Abstract
Canine distemper virus (CDV) is a common cause of a multisystemic disease in both domestic dogs and wildlife species, including raccoons and foxes. Outbreaks of CDV in domestic dogs in eastern Tennessee have occurred since 2012, and it was determined that these outbreaks resulted from a novel genotype of CDV. We hypothesized that this virus is also infecting area wildlife and may be a source of the virus for these outbreaks in dogs. From 2013 to 2014, autopsies were performed and tissues collected from raccoons (Procyon lotor; n = 50) and gray foxes (Urocyon cinereoargenteus; n = 8) for CDV testing. A real-time reverse transcription PCR was used to document the presence of CDV in tissue samples, and a portion of the virus was subsequently sequenced for phylogenetic analysis. A high percentage of wildlife, both with (86%) and without (55%) clinical signs, tested positive for CDV, with the majority (77%) testing positive for the novel genotype. Microscopic findings, including syncytia in the lungs and viral inclusion bodies in urothelium, astrocytes, neurons, and bronchiolar epithelium, were also consistent with canine distemper. Minimal inflammation in the central nervous system of affected animals was indicative of the acute neurologic form of the disease. Pneumonia and parasitism were also commonly found in CDV-infected animals. Based on these results, CDV appears to be prevalent in eastern Tennessee wildlife. Subclinical or clinically recovered shedders are a potential source of this novel genotype for domestic dogs, and this genotype is genetically distinct from vaccine strains.
Introduction
Canine distemper virus (CDV; order Mononegavirales, family Paramyxoviridae, subfamily Paramyxovirinae, genus Morbillivirus) is a single-stranded, enveloped RNA virus that causes frequently fatal infections in domestic dogs. This virus also affects species in all families of terrestrial carnivores, including Canidae, Felidae, Hyaenidae, Mustelidae, Procyonidae, Ursidae, and Viverridae. 5 In addition, CDV is considered the most important cause of mortality in wild raccoons (Procyon lotor), gray foxes (Urocyon cinereoargenteus), and striped skunks (Mephitis mephitis) in various geographic areas in the United States, 23 and these terrestrial carnivores play an important role in the epidemiology of CDV. 5 As a multisystemic disease, CDV has a complex pathogenesis that can result in pathologic conditions ranging from pneumonia to enteritis to encephalitis. Mortality varies between species and within species. 1 Virulence depends on viral characteristics, which vary based on strains rather than lineages,19,26 and animal characteristics, including immune status, immune response, and age. 21 Mortality in juvenile dogs is higher than in adult dogs. 21
Incubation of CDV can vary from 1 to 4 weeks 2 and can fluctuate in disease presentation (i.e., from no clinical signs or subclinical infection in 25–75% of dogs to neurologic signs and then death). 10 Initially, CDV infects respiratory lymphoid tissue. The first viremic phase, which involves high viral replication and systemic dissemination throughout lymphoid tissues, results in immunosuppression and fever.2,21 A second viremic phase occurs 6–9 days after infection and involves generalized infection of parenchymal and epithelial cells throughout the body.2,15,21 An additional complicating factor is the development of secondary bacterial or protozoal infections as a result of immunosuppression. 21 Neurologic disease typically occurs 1–3 weeks following initial clinical signs and has both acute and chronic presentations. 15 The acute phase, occurring at ~20 days postinfection, is characterized by little inflammation, and the accompanying demyelination is proposed to be secondary to decreased myelin synthesis or metabolic dysfunction. 21
Regardless of clinical signs, animals shed virus in all secretions. 1 The virus has extended persistence in neurons, urothelium, foot pads, and uvea, 21 and animals that recover from acute infections can continue to shed virus in the urine 15 for up to 60–90 days. 10 With the development of antibodies, animals with an adequate immune response usually clear the virus by day 14. 10 However, an estimated 50–90% of raccoons and gray foxes do not develop a sufficient immune response with CDV infection.8,23 Epizootics in wildlife can result in spillover to domestic dogs, and CDV strains circulate between domestic dogs and raccoons. 15
Recent outbreaks of CDV in dogs in eastern Tennessee have been associated with a genotype of distemper that was newly described in the United States.22,29 This genotype was also detected in wildlife, and some of the affected dogs had a history of direct exposure to raccoons prior to the development of infection with CDV. 22 It is thought that direct animal-to-animal contact is required for spread of the virus. 21 The purpose of our study was to characterize the newly identified genotype (suggested America 4 genotype) 22 in wildlife in eastern Tennessee using real-time reverse transcription PCR (RT-PCR). 29 An additional aim of this study was to investigate whether these wildlife species could act as asymptomatic carriers for CDV by evaluating tissues from clinically healthy animals for the presence of the virus. Finally, we investigated gross anatomic and microscopic lesions associated with this novel genotype and compared them to previously documented lesions.
Materials and methods
Animals (raccoons and gray foxes) were selected from cases seen by the Avian, Exotic, and Zoological (AEZ) Medicine and Surgical service at the University of Tennessee Veterinary Medical Center; from the Wildlife Services’ Enhanced Rabies Surveillance Program (part of the U.S. Department of Agriculture, National Rabies Management Plan); and from nuisance animals trapped and euthanized around the Knoxville Zoological Gardens. Animals were collected from November 2013 through August 2014 and included those with and without clinical signs. Clinically ill animals were selected based on the presence of nasal or ocular discharge and/or presence of neurologic signs without a positive rabies test. Healthy animals were defined as animals lacking clinical signs or lacking any history of abnormal behavior. Healthy animals were chosen from counties from which clinically ill animals had previously tested positive for CDV. All healthy animals were also negative for Rabies virus.
RNA was extracted from supernatant obtained from a 1:1 homogenized a mixture of tissues (renal pelvis, urinary bladder, spleen, cerebellum, pons, and lung) and phosphate buffered saline, according to the manufacturer’s instructions. b A real-time RT-PCR assay 7 was used for initial detection of CDV. A portion of the CDV matrix gene and matrix-fusion intergenic region were amplified from each positive sample and sequenced in both forward and reverse directions, as previously described. 29
Samples containing more than 1 strain were cloned to isolate individual sequences. The PCR products were cloned into a plasmid vector c and transformed into Escherichia coli. d Plasmids from several colonies were purified e and then sequenced using the M13 reverse primer at the Molecular Biology Core Facility, University of Tennessee, Knoxville, Tennessee.
Sequences were curated, assembled, and analyzed (primer regions removed). f Sequence alignment was performed using MAFFT, 16 and phylogenetic trees were generated using the Jukes–Cantor 13 and UPGMA (unweighted pair-group method with arithmetic) 28 model. Evaluation of the alignment confirmed the low rates of divergence that are traditionally associated with this virus. Jukes–Cantor and UPGMA were used because CDV has no proofreading capability, and the sequenced area is short and noncoding, so rates of divergence are most likely equal across all clades. Phylogenetic trees were edited using FigTree (version 1.4.2, http://tree.bio.ed.ac.uk/software/figtree/).
A thorough autopsy was performed on all clinically ill animals, and routine tissues were collected into 10% neutral-buffered formalin for histology. The fixed tissues were routinely processed, embedded in paraffin, sectioned, mounted on glass slides, stained with hematoxylin and eosin (H&E), and examined by light microscopy. The H&E tissue sections included brain with cerebral cortex, thalamus, hippocampus, cerebellum and pons, lung, heart, liver, both kidneys including renal pelvis, urinary bladder, mesenteric lymph node, jejunum, and colon. Any additional organs with gross anatomic lesions were examined by authors (pathologist DL Miller and pathology resident JP Pope).
An analysis of variance (ANOVA) was used to compare mean quantification cycle (Cq) values between clinically healthy and clinically ill animals. An ANOVA and Tukey post hoc test were used to compare mean Cq values between different CDV strains detected. p < 0.05 was considered significant.
Results
A total of 58 animals (50 raccoons and 8 gray foxes) were tested for CDV, and 43 (74%) tested positive. This included 31 of 36 (86%) clinically ill animals and 12 of 22 (55%) clinically healthy animals. Of the clinically ill animals, 16 were males and 15 were females. Of the 43 raccoons and foxes testing positive for CDV, 33 had a genotype of CDV that had not been previously detected in the United States or elsewhere until recently (13-1941 reference genotype, Wilkes), 22 including 23 clinically ill and 10 clinically healthy animals (Fig. 1). This genotype (proposed new lineage America 4) was detected in animals originating in 10 different counties in eastern Tennessee (Fig. 2). A different strain was found in 23% of the positive animals (7 clinically ill and 3 healthy), representing detection of this strain in 5 eastern Tennessee counties. The latter strain groups with a virus that we previously identified from a dog (13-1837, Wilkes) 29 and resembles a virus that was reported in association with an outbreak in Wyoming, which was linked to a domestic dog-breeding facility in Kansas 24 (Fig. 1). Unique strains (similar sequences not previously reported) were detected from 3 animals (2 raccoons and 1 fox), and these strains grouped together in a separate clade (Fig. 1). Interestingly, 5 of 43 (9%) animals (all raccoons) tested positive for 2 concurrent strains of CDV (animals 005, 007, 365, 021; Fig. 1), and the majority (n = 3) of these animals were considered clinically healthy. Three of these animals were infected with the newly described U.S. genotype 22 concurrently with another distemper virus strain.
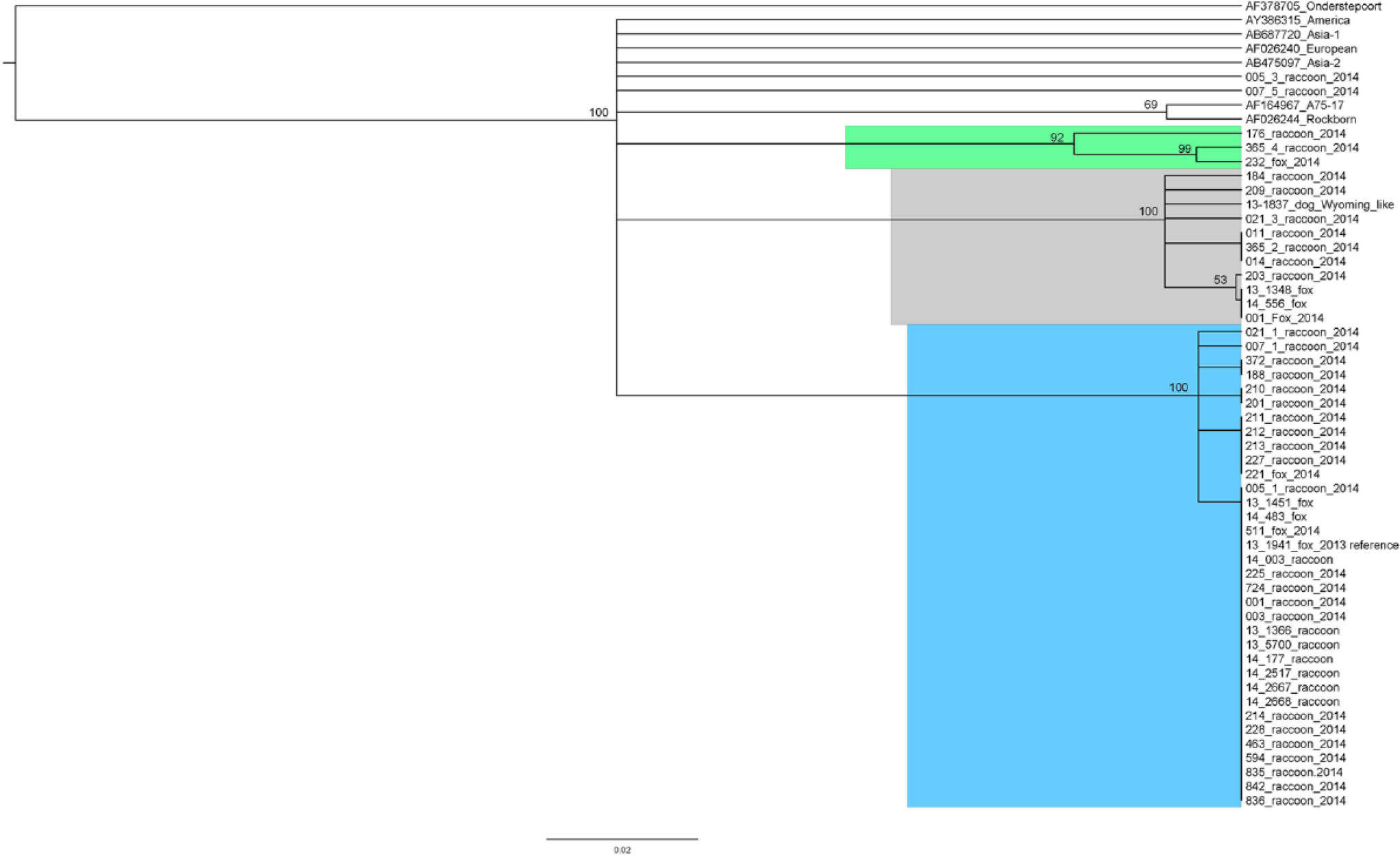
Phylogenetic tree showing strains of Canine distemper virus (CDV) detected in (Procyon lotor) and gray foxes (Urocyon cinereoargenteus) in eastern Tennessee. The majority of identified strains group with a newly described U.S. genotype (blue clade) previously detected in dogs and a fox (reference 13-1941). 22 Some sequences (gray clade) resemble a published strain from a CDV outbreak in Wyoming 24 and group with a strain previously described from a dog (reference 13-1837). 29 Unique sequences were obtained from 2 raccoons and a fox and form another individual clade (green clade). Consensus phylogenetic trees were generated within Geneious software f using the Jukes–Cantor 13 and UPGMA (unweighted pair-group method with arithmetic) model 28 and 1,000 bootstrap replicates. Trees were edited in FigTree. g
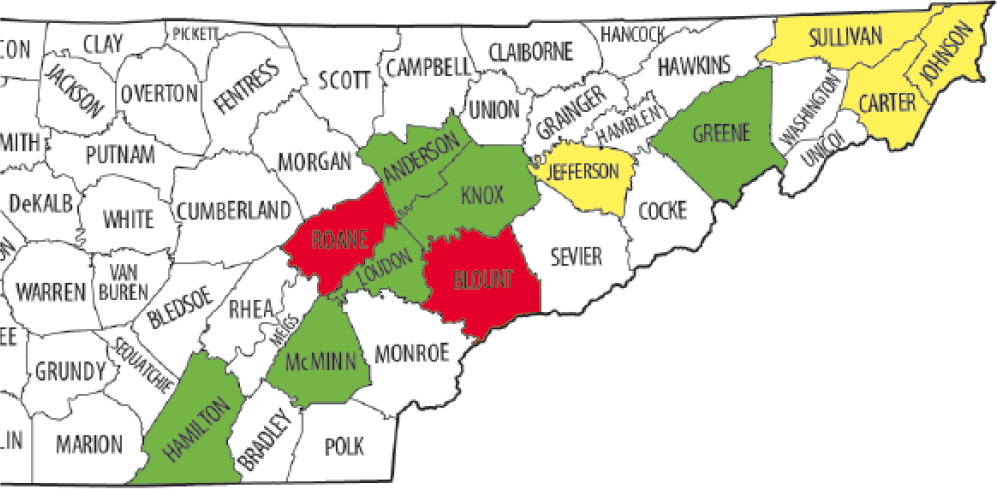
Map of Tennessee counties with Canine distemper virus (CDV)-positive raccoons (Procyon lotor) and gray foxes (Urocyon cinereoargenteus). Yellow indicates counties with animals positive for the newly described genotype. 22 Green indicates counties with animals positive for multiple strains, including the novel genotype. Red indicates counties with animals positive for the Wyoming-like strain. 24
Real-time RT-PCR Cq values were available for 34 of the CDV-positive animals, and 9 were lost because of a mechanical issue with the machine. The average Cq value of the clinically healthy animals tested was 28.75, and the average Cq value of the clinically ill animals was 16.28. There was a significant difference in the mean score between healthy and sick animals (F1.33 = 35.72; p < 0.001). Mean Cq values obtained from the different strains detected were also compared, but no significant difference was detected (p = 0.064).
Clinical signs were similar in both species and for all infecting strains and varied in severity from strictly respiratory symptoms (nasal discharge) to neurologic signs, including confusion and altered behavior, as well as severe neurologic signs such as jaw snapping. Gross anatomic lesions were similar in both species for all infecting strains (Table 1), and included ocular and/or nasal discharge (n = 10), pneumonia (n = 8), endoparasitism (n = 12, including 2 animals with multiple parasites), ectoparasitism (n = 4), or no gross anatomic lesions (6 animals), with the first 2 lesions being indicative of distemper.
Gross anatomic lesions in 19 raccoons (Procyon lotor) and 4 gray foxes (Urocyon cinereoargenteus) infected with a novel genotype of Canine distemper virus.
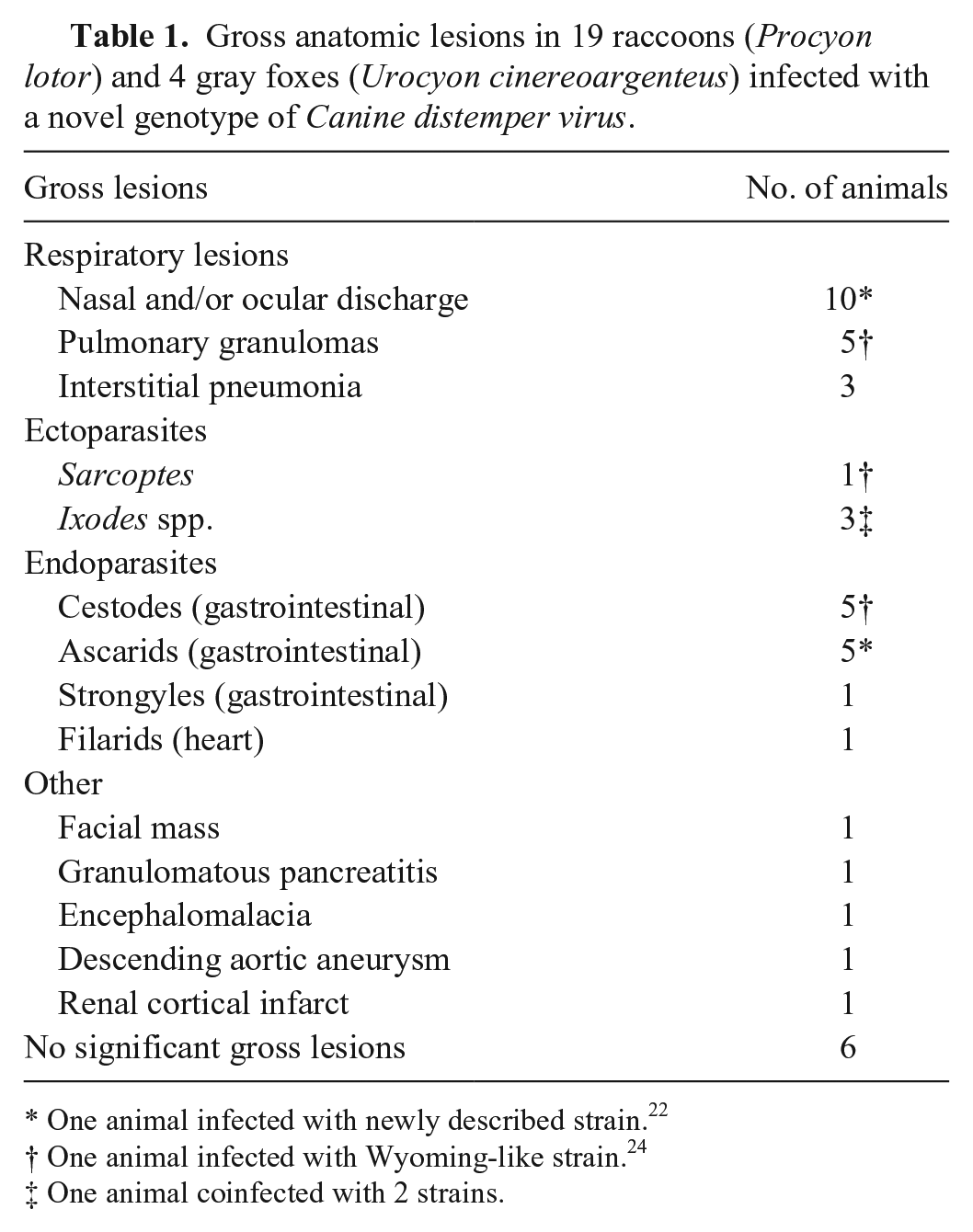
One animal infected with newly described strain. 22
One animal infected with Wyoming-like strain. 24
One animal coinfected with 2 strains.
Histologic lesions were similar in both species and for all infecting strains (Table 2, Fig. 3). Inclusion bodies were observed in 14 animals, including 11 raccoons and 3 gray foxes. Inclusions were observed most commonly in the urothelium (n = 10), lung (n = 10), and brain (n = 8). Inclusions in the urothelium were more commonly intracytoplasmic (n = 10; Fig. 3B). Inclusions in the brain were more commonly intranuclear and in astrocytes (n = 6; Fig. 3C). Inclusions in the lung were often both intranuclear and intracytoplasmic (n = 5), and an additional 5 animals had syncytial cells (Fig. 3A). Despite neurologic signs being a common presenting sign, lesions in the brain were frequently minimal, indicative of the acute phase of the disease. Lesions in the brain more commonly consisted of glial nodules or microgliosis (n = 10), and less commonly minimal lymphoplasmacytic perivascular encephalitis (n = 4), which is common in raccoons infected with distemper.
Number of animals with microscopic lesions by organ system in 19 raccoons (Procyon lotor) and 4 gray foxes (Urocyon cinereoargenteus) infected with Canine distemper virus.

IC = intracytoplasmic inclusion bodies; IN = intranuclear inclusion bodies.
One animal infected with Wyoming-like strain. 24
One animal infected with newly described strain. 22
One animal coinfected with 2 strains.
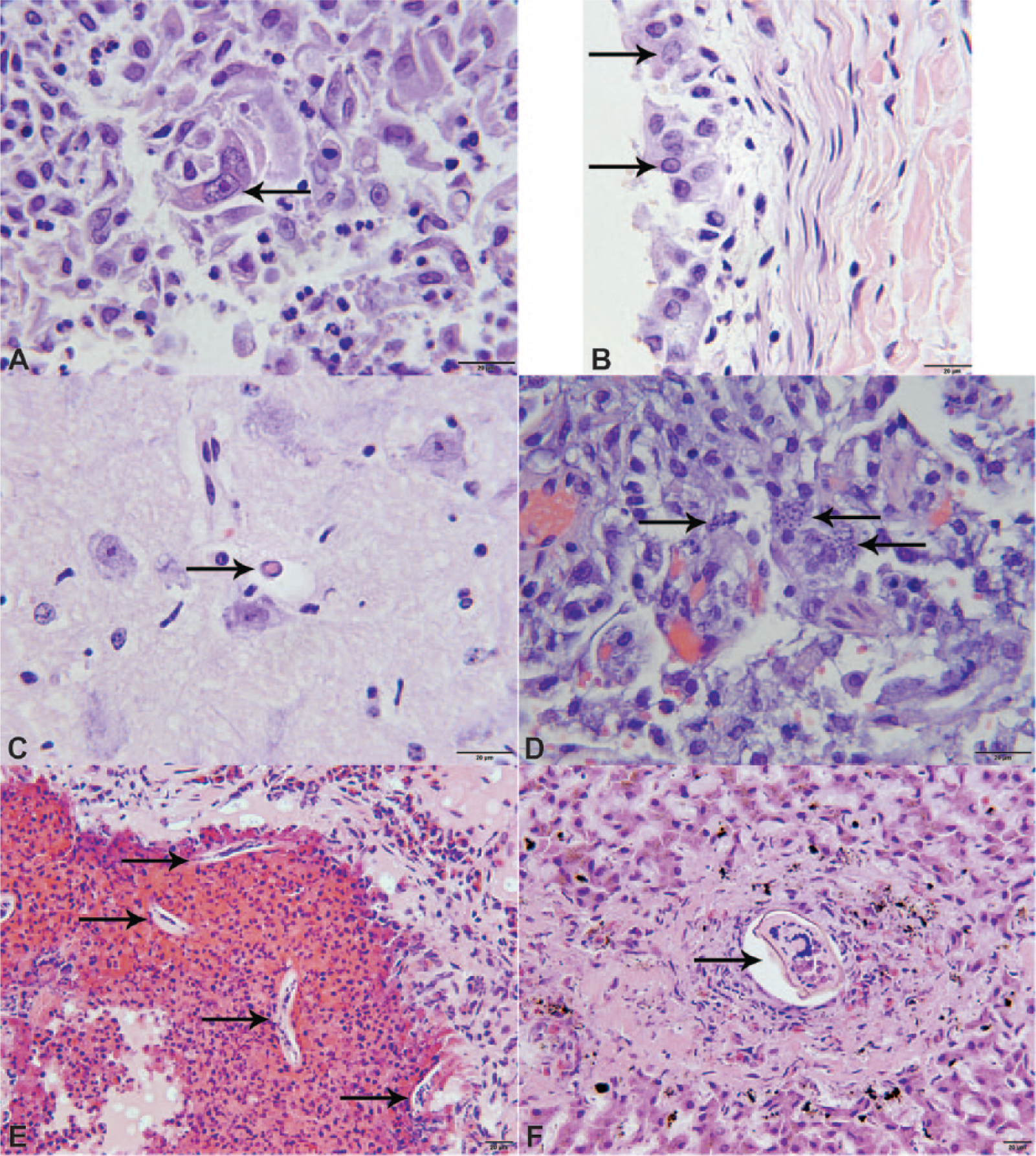
Microscopic lesions in raccoons (Procyon lotor) and gray foxes (Urocyon cinereoargenteus) infected with a novel Canine distemper virus genotype.
Lung lesions were observed in 17 animals and included eosinophilic to granulomatous (n = 11), granulomatous to pyogranulomatous (n = 6), and interstitial (n = 2) pneumonia. Hepatic lesions were observed in 11 animals, and the most common lesion was portal to bridging fibrosis (n = 8; Table 2). Cardiovascular lesions were observed in 4 animals and included necrotizing eosinophilic myocarditis and lymphocytic myocarditis (Table 2). Renal lesions generally consisted of mild inflammation (n = 8; Table 2).
Parasitism was a common microscopic finding (Table 2). Protozoa, including both tachyzoites and cysts containing bradyzoites consistent with Toxoplasma gondii (Fig. 3D), were observed in 3 animals. The protozoa were subsequently confirmed by quantitative real-time (q)PCR g to be T. gondii in 1 animal (Table 2). An additional 4 animals were suspected to have Toxoplasma based on the eosinophilic character of the inflammation in various organs, but qPCR for Toxoplasma was negative. Other parasites included larval metastrongyles in the lung (Fig. 3E) and trematodes in the liver (Fig. 3F).
Discussion
Both clinically healthy and clinically ill raccoons and gray foxes tested in our study in eastern Tennessee were infected frequently with CDV. All animals testing positive for CDV were confined to eastern Tennessee; therefore, this data cannot be extrapolated to the rest of the state of Tennessee or surrounding states. There was no sex predilection in our study, which is similar to results from previous studies.14,23 Over half (55%) of the clinically healthy animals were positive for CDV, which is similar to the serologic assessment in raccoons (54.1%) near a large zoological park in St. Louis, Missouri in 2000–2005. 14 Seroprevalence in raccoons may vary by year 14 and by geographic region. 12 Epizootic outbreaks have been reported to occur in 4-year cycles, likely resulting from a combination of the emptying of host reservoirs as well as failure of raccoons to develop an adequate immune response. 23 Periodic outbreaks in raccoons can lead to a spillover/spillback into domestic dogs and spillover to other wildlife species. 15 Although no increased incidence of distemper has been observed in domestic dogs associated with previous epizootics in raccoons, 12 wildlife species have been implicated as a source for CDV outbreaks in dogs, as in the outbreak in sled dogs in Greenland that was attributed to CDV originating from Artic foxes. 4 In Germany, circulating strains in wild carnivores, including red foxes (Vulpes vulpes) and mustelids (Mustelidae spp.), were phylogenetically related to the CDV strains circulating simultaneously in the domestic dog population. 9 This is similar to the current situation in eastern Tennessee, except that raccoons and gray foxes are the wildlife species involved in this region. Unlike previous reports, 12 there is an apparent increase in clinical distemper in domestic dogs in eastern Tennessee associated with this new genotype, 22 but the original source of the genotype (whether dogs or wildlife) cannot be determined by the current data.
Coinfection with more than 1 strain of CDV in raccoons in our study was a unique finding and suggests that raccoons may serve as a host that would allow for genetic recombination of the virus, resulting in new genotypes that could result in outbreaks in domestic dogs. Coinfection with 2 separate strains in the current study may be partially explained by persistent infection of the initial infecting strain. Some researchers suggest that CDV does not result in persistent infections, 19 whereas others suggest that animals that recover from acute infections can be persistently infected and continue to shed virus for an extended period of time.10,15 T-cell immunity is important for elimination of the virus, and animals with an appropriate immune response typically develop an antibody response 10–14 days after infection. 15 Raccoons are proposed to develop a poor immune response to CDV, and this may allow for infection with multiple strains.8,23 However, interestingly, the majority of the animals in our study with 2 strains were clinically healthy.
In the current study, this lack of clinical signs raises questions regarding the raccoons and foxes that were deemed healthy. It is possible (but unlikely) that the healthy animals had clinical signs that were missed; for example, lethargy, which may be difficult to detect, has been reported as the most common clinical sign in raccoons. 23 Potentially, these clinically healthy animals were in the incubation phase of the disease, but this is also unlikely given the detection of the virus in the tissues. Most likely, the animals were subclinical carriers or recovered but persistently shedding, especially given the statistically significant differences in Cq values between clinically healthy and clinically ill animals. Both carriers and persistent shedders could serve as a source of the virus. Persistent shedders may be particularly significant in colder months, when the virus can survive longer outside the host. 10
Lesions previously reported in raccoons infected with CDV include lymphoid depletion, interstitial pneumonia, cerebellar necrosis, and viral inclusions in multiple tissues, as well as secondary infections.6,11,25,30 Generally, inflammation has been reported as minimal both against the virus itself as well as the opportunistic infections that are believed to be secondary to immunosuppression from infection with CDV. 11 In our study, we observed minimal inflammation in the central nervous system (CNS) of the raccoons and foxes, which is similar to previous reports in raccoons. 11 In a previous study on CDV in wild carnivores, approximately half of infected mustelids (stone martens [syn. beach martens, Martes foina], badgers [Meles meles], polecats [Mustela putorius], and weasels [Mustela nivalis]) had CNS lesions consistent with distemper and included mononuclear inflammation (17%) or demyelination (7%). 27 Inflammation in the acute neurologic manifestation of distemper in domestic dogs is also often minimal, and clinical signs are thought to be associated with direct viral replication and injury. 10 An additional study in domestic dogs reported that 32% of dogs with the acute neurologic manifestation of distemper had inclusion bodies in the CNS, 18 which is similar to our study (6/23, 26%). Comparison with previous findings support that CDV-infected raccoons and gray foxes in the current study likely had the acute neurologic phase of CDV, and that this newly described genotype 22 results in similar lesions, as previously reported.10,11,27 Interestingly, Cq value was not associated with the presence of inclusion bodies in any tissues. Inclusion bodies are typically observed in the acute phase of the infection, but this finding would suggest that the presence of inclusion bodies may not be dependent on the stage of the disease, and, even in acute infections, they might not be observed in tissues.
Additional microscopic lesions of pneumonia, protozoal infections, and icterus were similar to previous reports in CDV-infected wildlife,3,6,17,25,30 and coinfection with T. gondii secondary to immunosuppression is commonly documented in domestic dogs.10,20 Other lesions, including parasitism and hepatic lesions, were considered unrelated to CDV infection and were consistent with other reports in raccoons.11,14
Footnotes
Acknowledgements
We thank Richard Gerhold for parasite identification; Erin Patrick from the USDA APHIS and personnel from the Knoxville Zoological Gardens for collection of animals; Ann Reed for statistical analysis; veterinary students Tim Williams and Gordon Ehrensing and visiting extern Alex Myers for help with autopsies and collection of tissues; Raven Cole for assistance with PCR; Misty Bailey (paid by the University of Tennessee College of Veterinary Medicine) for providing technical editing support; and Joy Chambers (paid by the University of Tennessee College of Veterinary Medicine) for graphic design support.
Authors’ note
The findings and opinions expressed herein belong to the authors and do not necessarily reflect the official views of the United States Army or the Department of Defense.
Authors’ contributions
JP Pope contributed to design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. DL Miller contributed to design of the study, and contributed to acquisition, analysis, and interpretation of data. MC Riley and E Anis contributed to analysis and interpretation of data. RP Wilkes contributed to conception and design of the study, and contributed to acquisition, analysis, and interpretation of data. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Stomacher lab blender, Tekmar Co., Cincinnati, OH.
b.
Viral RNA mini kit, Qiagen Inc., Valencia, CA.
c.
pCR2.1, TA cloning kit, Invitrogen, Life Technologies, Grand Island, NY.
d.
Invitrogen One Shot Top 10 F’, Life Technologies, Grand Island, NY.
e.
Wizard Plus SV minipreps, Promega Corp., Madison, WI.
f.
Geneious (8.1.6 Build 2015-06-16 09:44 Java version 1.7.0_51-b13; 64 bit), Biomatters Limited, Auckland, New Zealand.
g.
Clinical Immunology Laboratory, University of Tennessee Veterinary Medical Center, Knoxville, TN.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Biomedical and Diagnostic Sciences Education Advancement and Research Fund, College of Veterinary Medicine, The University of Tennessee.
