Abstract
A 0.5-cm nodular thickening of the vagina was observed in a flying fox (Pteropus poliocephalus) that had been rescued after becoming entangled in fruit netting. Over the following 6 mo, the thickening progressed to diffuse multinodular mucosal thickening of both the vagina and anus. The proliferative lesions were removed surgically. Histologically, the thickened mucosa was arranged in numerous small exophytic papillomas. Cells within the basilar layers were crowded and basophilic. Rarely, enlarged cells that contained increased quantities of pale, smudged eosinophilic cytoplasm [consistent with papillomavirus (PV)-induced cellular changes] were visible. PCR amplified a PV DNA sequence from a sample of affected vaginal mucosa; the sequence was ~90% similar to a PV DNA sequence previously detected as a subclinical infection in an African species of fruit bat. The lesion had not recurred within 6 mo of surgical excision. This is the second report of PV-associated disease in bats; to our knowledge, PV infection has not been reported previously in association with anogenital lesions in bats. Additionally, to our knowledge, a PV has not been identified previously in an Australian bat species.
Papillomaviruses (PVs) are well established as causes of a variety of hyperplastic and neoplastic diseases within numerous animal species.11,12 Metagenomics has identified PV DNA in feces and pharyngeal samples from diverse bat species.1,2,5,15 However, there is only a single report of disease associated with PV infection in bats; an Egyptian fruit bat developed cutaneous basosquamous carcinomas and a squamous cell carcinoma in association with PV infection. 7 The associated PV was subsequently fully sequenced and classified as Rousettus aegyptiacus papillomavirus 1 (RaPV1; Papillomaviridae, Psipapillomavirus 1). 13
We describe here a grey-headed flying fox (Pteropus poliocephalus, an Australian species of fruit bat) that developed progressive diffuse papillomatosis within the anogenital mucosa. In addition to the rarity of PV-induced disease described in bats, the anogenital distribution and diffuse papillomatous appearance of these lesions were highly unusual for any of the domestic species. DNA from a novel PV was amplified by PCR from the lesions
A female grey-headed flying fox of unknown age was rescued after becoming entangled in netting surrounding fruit trees. The bat was judged to be in a poor state of nutrition with cutaneous mite infestation and poor fur quality. A poorly defined, 0.5-cm, reddened vaginal nodule was initially interpreted by the wildlife rehabilitator to be a vaginal prolapse, and the bat was monitored over time at a rescue facility. Over the following months, the lesion became progressively larger and more exophytic. Six months after admission into the rescue facility, the mucosa of both the vagina and the anus were reddened and markedly irregularly thickened with the mucosa protruding above the surface of the surrounding skin (Fig. 1A). Differential diagnoses considered at this time included proliferation due to pruritus (e.g., secondary to parasitic infestation) or proliferation due to previous trauma (e.g., after giving birth). However, few processes were considered likely to affect both the vagina and the anus; hence, due to concerns that the bat’s health may be worsening, the lesions due to self-trauma and concerns that the bat may be unable to pass feces through the thickened mucosa, the bat was admitted to a wildlife hospital. Here the thickened tissue from the vagina and anus was surgically excised. Samples of the excised tissue were fixed in formalin and submitted for histology. No bacterial cultures, evaluations for parasitism, or other diagnostic testing were performed in this case
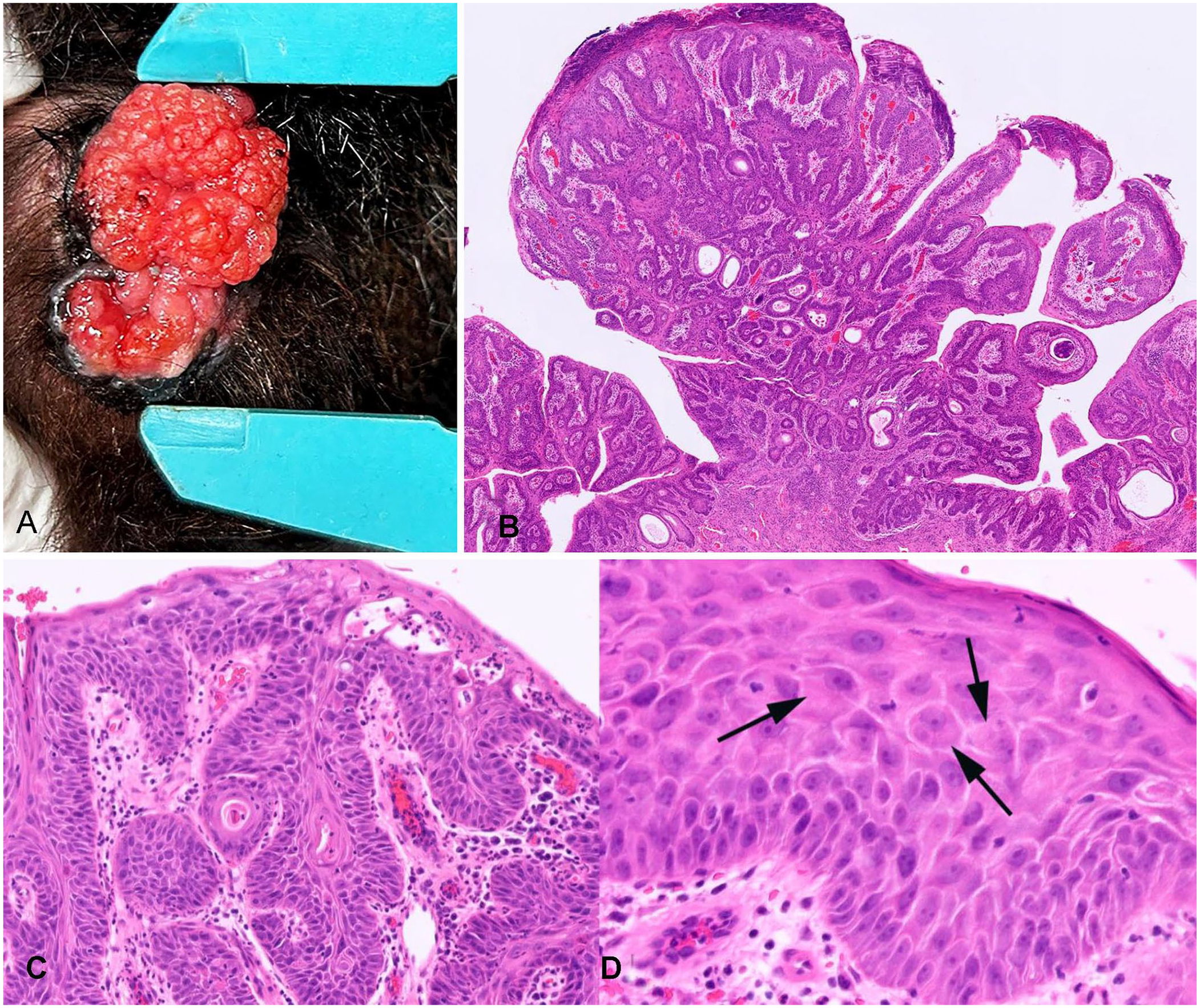
Anogenital papillomatosis in a grey-headed flying fox.
Histology revealed diffuse changes affecting the vaginal and anal mucosa. Subgrossly, multiple papillomatous areas of folded epithelium were supported by a stroma of fibrous tissue. Folding of the epithelium had resulted in cystic cavities that variably contained small quantities of keratin (Fig. 1B). Basilar cells within both mucosae were crowded, with loss of normal orderly arrangement and maturation. Epithelial cells within all layers had increased variability in cell size and shape (Fig. 1C). A small number of cells within the thickened epithelium contained increased quantities of smudged eosinophilic cytoplasm (Fig. 1D). Multifocally, the lesions were covered by a variably thick serocellular crust of parakeratotic epithelium, inflammatory cell debris, and bacteria. The underlying submucosa, especially that of the vagina, was infiltrated by large numbers of inflammatory cells, predominantly neutrophils with smaller numbers of macrophages and lymphocytes. The histologic diagnosis was diffuse vaginal and anal papillomatosis. There has been no evidence of lesion recurrence in the 6 mo following surgical excision.
DNA was extracted from a formalin-fixed paraffin-embedded tissue scroll of an area of vaginal papillomatosis (NucleoSpin DNA FFPE XS kit; Macherey-Nagel) according to the manufacturer’s instructions, and MY09/11 consensus PCR primers were used to amplify ORF L1 PV DNA. 9 No template was added to the negative control reactions. A 387-bp sequence of DNA was amplified from our bat sample and compared to other sequences in GenBank using a BLAST search (https://blast.ncbi.nlm.nih.gov/Blast.cgi). The sequence was most similar to a sequence of PV DNA amplified from a straw-colored fruit bat (Eidolon helvum; KX276955); 15 the 2 sequences were 88.5% similar over the 387-bp length. The next most similar sequences were Sus scrofa (domestic pig) papillomavirus 1, Manis pentadactyla (Chinese pangolin) papillomavirus 1, and Saimiri sciureus (squirrel monkey) papillomavirus 2, which were all ~65% similar to our bat PV sequence. In contrast, our novel sequence was only ~57% similar to RaPV1. We submitted our novel sequence to GenBank (PV054612).
As PVs have been shown to infect bats subclinically, 1 the detection of PV DNA in the papillomatous lesions does not prove causality. However, PVs are well-known to induce epithelial hyperplasia as part of their life cycle, consistent with the lesions observed in our case. 11 In addition, cell enlargement due to increased quantities of smudged cytoplasm is consistent with PV-induced cell changes. 10 Furthermore, the histology of the lesions in our case had some similarities to those described for the lesions associated with PV infection in an Egyptian fruit bat, although, unlike the fruit bat case, there was no evidence of invasion of underlying tissues. 7 Overall, the detected PV appears to be the most likely cause of the lesions in our case. However, no other testing was performed in our case, and an undetected bacterial or parasitic cause of the lesions cannot be excluded.
A search of PubMed revealed that our report is the second report of PV-associated disease in bats. Both reports of disease are in megabat species, which are fruit-eating bats within the family Pteropodidae. 3 Papillomaviral lesions have not been reported in any of the smaller species of insectivorous bats, despite PVs being detected in these species. Whether the larger size of the fruit bats means lesions are more likely to be observed, or whether the megabats are truly more susceptible to developing PV-induced lesions, is uncertain.
The lesions in our case were restricted to the vaginal and anal mucosa. This distribution of PV-associated lesions is common in humans, but unusual in the domestic species. While genital disease caused by PVs is seen in horses and cattle, disease in both species involves the penis much more commonly than the vagina or anus.8,14 In humans, the restriction of lesions to the anogenital areas is due to strict tissue tropism of the PV to these areas. 6 The similar restriction of lesions in the first reported bat case suggests that this PV may also have a tropism for anogenital epithelium. The diffuse nature of the lesions in our case was also highly unusual. Diffuse papillomatosis of the equine penis has been reported, 4 but single or clusters of distinct papillomas are more typically reported due to PV infection in most species.
A search of PubMed revealed that our novel PV sequence is the first PV DNA to be amplified and reported from an Australian bat. As ORF L1 is conserved in PVs, and as this region is used for classification, we used primers to amplify a portion of this gene. However, definitive classification of a novel PV is not possible without amplification of the entire ORF L1, which was not possible using a formalin-fixed sample. When the novel sequence was compared to others, it was most similar to a sequence detected from a straw-colored fruit bat, another megabat species. 15 As megabats share a common evolutionary ancestor, 3 it is unsurprising that PVs from different megabat species are closely related. However, the novel PV sequence that we found was dissimilar to RaPV1, the PV type that was associated with skin neoplasia in an Egyptian fruit bat. 13 This was surprising considering that Egyptian fruit bats are also megabats and considering that the PVs caused epithelial proliferation in both megabat species. 7 Unlike our case, PV infection was associated with invasive neoplasms that developed over several years in the previously reported Egyptian fruit bat. 7
Although there has been no evidence of lesion recurrence 6 mo after excision in our case, we suggest that the bat should be carefully observed for lesion recurrence, the development of new lesions, and progression of lesions to neoplasia. Why the lesions developed in our case is uncertain. However, the bat was noted to be in poor body condition when rescued. This poor body condition could have resulted in reduced immunity and, therefore, allowed progression of the PV infection. It is possible that the lesions have not recurred, at least in part, because the bat has been kept in captivity and is now in a better state of nutrition.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article
