of breast cancer has led to the evolution of breast cancer treatment over the years resulting in de-escalation of radical mastectomy and/or radiotherapy. Progress has been achieved by increasing implementation of multimodal treatment comprising chemotherapy, endocrine therapy, targeted therapy, and immunotherapy.3,4
Clinical Outcomes of Various Adjuvant Therapies in Early Breast Cancer Patients.
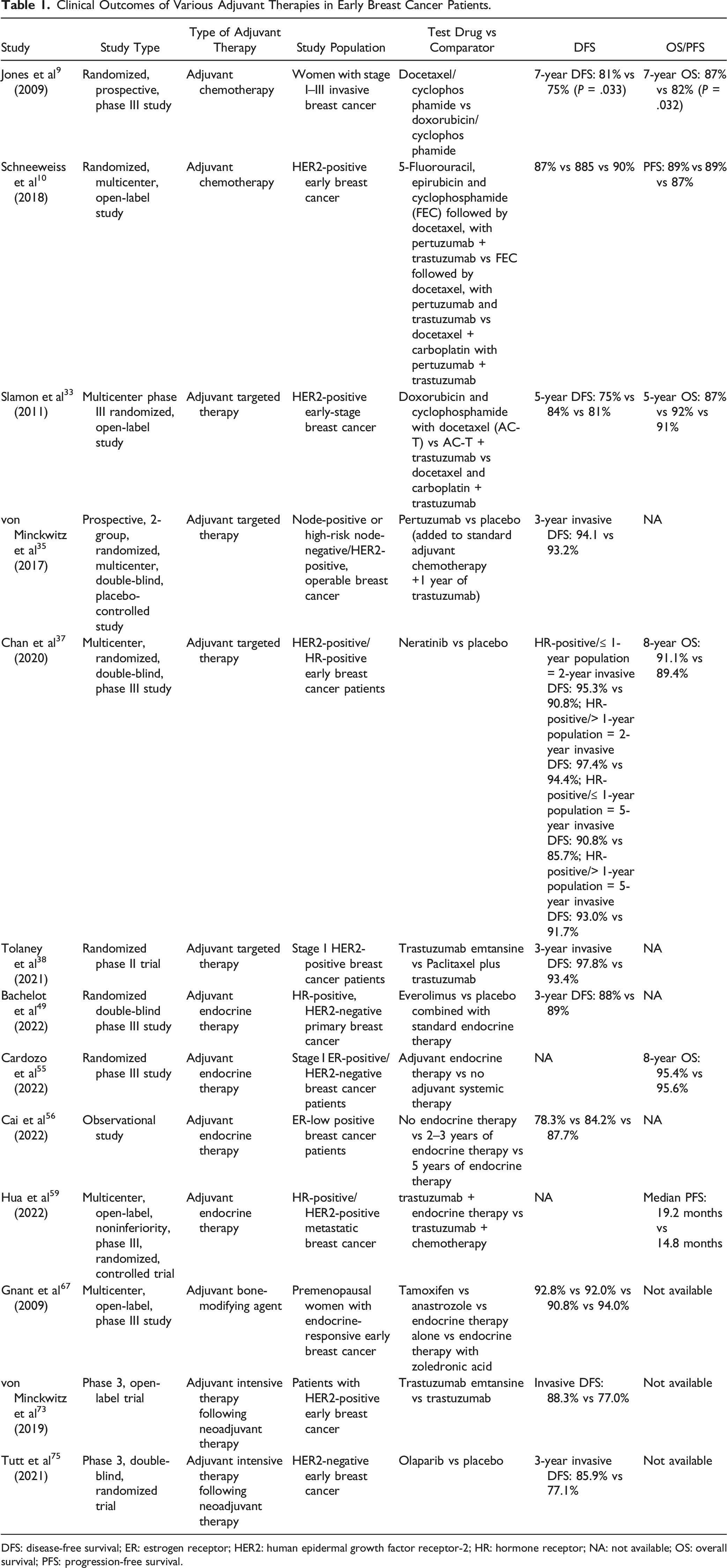
DFS: disease-free survival; ER: estrogen receptor; HER2: human epidermal growth factor receptor-2; HR: hormone receptor; NA: not available; OS: overall survival; PFS: progression-free survival.
Adjuvant Chemotherapy
Chemotherapy plays an important role in the treatment of breast cancer in all stages. Chemotherapy can prolong the survival and improve the prognosis of early breast cancer. However, adverse reactions and drug resistance caused by chemotherapy are a cause of concern and need to be resolved. 6 Anthracycline-containing adjuvant chemotherapy regimen is often recommended for women with resected node-positive or high-risk node-negative breast cancer. Anthracyclines have been shown to be effective in several large cohort studies. Improved disease-free survival (DFS) and overall survival (OS) were observed in early breast cancer patients by adding taxane to an anthracycline-based regimen in the adjuvant setting. 7 According to the National Comprehensive Cancer Network (NCCN), anthracycline-based regimens have been the cornerstone of adjuvant chemotherapy for the last 3 decades but are associated with long-term cardiotoxicity. Thus, in order to prevent cardiotoxicities, non-anthracycline-containing regimens are increasingly sought after for patients with early-stage breast cancer. 8
The US Oncology Trial 9735 showed DFS (81% vs 75%) and OS (87% vs 82%) rates as well as reduced cardiotoxicity in early breast cancer patients treated with anthracycline-free docetaxel with cyclophosphamide regimen compared to anthracycline-containing epirubicin with cyclophosphamide regimen. 9 Similarly, TRYPHAENA trial also demonstrated better outcomes with the addition of docetaxel–carboplatin-based neoadjuvant chemotherapy to dual HER2 blockade regimen. About 58% of patients showed total pathological complete response (pCR) and they were more likely to be disease free at 3 years (HR = .27; 95% CI, .11–.64) with better DFS (∼90%) and PFS rates (∼89%). 10 However, NATT trial demonstrated anthracycline-based chemotherapy to be more beneficial for patients with early breast cancer than “anthracycline-free” chemotherapy. The pCR rates were lower in patients treated with docetaxel plus cyclophosphamide (6.8%) than patients treated with docetaxel, anthracycline, and cyclophosphamide (17.6%). 11
The results of an individual patient-level meta-analysis by the Early Breast Cancer Trialists’ Collaborative Group (EBCTCG) comparing anthracycline-based vs anthracycline-free regimens confirmed that anthracycline-based chemotherapy, especially dose-dense regimens, can lead to a favorable survival benefit. It reduced the mortality rate by 57% compared to 45% by anthracycline-free regimens in early breast cancer patients. 12 Clinical management of breast cancer with anthracycline is still controversial, and accurately treating anthracycline-sensitive patients continues to remain as a challenge. Nevertheless, anthracyclines are still preferable due to shorter treatment duration, less patient visits, shorter duration of antiemetic usage, and therefore became standard of care in early breast cancer treatment. 13
Triple-Negative Breast Cancer
Chemotherapy has long been the only effective treatment strategy available for triple-negative breast cancers (TNBCs) commonly characterized by heterogeneity, aggressiveness, and poor prognosis.14,15 Even though programmed cell death 1 (PD1), programmed cell death ligand 1 (PD-L1), and poly (ADP-ribose) polymerase (PARP) inhibitor have been found to be effective in the neoadjuvant phase, chemotherapy remains the fundamental treatment modality for TNBC. 15 Adjuvant chemotherapy for early TNBC emphasizes the use of standardized chemotherapy regimens and development for reasonable management of adverse reactions. BGIRG005 trial studied AC-T (doxorubicin + cyclophosphamide followed by docetaxel) vs TAC (docetaxel + doxorubicin + cyclophosphamide) as adjuvant therapy for early triple-negative breast cancer. There was no significant difference in DFS and OS in terms of efficacy, but AC-T showed significantly higher hematological toxicity than the combination therapy. Due to low tolerability and toxicity, adjuvant chemotherapy with AC-T can be preferred in high-risk TNBC patients. 16 In SYSUCC-001 trial, patients with early triple-negative breast cancer who completed standardized adjuvant therapy were given capecitabine maintenance therapy for 1 year. After a median follow-up of 56.5 months, the 5-year DFS of capecitabine (82.8%) was significantly superior to that of the control group (73.0%). Therefore, the choice of standard therapy followed by 1 year of capecitabine therapy may reduce the risk of recurrence. 17 Meta-analysis on 9 randomized clinical trials in TNBC patients treated with combined capecitabine regimens in neoadjuvant and adjuvant chemotherapy showed significantly improved DFS (HR = .75; 95% CI; .65–.86; P < .001) and OS (HR = .63; 95% CI; .53–.77; P < .001). 15 Chemotherapy combined with immunotherapy has been implicated as neoadjuvant treatment for early breast cancer and as salvage treatment for metastatic breast cancer. 18 However, the optimal combination and management of adverse events when using combination therapies are still being explored and no studies have been published in early TNBC adjuvant settings.
Hormone Receptor-Positive Breast Cancer
Hormone receptor (HR)-positive (ie, estrogen receptor (ER)- and/or progesterone receptor (PR)-positive) and human epidermal growth factor receptor 2 (HER2)-negative breast cancer contributes to 70% of breast cancer cases in the Western countries. Only 15% of these breast cancer patients develop recurrence in <10 years when treated only with adjuvant endocrine therapy. 19 Adjuvant chemotherapy helps to reduce the risk of recurrence of breast cancer in these patients. This led the National Institutes of Health consensus recommend adjuvant chemotherapy for most patients which decreased the mortality of breast cancer patients. 20 Given the low relative risk of recurrence and partial chemotherapy effectives, a majority of patients receive adjuvant chemotherapy unnecessarily and 85% of women can be spared from treatment.19,20 Consequently, a method to screen for and identify breast cancer patients who could benefit from chemotherapy has emerged as a key area of current research. The risk assessment of postoperative recurrence is mainly based on the clinicopathologic characteristics of patients and the results of multi-gene testing. The advent of multi-gene testing provides more auxiliary tools for the prognosis, prediction, and treatment of early breast cancer. By detecting the expression of specific genes, the prognosis of different patients can be evaluated, and the therapeutic effect can be predicted, to guide the decision making of adjuvant chemotherapy, radiotherapy, and endocrine therapy. At present, the multi-gene assays mainly recommended by domestic and foreign consensus include Oncotype Dx® (21 genes), MammaPrint® (70 genes), RecurIndex® (28 genes), and Breast Cancer Index® (7 genes).20-22
The pursuit of standard chemotherapy regimen optimization in the treatment of HR-positive breast cancer is continuously ongoing. Many trials have recently examined the efficacy of anthracycline-free chemotherapy regimens, especially in patients with HER2-negative breast cancer, which was found to be a viable alternative.23-25 Phase III ELEGANT study is the first prospective, randomized, open-label clinical trial to compare the safety profile of epirubicin and cyclophosphamide (EC) with docetaxel and cyclophosphamide (TC). The study first reported similar efficacy results of EC and TC with no significant difference in survival, providing favorable evidence for chemotherapy regimens in patients with node-negative low-risk luminal breast cancer. 26 A meta-analysis of 37 298 patients from 26 clinical trials included by the EBCTCG compared the efficacies of dose-dense and standard chemotherapy regimens. The latest analysis of the data revealed that dose-dense regimen (every 2 weeks) containing taxanes could lower the recurrence rate (24.0% vs 28.3%) and reduce breast cancer mortality (16.8% vs 19.6%) as against the standard regimen (every 3 weeks). 27 Dose-dense chemotherapy could improve the long-term survival benefit of patients with early high-risk breast cancer and shorten the duration of treatment. Studies are required to evaluate the usefulness of dose-dense chemotherapy approach in HR-positive early breast cancer settings. Currently, dose-dense chemotherapy has been incorporated into the domestic and foreign guidelines as routine postoperative adjuvant chemotherapy for high-risk patients. Notably, the dose-dense regimen breaks the balance between efficacy and safety to some extent, and this trade-off puts higher demands on clinicians.
HER2-Positive Breast Cancer
In recent years, a growing number of studies have begun to explore whether anthracyclines can be avoided without affecting prognosis of breast cancer patients, considering their high toxicity and adverse effects. This approach was first initiated in low-risk HER2-positive early breast cancer patients. The HERTC study exploring the TCH (docetaxel + cyclophosphamide + trastuzumab) regimen in HER2-positive early breast cancer patients found that 2-year DFS rate was 100% and 98.1% in axillary node-negative patients with tumor diameters of <1 cm and 1.1–2.0 cm, respectively. This study confirms the efficacy of TCH regimen as adjuvant therapy for HER2-positive breast cancer patients. 28 The single-arm APT study explored the efficacy and safety of adjuvant paclitaxel plus TH in patients with node-negative, early HER2-positive breast cancer with tumors smaller than 3 cm. Propensity-score matching (PSM) showed that patients who received TH and non-paclitaxel chemotherapy had similar invasive DFS (IDFS) (96.5% vs 92.9%) and OS (99.3% vs 97.4%) as those who received paclitaxel and TH, although the incidence of adverse reactions was lower in the latter group. 29 For the purpose of reducing impact of adverse reactions on patients and improve the quality of life, chemotherapy “subtraction” on the premise of ensuring the efficacy has gradually started to show advantages.
Adjuvant Targeted Therapy
Adjuvant targeted therapy that targets molecular pathways was used to reduce the risk of recurrence especially in HER2-positive breast cancer patients. 30 Approximately 20%–30% of breast cancer tumors overexpress or amplify HER2. 31 This molecular subtype is highly aggressive and malignant and contributes to high rates of metastasis and mortality. 32 Anti HER-2 monoclonal antibody TH was shown to improve DFS (84%) and OS (92%) in HER2-positive early breast cancer patients when used in combination with chemotherapy in adjuvant settings. 33 Earlier TH was the only anti-HER2 drug available but in the past 20 years, landscape of HER2-targeted drugs has been constantly evolving. This led to the emergence of new drugs such as pertuzumab, lapatinib, trastuzumab emtansine (T-DM1), and trastuzumab deruxtecan (DS8201) that have significantly improved the therapeutic effect and prognosis of HER2-positive breast cancer patients. 34
APHINITY study found that the addition of pertuzumab to TH and chemotherapy decreases the recurrence rate (7.1% vs 8.7%, P = 0. 045) and prolongs the IDFS (94.1% vs 93.2%) in early HER2-positive breast cancer patients. 35 This study establishes the role of dual-targeted therapy in the standard of care for HER2-positive breast cancers. A subsequent update on the APHINITY study published the data of 8.4-year follow-up that showed an OS hazard ratio (HR) of .83 (95% CI: .68–1.02; P = .078) with an 8-year OS rate of 92.7% vs 92.0% for single-targeted vs dual-targeted therapy, respectively. The updated IDFS HR was .77 (95% CI: .66–.91), and the node-positive (N+) cohort IDFS HR was .72 (95% CI: .60–.87). Updated study results suggested that IDFS benefit from pertuzumab was maintained in HER2-positive early breast cancer, while patients with node-positive breast cancer were benefited more from dual-targeted therapy. 36
The ExteNET multi-center, randomized, double-blind phase III clinical trial enrolled 2840 HER2-positive early breast cancer patients treated with trastuzumab in combination with neoadjuvant/adjuvant chemotherapy. Intensive adjuvant therapy with neratinib initiated in HR-positive in ≤1 year showed significantly improved outcomes in terms of 5-year IDFS (7.4%) and 8-year OS (9.1%) with fewer central nervous system adverse events. 37
ATEMPT study enrolled 497 patients with stage I HER2-positive breast cancer patients who were randomized to receive either the trastuzumab regimen or trastuzumab emtansine monotherapy for 1 year. The results indicated 1 year of adjuvant trastuzumab emtansine was associated with excellent 3-year iDFS. The number of recurrent events was lower in the trastuzumab emtansine group with a 3-year IDFS rate of 97.8% and relapse-free survival (RFS) rate of 99.2%. Although clinically relevant toxicities were not significantly reduced, it is suggested that adjuvant targeted therapy without chemotherapy can serve as an alternative to taxane-based trastuzumab regimen without compromising efficacy. 38 With the advent of antibody drug conjugates, adjuvant therapy options for treating early HER2-positive breast cancer patients have become enriched. 39 Evolution of HER2-targeted therapies for patients with HER2-positive breast cancer is an important milestone in breast cancer treatment and enables clinicians to design more beneficial personalized treatments.
Adjuvant Endocrine Therapy
HR-positive breast cancer is the most common subtype of breast cancer mostly diagnosed with early-stage disease. Optimization of adjuvant systemic therapy has the potential to exert a broad impact on the treatment of HR-positive early breast cancer patients. Currently, adjuvant therapy for early-stage HR-positive breast cancer includes endocrine therapy, with or without chemotherapy.40,41 Endocrine therapy had long been recognized as a powerful and important adjuvant treatment for women with HR-positive early-stage breast cancer. In 1982, adjuvant tamoxifen given for 2 years was shown to reduce the risk of recurrence. Subsequently, several randomized clinical trial data have established the benefits of ovarian function suppression (OFS), tamoxifen, and aromatase inhibitors (AIs) alone or in combination with other agents. The survival benefits of endocrine therapy for a minimum of 5 years in stage I through III pre- and postmenopausal women with HR-positive breast cancer were further confirmed by meta-analyses studies which assessed multiple clinical trial data. In postmenopausal women, AIs were shown to be more effective while in patients with higher-stage/risk disease, prolonged adjuvant therapy for up to 10 years was reported to be useful. Ovarian suppression plus an AI was shown to be more beneficial than tamoxifen in premenopausal women with HR-positive breast cancer. Recent trials evaluating adjuvant endocrine therapy with CDK-4/-6 inhibitors show marked survival benefits in combination with endocrine therapy in high- and intermediate-risk HR-positive early breast cancer patients.2,42 The development and clinical practice of endocrine therapy has further enabled the improvement of clinical outcomes and lowered the risk of relapse and mortality and also reduces the burden of overtreatment in breast cancer patients. 40
HR-Positive/HER2-Negative Breast Cancer
Seventy percent of breast cancers belong to HR-positive and human epidermal growth HER2-negative subtype at initial diagnosis.43-45 Since metastatic recurrence of HR-positive breast cancer is associated with high mortality, researchers are constantly exploring new and more effective drugs targeting endocrine-related pathways in addition to standard treatment regimens. According to the ATAC study, a 5-year AI therapy significantly increased DFS and decreased the chance of recurrence compared to a 5-year tamoxifen treatment in postmenopausal individuals (HR 0.81; 95% CI, 0.67–0.98; P = 0.03), demonstrating the importance of AI as a standard adjuvant therapy for postmenopausal early breast cancer. 46 As a result of intensive clinical research on CDK4/6 inhibitors for early breast cancer, abemaciclib in combination with endocrine therapy was approved by the US Food and Drug Administration (FDA)for the adjuvant treatment of HR-positive, HER2-negative, node-positive, early breast cancer patients at a high risk of recurrence and patients with a Ki-67 score ≥20%.47,48
The UNIRAD study assessed 2-year efficacy of mammalian target of rapamycin (mTOR) inhibitor everolimus in combination with adjuvant endocrine therapy vs endocrine therapy alone (AI or tamoxifen based on physician’s choice) in 1278 patients with ER+/HER2-early breast cancer. 49 The study was suspended due to a high discontinuation rate of 53.4% in the everolimus group and 22.3% in the placebo group, even before the first planned interim analysis. This emphasizes the necessity for drugs provided in combination with endocrine therapy in patients with early breast cancer to have long-term tolerability. An oral PI3Kα-selective inhibitor, alpelisib, showed a 7.9-month numeric improvement in median OS when alpelisib was added to fulvestrant treatment in PIK3CA-mutated, HR-positive, HER2-negative advanced breast cancer patients in SOLAR-1 trial. 50 Alpelisib in combination with fulvestrant is the first drug to be approved by the FDA for the treatment of PIK3CA-mutated advanced or metastatic breast cancer patients, 51 and the efficacy of alpelisib in ER-positive early breast cancer patients remains to be explored. CoopERA study, which provided the first clinical evidence of oral selective ER degraders (SERDs) in early ER-positive/HER2-negative breast cancer, compared giredestrant with anastrozole in neoadjuvant settings. This study found that, giredestrant treatment resulted in further reduction of Ki67 expression from baseline compared to anastrozole (−81%; 95% CI, −86%, −75% vs −74%; 95% CI, −80%, −67%). 52 Several large adjuvant trials comparing oral SERDs with standard endocrine therapy have been initiated.53,54 Superior clinical potential of these drugs will be reflected by their rapid transition for the treatment of early-stage disease prior to approval of advanced breast cancer.
At the same time, the optimal regimens for applicable population of endocrine therapy are constantly being explored. Results from an 8-year follow-up of the MINDACT trial showed that, more locoregional recurrences and contralateral breast cancer were observed in patients with stage I low-risk breast cancer who did not receive adjuvant endocrine therapy (4.7% and 4.6% in patients receiving no adjuvant systemic therapy vs 1.4% and 1.5% in patients receiving only endocrine therapy). Overall, significantly fewer breast cancer events were observed in patients receiving adjuvant endocrine therapy, although the effect of endocrine therapy on the distant metastasis-free interval was limited. 55 A recent propensity-matched study compared survival differences between patients with weakly ER-positive early breast cancer who did not receive endocrine therapy and those who received 2–3 years or 5 years of endocrine therapy. No significant differences in 5-year DFS rates between the 2–3-year group and the 5-year group were observed after a median follow-up of 65 months (HR .82; 95% CI, .51–1.33; P = .43). Short-term adjuvant endocrine therapy for 2–3 years may serve as an alternative to the standard 5-year adjuvant endocrine therapy for patients with weakly ER-positive early breast cancer. 56
HR-Positive/HER2-Positive Breast Cancer
HR-positive and HER2-positive breast cancer is a unique subtype and differs from other subtypes in clinical behavior, molecular function, signaling pathway, and treatment sensitivity. Optimal endocrine therapeutic approach for HR-positive/HER2-positive breast cancer remains controversial. 57 The currently recommended adjuvant therapy for HR-positive/HER2-positive breast cancer is anti-HER2 targeted therapy in combination with chemotherapy followed by endocrine therapy. 58 The SYSUCC-002 trial studied the efficacy and safety of TH in combination with endocrine therapy or chemotherapy as first-line treatment in patients with HR-positive/HER2-positive metastatic breast cancer. A median progression-free survival (PFS) of 19.2 months (95% CI: 16.7–21.7) in the endocrine therapy arm and 14.8 months (12.8–16.8) in the chemotherapy arm (hazard ratio of .88, 95% CI: .71–1.09; P non-inferiority <.0001) was observed. Trastuzumab plus endocrine therapy was found to be non-inferior to trastuzumab plus chemotherapy, and a significantly higher incidence of toxicity was observed in the chemotherapy arm than the endocrine therapy arm. 59 Still there is a lack of relevant evidence on the adjuvant therapy of early breast cancer, but for low-risk patients who do not need chemotherapy, or those who need chemotherapy but have issues in tolerability, endocrine therapy in combination with targeted therapy can be considered.
Adjuvant Bone-Modifying Agent
In recent years, the value of the third-generation bisphosphonates in bone protection and treatment of breast cancer bone metastasis have been gradually explored. In 2022, the American Society of Clinical Oncology (ASCO) introduced guidelines for adjuvant therapy of bone-modifying agents in early breast cancer. It highlighted that, despite the EBCTCG meta-analysis finding advantages for bisphosphonates in all subgroups of postmenopausal early breast cancer patients, the benefit was rather marginal. 60
EBCTCG meta-analysis found highly significant reductions in recurrence (rate ratio [RR] 0.86; 95% CI 0.78–0.94; P = 0.002), distant recurrence (RR 0.82; 95% CI 0.74–0.92; P = 0.0003), bone recurrence (RR 0.72; 95% CI 0.60–0·86; P = 0.0002), and breast cancer mortality (RR 0.82; 95% CI 0.73–0.93; P = 0.002) in postmenopausal women while in premenopausal women the treatment had no apparent effect on any outcome. 61 Basic studies have shown that bisphosphonates can inhibit proliferation of breast cancer cells and induce apoptosis and have anti-angiogenic properties in terms of anti-tumor capacity, with direct anti-tumor effects.62-65 Of these, zoledronic acid was identified to be the most potent bisphosphonate. 66 Several studies have established the anti-tumor status of zoledronic acid in the treatment of early breast cancer as it may reduce the risk of recurrence and improve OS. Zoledronic acid was recommended by the 2022 ASCO guidelines for adjuvant therapy in early breast cancer patients.
Regardless of molecular typing, anti-tumor effects of zoledronic acid in breast cancer patients have been demonstrated by several randomized, open-label, phase III studies. The ABCSG-12 trial enrolled 1800 premenopausal patients with HR-positive early breast cancer who received zoledronic acid combined with endocrine therapy. This trial showed an absolute increase in DFS rate of 3.4% (88.4% vs 85.0%), improved OS rate (96.7% vs 94.5%), and a 23% reduction in the risk of relapse (HR = .77; 95% CI, .60–.99, P = .042), when compared with endocrine therapy alone. 67 The AZURE study analyzed the efficacy of zoledronic acid in combination with adjuvant systemic therapy in 3360 women with breast cancer (all molecular subtypes included). In postmenopausal patients, systemic therapy combined with zoledronic acid reduced the risk of disease recurrence by 18% (HR = .82; 95% CI, .67–1.00), the risk of developing invasive disease by 22% (HR = .78; 95% CI, .64–.94), and the risk of death by 31% (HR = .69; 95% CI, .50–.94; P = .019). 68 The results of ZO-FAST study, which enrolled 1065 HR-positive postmenopausal early breast cancer patients, made comparative efficacy evaluation between early zoledronic acid plus endocrine therapy and delayed zoledronic acid plus endocrine therapy. The findings showed that the early use of zoledronic acid increased DFS (HR = .66; 95% CI, .44–.97; P = .0375) and decreased the risk of disease recurrence by 34% as compared to delayed use (P < .05). 69 SWOG S0307 showed no difference in efficacy between oral clodronic acid and oral ibandronic acid with zoledronic acid in the adjuvant therapy of early breast cancer, despite an increased rate of osteonecrosis of the jaw with zoledronic acid. 70
Studies exploring the efficacy of denosumab, an anti-receptor activator of nuclear factor-κB ligand (RANKL) monoclonal antibody, in the adjuvant therapy of breast cancer have also reached the study endpoints in recent years. An international, double-blind, randomized, placebo-controlled phase III trial, D-CARE, enrolled 4509 breast cancer patients to receive either denosumab (120 mg) or matching placebo, starting with neoadjuvant or adjuvant chemotherapy. Primary analysis showed that bone metastasis-free survival was not significantly different between the treatment groups. According to this study, RANKL inhibition might delay bone metastases in early breast cancer patients but did not enhance disease-related outcomes for high-risk early breast cancer patients. 71
Another prospective, double-blind, placebo-controlled phase III clinical study (ABCSG-18) explored the efficacy of denosumab in adjuvant settings for breast cancer. Long-term follow-up demonstrated that denosumab (60 mg) improved DFS and OS, with an absolute difference of 5.4% in DFS at 11 years, and an improvement rate by 17% in risk of progressive disease (HR = .83; 95% CI, .71–.97; P = .02), compared to placebo. Further statistical analysis showed significant OS benefit, with the risk of death decreased by 26% (HR = .74; 95% CI, .58–.94; P = .013). 52 Conflicting results of these 2 studies call into question the anti-tumor efficacy of denosumab in early breast cancer patients. Therefore, denosumab is not currently recommended for adjuvant therapy of early breast cancer and requires further evaluations.
Adjuvant Intensive Therapy Following Neoadjuvant Therapy
Presence of residual disease after neoadjuvant therapy indicates the existence of partial treatment resistance in the tumor. After neoadjuvant therapy, achieving a pathologic complete response is associated with better prognosis in breast cancer patients. Additional anti-cancer treatments might help breast cancer patients with residual disease after neoadjuvant chemotherapy. 72 Many post-neoadjuvant therapy trials evaluating additional chemotherapy with different agents, bisphosphonates, and poly ADP-ribose polymerase inhibitors (PARPi) have been conducted.
Results of KATHERINE study revealed that 14 cycles of adjuvant intensive T-DM1 therapy after surgery further improved iDFS in the T-DM1 group compared to the TH group (88.3% vs 77.0%; HR = .50, 95% CI, .39–.64; P < .0001) in HER2-positive breast cancer patients who did not achieve pCR after neoadjuvant therapy. 73 CREATE-X study showed that postoperative administration of 6–8 cycles of capecitabine could significantly improve 5-year DFS and OS, with good safety and tolerable toxicity in triple-negative breast cancer patients failing to achieve pCR. 74 The OlympiA study was designed to evaluate the efficacy and safety of olaparib and placebo as adjuvant therapy after standard of care for patients with germline BRCA (gBRCA)-mutated, HER2-negative early breast cancer at a high risk for recurrence. Results revealed that adjuvant intensive therapy with olaparib significantly improved 3-year distant DFS (87.5% vs 80.4%; P < .001) and reduced mortality after 1 year (3.21% vs 4.68%; P = .02). 75 The interim analysis of KEYNOTE-522 revealed that patients with triple-negative breast cancer who had been treated with PD-1 inhibitors in the neoadjuvant settings, regardless of their pCR status after surgery, had a lower risk of distant recurrence after continued use of PD-1 inhibitors for 1 year. 76 DESTINY-Breast05 explored the efficacy and safety of ADC drug DS8201 vs T-DM1 in adjuvant intensive therapy of HER2-positive breast cancer with neoadjuvant non-pCR, with the primary endpoint of investigator-assessed IDFS. Although the study is still ongoing, DS8201-based ADC drugs have shown breakthrough efficacy in the past. Hence, this study is expected to show improvement in the cure rate and further redefine its standard of care for HER2-positive breast cancer patients. Several interesting strategies are currently being evaluated in the post-neoadjuvant setting and a precise understanding on tumor characteristics will improve adjuvant intensive therapy strategies.
Future Prospects
Postoperative adjuvant therapy is a key part of the comprehensive breast cancer treatment. Standardized adjuvant therapy might help in the possibility of early breast cancer cure, significantly prolonging the survival time and reducing the risk of recurrence. The commonly used anthracyclines for adjuvant therapy serve potential survival benefits for early breast cancer patients but their long-term cytotoxicity led researchers to seek alternative therapeutic approaches. Different treatment initiatives have been studied for different breast cancer types. In the absence of standard chemotherapy for HR-positive early breast cancer, dose-density chemotherapy has shown promise but the equilibrium between efficacy and safety cannot be maintained, which poses a concern. Meanwhile, targeted therapy with monoclonal antibody demonstrated equal as well as superior efficacy and safety.
Single and dual-targeted antibody therapies showed promise in the last 2 decades and are also constantly evolving. It also remains as a milestone in personalized breast cancer treatment approaches. Endocrine therapy also showed improved clinical outcomes which can be useful in negating high-toxicity profiles of anthracyclines. Recently, PARPi therapy exhibited significant benefits in post-neoadjuvant intensive therapy settings. Consistent alternative measures have been taken so far in reducing the toxicities by adjuvant chemotherapy using anthracyclines. Hence, further studies should be encouraged to test the clinical outcomes of anthracycline-free regimens including different types of early breast cancer patients to identify their benefits and shortcomings. Moreover, studies on personalized adjuvant treatment approaches using advanced diagnostic tools to accurately identify the breast cancer type should be given additional consideration since early breast cancer detection and clinical management are highly important in eradicating further recurrences. With the deeper scientific understanding on the mechanism of breast cancer occurrence and development, and the continuous clinical trial investigations on new drugs and treatment regimens, adjuvant therapy for breast cancer will improve the therapeutic effect, thus bringing more survival benefits to early breast cancer patients.
Footnotes
Acknowledgments
The authors acknowledge Swathirajan CR, PhD, of Indegene Pvt. Ltd. for providing medical writing and editorial services on the basis of the authors’ inputs and direction.
Author Contributions
W.H. and N.L. contributed to the idea, design, and conceptualization of the study, and W.H. and D.X. read and approved the final manuscript. All authors reviewed the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
